Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (34): 8970-8977.doi: 10.12307/2026.891
Previous Articles Next Articles
Establishment and identification of a patient-derived organoid model for esophagogastric junction adenocarcinoma
Gao Zhendong1, 2, Zhang Li2, Chen Pan2, Yin Xiyao2, Li Jiayi2, Yang Pingjuan2, Zhang Min1, Liao Shuxin2, Shi Linlin2, Gao Shegan2
- 1School of Basic Medical Sciences and Forensic Medicine, Henan University of Science and Technology, Luoyang 471023, Henan Province, China; 2The First Affiliated Hospital, College of Clinical Medicine, Henan Key Laboratory of Microbiome and Esophageal Cancer Prevention and Treatment, Henan University of Science and Technology, Luoyang 471003, Henan Province, China
-
Received:2025-09-23Revised:2026-02-14Online:2026-12-08Published:2026-04-14 -
Contact:Shi Linlin, PhD, Associate professor, Associate chief physician, The First Affiliated Hospital, College of Clinical Medicine, Henan Key Laboratory of Microbiome and Esophageal Cancer Prevention and Treatment, Henan University of Science and Technology, Luoyang 471003, Henan Province, China Co-corresponding author: Gao Shegan, PhD, Professor, Chief physician, The First Affiliated Hospital, College of Clinical Medicine, Henan Key Laboratory of Microbiome and Esophageal Cancer Prevention and Treatment, Henan University of Science and Technology, Luoyang 471003, Henan Province, China -
About author:Gao Zhendong, MS, School of Basic Medical Sciences and Forensic Medicine, Henan University of Science and Technology, Luoyang 471023, Henan Province, China; The First Affiliated Hospital, College of Clinical Medicine, Henan Key Laboratory of Microbiome and Esophageal Cancer Prevention and Treatment, Henan University of Science and Technology, Luoyang 471003, Henan Province, China -
Supported by:National Natural Science Foundation of China, No. 82302966 (to SLL); Joint Fund of National Key Clinical Specialty Construction Project, No. ZLKFJJ20230401 (to SLL); Henan Provincial Training Program for Clinical Medical Scientists, No. HNCMS202438 (to SLL); Key Project of Henan Provincial Medical Science and Technology Research, No. LHGJ20230455 (to CP)
CLC Number:
Cite this article
Gao Zhendong, Zhang Li, Chen Pan, Yin Xiyao, Li Jiayi, Yang Pingjuan, Zhang Min, Liao Shuxin, Shi Linlin, Gao Shegan. Establishment and identification of a patient-derived organoid model for esophagogastric junction adenocarcinoma[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(34): 8970-8977.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
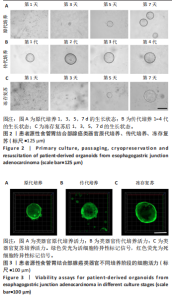
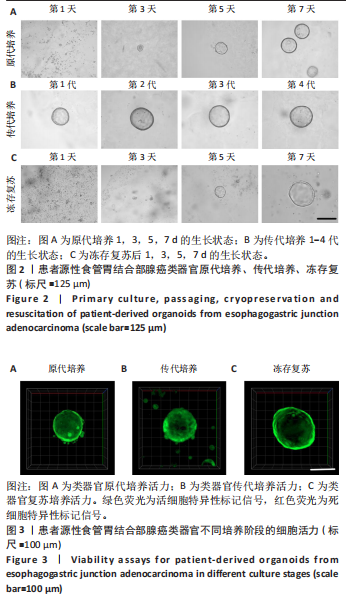
2.1 患者源性食管胃结合部腺癌类器官的原代培养、传代培养及冻存复苏特征 在原代培养体系中,细胞于接种后展现出逐步发育的特征。在培养第1天,以分散的细胞碎片和单细胞为主,类器官结构尚未成形,处于原代细胞适应培养微环境的起始阶段;培养第3天,细胞发生聚集与增殖,类器官雏形开始出现;培养第5天,类器官结构较为清晰,边界逐渐完整,体积较第3天显著增大,显示出原代培养阶段类器官的持续发育过程;培养第7天,类器官数量增多、体积进一步增大,呈现出典型的类圆形三维结构(图2A)。 在传代培养体系中,第1代到第4代的各代类器官均维持类圆形、边界清晰的三维结构,形态学特征高度相似,上述结果验证了患者源性食管胃结合部腺癌类器官在传代培养条件下,具备稳定的自我更新能力与遗传表型维持特性,可满足长期传代研究对模型稳定性的需求(图2B)。 在冻存复苏体系中,随培养时间延长,类器官轮廓逐渐清晰,部分单细胞或小细胞团开始聚集增殖;复苏后第5天,类器官结构清晰度进一步提升,体积缓慢增长,说明冻存复苏未破坏类器官的再生潜能;复苏后第7天,类器官形态接近成熟状态,从形态学角度证实冻存复苏技术可保留类器官的培养活性,为类器官生物样本库构建及相关研究提供技术支撑(图2C)。 以上结果证明成功建立了患者源性食管胃结合部腺癌类器官原代培养、传代培养、冻存复苏的可靠方法且可以保持其培养的稳定性。 2.2 患者源性食管胃结合部腺癌类器官在不同培养阶段的细胞活力 类器官在3种培养阶段下均呈现清晰连续的绿色荧光信号,无红色荧光信号。其中,原代培养类器官绿色荧光分布均匀(图3A),表明肿瘤细胞在原代培养体系维持较高的活性;传代培养类器官荧光强度稳定、形态规则(图3B),表明传代未显著损伤细胞活力,其自我更新与代谢功能持续;冻存复苏类器官荧光完整覆盖结构(图3C),说明冻存复苏后的类器官修复充分,细胞活力得到有效保留。活死荧光染色结果从功能层面证实所构建的类器官在原代培养、传代培养、冻存复苏全流程中均保持较好的细胞活性。 2.3 患者源性食管胃结合部腺癌类器官的组织病理学鉴定分析 为了验证所培养的患者源性食管胃结合部腺癌类器官与原发肿瘤组织形态学和糖原表达的一致性,分别进行了苏木精-伊红"
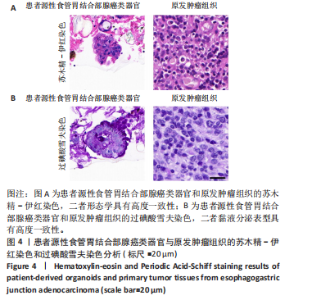
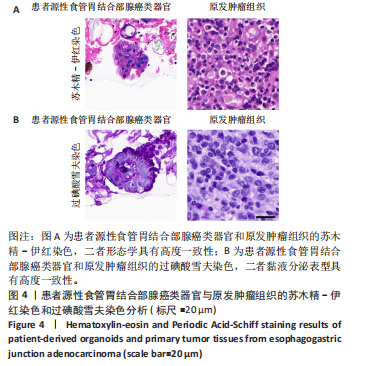
染色和过碘酸雪夫染色实验。苏木精-伊红染色显示,患者源性食管胃结合部腺癌类器官与原发肿瘤组织呈现形态学一致性(图4A)。类器官可见腺管样结构与不规则细胞团,细胞排列紊乱、核质比失调,核形态多样且存在异型性;原发肿瘤组织同样展现紊乱的细胞排列模式,腺管结构破坏伴细胞异型性,二者在组织架构、细胞形态及排列紊乱程度上高度契合。过碘酸雪夫染色显示,患者源性食管胃结合部腺癌类器官与原发肿瘤组织均呈现阳性信号,且染色分布模式、表达强度具有一致性(图4B):类器官的腺管样结构及细胞胞质胞膜区域着色清晰,提示糖原富集;原发肿瘤组织在对应细胞区同样具有阳性信号,说明患者源性食管胃结合部腺癌类器官保留了原发肿瘤的糖原代谢表型。上述结果证实此研究所构建的患者源性食管胃结合部腺癌类器官模型在组织形态、细胞功能层面与患者原发肿瘤具有高度一致性。 2.4 患者源性食管胃结合部腺癌类器官免疫组织化学染色结果 Ki67免疫组织化学染色显示(图5A),患者源性食管胃结合部腺癌类器官与原发肿瘤组织均呈现阳性表达,且阳性细胞百"
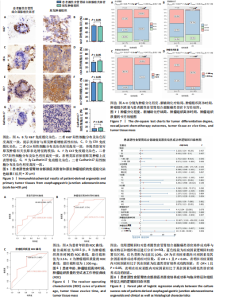
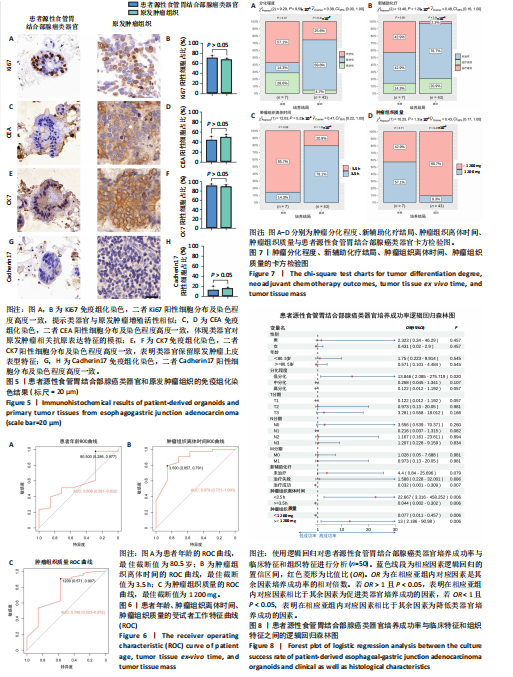
分比相近(图5B)。CEA免疫组织化学染色显示,患者源性食管胃结合部腺癌类器官和原发肿瘤组织都呈现阳性表达,且类器官中阳性细胞的分布密度、染色范围及着色深浅与肿瘤组织切片中对应阳性细胞特征相匹配(图5C)。定量分析结果显示(图5D),二者CEA阳性细胞的比例无显著差异。CK7免疫组织化学染色显示(图5E,F),患者源性食管胃结合部腺癌类器官和原发肿瘤组织均呈现阳性表达且与原发肿瘤组织染色强度、染色范围及阳性细胞比例均具有较高的一致性。 Cadherin17免疫组织化学染色显示,虽然与Ki67、CEA、CK7相比可见Cadherin17阳性表达程度较低,但患者源性食管胃结合部腺癌类器官和原发肿瘤组织均呈现阳性表达且阳性细胞比例无显著差异(图5G,H)。 以上结果进一步证明所培养的患者源性食管胃结合部腺癌类器官与原发肿瘤组织在各指标染色分布、着色特征上具有一致性,提示较好地保留了原发肿瘤组织部分免疫表型特征,为其作为肿瘤研究模型提供实验依据。 2.5 患者源性食管胃结合部腺癌类器官培养成功率与患者临床特征和组织特征的相关性分析 此研究所构建的患者源性食管胃结合部腺癌类器官原代培养成功率为86%(43/50)。 根据受试者工作特征曲线分析结果,使用最佳截断值将年龄分为< 80.5岁组及≥80.5岁组(图6A),将肿瘤组织离体时间分为< 3.5 h组及≥3.5 h组(图6B),将肿瘤组织质量分为< 1 200 mg组及≥1 200 mg组(图6C)。卡方检验结果初步表明肿瘤分化程度、新辅助治疗结局、肿瘤组织离体时间及肿瘤组织质量与培养成功率有关(图7A-D)。随后将培养成功率作为因变量对性别、年龄、肿瘤分化程度、T分期、N分期、M分期进行单因素逻辑回归以量化各类因素对培养结局的影响。 图8表明,肿瘤低分化组相较于肿瘤中、高分化组,培养成功率提高13.846倍(P=0.02),肿瘤组织离体时间< 3.5 h组相"
| [1] 刘凯,杨玉赏,杨昆,等.食管胃结合部腺癌外科治疗中国专家共识(2024年版)[J].消化肿瘤杂志(电子版),2024,16(2):133-152. [2] ARNOLD M, FERLAY J, VAN BERGE HENEGOUWEN MI, et al. Global burden of oesophageal and gastric cancer by histology and subsite in 2018. Gut. 2020;69(9): 1564-1571. [3] LIU K, YANG K, ZHANG W, et al. Changes of Esophagogastric Junctional Adenocarcinoma and Gastroesophageal Reflux Disease Among Surgical Patients During 1988-2012: A Single-institution, High-volume Experience in China. Ann Surg. 2016;263(1):88-95. [4] RUBENSTEIN JH, FONTAINE S, MACDONALD PW, et al. Predicting Incident Adenocarcinoma of the Esophagus or Gastric Cardia Using Machine Learning of Electronic Health Records. Gastroenterology. 2023;165(6):1420-1429.e10. [5] PUCHER PH, PARK MH, CROMWELL DA, et al. Diagnosis and treatment for gastro-oesophageal cancer in England and Wales: analysis of the National Oesophago-Gastric Cancer Audit (NOGCA) database 2012-2020. Br J Surg. 2023;110(6):701-709. [6] THRIFT AP, WENKER TN, EL-SERAG HB. Global burden of gastric cancer: epidemiological trends, risk factors, screening and prevention. Nat Rev Clin Oncol. 2023;20(5):338-349. [7] TAKEDA FR, KODAMA PERTILLE RAMOS MF, PEREIRA MA, et al. Predictive factors of recurrence in adenocarcinoma of the esophagogastric junction in the multimodal era. Am J Surg. 2021;221(3):631-636. [8] GAMBARDELLA V, FLEITAS T, TARAZONA N, et al. Towards precision oncology for HER2 blockade in gastroesophageal adenocarcinoma. Ann Oncol. 2019; 30(8):1254-1264. [9] QU S, XU R, YI G, et al. Patient-derived organoids in human cancer: a platform for fundamental research and precision medicine. Mol Biomed. 2024;5(1):6. [10] DUTTA D, HEO I, CLEVERS H. Disease Modeling in Stem Cell-Derived 3D Organoid Systems. Trends Mol Med. 2017;23(5):393-410. [11] VENINGA V, VOEST EE. Tumor organoids: Opportunities and challenges to guide precision medicine. Cancer Cell. 2021;39(9):1190-1201. [12] CORRÒ C, NOVELLASDEMUNT L, LI VSW. A brief history of organoids. Am J Physiol Cell Physiol. 2020;319(1):C151-C165. [13] SATO T, VRIES RG, SNIPPERT HJ, et al. Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature. 2009;459(7244):262-265. [14] CLEVERS H. Modeling Development and Disease with Organoids. Cell. 2016; 165(7):1586-1597. [15] YANG R, YU Y. Patient-derived organoids in translational oncology and drug screening. Cancer Lett. 2023;562:216180. [16] RICE TW, PATIL DT, BLACKSTONE EH. 8th edition AJCC/UICC staging of cancers of the esophagus and esophagogastric junction: application to clinical practice. Ann Cardiothorac Surg. 2017;6(2):119-130. [17] PEDRAZZANI C. Should adenocarcinoma of the esophagogastric junction be classified as gastric or esophageal cancer, or else as a distinct clinical entity? Ann Surg. 2015;261(4):e107-108. [18] ORDITURA M, GALIZIA G, LIETO E, et al. Treatment of esophagogastric junction carcinoma: an unsolved debate. World J Gastroenterol. 2015;21(15):4427-4431. [19] 宋梦欣,闫君雅,仓顺东.食管胃结合部腺癌研究进展[J].中华实用诊断与治疗杂志,2021,35(9):889-891. [20] WANG J, WU Z, FREUND JE, et al. Comparison between AJCC 8th edition gastric and esophageal ypTNM classification system in Siewert II esophagogastric junction adenocarcinomas: A retrospective study based on Eastern and Western Data. Eur J Surg Oncol. 2024;50(12):108730. [21] 朱甲明,孙景旭,王鑫,等.食管胃结合部腺癌围手术期综合治疗进展[J]. 中国实用外科杂志,2024,44(10):1186-1191. [22] WHELESS MC, COMER M, GIBSON MK. Evolving Treatment Landscape for Advanced Esophageal and Gastroesophageal Junction Adenocarcinoma. Curr Oncol Rep. 2024;26(11):1469-1488. [23] QIAN M, FANG Y, XIANG Z, et al. The efficacy of neoadjuvant immunotherapy in gastric cancer, adenocarcinoma of the esophagogastric junction, and esophageal cancer: a meta-analysis. Front Oncol. 2024;14:1502611. [24] LESAVAGE BL, ZHANG D, HUERTA-LÓPEZ C, et al. Engineered matrices reveal stiffness-mediated chemoresistance in patient-derived pancreatic cancer organoids. Nat Mater. 2024;23(8):1138-1149. [25] DUAN X, ZHANG T, FENG L, et al. A pancreatic cancer organoid platform identifies an inhibitor specific to mutant KRAS. Cell Stem Cell. 2024;31(1):71-88.e8. [26] SENKOWSKI W, GALL-MAS L, FALCO MM, et al. A platform for efficient establishment and drug-response profiling of high-grade serous ovarian cancer organoids. Dev Cell. 2023;58(12):1106-1121.e7. [27] KIM M, MUN H, SUNG CO, et al. Patient-derived lung cancer organoids as in vitro cancer models for therapeutic screening. Nat Commun. 2019;10(1):3991. [28] ZHAO Y, LI S, ZHU L, et al. Personalized drug screening using patient-derived organoid and its clinical relevance in gastric cancer. Cell Rep Med. 2024;5(7): 101627. [29] LIN M, GAO M, CAVNAR MJ, et al. Utilizing gastric cancer organoids to assess tumor biology and personalize medicine. World J Gastrointest Oncol. 2019;11(7):509-517. [30] MAO Y, WANG W, YANG J, et al. Drug repurposing screening and mechanism analysis based on human colorectal cancer organoids. Protein Cell. 2024;15(4): 285-304. [31] MO S, TANG P, LUO W, et al. Patient-Derived Organoids from Colorectal Cancer with Paired Liver Metastasis Reveal Tumor Heterogeneity and Predict Response to Chemotherapy. Adv Sci (Weinh). 2022;9(31):e2204097. [32] SACHS N, DE LIGT J, KOPPER O, et al. A Living Biobank of Breast Cancer Organoids Captures Disease Heterogeneity. Cell. 2018;172(1-2):373-386.e10. [33] WANG HM, ZHANG CY, PENG KC, et al. Using patient-derived organoids to predict locally advanced or metastatic lung cancer tumor response: A real-world study. Cell Rep Med. 2023;4(2):100911. [34] SEIDLITZ T, MERKER SR, ROTHE A, et al. Human gastric cancer modelling using organoids. Gut. 2019;68(2):207-217. [35] TAN R, HONG F, WANG T, et al. Standard: Human gastric cancer organoids. Cell Regen. 2024;13(1):33. [36] HARADA K, SAKAMOTO N, UKAI S, et al. Establishment of oxaliplatin-resistant gastric cancer organoids: importance of myoferlin in the acquisition of oxaliplatin resistance. Gastric Cancer. 2021;24(6):1264-1277. [37] HUO C, ZHANG X, GU Y, et al. Organoids: Construction and Application in Gastric Cancer. Biomolecules. 2023;13(5):875. [38] SEVEN D, BARON S, FARIN HF. Establishment of Patient-Derived Organoids from Colorectal Cancer Resection Samples. Methods Mol Biol. 2025;2951:245-255. [39] FARIN HF, MOSA MH, NDRESHKJANA B, et al. Colorectal Cancer Organoid-Stroma Biobank Allows Subtype-Specific Assessment of Individualized Therapy Responses. Cancer Discov. 2023;13(10):2192-2211. [40] WITKOWSKI TA, LI B, ANDERSEN JG, et al. Y-27632 acts beyond ROCK inhibition to maintain epidermal stem-like cells in culture. J Cell Sci. 2023;136(17):jcs260990. [41] WANG P, SHEN S, GUO Y, et al. Rho kinase inhibitor Y-27632 and dual media culture approach promote the construction and transplantation of rabbit limbal epithelial cell sheets via cell spheroid culture and auto-bioprinting. Acta Biomater. 2025;194:140-152. [42] WANG W, YANG T, CHEN S, et al. Tissue engineering RPE sheet derived from hiPSC-RPE cell spheroids supplemented with Y-27632 and RepSox. J Biol Eng. 2024;18(1):7. |
| [1] | Jiang Xinghai, Song Yulin, Li Dejin, Shao Jianmin, Xu Junzhi, Liu Huakai, Wu Yingguo, Shen Yuehui, Feng Sicheng. Vascular endothelial growth factor 165 genes transfected into bone marrow mesenchymal stem cells to construct a vascularized amphiphilic peptide gel module [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 1903-1911. |
| [2] | . Regional specificity of brain organoids and their application in ischemic stroke modeling and drug development [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(34): 9008-9016. |
| [3] | He Renda, Ma Wei, Sun Yongsi, Mo Xueni. Future medical research on brain organoids: interdisciplinary training, bioengineering technologies, and optimized model maturity [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(34): 9088-9094. |
| [4] | Yu Jinye, Jiang Nan, Zhao Yixun, Huang Mengjing, Yang Jie, Sun Rui, Feng Suolan, Jiang Hui, Yang Jun. Ready-to-use sodium alginate@paper material for three-dimensional cell culture [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6873-6879. |
| [5] | Mou Jiancheng, Luo Jie, Liu Haotian, Yang Zhuotao, Mu Yuxiao, Qian Da, Meng Xuli. Application and prospects of precision-medicine-driven breast cancer organoids in therapeutic drug discovery [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(19): 5033-5039. |
| [6] |
Zhang Yueting, Li Jinglin, Fu Zhenyi, Yan Fei, Gao Yu, Liu Jiaxin.
Endoplasmic reticulum stress promotes ferroptosis and aggravates cerebral ischemia-reperfusion injury#br#
#br#
[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(11): 2806-2813.
|
| [7] | Wu Jiazhou, Qian Tao, Liu Zexian, Wu Yanbin, He Ying, Li Yazhou, Peng Jiang. Three-dimensional culture of stromal vascular fraction self-assembles into complex vascularized osteogenic organoids [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(11): 2681-2690. |
| [8] | Zhao Qianwei, Sun Guangyuan . Intestinal organoids: a bibliometric analysis of the latest trends in tissue/organ biology, disease modeling, and clinical applications [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(1): 238-247. |
| [9] | Yuan Weibo, Liu Chan, Yu Limei. Potential application of liver organoids in liver disease models and transplantation therapy [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(8): 1684-1692. |
| [10] | Li Dijun, Jiu Jingwei, Liu Haifeng, Yan Lei, Li Songyan, Wang Bin. Three-dimensional gelatin microspheres loaded human umbilical cord mesenchymal stem cells for chronic tendinopathy repair [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(7): 1356-1362. |
| [11] | Huang Haina, Yu Yanrong, Bi Jian, Huang Miao, Peng Weijie. Epigenetic characteristics of hepatogenic differentiation of mesenchymal stem cells in three-dimensional culture [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(36): 7848-7855. |
| [12] | Liu Lu, Zhong Chang, Yu Xin, Ren Chenyuan, Gong Yangyang, Zhou Ping, Wang Yingbin. Academic progress and clinical application of in vitro synthetic microenvironment to promote maturation of human pluripotent stem cell-derived cardiomyocytes [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(36): 7856-7862. |
| [13] | Wang Zhao, Gong Lin, Piao Yongjun. Research on hair follicle organoids: current status, challenges and prospects [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(31): 6733-6742. |
| [14] | Fan Hongkai, Guan Yingying, Wang Lumin, Zeng Fanwei, Yin Yirui. Feasibility of gastric cancer organoid models for personalized drug screening [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(25): 5345-5350. |
| [15] | Lin Zhiguang, Rao Qi, Liang Shanshan, Wang Ruoyu, Yu Weiting. Key role of calcium ion in sodium alginate based composite hydrogel for breast cancer organoid culture [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(22): 4702-4709. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

