Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (34): 8962-8969.doi: 10.12307/2026.897
Previous Articles Next Articles
Mazdutide improves cognitive function in APP/PS1/Tau triple transgenic mice
Yuan Jingjing1, Zhang Xiaomin2, Du Pengyang2, Wang Weifeng1
- 1Department of Cerebral Diseases I, Shanxi Provincial Hospital of Acupuncture and Moxibustion, Taiyuan 030001, Shanxi Province, China; 2Department of Neurology, The Second Hospital of Shanxi Medical University, Taiyuan 030001, Shanxi Province, China
-
Received:2025-10-15Revised:2026-02-28Online:2026-12-08Published:2026-04-14 -
Contact:Wang Weifeng, Chief physician, Department of Cerebral Diseases I, Shanxi Provincial Hospital of Acupuncture and Moxibustion, Taiyuan 030001, Shanxi Province, China -
About author:Yuan Jingjing, MS, Attending physician, Department of Cerebral Diseases I, Shanxi Provincial Hospital of Acupuncture and Moxibustion, Taiyuan 030001, Shanxi Province, China -
Supported by:Shanxi Provincial Administration of Traditional Chinese Medicine, No. 024ZYY2B014 (to YJJ); Shanxi Provincial Department of Education Project for Shanxi University of Chinese Medicine in 2020, No. 2020L0436 (to YJJ); Shanxi Provincial Department of Education Project, No. 2022L156 (to ZXM)
CLC Number:
Cite this article
Yuan Jingjing, Zhang Xiaomin, Du Pengyang, Wang Weifeng. Mazdutide improves cognitive function in APP/PS1/Tau triple transgenic mice[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(34): 8962-8969.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

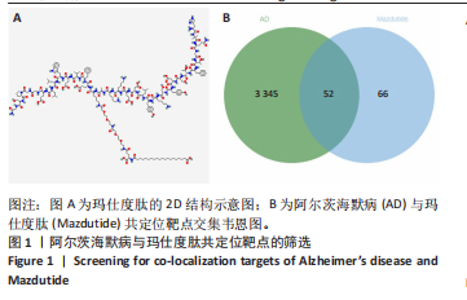
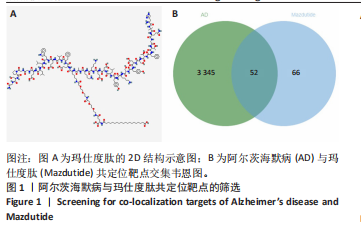
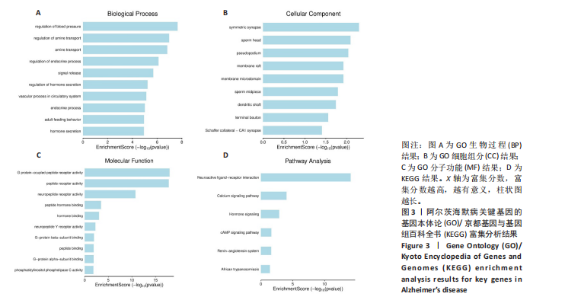
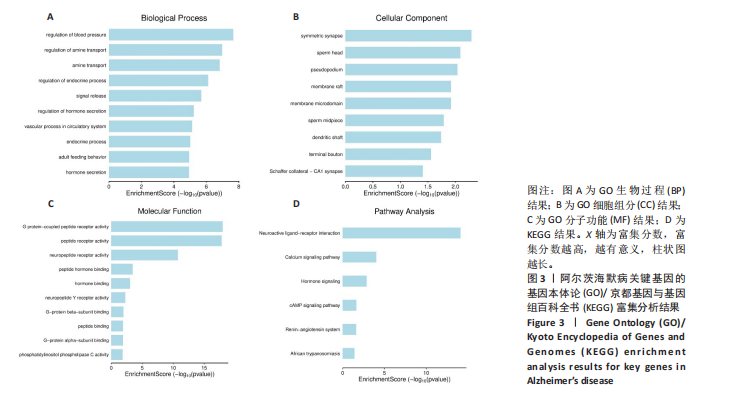
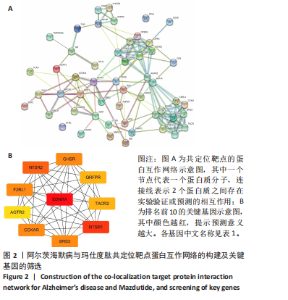
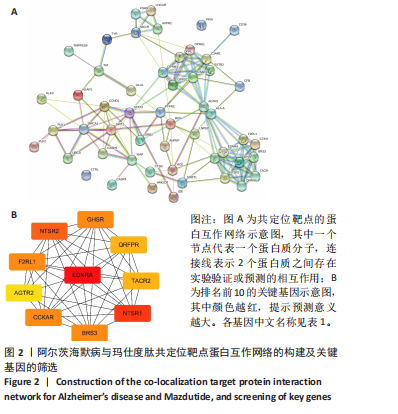
2.1 阿尔茨海默病与玛仕度肽共定位靶点的筛选 从DisGeNET数据库筛选出阿尔茨海默病相关联的基因,共得到3 397个关联基因。从PubChem上获取玛仕度肽的2D结构(图1A)和SMILES 编码结构式,随后将SMILES 编码结构式输入至SEA 数据库中,得到118个玛仕度肽的潜在作用靶点。随后使用R 语言筛选出阿尔茨海默病与玛仕度肽共定位靶点,共52个,并使用R 语言进行可视化(图1B)。 2.2 蛋白互作网络的构建及关键基因的筛选 基于前期确定的 52 个共定位靶蛋白,此次研究首先利用STRING在线数据库构建了相应的蛋白互作网络(图 2A)。随后,借助Cytoscape软件中的cytoHubba插件,依据网络中蛋白节点的连接度(Degree)这一核心拓扑学参数进行筛选,最终鉴别出网络连通性最高的10个核心枢纽基因(Hub gene):EDNRA、NTSR1、NTSR2、BRS3、CCKAR、GHSR、F2RL1、TACR2、QRFPR 和 AGTR2(图2B、表1)。在完成对这些关键枢纽基因功能的初步文献查阅与分析后,此次研究聚焦于其中与阿尔茨海默病病理过程及玛仕度肽生物学效应相关联的前10位基因,将其确定为后续基因本体论(gene ontology,GO)功能注释及京都基因与基因组百科全书(Kyoto encyclopedia of genes and genomes,KEGG)通路富集分析的研究目标。"

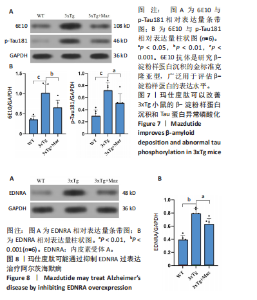
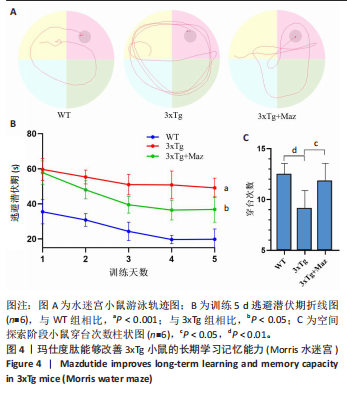
和伪足结构(pseudopodium)是主要富集位点;分子功能方面,G蛋白偶联肽受体活性(G protein-coupled peptide receptor activity)、肽受体活性(peptide receptor activity)及神经肽受体活性(neuropeptide receptor activity)占据主导地位(图3A-C)。KEGG通路分析中,神经活性配体-受体相互作用(Neuroactive ligand-receptor interaction)、钙信号通路(Calcium signaling pathway)和激素信号传导(Hormone signaling)为最显著的3大通路(图 3D)。提示玛仕度肽可能通过改善神经元功能治疗阿尔茨海默病,后续对此展开讨论。 2.4 行为学实验结果 2.4.1 玛仕度肽可以改善APP/PS1/Tau小鼠的长期学习记忆能力 图4A为水迷宫小鼠游泳轨迹图。如图4B所示,WT组、3xTg组、3xTg+Maz组的逃避潜伏期分别为(19.83±5.84) s、(49.17±5.52) s、(36.83±7.33) s。研究发现,3xTg组小鼠的逃避潜伏期明显长于WT组,提示3xTg小鼠模型的长期学习记忆能力明显下降(P < 0.001);3xTg+Maz组小鼠的"
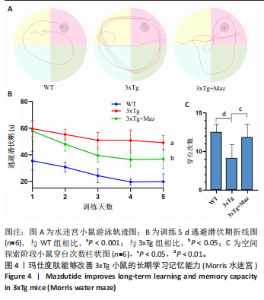
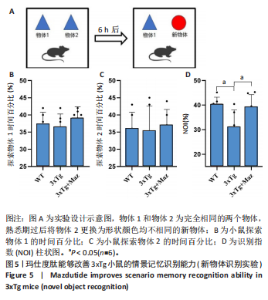
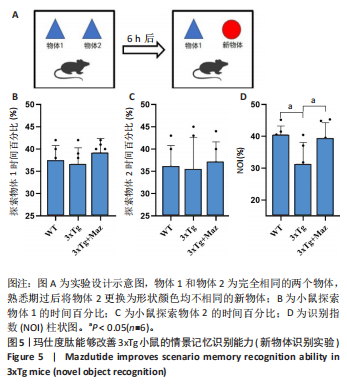
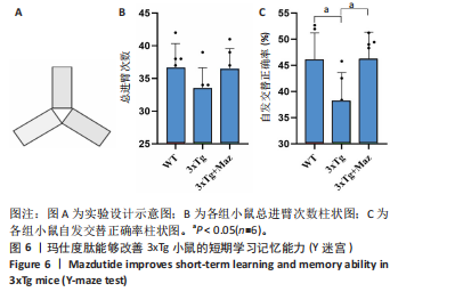
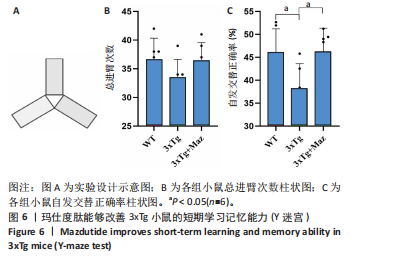
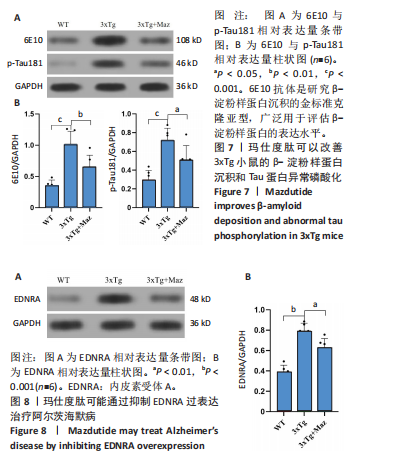
显增加(P=0.022 3),提示玛仕度肽可以改善3xTg小鼠的长期学习记忆能力。 2.4.2 玛仕度肽可以改善APP/PS1/Tau小鼠的情景记忆识别能力 实验设计示意图见图5A。如图5B-D所示,WT组、3xTg组、3xTg+Maz组探索物体1的时间百分比分别为(37.50±3.27)%、(36.67±3.61)%、(39.17±3.18)%;探索物体2的时间百分比分别为(36.17±4.62)%、(35.50±7.14)%、(37.17±4.44)%,各组小鼠探索物体1和物体2的时间无统计学差异。WT组、3xTg组、3xTg+Maz组的识别指数分别为(40.51±2.75)%、(31.28±6.76)%、(39.44±4.86)%,3xTg组的识别指数明显低于WT组(P=0.016 9),提示3xTg小鼠的情景记忆识别能力明显下降;3xTg+Maz组的识别指数明显高于3xTg组(P=0.034 8),提示玛仕度肽可以改善3xTg小鼠的情景记忆识别能力。 2.4.3 玛仕度肽可以改善APP/PS1/Tau小鼠的短期学习记忆能力 实验设计示意图见图6A。如图6B所示,WT组、3xTg组、3xTg+Maz组小鼠的总进臂次数分别为36.00±3.67,33.00±3.14,36.00±3.08,各组小鼠总进臂次数无统计学差异。如图6C所示,各组小鼠自发交替正确率分别为(46.13±5.08)%、(38.12±5.42)%、(46.29±5.08)%,3xTg组小鼠的自发交替正确率明显低于WT组(P=0.046 2),提示3xTg小鼠的短期学习记忆能力明显下降;3xTg+Maz组小鼠的自发交替正确率明显高于3xTg组(P=0.041 8),提示玛仕度肽可以改善3xTg小鼠的短期学习记忆能力。 2.4.4 玛仕度肽可以改善APP/PS1/Tau小鼠的β-淀粉样蛋白沉积和Tau蛋白异常磷酸化 6E10抗体是研究β-淀粉样蛋白沉积的金标准克隆亚型,广泛用于评估β-淀粉样蛋白的表达水平。Tau蛋白是神经元内主要的微管相关蛋白之一,属于低分子质量含磷糖蛋白。在阿尔茨海默病的早期阶段,血浆磷酸化Tau(p-Tau)水平呈进行性升高趋势。大规模临床研究证实,血浆p-Tau能有效区分阿尔茨海默病痴呆与非阿尔茨海默病神经退行性疾病相关的痴呆。此外,在轻度认知障碍患者中,p-Tau(尤其是Thr181位点磷酸化的亚型p-Tau181)已被证明可准确预测未来2-6年内出现的认知能力下降及其向阿尔茨海默病痴呆的转化。因而此次研究选用6E10和p-Tau181以评估玛仕度肽对3xTg小鼠脑内典型阿尔茨海默病病理表现的改善作用。 如图7所示,6E10在WT组、3xTg组、3xTg+Maz组小鼠脑海马组织中的相对表达量分别为0.35±0.08,1.02±0.20,0.65± 0.18,3xTg组小鼠脑海马组织中6E10的表达量明显高于WT组(P < 0.001),提示阿尔茨海默病小鼠模型中存在大量β-淀粉样蛋白1-16沉积;3xTg+Maz组小鼠脑海马组织中6E10的表达量明显低于3xTg组(P=0.004 2),提示玛仕度肽可以改善3xTg小鼠脑海马组织中的β-淀粉样蛋白1-16沉积。 p-Tau181在WT组、3xTg组、3xTg+Maz组小鼠脑海马组织中的相对表达量分别为0.29±0.09,0.72±0.12,0.51±0.14,3xTg组小鼠脑海马组织中p-Tau181的表达量明显高于WT组(P < 0.001),提示阿尔茨海默病小鼠模型中存在大量异常磷酸化Tau蛋白;3xTg+Maz组小鼠脑海马组织中p-Tau181的表达量明显低于3xTg组(P=0.026 7),提示玛仕度肽可以改善3xTg小鼠脑海马组织中的Tau蛋白异常磷酸化。 以上得出结论,玛仕度肽可以改善3xTg小鼠的典型阿尔茨海默病病理表现:β-淀粉样蛋白沉积和Tau蛋白异常磷酸化。 2.4.5 玛仕度肽可能通过抑制EDNRA过表达治疗阿尔茨海默病 EDNRA为前期网络药理学筛选出的TOP1核心靶点。如图8所示,EDNRA在WT组、3xTg组、3xTg+Maz组小鼠脑组织中的相对表达量分别为0.39±0.06,0.79±0.07,0.63±0.08,与对照WT组相比,3xTg组EDNRA的相对表达量明显上调(P < 0.001),提示阿尔茨海默病小鼠模型存在异常过表达的EDNRA;而3xTg+Maz组EDNRA的相对表达量较3xTg组明显下调(P=0.026 7),提示玛仕度肽可以抑制EDNRA的过表达。说明玛仕度肽可能通过抑制EDNRA过表达治疗阿尔茨海默病,EDNRA可能为玛仕度肽治疗阿尔茨海默病的潜在靶点,后文将围绕此靶点深入探讨。"
| [1] ZHANG Z, XUE P, BENDLIN BB, et al. Melatonin: a potential nighttime guardian against Alzheimer’s. Mol Psychiatry. 2025;30(1):237-250. [2] LANE CA, HARDY J, SCHOTT JM, et al. Alzheimer’s disease. Eur J Neurol. 2018; 25(1):59-70. [3] ZHENG Q, WANG X. Alzheimer’s disease: insights into pathology, molecular mechanisms, and therapy. Protein Cell. 2025;16(2):83-120. [4] HARDY J. Alzheimer’s Disease: Treatment Challenges for the Future. J Neurochem. 2025;169(8):e70176. [5] WANG R, REDDY PH. Role of Glutamate and NMDA Receptors in Alzheimer’s Disease. J Alzheimers Dis. 2017;57(4):1041-1048. [6] LIANG Y, DORE V, ROWE CC, et al. Clinical Evidence for GLP-1 Receptor Agonists in Alzheimer’s Disease: A Systematic Review. J Alzheimers Dis Rep. 2024;8(1):777-789. [7] JI L, JIANG H, BI Y, et al. Once-Weekly Mazdutide in Chinese Adults with Obesity or Overweight. N Engl J Med. 2025;392(22):2215-2225. [8] VARGAS-SORIA M, CARRANZA-NAVAL MJ, DEL MARCO A, et al. Role of liraglutide in Alzheimer’s disease pathology. Alzheimers Res Ther. 2021;13(1):112. [9] WANG W, WANG Q, QI X, et al. Associations of semaglutide with first-time diagnosis of Alzheimer’s disease in patients with type 2 diabetes: Target trial emulation using nationwide real-world data in the US. Alzheimers Dement. 2024; 20(12):8661-8672. [10] WANG ZJ, LI XR, CHAI SF, et al. Semaglutide ameliorates cognition and glucose metabolism dysfunction in the 3xTg mouse model of Alzheimer’s disease via the GLP-1R/SIRT1/GLUT4 pathway. Neuropharmacology. 2023;240:109716. [11] SALAMEH TS, RHEA EM, TALBOT K, et al. Brain uptake pharmacokinetics of incretin receptor agonists showing promise as Alzheimer’s and Parkinson’s disease therapeutics. Biochem Pharmacol. 2020;180:114187. [12] DONG W, BAI J, YUAN Q, et al. Mazdutide, a dual agonist targeting GLP-1R and GCGR, mitigates diabetes-associated cognitive dysfunction: mechanistic insights from multi-omics analysis. EBioMedicine. 2025;117:105791. [13] DING K, ZHANG Z, HAN Z, et al. Liver ALKBH5 regulates glucose and lipid homeostasis independently through GCGR and mTORC1 signaling. Science. 2025;387(6737):eadp4120. [14] ZIMMERMANN T, THOMAS L, BAADER-PAGLER T, et al. BI 456906: Discovery and preclinical pharmacology of a novel GCGR/GLP-1R dual agonist with robust anti-obesity efficacy. Mol Metab. 2022;66:101633. [15] PINERO J, SAUCH J, SANZ F, et al. The DisGeNET cytoscape app: Exploring and visualizing disease genomics data. Comput Struct Biotechnol J. 2021;19:2960-2967. [16] WANG Y, XIAO J, SUZEK TO, et al. PubChem’s BioAssay Database. Nucleic Acids Res. 2012;40(Database issue):D400-D412. [17] SHANNON P, MARKIEL A, OZIER O, et al. Cytoscape: a software environment for integrated models of biomolecular interaction networks. Genome Res. 2003; 13(11):2498-2504. [18] CHIN CH, CHEN SH, WU HH, et al. CytoHubba: identifying hub objects and sub-networks from complex interactome. BMC Syst Biol. 2014;8 Suppl 4(Suppl 4):S11. [19] CHEN T, SUN T, BIAN Y, et al. The Design and Optimization of Monomeric Multitarget Peptides for the Treatment of Multifactorial Diseases. J Med Chem. 2022;65(5):3685-3705. [20] BERTONI-FREDDARI C, SENSI SL, GIORGETTI B, et al. Decreased presence of perforated synapses in a triple-transgenic mouse model of Alzheimer’s disease. Rejuvenation Res. 2008;11(2):309-313. [21] WANG ZJ, HAN WN, CHAI SF, et al. Semaglutide promotes the transition of microglia from M1 to M2 type to reduce brain inflammation in APP/PS1/tau mice. Neuroscience. 2024;563:222-234. [22] MEKADA K, YOSHIKI A. Substrains matter in phenotyping of C57BL/6 mice. Exp Anim. 2021;70(2):145-160. [23] CABRAL-MARQUES O, MARQUES A, GIIL LM, et al. GPCR-specific autoantibody signatures are associated with physiological and pathological immune homeostasis. Nat Commun. 2018;9(1):5224. [24] XU Z, ZHOU Q, LIU C, et al. EDNRA affects susceptibility to large artery atherosclerosis stroke through potential inflammatory pathway. Sci Rep. 2024; 14(1):25173. [25] WANG M, WANG L, LI X, et al. EDNRA regulates the tumour immune environment and predicts the efficacy and prognosis of cancer immunotherapy. J Cell Mol Med. 2024;28(22):e70172. [26] LING W, JOHNSON SK, MEHDI SJ, et al. EDNRA-Expressing Mesenchymal Cells are Expanded in Myeloma Interstitial Bone Marrow and Associated with Disease Progression. Cancers (Basel). 2023;15(18):4519. [27] LEE YJ, JUNG E, CHOI J, et al. The EDN1/EDNRA/beta‑arrestin axis promotes colorectal cancer progression by regulating STAT3 phosphorylation. Int J Oncol. 2023;62(1):13. [28] PALMER JC, BARKER R, KEHOE PG, et al. Endothelin-1 is elevated in Alzheimer’s disease and upregulated by amyloid-beta. J Alzheimers Dis. 2012;29(4):853-861. [29] MINERS JS, PALMER JC, TAYLER H, et al. Abeta degradation or cerebral perfusion? Divergent effects of multifunctional enzymes. Front Aging Neurosci. 2014;6:238. [30] ZHANG J, WANG YJ, WANG X, et al. PKC-Mediated Endothelin-1 Expression in Endothelial Cell Promotes Macrophage Activation in Atherogenesis. Am J Hypertens. 2019;32(9):880-889. [31] YANG W, LIU Y, XU QQ, et al. Sulforaphene Ameliorates Neuroinflammation and Hyperphosphorylated Tau Protein via Regulating the PI3K/Akt/GSK-3beta Pathway in Experimental Models of Alzheimer’s Disease. Oxid Med Cell Longev. 2020;2020:4754195. [32] PAHLAVANI HA. Exercise therapy to prevent and treat Alzheimer’s disease. Front Aging Neurosci. 2023;15:1243869. [33] RAJABLI F, BENCHEK P, TOSTO G, et al. Multi-ancestry genome-wide meta-analysis of 56,241 individuals identifies known and novel cross-population and ancestry-specific associations as novel risk loci for Alzheimer’s disease. Genome Biol. 2025;26(1):210. [34] SINGH N, NANDY SK, JYOTI A, et al. Protein Kinase C (PKC) in Neurological Health: Implications for Alzheimer’s Disease and Chronic Alcohol Consumption. Brain Sci. 2024;14(6):554. [35] VERMA A, CHAUDHARY S, SOLANKI K, et al. Exendin-4: a potential therapeutic strategy for Alzheimer’s disease and Parkinson’s disease. Chem Biol Drug Des. 2024;103(1):e14426. [36] KANG X, WANG D, ZHANG L, et al. Exendin-4 ameliorates tau hyperphosphorylation and cognitive impairment in type 2 diabetes through acting on Wnt/beta-catenin/NeuroD1 pathway. Mol Med. 2023;29(1):118. [37] RAJABI H, AHMADI M, ASLANI S, et al. Exendin-4 as a Versatile Therapeutic Agent for the Amelioration of Diabetic Changes. Adv Pharm Bull. 2022;12(2):237-247. [38] ESPARZA-SALAZAR FJ, LEZAMA-TOLEDO AR, RIVERA-MONROY G, et al. Exendin-4 for Parkinson’s disease. Brain Circ. 2021;7(1):41-43. [39] WEI R, ZHANG L, HU W, et al. Zeb2/Axin2-Enriched BMSC-Derived Exosomes Promote Post-Stroke Functional Recovery by Enhancing Neurogenesis and Neural Plasticity. J Mol Neurosci. 2022;72(1):69-81. [40] YANG G, PEI YN, SHAO SJ, et al. [Effects of electroacupuncture at “Baihui” and “Yongquan” on the levels of synaptic plasticity related proteins postsynaptic density-95 and synaptophysin in hippocampus of APP/PS1 mice]. Zhen Ci Yan Jiu. 2020;45(4):310-314. [41] VE H, CABANA VC, GOUSPILLOU G, et al. Quantitative Immunoblotting Analyses Reveal that the Abundance of Actin, Tubulin, Synaptophysin and EEA1 Proteins is Altered in the Brains of Aged Mice. Neuroscience. 2020;442:100-113. [42] WANG H, SHEN Z, WU CS, et al. Neuronal ablation of GHSR mitigates diet-induced depression and memory impairment via AMPK-autophagy signaling-mediated inflammation. Front Immunol. 2024;15:1339937. [43] ZHANG LQ, ZHANG W, LI T, et al. GLP-1R activation ameliorated novel-object recognition memory dysfunction via regulating hippocampal AMPK/NF-kappaB pathway in neuropathic pain mice. Neurobiol Learn Mem. 2021;182:107463. [44] ZHANG QH, HAO JW, LI GL, et al. Proinflammatory switch from Galphas to Galphai signaling by Glucagon-like peptide-1 receptor in murine splenic monocyte following burn injury. Inflamm Res. 2018;67(2):157-168. [45] ATEF MM, ABOU HN, HAFEZ YM, et al. The potential protective effect of liraglutide on valproic acid induced liver injury in rats: Targeting HMGB1/RAGE axis and RIPK3/MLKL mediated necroptosis. Cell Biochem Funct. 2023;41(8):1209-1219. [46] JI L, JIANG H, CHENG Z, et al. A phase 2 randomised controlled trial of mazdutide in Chinese overweight adults or adults with obesity. Nat Commun. 2023;14(1):8289. [47] BHATTACHAR SN, THAM LS, LI Y, et al. Mazdutide reduces body weight in adults with overweight or obesity: a high-dose Phase 1 trial. Diabetes Obes Metab. 2025;27(11):6460-6469. [48] AYESH H, AYESH S, NISWENDER K. Mazdutide Versus Dulaglutide for Weight Loss and Diabetes Management: Meta-Analysis of Randomized Clinical Trials. Am J Ther. 2024;31(5):e619-e622. [49] DONG W, BAI J, YUAN Q, et al. Mazdutide, a dual agonist targeting GLP-1R and GCGR, mitigates diabetes-associated cognitive dysfunction: mechanistic insights from multi-omics analysis. EBioMedicine. 2025;117:105791. [50] JI L, JIANG H, BI Y, et al. Once-Weekly Mazdutide in Chinese Adults with Obesity or Overweight. N Engl J Med. 2025;392(22):2215-2225. |
| [1] | Zhou Sirui, Xu Yukun, Zhao Kewei. Ideas and methods of anti-melanogenesis of Angelica dahurica extracellular vesicles [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1747-1754. |
| [2] | Chen Yulin, He Yingying, Hu Kai, Chen Zhifan, Nie Sha Meng Yanhui, Li Runzhen, Zhang Xiaoduo , Li Yuxi, Tang Yaoping. Effect and mechanism of exosome-like vesicles derived from Trichosanthes kirilowii Maxim. in preventing and treating atherosclerosis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1768-1781. |
| [3] | Peng Zhiwei, Chen Lei, Tong Lei. Luteolin promotes wound healing in diabetic mice: roles and mechanisms [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1398-1406. |
| [4] | Chen Yixian, Chen Chen, Lu Liheng, Tang Jinpeng, Yu Xiaowei. Triptolide in the treatment of osteoarthritis: network pharmacology analysis and animal model validation [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 805-815. |
| [5] | Zhao Canbin, Zeng Ping, Shi Weiqi, Liu Jinfu, Ding Qiang, Guo Liang, Wang Weiwei, Tao Hongcheng, Guo Yafeng, Qin Ying. Mechanism by which luteolin regulates macrophage polarization in the treatment of knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(34): 8868-8877. |
| [6] | Jing Kun, Wang Yulu, Liang Hao, Huo Yuhang, Cong Longxu, . Mechanisms by which mangiferin alleviates pain in osteoarthritis: integration of microarray data analysis, network pharmacology, and experimental validation in a rat model [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(34): 8953- 8961. |
| [7] | Wang Shaona, Li Ruiyang, An Lanhua, Zhang Jinsheng. Role of non-coding RNAs in Alzheimer’s disease and treatment with traditional Chinese medicine [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(29): 7663-7672. |
| [8] | Qi Yuxin, Dang Yifan, Dai Liming, Zhang Xiaoling. Function and molecular mechanism of physcion in regulating bone homeostasis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(28): 7237-7244. |
| [9] | Deng Qian, Peng Zining, Meng Fanyu, Huang Yuanbo, Liu Nian, Yan Weitian, Li Zhaofu, Peng Jiangyun. Molecular mechanisms of Toddalia asiatica against rheumatoid arthritis: bioinformatics and molecular dynamics simulation [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(28): 7267-7279. |
| [10] | Chen Chaoqi, Liu Fei, Song Chao, Shen Baoxin, Huang Wutao, Chen Feng, Yang Lei. Yanggan Roujin Decoction delays intervertebral disc degeneration: network pharmacological analysis and experimental validation in rat models [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(25): 6533-6543. |
| [11] | Zhao Fangfang, Guo Yanke, Wang Xueke, Pang Botong, Zhu Yanqiang, Qin Yang, Cui Yinglin. A new approach to intervene in Alzheimer's disease through regulating the silencing information regulator 1 signaling pathway with traditional Chinese medicine [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(25): 6621-6631. |
| [12] | He Wenyang, Chen Weijian, Ran Qingzhi, Huang Yidie, Lin Xiaodong, Xu Xuemeng, Guo Meirong, Liu Wengang. Action mechanisms of Bushen Qiangjin capsule in regulating inflammatory signaling pathways and improving knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(25): 6632-6642. |
| [13] | Xu Dongfang, Zhao Kun, Lu Changzhu, Wang Yuge, Bai Lianjie, Meng Fanmou, Wang Yang, , Yao Hongbo. m6A-related ferroptosis gene expression and its association with immune infiltration in Alzheimer’s disease: machine learning and molecular biology validation [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6421-6432. |
| [14] | Zhang Xiaomin, Du Pengyang, Zhang Xiuping, Xue Guofang. Potential targets of glucagon-like peptide 1 receptor agonist ticagrelor in the treatment of Alzheimer’s disease [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(23): 6122-6133. |
| [15] | Hu Yalin, Huang Fengqin, Yang Boyin, Luo Xingmei. Transcription factor EB improves Alzheimer’s disease via the autophagy-lysosome pathway [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(22): 5844-5858. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||