Chinese Journal of Tissue Engineering Research ›› 2017, Vol. 21 ›› Issue (2): 260-267.doi: 10.3969/j.issn.2095-4344.2017.02.018
Previous Articles Next Articles
Preparation of span-poly(ethylene glycol) ultrasound contrast agent microbubbles combined with folate-carbon nano tube-paclitaxel
- 1Pharmaceutical Research Institute of Heilongjiang Province, Jiamusi University, Jiamusi 154007, Heilongjiang Province, China; 2Department of Neurology, First Affiliated Hospital of Harbin Medical University, Harbin 150081, Heilongjiang Province, China; 3Department of Pharmacy, Heilongjiang General Hospital of Arm Police Force, Harbin 150070, Heilongjiang Province, China
-
Received:2016-12-09Online:2017-01-18Published:2017-02-27 -
Contact:Zhang Jie, M.D., Lecturer, Pharmaceutical Research Institute of Heilongjiang Province, Jiamusi University, Jiamusi 154007, Heilongjiang Province, China; Department of Neurology, First Affiliated Hospital of Harbin Medical University, Harbin 150081, Heilongjiang Province, China Zhang Yu, Master, Professor, Pharmaceutical Research Institute of Heilongjiang Province, Jiamusi University, Jiamusi 154007, Heilongjiang Province, China -
About author:Liu Jun-xi, Studying for master’s degree, Pharmaceutical Research Institute of Heilongjiang Province, Jiamusi University, Jiamusi 154007, Heilongjiang Province, China -
Supported by:the National Natural Science Foundation of China, No. 81601616; the Post Doctoral Foundation of Heilongjiang Province, No. LBH-Z15164; the Natural Science Foundation of Heilongjiang Province, No. H2016086; the Scientific and Technologic Innovation Program for the Graduates of Jiamusi University, No. LM2015-091; the Scientific Innovation Group of Jiamusi University, No. CXTD-2013-05
CLC Number:
Cite this article
Liu Jun-xi, Zhang Jie, Zhang Yu, Zhao Yue, Wan Guo-jing, Li Guo-zhong.
share this article
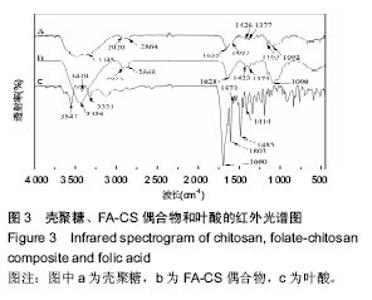
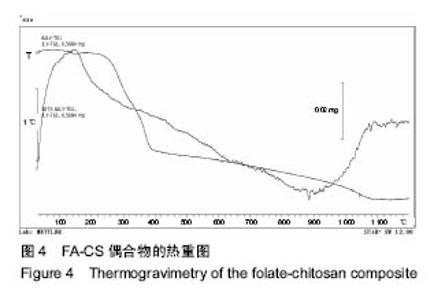
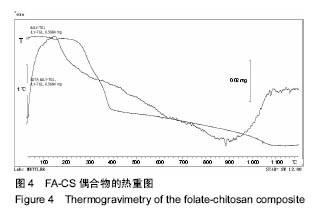
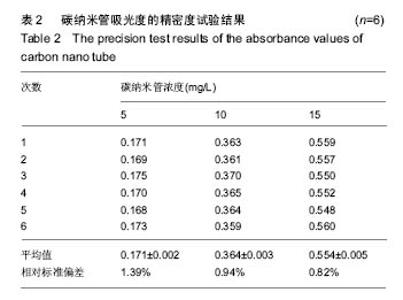
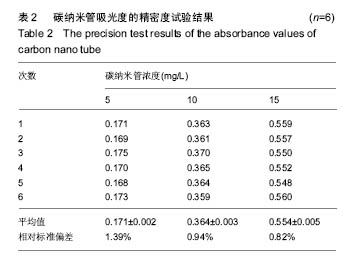
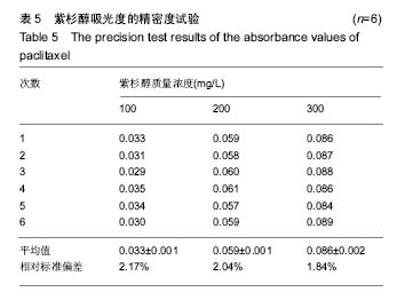
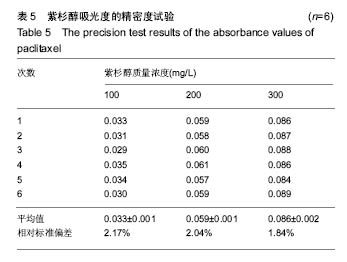
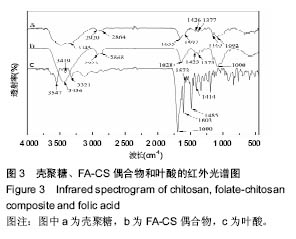
2.2 复合物中间体 2.2.1 FA-CS偶合物 壳聚糖的红外光谱中在3 345 cm-1附近存在较宽的一段吸收峰,这是由于壳聚糖中O-H和N-H的伸缩振动叠加造成的,在2 920 cm-1 和2 864 cm-1 处出现的是C-H伸缩振动吸收峰,在1 655 cm-1和1 597 cm-1吸收峰分别是C-N伸缩振动和氨基(-NH2)的特征峰(图3a)。叶酸的红外光谱中在3 547 cm-1处出现叶酸中羟基(O-H)的吸收峰,在3 419 cm-1和3 321 cm-1处吸收峰是由叶酸中氨基、-NH的伸缩振动引起的(图3c)。FA-CS偶合物红外光谱中在3 436 cm-1附近只有一个吸收峰,为仲酰胺的特征峰,这是由于壳聚糖上的氨基基团与叶酸上的羧基基团(-COOH)发生静电自组装所致;在2 923 cm-1和2 868 cm-1 出现了壳聚糖中C-H伸缩振动吸收峰;在1 655 cm-1和 1 597 cm-1处的吸收峰明显减弱,在1 597 cm-1 处的N-H变形振动峰移动到了高频的1 628 cm-1[35],这是由于FA-CS偶合物中壳聚糖氨基被叶酸部分取代(图3b)。"
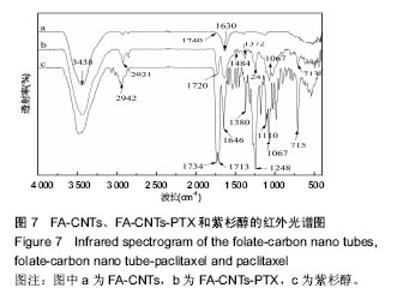
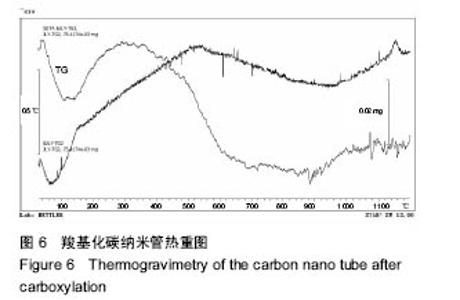
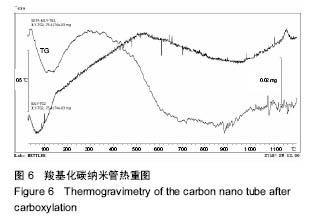
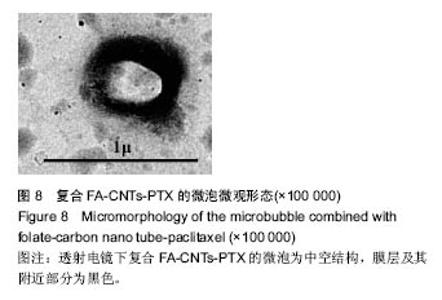
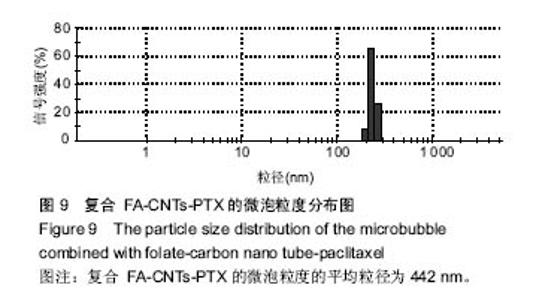
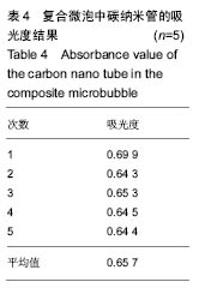
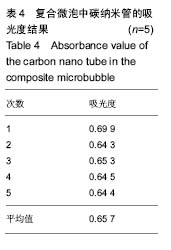
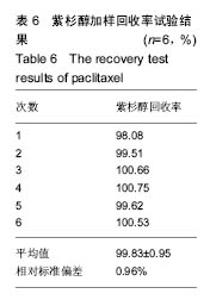
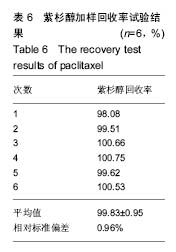
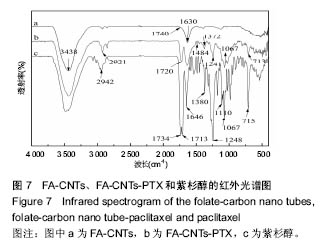
2.2.3 静电自组装产物(FA-CNTs)负载紫杉醇 由图3b,7a可知,静电自组装产物FA-CNTs在3 438 cm-1处出现了FA-CS中仲酰胺的特征峰;在1 740 cm-1处出现了一个小峰,此为酯键上的C=O伸缩振动峰;在1 630 cm-1处为羧基的C=O伸缩振动,这是由于FA-CS在1 628 cm-1处的N-H变形振动峰所致。这说明已成功将FA-CS静电自组装至碳纳米管上。 紫杉醇的红外光谱中,在3 500 cm-1附近的O-H伸缩振动吸收峰与N-H伸缩振动峰重叠;在2 942 cm-1处为苯环上的C-H伸缩振动峰,在1 734 cm-1和1 713 cm-1处出现特征吸收峰为酮羰基C=O的2个裂分峰,在1 645 cm-1处为酰胺基的羰基峰,在1 500 cm-1附近为苯环的骨架振动所产生的吸收峰;在1 380 cm-1处为甲基产生的C-H面内弯曲振动, 1 248 cm-1处为C-N键伸缩振动,在1 067 cm-1、 1 110 cm-1处为仲醇和叔醇中的C-O伸缩振动产生的吸收峰;在900-690 cm-1处为苯环C-H的面外弯曲振动所产生的吸收峰,其中715 cm-1为紫杉醇单取代苯的特征峰(图7c)。 复合物FA-CNTs-PTX在3 438 cm-1处的一个单峰,是图7(a)FA-CNTs中仲酰胺的特征峰,在2 921 cm-1苯环上的C-H伸缩振动峰;在1 372 cm-1处为紫杉醇甲基产生的C-H面内弯曲振动吸收峰,在1 241 cm-1处为紫杉醇的C-N键伸缩振动吸收峰,在1 067 cm-1处为紫杉醇的叔醇中的C-O伸缩振动吸收峰;在713 cm-1为紫杉醇单取代苯的特征峰产生的吸收峰(图7b)。这些说明已成功将紫杉醇负载到FA-CNTs上。 综上可知,成功得到复合物FA-CNTs-PTX。 2.3 复合FA-CNTs-PTX的微泡的制备结果"
| [1]Gramiak R, Shah PM. Echocardiography of the aortic root. Invest Radiol. 1968;3(5):356-366.[2]Sheeran PS, Luois S, Dayton PA, et al. Formulation and acoustic studies of a new phase-shift agent for diagnostic and therapeutic ultrasound. Langmuir. 2011;27(17):10412-10420.[3]Eisenbrey JR, Burstein OM, Kambhampati R, et al. Development and optimization of a doxorubicin loaded poly(lactic acid) contrast agent for ultrasound directed drug delivery. J Control Release. 2010;143(1):38-44.[4]Yan F, Li X, Jin Q, et al. Therapeutic ultrasonic microbubbles carrying paclitaxel and LyP-1 peptide: preparation, characterization and application to ultrasound-assisted chemotherapy in breast cancer cells. Ultrasound Med Biol. 2011;37(5):768-779.[5]Unger EC, Hersh E, Vannan M, et al. Local drug and gene delivery through microbubbles. Prog Cardiovasc Dis. 2001; 44(1):45-54.[6]Eisenbrey JR, Burstein OM, Kambhampati R, et al. Development and optimization of a doxorubicin loaded poly(lactic acid) contrast agent for ultrasound directed drug delivery. J Control Release. 2010;143(1):38-44.[7]Wicki A, Rochlitz C, Orleth A, et al. Targeting tumor-associated endothelial cells: anti-VEGFR2 immunoliposomes mediate tumor vessel disruption and inhibit tumor growth. Clin Cancer Res. 2012;18(2):454-464.[8]Gao ZG, Fain HD, Rapoport N. Controlled and targeted tumor chemotherapy by micellar-encapsulated drug and ultrasound. J Control Release. 2005;102(1):203-222.[9]Wang L, Li L, Guo Y, et al. Construction and in vitro/in vivo targeting of PSMA-targeted nanoscale microbubbles in prostate cancer. Prostate. 2013;73(11):1147-1158.[10]Yang K, Hu L, Ma X, et al. Multimodal imaging guided photothermal therapy using functionalized graphene nanosheets anchored with magnetic nanoparticles. Adv Mater. 2012;24(14):1868-1872.[11]Janib SM, Moses AS, MacKay JA. Imaging and drug delivery using theranostic nanoparticles. Adv Drug Deliv Rev. 2010; 62(11):1052-1063.[12]Barua A, Yellapa A, Bahr JM, et al. Interleukin 16- (IL-16-) Targeted Ultrasound Imaging Agent Improves Detection of Ovarian Tumors in Laying Hens, a Preclinical Model of Spontaneous Ovarian Cancer. Biomed Res Int. 2015;2015: 567459.[13]Mancini M, Greco A, Salvatore G, et al. Imaging of thyroid tumor angiogenesis with microbubbles targeted to vascular endothelial growth factor receptor type 2 in mice. BMC Med Imaging. 2013;13:31.[14]Mannaris C, Averkiou MA. Investigation of microbubble response to long pulses used in ultrasound-enhanced drug delivery. Ultrasound Med Biol. 2012;38(4):681-691.[15]Rapoport N, Nam KH, Gupta R, et al. Ultrasound-mediated tumor imaging and nanotherapy using drug loaded, block copolymer stabilized perfluorocarbon nanoemulsions. J Control Release. 2011;153(1):4-15.[16]Tong R, Cheng J. Paclitaxel-initiated, controlled polymerization of lactide for the formulation of polymeric nanoparticulate delivery vehicles. Angew Chem Int Ed Engl. 2008;47(26):4830-4834.[17]Singh RK, Kim TH, Patel KD, et al. Biocompatible magnetite nanoparticles with varying silica-coating layer for use in biomedicine: physicochemical and magnetic properties, and cellular compatibility. J Biomed Mater Res A. 2012;100(7): 1734-1742.[18]Wu H, Liu G, Zhuang Y, et al. The behavior after intravenous injection in mice of multiwalled carbon nanotube / Fe3O4 hybrid MRI contrast agents. Biomaterials. 2011;32(21): 4867-4876.[19]Lacerda L, Russier J, Pastorin G, et al. Translocation mechanisms of chemically functionalised carbon nanotubes across plasma membranes. Biomaterials. 2012;33(11): 3334-3343.[20]Jiang H. Chemical preparation of graphene-based nanomaterials and their applications in chemical and biological sensors. Small. 2011;7(17):2413-2427.[21]Singh RK, El-Fiqi AM, Patel KD, et al. A novel preparation of magnetic hydroxyapatite nanotubes. Mater Lett. 2012;75(1): 130-133.[22]Prakash S, Malhotra M, Shao W, et al. Polymeric nanohybrids and functionalized carbon nanotubes as drug delivery carriers for cancer therapy. Adv Drug Deliv Rev. 2011;63(14-15): 1340-1351.[23]Kesharwani P, Tekade RK, Gajbhiye V, et al. Cancer targeting potential of some ligand-anchored poly(propylene imine) dendrimers: a comparison. Nanomedicine. 2011;7(3):295-304.[24]Pruthi J, Mehra NK, Jain NK. Macrophages targeting of amphotericin B through mannosylated multiwalled carbon nanotubes. J Drug Target. 2012;20(7):593-604.[25]Biju V. Chemical modifications and bioconjugate reactions of nanomaterials for sensing, imaging, drug delivery and therapy. Chem Soc Rev. 2014;43(3):744-764.[26]Madani SY, Naderi N, Dissanayake O, et al. A new era of cancer treatment: carbon nanotubes as drug delivery tools. Int J Nanomedicine. 2011;6:2963-2979.[27]Rapoport N, Gao Z, Kennedy A. Multifunctional nanoparticles for combining ultrasonic tumor imaging and targeted chemotherapy. J Natl Cancer Inst. 2007;99(14):1095-1106.[28]Liu Z, Yan K g, Lee ST. Single-walled carbon nanotubes in biomedical imaging. J Mater Chem. 2010;21(3):586-598.[29]Wang X, Wang C, Cheng L, et al. Noble metal coated single-walled carbon nanotubes for applications in surface enhanced Raman scattering imaging and photothermal therapy. J Am Chem Soc. 2012;134(17):7414-7422.[30]Tucker-Schwartz JM, Hong T, Colvin DC, et al. Dual-modality photothermal optical coherence tomography and magnetic-resonance imaging of carbon nanotubes. Opt Lett. 2012;37(5):872-874.[31]de la Zerda A, Liu Z, Bodapati S, et al. Ultrahigh sensitivity carbon nanotube agents for photoacoustic molecular imaging in living mice. Nano Lett. 2010;10(6):2168-2172.[32]El-Sayed R, Eita M, Barrefelt A, et al. Thermostable luciferase from Luciola cruciate for imaging of carbon nanotubes and carbon nanotubes carrying doxorubicin using in vivo imaging system. Nano Lett. 2013;13(4):1393-1398.[33]Lee N, Cho HR, Oh MH, et al. Multifunctional Fe3O4/TaO(x) core/shell nanoparticles for simultaneous magnetic resonance imaging and X-ray computed tomography. J Am Chem Soc. 2012;134(25):10309-10312.[34]Delogu LG, Vidili G, Venturelli E, et al. Functionalized multiwalled carbon nanotubes as ultrasound contrast agents. Proc Natl Acad Sci U S A. 2012;109(41):16612-16617.[35]Zhou M, Li PW, Wang G, et al. Preparation of nanoparticles by crosslinking folate conjugated chitosan with vanillin and its characterization. Adv Mater Res. 2012;466-467: 454-457.[36]Kuang SP, Wang ZZ, Liu J, et al. Preparation of triethylene-tetramine grafted magnetic chitosan for adsorption of Pb(II) ion from aqueous solutions. J Hazard Mater. 2013; 260:210-219.[37]Leamon CP, Reddy JA. Folate-targeted chemotherapy. Adv Drug Deliv Rev. 2004;56(8):1127-1141. |
| [1] | Yao Xiaoling, Peng Jiancheng, Xu Yuerong, Yang Zhidong, Zhang Shuncong. Variable-angle zero-notch anterior interbody fusion system in the treatment of cervical spondylotic myelopathy: 30-month follow-up [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1377-1382. |
| [2] | Zhang Jinglin, Leng Min, Zhu Boheng, Wang Hong. Mechanism and application of stem cell-derived exosomes in promoting diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1113-1118. |
| [3] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [4] | He Yunying, Li Lingjie, Zhang Shuqi, Li Yuzhou, Yang Sheng, Ji Ping. Method of constructing cell spheroids based on agarose and polyacrylic molds [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 553-559. |
| [5] | He Guanyu, Xu Baoshan, Du Lilong, Zhang Tongxing, Huo Zhenxin, Shen Li. Biomimetic orientated microchannel annulus fibrosus scaffold constructed by silk fibroin [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 560-566. |
| [6] | Chen Xiaoxu, Luo Yaxin, Bi Haoran, Yang Kun. Preparation and application of acellular scaffold in tissue engineering and regenerative medicine [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 591-596. |
| [7] | Kang Kunlong, Wang Xintao. Research hotspot of biological scaffold materials promoting osteogenic differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 597-603. |
| [8] | Shen Jiahua, Fu Yong. Application of graphene-based nanomaterials in stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 604-609. |
| [9] | Zhang Tong, Cai Jinchi, Yuan Zhifa, Zhao Haiyan, Han Xingwen, Wang Wenji. Hyaluronic acid-based composite hydrogel in cartilage injury caused by osteoarthritis: application and mechanism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 617-625. |
| [10] | Li Hui, Chen Lianglong. Application and characteristics of bone graft materials in the treatment of spinal tuberculosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 626-630. |
| [11] | Gao Cangjian, Yang Zhen, Liu Shuyun, Li Hao, Fu Liwei, Zhao Tianyuan, Chen Wei, Liao Zhiyao, Li Pinxue, Sui Xiang, Guo Quanyi. Electrospinning for rotator cuff repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 637-642. |
| [12] | Guan Jian, Jia Yanfei, Zhang Baoxin , Zhao Guozhong. Application of 4D bioprinting in tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(3): 446-455. |
| [13] | Liu Jiali, Suo Hairui, Yang Han, Wang Ling, Xu Mingen. Influence of lay-down angles on mechanical properties of three-dimensional printed polycaprolactone scaffolds [J]. Chinese Journal of Tissue Engineering Research, 2022, 10(16): 2612-2617. |
| [14] | Huang Bo, Chen Mingxue, Peng Liqing, Luo Xujiang, Li Huo, Wang Hao, Tian Qinyu, Lu Xiaobo, Liu Shuyun, Guo Quanyi . Fabrication and biocompatibility of injectable gelatin-methacryloyl/cartilage-derived matrix particles composite hydrogel scaffold [J]. Chinese Journal of Tissue Engineering Research, 2022, 10(16): 2600-2606. |
| [15] | Li Xuan, Sun Yimin, Li Longbiao, Wang Zhenming, Yang Jing, Wang Chenglin, Ye Ling. Manufacturing of nano-modified polycaprolactone microspheres and its biological effects in dental pulp cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(10): 1530-1536. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||