Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (28): 7437-7446.doi: 10.12307/2026.821
Previous Articles Next Articles
Association between plasma metabolites and osteoarthritis
Li Yunpeng1, 2, Lyu Yuqiang2, Zhang Jialin2, Tang You2, Wang Kai3, Zhao Wenzhi1
- 1Second Affiliated Hospital of Dalian Medical University, Dalian 116000, Liaoning Province, China; 2Binzhou People’s Hospital, Binzhou 256600, Shandong Province, China; 3Binzhou Medical University, Binzhou 256600, Shandong Province, China
-
Received:2025-08-25Revised:2025-12-08Online:2026-10-08Published:2026-02-26 -
Contact:Zhao Wenzhi, Professor, Chief physician, Doctoral supervisor, Second Affiliated Hospital of Dalian Medical University, Dalian 116000, Liaoning Province, China -
About author:Li Yunpeng, Attending physician, MD candidate, Second Affiliated Hospital of Dalian Medical University, Dalian 116000, Liaoning Province, China; Binzhou People’s Hospital, Binzhou 256600, Shandong Province, China -
Supported by:Shandong Provincial Natural Science Foundation Project, No. ZR2022QH391 (to WK); Dalian Key Laboratory of Digital Orthopedics Project (to ZWZ); Binzhou People’s Hospital Project, No. XJ2024000803 (to LYP)
CLC Number:
Cite this article
Li Yunpeng, Lyu Yuqiang, Zhang Jialin, Tang You, Wang Kai, Zhao Wenzhi. Association between plasma metabolites and osteoarthritis[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(28): 7437-7446.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
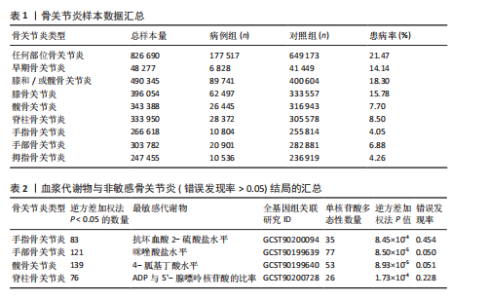
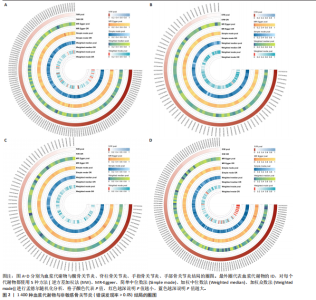
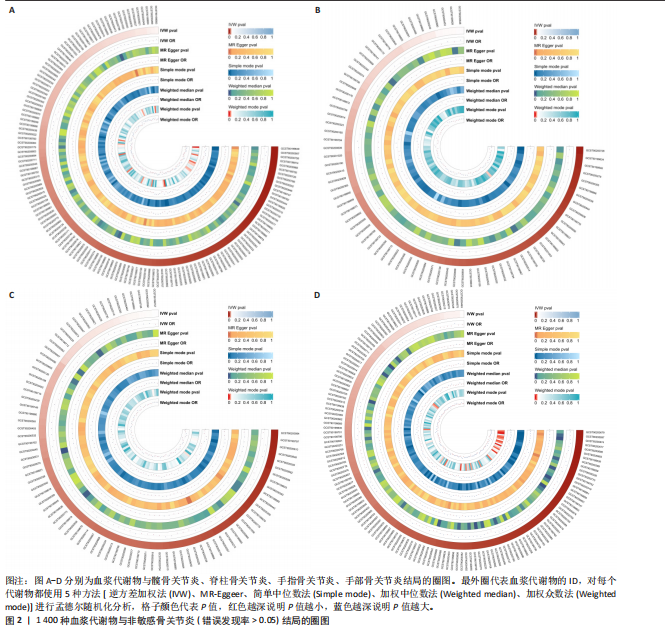
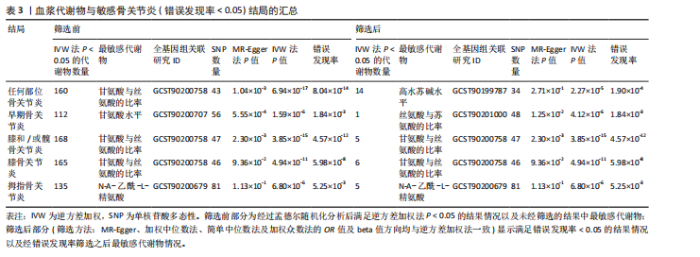
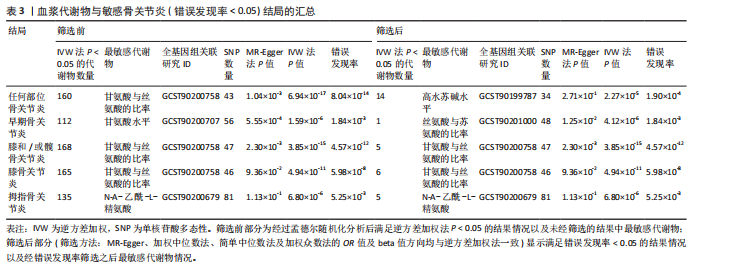
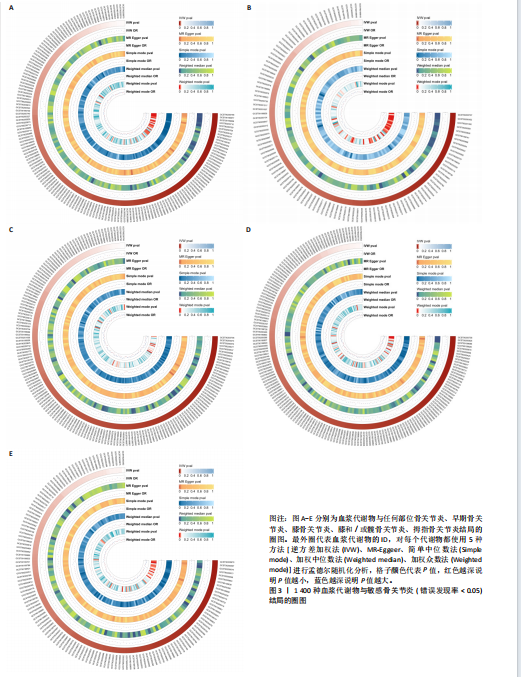
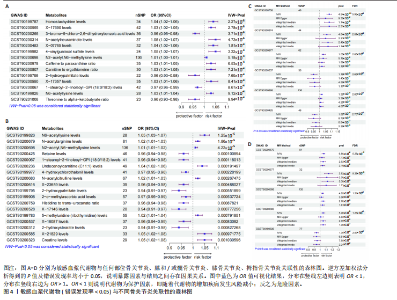
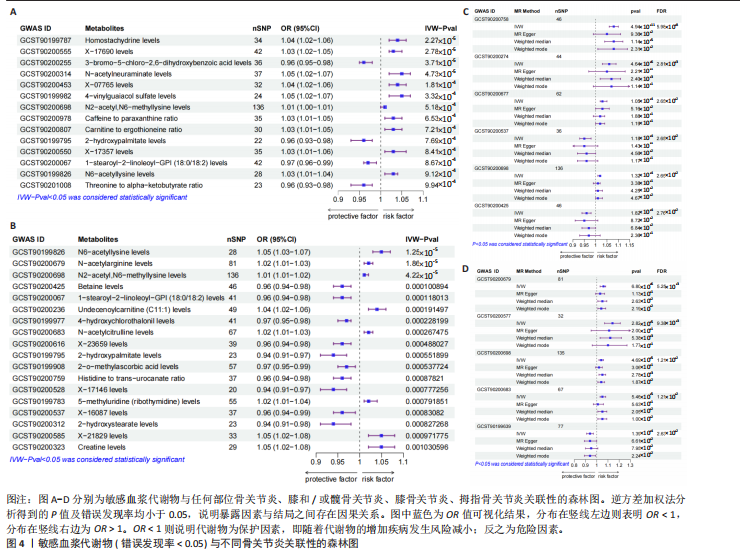
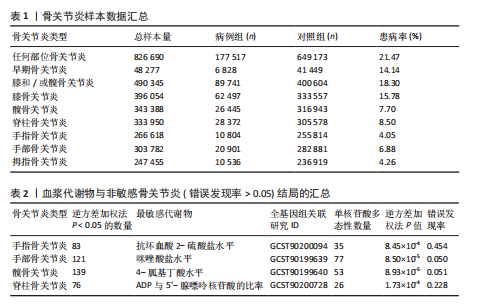
2.1 工具变量筛选结果 根据标准对1 400种血浆代谢物进行筛选,每个代谢物得到的单核苷酸多态性数量从10-169个不等,得到的工具变量F > 10,说明所得到的单核苷酸多态性存在弱工具变量的可能性较小,均可进行下一步孟德尔随机化分析。 2.2 孟德尔随机化分析结果 以逆方差加权法作为孟德尔随机化的主要分析方法,筛选PIVW < 0.05的结果,将获得的结局数据以圈图(图2,3)及表格(表2,3)的形式呈现,圈图中最外圈代表代谢物的ID,对每个代谢物都使用5种方法(逆方差加权法、MR-Eggeer、简单中位数法、加权中位数法、加权众数法) 进行孟德尔随机化分析,得到5种方法的P值及置信区间(OR),格子颜色代表P值,红色越深说明P值越小,蓝色越深说明P值越大。 经过FDR矫正之后,手指骨关节炎、手部骨关节炎、髋骨关节炎、脊柱骨关节炎均无符合FDR < 0.05的数据;早期骨关节炎筛选后仅丝氨酸与苏氨酸的比率符合FDR < 0.05;任何部位骨关节炎、膝和/或髋骨关节炎、膝骨关节炎、拇指骨关节炎均有数量不等满足FDR < 0.05的血浆代谢物,绘制相应的森林图进行呈现(图4)。 任何部位骨关节炎:符合逆方差加权法P < 0.05的血浆代谢物有160个,最敏感的血浆代谢物为甘氨酸与丝氨酸的比率(FDR=8.04×10-14);经筛选之后,符合逆方差加权法P < 0.05的血浆代谢物有14个,最敏感血浆代谢物为高水苏碱水平(FDR=1.90×10-4)。 早期骨关节炎:符合逆方差加权法P < 0.05的血浆代谢物有112个,最敏感的血浆代谢物为甘氨酸水平(FDR=1.84×10-3);经筛选之后,符合逆方差加权法P < 0.05 的血浆代谢物有1个,为丝氨酸与苏氨酸的比率(FDR=1.84×10-3)。 膝骨关节炎:符合逆方差加权法P < 0.05 的血浆代谢物有165个,最敏感的血浆代谢物为甘氨酸与丝氨酸的比率(FDR=5.98×10-8);经筛选之后,符合逆方差加权法P < 0.05 的血浆代谢物有6个,最敏感血浆代谢物仍为甘氨酸与丝氨酸的比率(FDR=5.98×10-8)。 膝和/或髋骨关节炎:符合逆方差加权法P < 0.05的血浆代谢物有168个,最敏感的血浆代谢物为甘氨酸与丝氨酸的比率(FDR=4.57×10-12);经筛选之后,符合逆方差加权法P < 0.05的血浆代谢物有5个,最敏感血浆代谢物仍为甘氨酸与丝氨酸的比率(FDR=4.57×10-12)。 髋骨关节炎:符合逆方差加权法P < 0.05 的血浆代谢物有139个,最敏感血浆代谢物为4-胍基丁酸水平(逆方差加权法P=8.93×10-5、FDR=0.051)。 脊柱骨关节炎:符合逆方差加权法P < 0.05的血浆代谢物有76个,最敏感血浆代谢物为ADP与AMP的比(逆方差加权法P=1.73×10-4、FDR=0.228)。 手指骨关节炎:符合逆方差加权法P < 0.05的血浆代谢物有83个,最敏感血浆代谢物为抗坏血酸2-硫酸盐水平(逆方差加权法P=8.45×10-4、FDR=0.454)。 手部骨关节炎:符合逆方差加权法P < 0.05的血浆代谢物有121个,最敏感血浆代谢物为咪唑酸盐水平(逆方差加权法P=8.50×10-5、FDR=0.050)。"
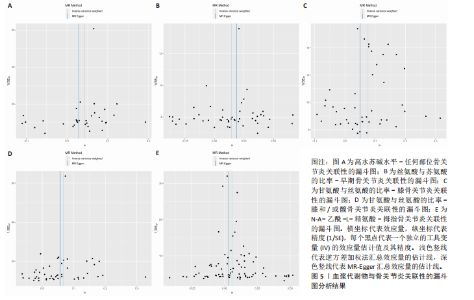
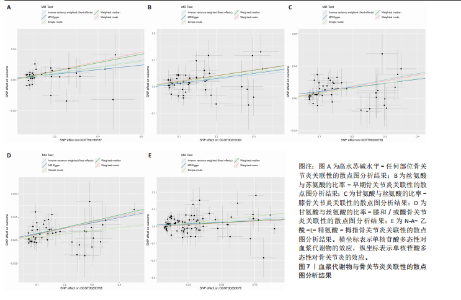
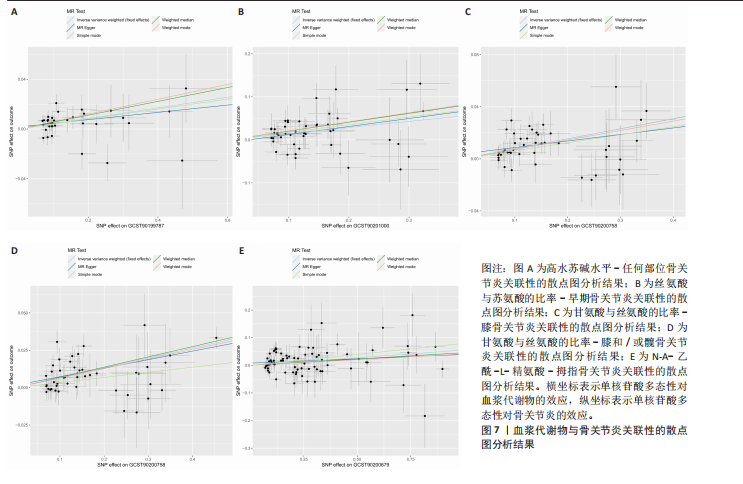
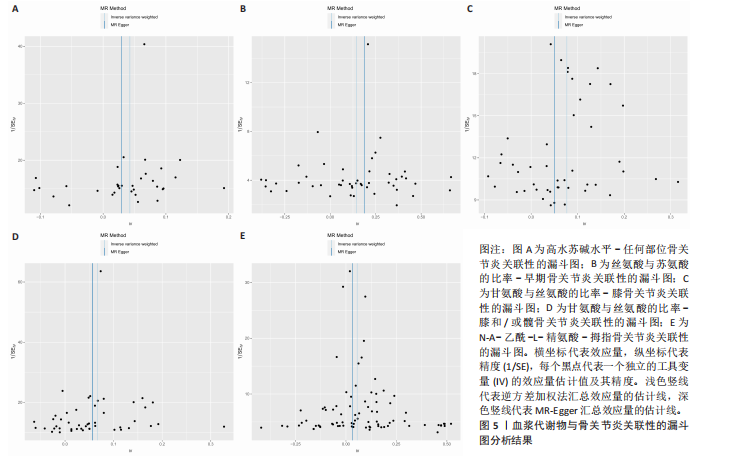
拇指骨关节炎:符合逆方差加权法P < 0.05的血浆代谢物有135个,最敏感血浆代谢物为N-A-乙酰-L-精氨酸(FDR=5.25×10-3);经筛选之后,符合逆方差加权法P < 0.05的血浆代谢物有5个,最敏感血浆代谢物仍为N-A-乙酰-L-精氨酸(FDR=5.25×10-3)。 此次研究通过漏斗图(图5)来检测遗传变异间的异质性,所绘漏斗图均呈现对称形状,这意味着没有明显的异质性,也即研究效应与其准确性之间不存在系统性偏差。绘制留一图(图6)进行敏感性分析,每次分析都去除一个单核苷酸多态性,然后使用剩下的单核苷酸多态性进行孟德尔随机化分析,图中显示去除一个单核苷酸多态性之后,其他单核苷酸多态性得到的综合效应值与所有单核苷酸多态性得到的主效应值一致,说明去除单个单核苷酸多态性对本孟德尔随机化分析不会产生过多的影响。 绘制散点图(图7)可视化展示工具变量(单核苷酸多态性)对代谢物与骨关节炎之间关系的效应估计及其不确定性,通过散点图可以直观地分析每个工具变量的具体影响方向(正向或负向关联)以及置信区间范围,进一步辅助证实因果关系的可靠性。"
| [1] XIA B, DI C, ZHANG J, et al. Osteoarthritis pathogenesis: a review of molecular mechanisms. Calcified Tissue Int. 2014;95(6):495-505. [2] CHO HJ, MOREY V, KANG JY, et al. Prevalence and Risk Factors of Spine, Shoulder, Hand, Hip, and Knee Osteoarthritis in Community-dwelling Koreans Older Than Age 65 Years. Clin Orthop Relat Res. 2015;473(10):3307-3314. [3] CIEZA A, CAUSEY K, KAMENOV K, et al. Global estimates of the need for rehabilitation based on the Global Burden of Disease study 2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet (London, England). 2021;396(10267):2006-2017. [4] CHEN X, LIU J, WANG G, et al. Regulating lipid metabolism in osteoarthritis: a complex area with important future therapeutic potential. Ann. Med. 2024;56(1):2420863. [5] ZHENG L, ZHANG Z, SHENG P, et al. The role of metabolism in chondrocyte dysfunction and the progression of osteoarthritis. Ageing Res Rev. 2021;66:101249. [6] ZHAI G. Clinical relevance of biochemical and metabolic changes in osteoarthritis. Adv Clin Chem. 2021;101:95-120. [7] SHOWIHEEN SAA, SUN AR, WU X, et al. Application of Metabolomics to Osteoarthritis: from Basic Science to the Clinical Approach. Curr Rheumatol Rep. 2019;21(6):26. [8] KETTUNEN J, DEMIRKAN A, WÜRTZ P, et al. Genome-wide study for circulating metabolites identifies 62 loci and reveals novel systemic effects of LPA. Nature communications. 2016;7:11122. doi:10.1038/ncomms11122. [9] CHEN Y, LU T, PETTERSSON-KYMMER U, et al. Genomic atlas of the plasma metabolome prioritizes metabolites implicated in human diseases. Nat Genet. 2023;55(1):44-53. [10] BOER CG, HATZIKOTOULAS K, SOUTHAM L, et al. Deciphering osteoarthritis genetics across 826,690 individuals from 9 populations. Cell. 2021; 184(18):4784-818.e17. [11] YANG X, XIE Y, WANG Y, et al. Arthritis is associated with an increased risk of incident diabetes in Chinese adults: A nationwide cohort study and updated meta-analysis. Diabetes Metab Res Rev. 2022;38(2):e3487. [12] DONG Q, LIU H, YANG D, et al. Diabetes mellitus and arthritis: is it a risk factor or comorbidity?: A systematic review and meta-analysis. Medicine. 2017;96(18):e6627. [13] DEVEREAUX D, TEWELDE SZ. Hyperthyroidism and thyrotoxicosis. Emerg Med Clin North Am. 2014;32(2):277-292. [14] CHEN S, SUN X, ZHOU G, et al. Association between sensitivity to thyroid hormone indices and the risk of osteoarthritis: an NHANES study. Eur J Med Res. 2022;27(1):114. [15] TO K, MAK C, ZHANG C, et al. The association between alcohol consumption and osteoarthritis: a meta-analysis and meta-regression of observational studies. Rheumatol Int. 2021;41(9): 1577-1591. [16] BURGESS S, BUTTERWORTH A, THOMPSON SG. Mendelian randomization analysis with multiple genetic variants using summarized data. Genet Epidemiol. 2013;37(7):658-665. [17] BOWDEN J, DEL GRECO MF, MINELLI C, et al. A framework for the investigation of pleiotropy in two-sample summary data Mendelian randomization. Stat Med. 2017;36(11):1783-1802. [18] BURGESS S, THOMPSON SG. Interpreting findings from Mendelian randomization using the MR-Egger method. Eur J Epidemiol. 2017;32(5):377-389. [19] BOWDEN J, DAVEY SMITH G, BURGESS S. Mendelian randomization with invalid instruments: effect estimation and bias detection through Egger regression. Int J Epidemiol. 2015; 44(2):512-525. [20] BOWDEN J, DAVEY SMITH G, HAYCOCK PC, et al. Consistent Estimation in Mendelian Randomization with Some Invalid Instruments Using a Weighted Median Estimator. Genet Epidemiol. 2016;40(4):304-314. [21] VERBANCK M, CHEN CY, NEALE B, et al. Detection of widespread horizontal pleiotropy in causal relationships inferred from Mendelian randomization between complex traits and diseases. Nat Genet. 2018;50(5):693-698. [22] LI XZ, ZHANG SN. Recent advance in treatment of osteoarthritis by bioactive components from herbal medicine. Chin Med. 2020;15:80. [23] KNIGHTS AJ, REDDING SJ, MAERZ T. Inflammation in osteoarthritis: the latest progress and ongoing challenges. Curr Opin Rheumatol. 2023;35(2): 128-134. [24] BLIDDAL H. Definition, pathology and pathogenesis of osteoarthritis. Ugeskr Laeger. 2020;182(42):V06200477. [25] LEE YM, SON E, KIM SH, et al. Protective Effects of Glycine soja Leaf and Stem Extract against Chondrocyte Inflammation and Osteoarthritis.Int J Mol Sci. 2023;24(5):4829. [26] LING H, LI M, YANG C, et al. Glycine increased ferroptosis via SAM-mediated GPX4 promoter methylation in rheumatoid arthritis. Rheumatology (Oxford, England). 2022;61(11):4521-4534. [27] WANG L, LUO J, MAO Z, et al. Glycine N-Acyltransferase deficiency in sensory neurons suppresses osteoarthritis pain. J Pain. 2025;33: 105408. [28] DE PAZ-LUGO P, LUPIÁÑEZ JA, MELÉNDEZ-HEVIA E. High glycine concentration increases collagen synthesis by articular chondrocytes in vitro: acute glycine deficiency could be an important cause of osteoarthritis. Amino Acids. 2018;50(10): 1357-1365. [29] GEERAERTS SL, HEYLEN E, DE KEERSMAECKER K, et al. The ins and outs of serine and glycine metabolism in cancer. Nat Metab. 2021;3(2):131-141. [30] MILNER JM, PATEL A, ROWAN AD. Emerging roles of serine proteinases in tissue turnover in arthritis. Arthritis Rheum. 2008;58(12):3644-3656. [31] CAO S, BUDINA E, RACZY MM, et al. A serine-conjugated butyrate prodrug with high oral bioavailability suppresses autoimmune arthritis and neuroinflammation in mice. Nat Biomed Eng. 2024;8(5):611-627. [32] LEWIS KA, OSIER N, CARRASCO R, et al. Serine, N-acetylaspartate differentiate adolescents with juvenile idiopathic arthritis compared with healthy controls: a metabolomics cross-sectional study. Pediatr Rheumatol Online J. 2022;20(1):12. [33] HE Z, LI P, LIU P, et al. Exploring stachydrine: from natural occurrence to biological activities and metabolic pathways. Front Plant Sci. 2024;15: 1442879. [34] CONNOR MA, STARK JB, FRITZ JC, et al. Stachydrine: content in alfalfa and biological activity in chicks. J Agric Food Chem. 1973;21(2): 195-198. [35] KUCHTA K, VOLK RB, RAUWALD HW. Stachydrine in Leonurus cardiaca, Leonurus japonicus, Leonotis leonurus: detection and quantification by instrumental HPTLC and 1H-qNMR analyses. Pharmazie. 2013;68(7):534-540. [36] SASSO S, DALMEDICO L, DELWING-DAL MAGRO D, et al. Effect of N-acetylarginine, a metabolite accumulated in hyperargininemia, on parameters of oxidative stress in rats: protective role of vitamins and L-NAME. Cell Biochem Funct. 2014; 32(6):511-519. [37] CAO S, LI Y, SONG R, et al. L-arginine metabolism inhibits arthritis and inflammatory bone loss. Ann Rheum Dis. 2024;83(1):72-87. [38] ONUORA S. L-arginine inhibits arthritis and bone loss by reprogramming osteoclast metabolism. Nat Rev Rheumatol. 2023;19(12):760. [39] MCHUGH J. Arginine restriction attenuates bone loss in arthritis. Nat Rev Rheumatol. 2020; 16(3):128. [40] JIN Z, CHANG B, WEI Y, et al. Curcumin exerts chondroprotective effects against osteoarthritis by promoting AMPK/PINK1/Parkin-mediated mitophagy. Biomed Pharmacother. 2022;151:113092. [41] REGINSTER JY, DEROISY R, ROVATI LC, et al. Long-term effects of glucosamine sulphate on osteoarthritis progression: a randomised, placebo-controlled clinical trial. Lancet (London, England). 2001;357(9252):251-256. [42] PAVELKÁ K, GATTEROVÁ J, OLEJAROVÁ M, et al. Glucosamine sulfate use and delay of progression of knee osteoarthritis: a 3-year, randomized, placebo-controlled, double-blind study. Arch Intern Med. 2002;162(18):2113-2123. [43] GREGORI D, GIACOVELLI G, MINTO C, et al. Association of Pharmacological Treatments With Long-term Pain Control in Patients With Knee Osteoarthritis: A Systematic Review and Meta-analysis. JAMA. 2018;320(24):2564-2579. [44] ANDRIANAKOS AA, KONTELIS LK, KARAMITSOS DG, et al. Prevalence of symptomatic knee, hand, and hip osteoarthritis in Greece. The ESORDIG study. J Rheumatol. 2006;33(12):2507-2513. [45] MAZUR BJ, CHUI CF, SMITH JK. Isolation and characterization of plant genes coding for acetolactate synthase, the target enzyme for two classes of herbicides. Plant Physiol. 1987; 85(4):1110-1117. [46] GANZ R, LEUNIG M, LEUNIG-GANZ K, et al. The etiology of osteoarthritis of the hip: an integrated mechanical concept. Clin Orthop Relat Res. 2008; 466(2):264-272. [47] TOMÉ I, ALVES-PIMENTA S, SARGO R, et al. Mechanical osteoarthritis of the hip in a one medicine concept: a narrative review. BMC Vet Res. 2023;19(1):222. [48] KERKHOF F, KENNEY D, OGLE M, et al. The biomechanics of osteoarthritis in the hand: Implications and prospects for hand therapy. J Hand Ther. 2022;35(3):367-376. |
| [1] | Yu Cenqi, Liu Yang, Yu Jiangfeng, Kang Kang, Deng Yaoge, Xia Xiaowei, Zhang Yijian, Zhu Xuesong. Biomimetic Black Phosphorus Nanosystem Regulates Synovial Macrophage Polarization for Osteoarthritis Treatment [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(在线): 1-13. |
| [2] | Zhang Zizheng, Luo Wang, Liu Changlu. Application value of finite element analysis on unicompartmental knee arthroplasty for medial knee compartmental osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(9): 2313-2322. |
| [3] | Chen Qiuhan, Yang Long, Yuan Daizhu, Wu Zhanyu, Zou Zihao, Ye Chuan. Peri-knee osteotomy for treatment of knee osteoarthritis: optimization of treatment strategies [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(9): 2303-2312. |
| [4] | Zhang Nan, Meng Qinghua, Bao Chunyu. Characteristics and clinical application of ankle joint finite element models [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(9): 2343-2349. |
| [5] | Li Qingbin, Lin Jianhui, Huang Wenjie, Wang Mingshuang, Du Jiankai, Lao Yongqiang. Bone cement filling after enlarged curettage of giant cell tumor around the knee joint: a comparison of subchondral bone grafting and non-grafting [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 1896-1902. |
| [6] | Li Linzhen, Jiao Hongzhuo, Chen Weinan, Zhang Mingzhe, Wang Jianlong, Zhang Juntao. Effect of icariin-containing serum on lipopolysaccharide-induced inflammatory damage in human chondrocytes [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1368-1374. |
| [7] | Chen Ju, Zheng Jinchang, Liang Zhen, Huang Chengshuo, Lin Hao, Zeng Li. Effect and mechanism of beta-caryophyllene in mice with osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1341-1347. |
| [8] | Lyu Guoqing, Aizimaitijiang·Rouzi, Xiong Daohai. Irisin inhibits ferroptosis in human articular chondrocytes: roles and mechanisms [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1359-1367. |
| [9] | Wu Zhilin, , He Qin, Wang Pingxi, Shi Xian, Yuan Song, Zhang Jun, Wang Hao . DYRK2: a novel therapeutic target for rheumatoid arthritis combined with osteoporosis based on East Asian and European populations [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1569-1579. |
| [10] | Liu Hongtao, Wu Xin, Jiang Xinyu, Sha Fei, An Qi, Li Gaobiao. Causal relationship between age-related macular degeneration and deep vein thrombosis: analysis based on genome-wide association study data [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1602-1608. |
| [11] | Li Hao, Tao Hongcheng, Zeng Ping, Liu Jinfu, Ding Qiang, Niu Chicheng, Huang Kai, Kang Hongyu. Mitogen-activated protein kinase signaling pathway regulates the development of osteoarthritis: guiding targeted therapy with traditional Chinese medicine [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1476-1485. |
| [12] | Guo Ying, Tian Feng, Wang Chunfang. Potential drug targets for the treatment of rheumatoid arthritis: large sample analysis from European databases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1549-1557. |
| [13] | Bu Yangyang, Ning Xinli, Zhao Chen. Intra-articular injections for the treatment of osteoarthritis of the temporomandibular joint: different drugs with multiple combined treatment options [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1215-1224. |
| [14] | Gao Zengjie, , Pu Xiang, Li Lailai, Chai Yihui, Huang Hua, Qin Yu. Increased risk of osteoporotic pathological fractures associated with sterol esters: evidence from IEU-GWAS and FinnGen databases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1302-1310. |
| [15] | Liu Fengzhi, Dong Yuna, Tian Wenyi, Wang Chunlei, Liang Xiaodong, Bao Lin. Gene-predicted associations between 731 immune cell phenotypes and rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1311-1319. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||