Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (28): 7316-7322.doi: 10.12307/2026.785
Previous Articles Next Articles
Evolution, development and molecular regulation of fish tooth
Wang Shuoran1, Huang Rui1, Dong Lingyue2, An Wei2, Huang Xiaofeng1
- 1Department of Stomatology, Beijing Friendship Hospital, Capital Medical University, Beijing 100050, China; 2Department of Cell Biology, Capital Medical University School of Basic Medical Sciences, Beijing 100069, China
-
Received:2025-08-25Revised:2025-11-16Online:2026-10-08Published:2026-02-11 -
Contact:Huang Xiaofeng, MD, Chief physician, Professor, Department of Stomatology, Beijing Friendship Hospital, Capital Medical University, Beijing 100050, China -
About author:Wang Shuoran, Doctoral candidate, Department of Stomatology, Beijing Friendship Hospital, Capital Medical University, Beijing 100050, China -
Supported by:Beijing Natural Science Foundation (General Program), No. 7252026 (to HXF); National Natural Science Foundation of China (General Program), No. 82071141 (to HXF)
CLC Number:
Cite this article
Wang Shuoran, Huang Rui, Dong Lingyue, An Wei, Huang Xiaofeng. Evolution, development and molecular regulation of fish tooth[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(28): 7316-7322.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
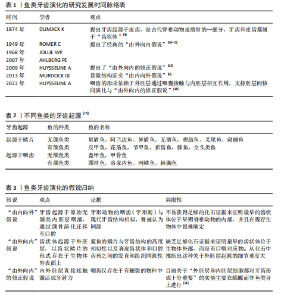
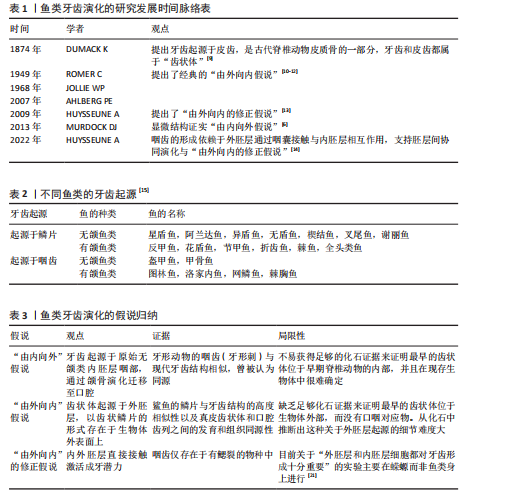
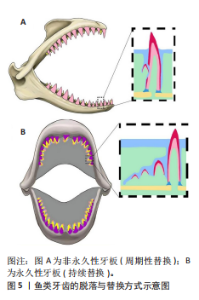
2.1 鱼类牙齿发育的起源与演化 DONOGHUE[3]发现,在最早的无颌鱼类头骨和鳞片中可见大量的真皮骨,其中可以发现齿状体的结构,被推测为“真牙”的前身。HARIDY等[4]发现在口腔中也存在类似的“口腔齿状体”,即牙齿或齿列,这种“口腔齿状体”在现存低等脊椎动物(比如鲨鱼和鳐鱼)的皮肤表面、口腔和咽部鳃弓等多个身体部位都可以被找到。 近2个世纪以来,鱼类牙齿演化的相关研究不断发展,学者们的观点也不断更新[5-11](表1),目前主要存在两种牙齿演化的假说(表2):①假说1:牙齿演化“由内向外”理论认为,随着咽齿的形成和颌骨的演化,起源于内胚层的牙齿向前移动到口腔。该理论的证据是牙形动物的硬刺。牙形动物(存在于寒武纪早期)无内外骨骼,有咽齿,称为牙形刺,它的物质构成与现代牙齿的结构和生长方式相似[5-6],即类釉质(似釉质)覆盖在牙本质上实现生长。牙形刺与现代牙齿的同源性是“由内向外”假说的重要依据。②假说2:牙齿演化“由外向内”理论认为有颌鱼类的牙齿由远古鱼类的真皮齿状体(鳞片)演化而来,这些原始硬骨鱼类的颅部存在大量的鳞片,其中口腔周围的一部分鳞片(即真皮齿状体)附着在颅骨上参与了捕食活动[7]。“由外向内”理论推测,这些由外胚层衍生的、折叠并融入口腔的真皮齿状体最终成为有颌脊椎动物的牙齿[8]。 在此基础上,HUYSSEUNE等[14]和FRASER等[16-18]提出了“由外向内”的修正假说,认为具备成牙潜能的外胚层组织通过口腔和鳃裂侵入咽腔,与神经嵴来源的间充质相互作用,形成了牙齿,这一假说也确认了真皮齿状体与牙齿的演化同源性。近期与基因表达相关的研究工作也支持真皮齿状体和口腔齿状体之间的同源性。DEBIAIS-THIBAUD等[19]通过分析与牙齿发生相关的基因在小点猫鲨口腔齿状体和真皮齿状体中的表达,发现口腔齿状体和真皮齿状体有着相同的发育模式,其中远端缺失同源框基因不仅参与了口腔齿状体的形成,也参与了真皮齿状体发育的调控,这一发现支持两者为系列同源物的观点。GILLIS等[20]通过细胞谱系示踪技术研究鲨鱼胚胎的细胞发育,发现鲨鱼的颅部鳞片和牙齿其实产生于相同类型的神经嵴细胞,说明鲨鱼的鳞片和牙齿之间存在同源关系,进一步确认了“由外向内”假说。 鱼类牙齿演化的假说归纳,见表3。 2.2 鱼类牙齿的形态、发生与发育 2.2.1 鱼类牙齿的形态与功能 鱼类牙齿大多由内部牙本质、表面高度矿化的牙釉质以及牙髓构成,牙齿形态、分布与其功能有关[22]。肉食性鱼类的牙齿都较尖锐,呈犬齿状、圆锥状或臼齿状;杂食或兼食性鱼类的牙齿呈切割形、磨形及刷形等;滤食性鱼类的牙齿都较细弱,呈绒毛状,集合排列成齿带。与哺乳类不同,鱼类牙齿可以分为口腔牙齿(颌齿)和咽齿,前者分布在口腔和上颚上,后者则分布在咽骨上。咽齿的形状也与食性有关,比如青鱼主要捕食螺蛳,咽齿呈臼齿状,可有力地碾碎螺蛳外壳;草鱼以水草为食,咽齿呈梳子状,便于切断植物。 2.2.2 鱼类牙齿的牙本质 基于目前的研究,鱼类牙齿的牙本质形态可分为正齿型(牙冠中央具有中空的髓腔,外层被正齿质包裹)、骨齿型(该类牙齿缺乏正齿质,整个牙冠完全由多孔性骨齿质构成)、假骨齿型(髓腔被骨齿质构成的齿质核心填充,外围仍包被正齿质)和血管齿质型[23](图4)。 大多数硬骨鱼只含有正齿质,形成中空状结构,这与哺乳类动物类似[24-25]。大多数软骨鱼,比如鲨鱼的牙本质主要是骨齿质,通常与颌骨产生骨性附着[19]。有学者研究了带鱼典型的假骨齿质结构特征和发育过程,发现假骨齿质的形成分2个阶段形成:由极化的成牙本质细胞分泌基质形成正齿质,位于牙本质的最外层与类釉质相连;由星状成牙本质细胞在牙髓中形成骨齿质[26]。而血管齿质是一种特殊的牙本质类型,它主要存在于硬骨鱼纲鳕科和鲽科的牙齿中。血管齿质中的毛细血管与位于成牙本质细胞层正下方的血管丛相连。牙髓组织由分散的间充质细胞组成,排列成网状并嵌入呈颗粒状的基质中,较大的血管出现在中央牙髓区域[27]。血管化是血液供应的关键,对组织维持、修复和生长至关重要。由于不同的摄食策略(如碾磨、撕裂、切割等)对牙齿施加不同的生物力学应力,所以特定的血管模式(如异齿鲨臼齿中平行的骨样小管)可能是为了抵抗特定类型的力,或为高应力区域提供有效营养"
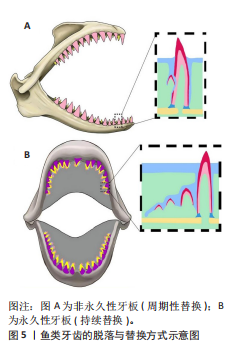
2.2.3 鱼类牙齿的牙釉质 牙釉质是一种广泛分布在脊椎动物中的硬组织,从发育特点上可以大致分为2类:一种是高等成体两栖动物及更高等动物(包括人类)的釉质,称为真牙釉质或外胚层牙釉质;另一种主要分布在鱼类中,称为类釉质[29]。鱼类的类釉质也由成釉细胞分泌形成,主要由羟基磷灰石组成,具有极高的硬度和耐磨性[30],但它与真牙釉质相比仍有许多不同特征。例如,类釉质的有机基质主要由Ⅰ型胶原纤维组成,形成大部分相互交织、错综复杂的粗大纤维束;而真牙釉质的有机基质不含胶原纤维,主要基质蛋白包括釉原蛋白、釉蛋白和成釉蛋白。同时与真牙釉质相比,类釉质中含氟量更多,以氟磷灰石的形式存在。与人类的真牙釉质发育有所不同,类釉质的发育由成釉细胞分泌基质矿化后形成[19]。而大部分鱼类的类釉质形成并非完全独立,而是与牙本质存在着联系。REIF[31]的扫描电镜数据显示类釉质需要间充质来源的成牙本质细胞参与,并非仅由成釉细胞形成[32]。在一些少数的硬骨鱼类(如雀鳝)中[33],牙釉质由纯粹的外胚层组织形成,与牙本质发育相对独立。 SASAGAWA[34]通过透射电镜技术、甲苯胺蓝染色、碱性磷酸酶活性检测和电子衍射分析揭示了软骨鱼类和硬骨鱼类的类釉质矿化机制,软骨鱼类的类釉质矿化过程可分为初始矿化和晶体生长2个关键阶段首先,成牙本质细胞通过其细胞突起分泌大量管状囊泡,这些囊泡直径12-30 nm,呈细长分支状结构,紧密分布于类釉质基质中;囊泡膜包裹的电子致密颗粒逐渐发育为细小晶体,随着矿化进展,晶体在囊泡内迅速生长,体积增大,形成规则的六边形截面,同时牙上皮细胞通过吞噬和降解作用逐步清除管状囊泡及残留的有机基质,最终形成高度矿化的类釉质。 硬骨鱼类的类釉质矿化则由更复杂的多个阶段组成。成牙本质细胞首先分泌直径40-100 nm的基质囊泡,分布于富含胶原纤维的类釉质基质中,从而启动初始矿化。与软骨鱼不同,在硬骨鱼的基质囊泡中常可检测到碱性磷酸酶活性,提示酶促反应可能参与矿化启动。随后,细长的晶体从囊泡中释放并沿Ⅰ型胶原纤维定向排列,形成带状或丝状结构,胶原纤维为晶体生长提供了空间模板和力学支撑。在矿化后期,晶体进一步增大并占据类釉质主体,牙上皮细胞则降解胶原纤维、清除残留囊泡,最终形成致密的矿化组织。因此,与软骨鱼相比,硬骨鱼类釉质的核心特征在于其有机基质的组成以胶原纤维为主,基质囊泡较少且仅辅助矿化启动,晶体形态亦受胶原排列的显著影响。在矿化过程中,这种囊泡与纤维的协同机制可能为高等脊椎动物(如哺乳动物)的釉质演化提供参考。 2.2.4 鱼类牙齿的发育 鱼类牙齿的整体发育过程与人类相似,都由牙上皮与颅神经嵴来源的间充质相互作用形成。首先,牙板上皮增殖,诱导下方的间充质增殖和分化形成牙胚。鱼类的牙胚发育可以明确地分为蕾状期、帽状期和钟状期[35],同时鱼类牙齿的发育与再生展现出高度保守的分子调控机制和显著的适应性特征。以软骨鱼类,如猫鲨为例[36],其牙齿发育涉及上皮-间充质的相互作用及Wnt、骨形态发生蛋白、成纤维细胞生长因子和音猬因子等信号通路的协同调控。鲨鱼牙齿保持终身再生的能力,这一过程的核心机制是牙板的持续存在,其内富集干细胞群,通过β-catenin、垂体同源框转录因子2等基因的动态表达调控多代牙齿的连续形成。鲨鱼的早期牙齿形态较为简单,呈简单锥形或三角片状,无牙根(端生齿),仅通过软组织附着颌骨;而成年鲨通过牙板输送带系统持续再生牙齿,形成复杂结构(如多尖齿),这一过程受釉结样信号中心调控,与哺乳动物牙齿形态发生的机制同源。此外,MA等[37]发现硬骨鱼类如斑石鲷的喙状愈合齿通过颌骨与牙齿的同步矿化形成,而鲤科鱼类的咽齿则通过高更新率适应捕食需求,体现了牙齿形态与功能的生态适应性。这些发现揭示了脊椎动物牙齿发育的古老遗传框架,并为理解牙齿再生机制提供了参考。 2.2.5 鱼类牙齿的脱落与替换 鱼类在牙齿更替机制上有很大不同。大多数硬骨鱼的牙齿附着于颌骨上,通过牙齿基底部分的吸收实现周期性替换,新生牙齿在内侧发育成熟后向外侧迁移替代旧齿[38],它们的每代替换牙形成不连续的短暂牙板,为非永久性牙板[39(图5A)。而鲨鱼等一些软骨鱼成熟的牙齿附着于颌骨软骨表面,与之连接的后续牙齿及其牙板为连续的上皮褶皱,唇舌向排列在口腔内,为永久性结构,在鲨鱼个体生命周期中持续存在,其牙齿的替换是通过整列更替实现牙齿的更新[40](图5B)。这一演化差异在化石记录中可得到有力的佐证。CHEN等[41]采用同步辐射显微CT技术对4.24亿年前的一种基干硬骨鱼类——安德雷奥莱鱼属进行三维重建,揭示了脊椎动物牙齿替换的最早证据:此种鱼类已具备通过基底部骨吸收实现牙齿更替的发育模式,这一发现修正了BOTELLA等[42]关于早期硬骨鱼牙齿不可更替的传统认知[43]。"

现代硬骨鱼的牙齿更替具有丰富的多样性。BEMIS等[44]发现蓝鱼的牙根深深嵌入牙槽,类似于人类牙槽窝,非常牢固,这导致其牙齿更替频率低于其他鱼类。与此同时,蓝鱼单个牙齿的替换模式十分高效,从启动吸收到完成更替的周期仅需48 h,快于多数其他鱼类,这揭示了脊椎动物通过调控牙齿替换节奏适应生存需求的新机制。 2.3 影响鱼类牙齿发育的信号分子 鱼类牙齿的发生和发育需要多种信号分子的协同调控,这些信号分子与相应受体结合调控特定基因的表达,进而调控细胞增殖和迁移、细胞分化、细胞凋亡等进程。不同的鱼类(比如:虹鳟、青鳉、墨西哥脂鲤、斑马鱼、慈鲷、肺鱼)通过各种分子信号通路的调控促进形成形态各异和数量不一的牙齿和牙列。最为重要的调控鱼类牙齿发育的信号分子及通路有成纤维细胞生长因子、音猬因子、视黄酸、Wnt等。 2.3.1 成纤维细胞生长因子 成纤维细胞生长因子信号通路作为发育的“启动开关”,其家族成员呈现阶段特异性表达:成纤维细胞生长因子8/成纤维细胞生长因子9最早在斑马鱼口腔上皮激活,随后成纤维细胞生长因子4/成纤维细胞生长因子9通过刺激釉质结周围细胞增殖,驱动牙胚的形态发生[45-46]。JACKMAN等[47]通过转基因技术使斑马鱼过表达成纤维细胞生长因子10 a,导致斑马鱼出现多尖齿和额外齿。另外值得注意的是,成纤维细胞生长因子3/成纤维细胞生长因子10的缺失虽然导致牙齿缩小与齿尖畸形,但不影响成釉细胞分化[48],说明成纤维细胞生长因子信号通路中不同成员负责牙齿发育的不同环节,彼此功能相对独立。 2.3.2 音猬因子 音猬因子信号通路的功能具有保守性,其核心功能在4亿年的演化历程中高度保守。音猬因子信号高度保守的核心功能主要有3方面:①调控上皮-间充质互作,比如音猬因子诱导间充质细胞聚集并表达Pax9、Msx1等转录因子,触发牙板形成(斑马鱼、青鳉鱼等硬骨鱼中保守);②音猬因子浓度梯度直接决定牙齿的形态极性,比如斑马鱼咽齿中音猬因子在牙蕾的远端(咬合面)高表达,驱动牙尖形成,近端(基底)低表达,驱动牙颈分化;③调控牙齿替换周期,音猬因子信号能激活Gli1+干细胞增殖,启动新牙胚发生(如斑马鱼第4-5代齿替换)。同时,音猬因子信号通路的表达特征与功能在不同物种中有显著差异,例如,在鲨鱼等软骨鱼类中,音猬因子精确标记单个牙齿萌出位点[49-50],这确保了它们的多排利齿能精确排列,控制齿列的规律性再生;而JACKMAN等[51]发现斑马鱼的音猬因子信号在咽部广泛表达,为咽齿的广泛分布提供发育指令,而且音猬因子突变体胚胎的牙齿不再发育,这表明音猬因子是牙齿启动阶段的关键因子。这种表达模式与小鼠音猬因子信号严格局限在牙上皮的模式类似[52]。 2.3.3 视黄酸(维甲酸) 视黄酸作为关键的形态发生因子,在脊椎动物的多种组织发育中发挥重要的作用,视黄酸信号通路的活性变化可调控颅骨形成、脊柱发育以及鱼类齿列的建立[53]。斑马鱼咽部牙齿的发育直接依赖视黄酸信号传导,视黄酸还同时参与调控内胚层咽囊的形态发生[54-55]。然而,这一机制具有物种特异性。EL SHAHAWY等[56]发现贝龙形宽脊鱼(日本青鳉)和墨西哥刺鱼的咽齿发育不受视黄酸信号影响,提示鲤科鱼类的视黄酸依赖性齿列诱导可能在演化过程中属于特殊现象。关于作用模式,HUYSSEUNE等[39]提出视黄酸可能通过调控咽囊-裂隙接触的形态发生间接影响牙齿发育,而GIBERT等[54]的实验结果显示视黄酸对牙齿的直接调控具有时空敏感性:视黄酸的空间浓度分布与音猬因子恰好相反,表现为牙颈区视黄酸浓度高,抑制细胞分化和矿化,形成牙颈部,而牙尖区视黄酸浓度低,让牙尖得以矿化延伸。在牙齿替换期需要启动牙齿再生过程中,激活Cyp26b1(视黄酸降解酶)导致局部视黄酸水平下降,从而解除对干细胞增殖的抑制;而牙胚启动期需要抑制成牙功能,此时由咽部间充质分泌高浓度视黄酸,抑制口腔上皮的成牙潜力,阻断牙板形成[57]。另外,胚胎期(受精后24 h)加入外源性视黄酸可使斑马鱼齿列前移且数量增加,但在发育后期(受精后56 h)加入外源性视黄酸仅会影响特定牙弓的牙齿数[58]。同时,DRAUT等[59]发现外源性视黄酸能突破斑马鱼主排牙齿的发育限制,诱导产生额外的第六颗牙齿。 视黄酸与音猬因子信号通路之间存在动态拮抗:音猬因子通过上调视黄酸分解酶Cyp26a1/c1限制视黄酸活性,而视黄酸又可反向调控音猬因子表达,这种拮抗作用在斑马鱼的鳍再生中确保鳍的形态和大小精确恢复,在比目鱼的咽部发育中则塑造出特定的齿列模式[40]。这些研究表明,视黄酸信号通路可以通过直接调控细胞分化和间接影响形态发生,促进鱼类的齿列发育。 2.3.4 Wnt 从牙胚起始到形态发生阶段,经典Wnt/β-catenin信号通路通过调控上皮-间充质相互作用,主导着器官发育进程,同时该信号通路的生物学效应具有剂量依赖性。SHIM等[60]通过斑马鱼模型揭示,Wnt 信号强度的精准调控会直接决定牙齿的形态发生能否成功,当Wnt活性维持在特定阈值范围时方可确保牙源性上皮与间充质细胞的正常作用。然而,这种调控机制在不同物种间存在差异,例如,斑马鱼的咽齿发育对Wnt拮抗表现出耐受性[61],而丽鱼科口腔牙齿的密度则与Wnt信号强度呈正相关[62]。因此,尽管Wnt通路在脊椎动物牙齿发育中具有演化保守性,但其具体调控网络可有物种特异性,与不同的生存需求相适应。 2.3.5 骨形态发生蛋白 骨形态发生蛋白6基因内含子区域的增强子存在多态性变异,这些变异是驱动鱼类牙齿数量演化的关键遗传基础。STEPANIAK等[63]的研究发现,将增强子存在差异的高齿型淡水帕克斯顿底栖种群(咽齿数量多,成年个体约40枚,显著多于海洋种群;齿列密集,利于处理硬质食物如甲壳类、螺类)与低齿型的海洋种群(咽齿数量少,成年个体约20枚,齿列稀疏,适应摄食浮游生物或软体猎物)杂交后,骨形态发生蛋白6基因在其F1代的牙齿发育组织中表达位置不同,这表明增强子的多态性很可能调控了该基因在牙齿发育过程中的空间表达模式。CLEVES等[64]发现,携带淡水种群特异性等位基因的个体,由于增强子的作用,它们的骨形态发生蛋白6表达量显著低于携带海洋等位基因的个体,但是这种表达的下降在演化上却与淡水种群牙齿数量的增加直接相关,具体表现为在牙齿发育的后期阶段牙齿数量出现明显增长,这一证据表明,增强子的多态性通过精确调控骨形态发生蛋白6的表达水进而影响了牙齿的发育数量。另外,JACKMAN等[47]通过骨形态发生蛋白抑制剂(如dorsomorphin)抑制斑马鱼骨形态发生蛋白信号,导致斑马鱼出现多尖齿和额外齿,增强了成纤维细胞生长因子过表达效果,表明骨形态发生蛋白和成纤维细胞生长因子具有协同调控作用。 2.3.6 Sox2 Sox2(SRY-Box Transcription Factor 2,Sox2)是在干细胞中表达的一种重要转录因子,参与干细胞的分化,也参与牙齿的发育。在鲨鱼牙齿的发育和再生区域特异性地高表达Sox2,这些区域的Sox2+干细胞能够周期性地激活上述保守的基因调控网络[65],激活后,干细胞经历增殖、分化等一系列步骤,最终在特定的位置生成新的牙齿,从而实现牙齿的终身持续更换。 2.3.7 外胚叶发育素A 外胚叶发育素A是一个编码肿瘤坏死因子家族配体的基因,是调控脊椎动物外胚层附属器官如牙齿、毛发、鳞片、汗腺发育的关键信号分子。外胚叶发育素A-A1亚型通过外胚叶发育素A受体/外胚叶发育素ARADD激活核因子κB通路调控牙齿发育,而外胚叶发育素A-A2的作用较弱。MIKKOLA[66]发现外胚叶发育素A和外胚叶发育素AR的突变会导致斑马鱼的咽齿缺失和鳞片缺陷。正常情况下,斑马鱼的咽齿仅存在于下咽部,如果通过转基因技术过表达外胚叶发育素A,斑马鱼的上咽部重新形成了牙齿,表明仅需恢复外胚叶发育素A信号即可逆转此区域的牙齿丧失;而将斑马鱼的外胚叶发育素A基因敲除后,它的下咽部牙齿也无法发育,证明外胚叶发育素A是牙齿形成的必要条件[67]。 2.3.8 垂体同源框转录因子2 垂体同源框转录因子2 基因作为硬骨鱼最早的牙上皮标记,它在斑马鱼中的表达不受成纤维细胞生长因子/音猬因子信号阻断影响,但垂体同源框转录因子2信号缺失仍导致牙胚形态异常,提示它可能位于调控网络的上游位点[68]。 不同鱼类牙齿发育演化相关的基因及其作用,见表4。 2.4 争议与未来方向 鱼类牙齿演化研究虽取得一些进展,仍存在关键争议亟待解决:①牙齿起源的同源性尚未定论:牙形动物的咽部牙形刺虽与现代牙齿存在显微结构相似性,但二者可能属趋同演化而非同源性状,削弱了“由内向外”假说的化石依据;而“由外向内”修正假说强调外胚层鳞片与内胚层间充质的协同成牙作用,其核心证据-胚层互作机制多源于蝾螈等非鱼类模型,在鱼类中缺乏直接实验支持。②化石记录的局限性制约了演化模型的完善:早期齿状体的解剖位置(体表抑或咽腔)难以通过碎片化化石确认,并且牙齿替换机制的演化节点存在分歧(如安德雷奥莱鱼属化石推翻早期硬骨鱼“牙齿不可替换”的传统认知),说明关键过渡物种的缺失。③分子调控机制的物种特异性引发新思考,如视黄酸信号在斑马鱼咽齿发育中不可或缺,却在青鳉和墨西哥刺鱼中不敏感,提示相同器官的发育策略可能因自然选择发生分化。④牙齿形态演化的适应性意义尚未系统阐明。关于鱼类牙齿形态和结构的问题,四类牙本质(正齿型/骨齿型/假骨齿型/血管齿质)的演化关系不明,类釉质矿化从软骨鱼类的“囊泡主导”向硬骨鱼类的“胶原模板”的过渡是否代表着普适规律,亦有待考证。 解决上述争议需多学科交叉创新。在技术层面上,同步辐射显微CT与三维重建技术可解析关键过渡物种如盾皮鱼类的齿状体微结构;单细胞时空转录组技术能精确定位胚层协同的信号中心(如咽囊-裂隙接触区)。斑马鱼单碱基突变细胞谱系追踪系统(SMAL)可以通过1 kb DNA条形码记录胚胎发育中的连续突变,实现单细胞分辨率的谱系重建[74]。在基因调控层面,跨物种单细胞转录组分析揭示了咽齿发育的演化可塑性。青鳉与斑马鱼咽齿高表达SCPP家族基因(SCPP5、SCPP9),其拷贝数扩张与鲤形目齿型多样性显著相关;而骨形态发生蛋白信号在斑马鱼中抑制咽齿数量,在青鳉中却无此功能,提示通路功能的物种分化[75]。YANG等[76]进一步在Characiformes鱼类中发现,釉质基质基因SCPP7在肉食性种类中特异性高表达,与穿刺齿型的适应性演化直接关联。此外,空间多组学技术拓展了形态演化的机制认知,WIBISANA等[77]结合单细胞染色质可及性测序(scATAC-seq)与空间转录组发现,斑石鲷喙状齿愈合依赖cyp27a1基因介导的钙沉积及Wnt/骨形态发生蛋白空间梯度调控。 在理论框架层面,应扩大比较发育学研究范畴,纳入基部硬骨鱼(雀鳝、弓鳍鱼)及无颌类(七鳃鳗),系统对比音猬因子/视黄酸/Wnt等通路的调控网络,结合基因编辑与类器官模型,模拟鳞片-牙齿转化的演化场景(如过表达远端缺失同源框基因基因验证同源调控"

| [1] ANDREEV PS, SANSOM IJ, LI Q, et al. The oldest gnathostome teeth. Nature. 2022;609(7929):964-968. [2] KOUSSOULAKOU DS, MARGARITIS LH, KOUSSOULAKOS SL. A curriculum vitae of teeth: evolution, generation, regeneration. Int J Biol Sci. 2009;5(3):226-243. [3] DONOGHUE PJC. Evolution of development of the vertebrate dermal and oral skeletons: unraveling concepts, regulatory theories,and homologies. Paleobiology. 2016;28(4):474-507. [4] HARIDY Y, GEE BM, WITZMANN F, et al. Retention of fish-like odontode overgrowth in Permian tetrapod dentition supports outside-in theory of tooth origins. Biol Lett. 2019;15(9):20190514. [5] 盖志琨,朱敏.无颌类演化史与中国化石记录[M].上海:上海科学技术出版社,2017. [6] MURDOCK DJ, DONG XP, REPETSKI JE, et al. The origin of conodonts and of vertebrate mineralized skeletons. Nature. 2013;502(7472):546-549. [7] CHEN D, BLOM H, SANCHEZ S, et al. The developmental relationship between teeth and dermal odontodes in the most primitive bony fish Lophosteus. Elife. 2020;9:e60985. [8] COOPER RL, NICKLIN EF, RASCH LJ, et al. Teeth outside the mouth: The evolution and development of shark denticles. Evol Dev. 2023; 25(1):54-72. [9] DUMACK K, GÖRZEN D, GONZÁLEZ-MIGUÉNS R, et al. Molecular investigation of Phryganella acropodia Hertwig et Lesser, 1874 (Arcellinida, Amoebozoa). Eur J Protistol. 2020;75:125707. [10] ROMER C. Treatment of abortus fever with sulphonamides and blood transfusion. Br Med J. 1949;1(4614):1035. [11] JOLLIE WP. Changes in the fine structure of the parietal yolk sac of the rat placenta with increasing gestational age. Am J Anat. 1968;122(3):513-531. [12] AHLBERG PE, SMITH MM, JOHANSON Z. Developmental plasticity and disparity in early dipnoan (lungfish) dentitions. Evol Dev. 2006;8(4): 331-349. [13] HUYSSEUNE A, SIRE JY, WITTEN PE. Evolutionary and developmental origins of the vertebrate dentition. J Anat. 2009;214(4):465-476. [14] HUYSSEUNE A, CERNY R, WITTEN PE. The conundrum of pharyngeal teeth origin: the role of germ layers, pouches, and gill slits. Biol Rev Camb Philos Soc. 2022;97(1):414-447. [15] DONOGHUE PC, RÜCKLIN M. The ins and outs of the evolutionary origin of teeth. Evol Dev. 2016; 18(1):19-30. [16] FRASER GJ, CERNY R, SOUKUP V, et al. The odontode explosion: the origin of tooth‐like structures in vertebrates. Bioessays. 2010;32(9): 808-817. [17] FRASER GJ, SMITH MM. Evolution of developmental pattern for vertebrate dentitions: an oro‐pharyngeal specific mechanism. J Exp Zool B Mol Dev Evol. 2011;316B(2):99-112. [18] FRASER GJ, HULSEY CD, BLOOMQUIST RF, et al. An ancient gene network is co-opted for teeth on old and new jaws. PLoS Biol. 2009;7(2):e31. [19] DEBIAIS-THIBAUD M, OULION S, BOURRAT F, et al. The homology of odontodes in gnathostomes: insights from Dlx gene expression in the dogfish, Scyliorhinus canicula. BMC Evol Biol. 2011;11:307. [20] GILLIS JA, ALSEMA EC, CRISWELL KE. Trunk neural crest origin of dermal denticles in a cartilaginous fish. Proc Natl Acad Sci U S A. 2017;114(50): 13200-13205. [21] GRAVESON AC, SMITH MM, HALL BK. Neural crest potential for tooth development in a urodele amphibian: developmental and evolutionary significance. Dev Biol. 1997;188:34-42. [22] NGOEPE N. Evolutionary flexibility to gain or lose tooth complexity sparks fish diversification. Nature. 2025;639(8056):869-870. [23] HUYSSEUNE A, SIRE JY. Evolution of patterns and processes in teeth and tooth-related tissues in non-mammalian vertebrates. Eur J Oral Sci. 1998;106 Suppl 1:437-481. [24] ENAX J, FABRITIUS HO, RACK A, et al. Characterization of crocodile teeth: correlation of composition, microstructure, and hardness. J Struct Biol. 2013;184(2):155-163. [25] VELASCO-HOGAN A, HUANG W, SERRANO C, et al. Tooth structure, mechanical properties, and diet specialization of Piranha and Pacu (Serrasalmidae): A comparative study. Acta Biomater. 2021;134:531-545. [26] LI T, YONGFENG L, RUIQI L, et al. Development and structural characteristics of pseudoosteodentine in the Pacific cutlassfish, Trichiurus lepturus. Tissue Cell. 2022;77:101847. [27] HEROLD RC. The fine structure of vasodentine in the teeth of the white hake, Urophycis tenuis (Pisces, Gadidae). Arch Oral Biol. 1970;15(4):311-322. [28] JAMBURA PL, TÜRTSCHER J, KINDLIMANN R, et al. Evolutionary trajectories of tooth histology patterns in modern sharks (Chondrichthyes, Elasmobranchii). J Anat. 2020;236(5):753-771. [29] WAKITA M. Current studies on tooth enamel development in lower vertebrates. Kaibogaku Zasshi. 1993;68(4):399-409. [30] SIRE JY, DONOGHUE PC, VICKARYOUS MK. Origin and evolution of the integumentary skeleton in non-tetrapod vertebrates. J Anat. 2009;214(4): 409-440. [31] REIF WE. Evolution of dermal skeleton and dentition in vertebrates//HECHT MK, WALLACE B, PRANCE GT. Evolutionary Biology. Springer, Boston, MA. 1982. [32] GILLIS JA, DONOGHUE PC. The homology and phylogeny of chondrichthyan tooth enameloid. J Morphol. 2007;268(1):33-49. [33] QU Q, HAITINA T, ZHU M, et al. New genomic and fossil data illuminate the origin of enamel. Nature. 2015;526(7571):108-111. [34] SASAGAWA I. Mechanisms of mineralization in the enameloid of elasmobranchs and teleosts. Connect Tissue Res. 1998;39(1-3):207-214; discussion 221-225. [35] 李谨,王涛,王娟,等.垂体同源框转录因子2基因在斑马鱼牙齿发育过程中早期表达的研究[J].口腔医学,2010,30(3):129-132. [36] RASCH LJ, MARTIN KJ, COOPER RL, et al. An ancient dental gene set governs development and continuous regeneration of teeth in sharks. Dev Biol. 2016;415(2):347-370. [37] MA Y, XIAO Y, XIAO Z, et al. Morphological characteristics of beak-like tooth in spotted knifejaw (Oplegnathus punctatus) and mechanisms of dental development regulation by the Wnt, BMP, FGF and Shh signalling pathways. Int J Biol Macromol. 2023;250:126188. [38] DEARDEN RP, GILES S. Diverse stem-chondrichthyan oral structures and evidence for an independently acquired acanthodid dentition. R Soc Open Sci. 2021;8(11):210822. [39] HUYSSEUNE A, WITTEN PE. Continuous tooth replacement: what can teleost fish teach us? Biol Rev Camb Philos Soc. 2024;99(3):797-819. [40] VAŠKANINOVÁ V, CHEN D, TAFFOREAU P, et al. Marginal dentition and multiple dermal jawbones as the ancestral condition of jawed vertebrates. Science. 2020;369(6500):211-216. [41] CHEN D, BLOM H, SANCHEZ S, et al. The stem osteichthyan Andreolepis and the origin of tooth replacement. Nature. 2016; 539(7628):237-241. [42] BOTELLA H, BLOM H, DORKA M, et al. Jaws and teeth of the earliest bony fishes. Nature. 2007;448(7153):583-586. [43] JOHANSON Z. Paleobiology: A Tooth for a Tooth. Curr Biol. 2017;27(3):R117-R119. [44] BEMIS WE, GIULIANO A, MCGUIRE B. Structure, attachment, replacement and growth of teeth in bluefish, Pomatomus saltatrix (Linnaeus, 1776), a teleost with deeply socketed teeth. Zoology (Jena). 2005;108(4):317-327. [45] JERNVALL J, KETTUNEN P, KARAVANOVA I, et al. Evidence for the role of the enamel knot as a control center in mammalian tooth cusp formation: non-dividing cells express growth stimulating Fgf-4 gene. Int J Dev Biol. 1994; 38(3):463-469. [46] KETTUNEN P, KARAVANOVA I, THESLEFF I. Responsiveness of developing dental tissues to fibroblast growth factors: expression of splicing alternatives of FgfR1, -2, -3, and of FgfR4; and stimulation of cell proliferation by Fgf-2, -4, -8, and -9. Dev Genet. 1998;22(4):374-385. [47] JACKMAN WR, DAVIES SH, LYONS DB, et al. Manipulation of Fgf and Bmp signaling in teleost fishes suggests potential pathways for the evolutionary origin of multicuspid teeth. Evol Dev. 2013;15(2):107-118. [48] JACKMAN WR, DRAPER BW, STOCK DW. Fgf signaling is required for zebrafish tooth development. Dev Biol. 2004;274(1):139-157. [49] SMITH MM, FRASER GJ, CHAPLIN N, et al. Reiterative pattern of sonic hedgehog expression in the catshark dentition reveals a phylogenetic template for jawed vertebrates. Proc Biol Sci. 2009;276(1660):1225-1233. [50] MAISEY JG, TURNER S, NAYLOR GJ, et al. Dental patterning in the earliest sharks: Implications for tooth evolution. J Morphol. 2014;275(5):586-596. [51] JACKMAN WR, YOO JJ, STOCK DW. Hedgehog signaling is required at multiple stages of zebrafish tooth development. BMC Dev Biol. 2010;10:119. [52] HARDCASTLE Z, MO R, HUI CC, et al. The Shh signalling pathway in tooth development: defects in Gli2 and Gli3 mutants. Development. 1998;125(15):2803-2811. [53] GIBERT Y, SAMARUT E, PASCO-VIEL E, et al. Altered retinoic acid signalling underpins dentition evolution. Proc Biol Sci. 2015;282(1802): 20142764. [54] GIBERT Y, BERNARD L, DEBIAIS-THIBAUD M, et al. Formation of oral and pharyngeal dentition in teleosts depends on differential recruitment of retinoic acid signaling. FASEB J. 2010;24(9):3298-3309. [55] KOPINKE D, SASINE J, SWIFT J, et al. Retinoic acid is required for endodermal pouch morphogenesis and not for pharyngeal endoderm specification. Dev Dyn. 2006;235(10):2695-2709. [56] EL SHAHAWY M, REIBRING CG, NEBEN CL, et al. Cell fate specification in the lingual epithelium is controlled by antagonistic activities of Sonic hedgehog and retinoic acid. PLoS Genet. 2017; 13(7):e1006914. [57] LARIONOVA D, HUYSSEUNE A. Differential retinoic acid sensitivity of oral and pharyngeal teeth in medaka (Oryzias latipes) supports the importance of pouch-cleft contacts in pharyngeal tooth initiation. Dev Dyn. 2024;253(12):1094-1105. [58] SERITRAKUL P, SAMARUT E, LAMA TT, et al. Retinoic acid expands the evolutionarily reduced dentition of zebrafish. FASEB J. 2012;26(12):5014-5024. [59] DRAUT H, LIEBENSTEIN T, BEGEMANN G. New Insights into the Control of Cell Fate Choices and Differentiation by Retinoic Acid in Cranial, Axial and Caudal Structures. Biomolecules. 2019; 9(12):860. [60] SHIM JS, KIM B, PARK HC, et al. Temporal Control of WNT Activity Regulates Tooth Number in Fish. J Dent Res. 2019;98(3): 339-346. [61] HUYSSEUNE A, SOENENS M, ELDERWEIRDT F. Wnt signaling during tooth replacement in zebrafish (Danio rerio): pitfalls and perspectives. Front Physiol. 2014;5:386. [62] BLOOMQUIST RF, PARNELL NF, PHILLIPS KA, et al. Coevolutionary patterning of teeth and taste buds. Proc Natl Acad Sci U S A. 2015;112(44):E5954-5962. [63] STEPANIAK MD, SQUARE TA, MILLER CT. Evolved Bmp6 enhancer alleles drive spatial shifts in gene expression during tooth development in sticklebacks. Genetics. 2021;219(4):iyab151. [64] CLEVES PA, ELLIS NA, JIMENEZ MT, et al. Evolved tooth gain in sticklebacks is associated with a cis-regulatory allele of Bmp6. Proc Natl Acad Sci U S A. 2014;111(38):13912-13917. [65] MARTIN KJ, RASCH LJ, COOPER RL, et al. Sox2+ progenitors in sharks link taste development with the evolution of regenerative teeth from denticles. Proc Natl Acad Sci U S A. 2016;113(51):14769-14774. [66] MIKKOLA ML. TNF superfamily in skin appendage development. Cytokine Growth Factor Rev. 2008;19(3-4):219-230. [67] AIGLER SR, JANDZIK D, HATTA K, et al. Selection and constraint underlie irreversibility of tooth loss in cypriniform fishes. Proc Natl Acad Sci U S A. 2014;111(21):7707-7712. [68] FRASER GJ, BLOOMQUIST RF, STREELMAN JT. A periodic pattern generator for dental diversity. BMC Biol. 2008;6:32. [69] FRASER GJ, GRAHAM A, SMITH MM. Conserved deployment of genes during odontogenesis across osteichthyans. Proc R Soc Lond B Biol Sci. 2024;271:2311-2317. [70] DEBIAIS-THIBAUD M, BORDAY-BIRRAUX V, GERMON I, et al. Development of oral and pharyngeal teeth in the mEdaka (Oryzias latipes): comparison of morphology and expression of eve1 gene. J Exp Zool B Mol Dev Evol. 2007;308(6):693-708. [71] STOCK DW, JACKMAN WR, TRAPANI J. Developmental genetic mechanisms of evolutionary tooth loss in cypriniform fishes. Development. 2006;133:3127-3137. [72] FRASER GJ, BLOOMQUIST RF, STREELMAN JT. A periodic pattern generator for dental diversity. BMC Biol. 2008;6:32. [73] SMITH MM, OKABE M, JOSS J. Spatial and temporal pattern for the dentition in the Australian lungfish revealed with sonic hedgehog expression profile. Proc Biol Sci. 2009;276(1657):623-631. [74] LIU Z, ZENG H, XIANG H, et al. Achieving single-cell-resolution lineage tracing in zebrafish by continuous barcoding mutations during embryogenesis. J Genet Genomics. 2024;51(9): 947-956. [75] 吴亚明.鱼类咽齿发育关键调控基因的筛选及其功能研究[D].武汉:华中农业大学,2023. [76] YANG X, SONG Y, ZHANG R, et al. Unravelling the genomic features, phylogeny and genetic basis of tooth ontogenesis in Characiformes through analysis of four genomes. DNA Res. 2023;30(5):dsad022. [77] WIBISANA JN, SALLAN RA, OTA T, et al. Modifiable Clinical Dental Impression Methods to Obtain Whole-Mouth and Detailed Dental Traits From Vertebrates. J Morphol. 2025;286(1):e70017. |
| [1] | Li Ruiqiang, Yin Chen, Ma Yan. Effect of carbamide peroxide and hydrogen peroxide bleaching agents on laser-induced fluorescence in dentin Raman spectroscopy [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(2): 296-302. |
| [2] | Li Qian, Qumanguli · Abudukelimu, Shao Ziyu, Hu Yang. Hard template construction of nano-beta-tricalcium phosphate and nano-hydroxyapatite root canal sealing materials [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(14): 3597-3608. |
| [3] | Li Tingyue, Guo Qian, He Wenxi, Wu Jiayuan. Long noncoding RNA TP53TG1 promotes odontogenic and osteogenic differentiation of stem cells from the apical papilla [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(36): 7776-7782. |
| [4] | Chen Jinjie, Li Geng, Jiang Yefan. Role of Siblings protein family in cardiovascular diseases [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(24): 5171-5178. |
| [5] | Yang Dujuan, Cheng Mengke, Liu Jia. Osteogenic/odontogenic differentiation ability of human dental pulp stem cells under photocrosslinked composite hydrogel scaffold [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(19): 4022-4028. |
| [6] | Dai Jieting, Ren Bihui, Xu Yehao, Guo Shuigen, Wei Hongwu. Reactive soft tissue preservation combined with demineralized dentin matrix for extraction site preservation [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(17): 3557-3565. |
| [7] | Israrguli · Maimeti, Jia Sen, Liu Jia. Bone morphogenetic protein 2-loaded hydrogel induces osteogenic differentiation of dental pulp stem cells [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(16): 3301-3310. |
| [8] | Cheng Ruiqing, Sun Honglei, Geng Shuangshuang, Wang Chao, Li Junke, Chen Yanfang. Effects of erythropoietin on restorative dentin formation and expression of bone morphogenetic protein 2 after pulp injury [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(11): 2231-2242. |
| [9] | Cheng Mengke, Yang Dujuan, Liu Jia. Preparation and performance of gelatin modified by methacrylic anhydride/treated dentin matrix bioactive scaffolds [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(22): 3555-3560. |
| [10] | Liu Huan, Li Han, Ma Yunhao, Zhong Weijian, Ma Guowu. Osteogenic capacity of partially demineralized dentin particles in the maxillary sinus lift [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 354-359. |
| [11] | Wang Ziyang, Zuo Enjun. Application of biological materials in vital pulp therapy [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 427-433. |
| [12] | Yu Lanning, Wang Qian, Jin Youshi, Fei Xiaowen, Wang Qingshan. Sealing effect of nano hydroxyapatite on dentinal tubules [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(25): 3964-3970. |
| [13] | Liang Hanying, Ma Yunhao, Li Han, Li Dongyang, Zhong Weijian, Ma Guowu. Osteogenic effects of partially demineralized autogenous dentin particles in implant site preservation [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(16): 2488-2492. |
| [14] | Sun Honglei, Qi Fengna, Cheng Ruiqing. ICON penetrating resin for treatment of dental fluorosis: 1-year follow-up [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(34): 5419-5424. |
| [15] | Chen Jiawen, Sun Tianyu, Liu Peng, Wu Buling, Wu Jingyi. Proteoglycans in tooth development and its role in regulating stem cell homeostasis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(25): 4064-4069. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

