Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (26): 6859-6867.doi: 10.12307/2026.382
Previous Articles Next Articles
Biocompatibility and preclinical experiments of a Chinese-made 3D-printed minimally invasive tungsten alloy needle electrodes
Li Yujin1, Ni Guansen2, Mao Weiqing3, Tang Jiayu1, Li Xueqing1
- 1Department of General Surgery, 3Department of Nursing, Shanghai Fifth People’s Hospital, Fudan University, Shanghai 200240, China; 2Department of Otolaryngology, Shanghai Fourth People’s Hospital, Shanghai 200434, China
-
Accepted:2025-09-25Online:2026-09-18Published:2026-03-12 -
Contact:Li Xueqing, Chief physician, Department of General Surgery, Shanghai Fifth People’s Hospital, Fudan University, Shanghai 200240, China -
About author:Li Yujin, MS, Attending physician, Department of General Surgery, Shanghai Fifth People’s Hospital, Fudan University, Shanghai 200240, China -
Supported by:Natural Science Research Fund of Minhang District, Shanghai, No. 2022MHZ094 (to LYJ)
CLC Number:
Cite this article
Li Yujin, Ni Guansen, Mao Weiqing, Tang Jiayu, Li Xueqing. Biocompatibility and preclinical experiments of a Chinese-made 3D-printed minimally invasive tungsten alloy needle electrodes[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6859-6867.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
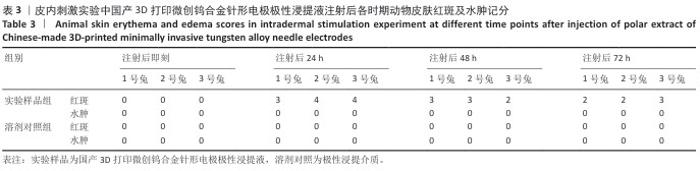
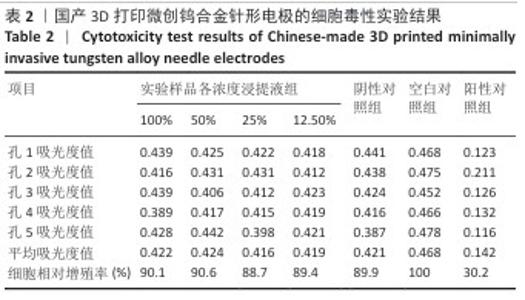
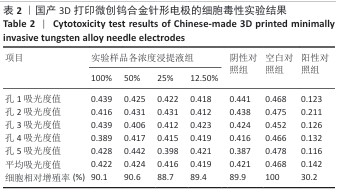
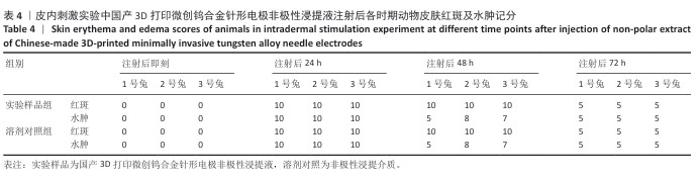
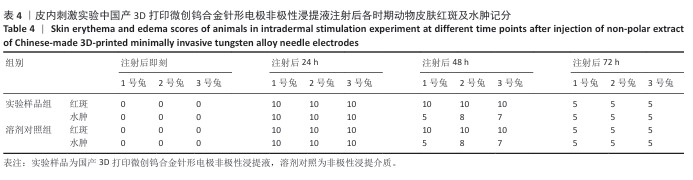
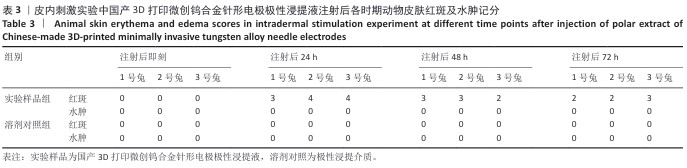
2.1.2 皮内刺激实验结果 国产3D打印微创钨合金针形电极极性浸提液注射后各时期动物皮肤红斑及水肿记分,见表3。国产3D打印微创钨合金针形电极非极性浸提液注射后各时期动物皮肤红斑及水肿记分,见表4。实验结果显示,实验样品极性浸提液组总平均记分为0.58[(26+0)/(15×3)=0.58],极性浸提介质组总平均记分为0[(0+0)/(15×3)=0],国产实验样品极性浸提液组最终记分为0.58-0=0.58。实验样品非极性浸提液组总平均记分为3.11,非极性浸提介质组总平均记分为3.11,实验样品非极性浸提液组最终记分为3.11-3.11=0。实验样品极性浸提液组和实验样品非极性浸提液组最终记分均< 1.0,符合实验要求,表明国产3D打印钨针电极浸提液无皮下刺激反应。"
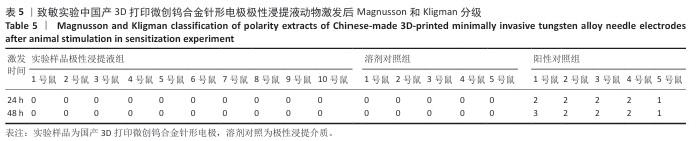
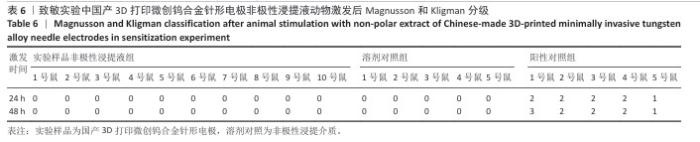
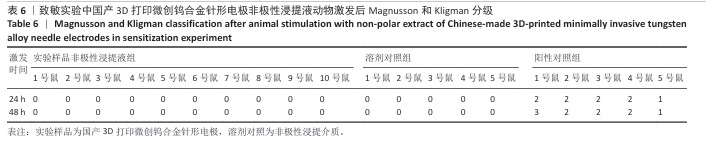
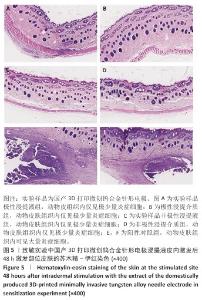
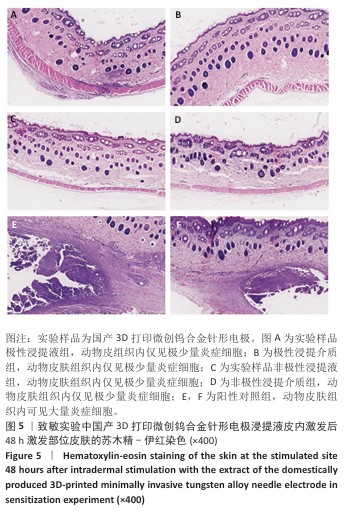
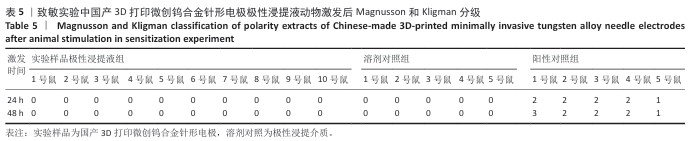
2.1.3 致敏实验结果 皮内注射14 d后,除去敷贴片后在自然光或全光谱光线下观察各组动物激发部位皮肤红斑情况并分级,实验样品极性浸提液组、实验样品非极性浸提液组、溶剂对照组、阳性对照组动物激发部位皮肤情况,见图4。各组动物激发部位皮肤Magnusson和Kligman分级,见表5,6。 结果显示,实验样品极性浸提液、非极性浸提液组动物皮肤未见红斑和水肿,动物皮肤红斑和水肿等级< 1;极性浸提介组与非极性浸提介组对照动物皮肤未见红斑和水肿,动物皮肤红斑和水肿等级< 1;阳性对照组动物皮肤可观察到明显的红斑,部分动物皮肤可观察到焦痂,该组动物皮肤致敏反应发生率为100%。 各组动物激发部位皮肤组织形态观察,见图5。苏木精-伊红染色显示,实验样品极性浸提液、非极性浸提液组激发部位皮肤组织内仅见极少量炎症细胞,阳性对照组皮肤组织内可见大量炎症细胞,表明国产3D打印微创钨合金针形电极浸提液无皮肤致敏反应。 "
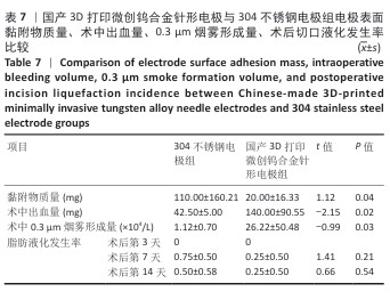
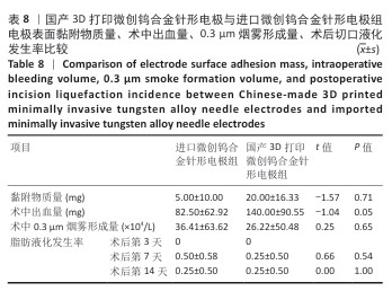
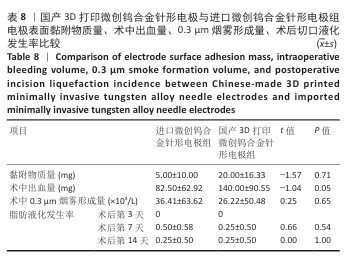
2.2 国产3D打印微创钨合金针形电极的临床前动物实验结果 2.2.1 实验动物数量分析 36只SD大鼠全部进入结果分析。 2.2.2 电极表面黏附物质量 国产3D打印微创钨合金针形电极组电极表面黏附物质量低于304不锈钢电极组(P < 0.05),与进口微创钨合金针形电极组比较差异无显著性意义(P > 0.05),见表7,8。结果表明,与304不锈钢电极相比,国产3D打印微创钨合金针形电极在减少术中组织黏附方面表现更优,可能有助于提高切割效率、降低手术风险,并且国产3D打印微创钨合金针形电极的低黏附性能已接近进口微创钨合金针形电极,但仍有优化空间。304不锈钢电极的表面化学稳定性较差,在高温状态下容易出现氧化,随着机械摩擦使表面变得更加粗糙,可能导致组织更容易附着,从而形成较大的黏附物质量。钨针电极(国产与进口)表面化学稳定性较好,具有更低的组织黏附性,减少了手术过程中组织碎屑的积累,从而降低了电极黏附物质量;这种低黏附性也可能与钨合金的高熔点、耐腐蚀性有关。另外,进口钨针电极经过精细加工和表面处理,可能进一步优化了它的抗黏附能力。国产3D打印微创钨合金针形电极目前处于初步研发阶段,表面精度可能略逊于进口电极,导致黏附物质量略高。未来可通过优化3D打印工艺、改进表面抛光和涂层技术进一步降低国产3D打印微创钨合金针形电极的黏附性,使它的性能接近或超越进口产品。"
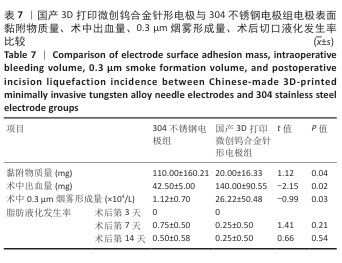
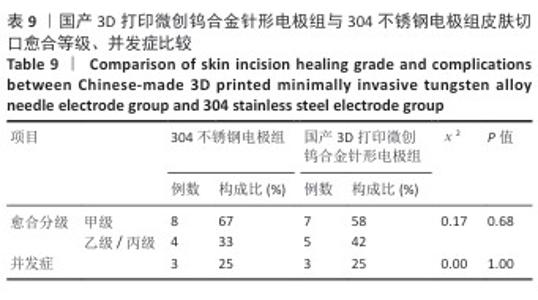
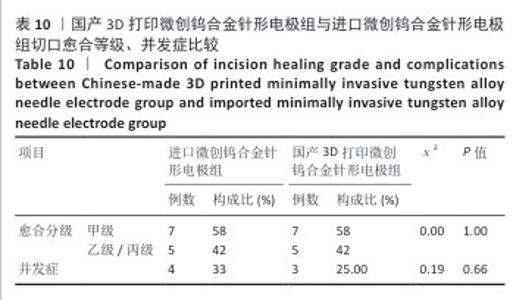
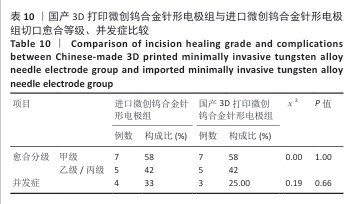
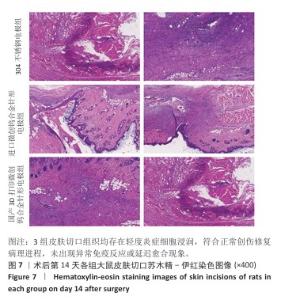
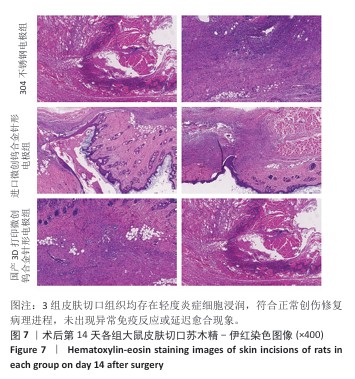
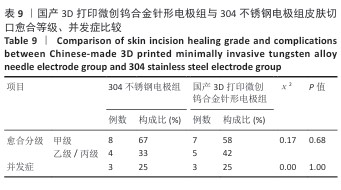
2.2.3 烟雾颗粒形成量 3组电极在术中产生的烟雾颗粒以0.3 μm烟雾颗粒为主。国产3D打印微创钨合金针形电极组术中0.3 μm烟雾形成量多于304不锈钢电极组(P < 0.05),与进口微创钨合金针形电极组比较差异无显著性意义(P > 0.05),见表7,8。结果表明,国产3D打印微创钨合金针形电极相较于304不锈钢电极会产生较多的细小烟雾颗粒,这与国产3D打印微创钨合金针形电极的热传导性能、表面化学特性等有关。304不锈钢电极由于热传导性较低导致局部温度容易过高,并且它的熔点相对较低,在高温切割过程中容易导致组织炭化,组织黏附在电极表面后进一步阻碍热量的传导,使得切割效率降低,产生的烟雾颗粒相对较少。另一方面,钨针电极具有高熔点(3 410 ℃)和低热膨胀系数,能够在高温下保持稳定的物理性能,它的表面相对光滑且化学惰性强,组织在切割过程中组织更容易被汽化,从而产生较多的烟雾颗粒;此外,钨针电极的高切割效率也使得组织在高温下汽化更为充分,进一步增加了烟雾颗粒的形成量,从侧面证明国产3D打印微创钨合金针形电极在减少组织黏附方面有优势,但这可能导致更多的烟雾形成。 2.2.4 术中出血量 国产3D打印微创钨合金针形电极组术中出血量多于304不锈钢电极组(P < 0.05),与进口微创钨合金针形电极组比较差异无显著性意义(P ≥ 0.05),见表7,8。304不锈钢电极的热传导性和电外科特性更适合组织凝固,而钨针电极主要通过高温汽化组织来实现切割,这种机制虽然能够实现更精细的切割效果,但对血管的热凝固作用较弱,所以导致术中止血效果较差。另外,进口微创钨合金针形电极组的术中出血量标准差较低,可能说明产品一致性较高。进口微创钨合金针形电极经过精细加工,可能在高频电外科手术中的热效应更稳定,从而减少术中出血量。国产3D打印微创钨合金针形电极组术中出血量的标准差较大,表明出血量波动较大,可能由于国产3D打印微创钨合金针形电极处于初步研发阶段,尚未经过精细化设计,从而导致术中切割效率和止血能力略逊于进口微创钨合金针形电极,但仍有优化空间。 2.2.5 脂肪液化评价 术后第3天3组均未发生脂肪液化情况;术后第7,14天,3组均发生脂肪液化情况,3组间脂肪液化发生率比较差异均无显著性意义(P > 0.05),见表7,8。 2.2.6 术后切口愈合评价 术后不同时间点3组大鼠皮肤切口愈合大体观,见图6所示。术后第3天,3组皮肤切口未完全愈合,创面边缘轻度红肿,整体愈合进程处于早期阶段;术后第7天,3组皮肤切口仍未完全闭合,局部积液,创面周围炎症反应有所缓解,愈合进程仍在继续;术后第14天,3组皮肤切口愈合良好,创面基本闭合,局部无明显积液,愈合区域未见明显感染或异常情况,整体愈合效果良好,其中进口微创钨合金针形电极组和国产3D打印微创钨合金针形电极组皮肤表面较为平整。国产3D打印微创钨合金针形电极组术后切口甲级愈合分级情况与304不锈钢电极组、进口微创钨合金针形电极组相比差异均无显著性意义(P > 0.05),见表9,10。 术后第14天3组大鼠皮肤切口苏木精-伊红染色结果,见图7。3组皮肤切口组织均存在轻度炎症细胞浸润,符合正常创伤修复病理进程,未出现异常免疫反应或延迟愈合现象,进一步佐证3类电极的生物安全性。 2.2.7 术后不良反应评价 3组术后不良反应发生率比较差异无显著性意义(P > 0.05),见表9,10。 "
| [1] HAJARI VR, BENKE AP, GOEL EO. Advances in High-Frequency Surgical Device Design and Safety. Darpan International Research Analysis. 2024;12(3):269-282. [2] VILOS GA, RAJAKUMAR C. Electrosurgical generators and monopolar and bipolar electrosurgery. J Minim Invasive Gynecol. 2013;20(3):279-287. [3] BAI Y, YANG F, LIU C, et al. Expert consensus on the clinical application of high‐frequency electrosurgery in digestive endoscopy (2020, Shanghai). J Dig Dis. 2022;23(1):2-12. [4] HAIPO C, JIAPING H, CHENGLI S, et al. Influence of different sizes on the temperature field of bipolar high-frequency electric knife. J Funct Mater Cailiao. 2018;49(9):9122-9126. [5] DUNDEKOVÁ S, ŠKORÍK V, HADZIMA B, et al. The influence of the temperature on the corrosion resistance of aisi 316l stainless steel. ANNALS of Faculty Engineering Hunedoara-International Journal of Engineering. 2015;13(1):203-206. [6] BARUA A, PRADHAN S, PRIYADARSHINI M, et al. Recent advancement in tungsten heavy alloy processing for different industrial applications. J Mater Eng Perform. 2024;34:8232-8252. [7] RYU H, SEO MH, ROGERS JA. Bioresorbable metals for biomedical applications: from mechanical components to electronic devices. Adv Healthc Mater. 2021; 10(17):2002236. [8] 付和林,郝汝飞,郑靖,等.高频电刀电凝模式下304不锈钢和钨两种电极表面的组织粘附行为[J].中国表面工程,2016,29(3):96-102. [9] WANG J, LIU Z, WU Y, et al. Cutting Performance and Tool Wear of AlCrN-and TiAlN-Coated Carbide Tools during Milling of Tantalum–Tungsten Alloy. Machines. 2024;12(3):170. [10] 李顺,张周然,郑昆鹏,等.TiVTa 系低活化多主元合金的微观结构及相稳定性[J].材料工程,2023,51(8):181-189. [11] WU M, DIAO G, XU Z, et al. Investigation of mechanical properties and wear resistance of A2/B2 type medium-entropy alloy matrix reinforced with tungsten particles by in-situ reaction. Metals. 2023;13(4):656. [12] 王利剑,陈文革.钨铜合金表面粗糙度对抗电弧烧蚀性能的影响[J].粉末冶金材料科学与工程,2016,21(5):802-808. [13] KURT MŞ, ARSLAN ME, YAZICI A, et al. Tribological, biocompatibility, and antibiofilm properties of tungsten–germanium coating using magnetron sputtering. J Mater Sci Mater Med. 2021;32(1):6. [14] AHERWAR A, BAHRAMINASAB M. Biocompatibility evaluation and corrosion resistance of tungsten added Co-30Cr-4Mo-1Ni alloy. Biomed Mater Eng. 2017; 28(6):687-701. [15] 蔡天智,陈婧婧.2015 年我国内窥镜贸易分析[J].中国医疗器械信息,2016, 22(6):21-25. [16] 尤颢,杨谦.电外科手术设备的进展[J].中国医疗器械杂志,2012,36(4): 285-287. [17] CHAROENKWAN K, CHOTIROSNIRAMIT N, RERKASEM K. Scalpel versus electrosurgery for abdominal incisions. Cochrane Database Syst Rev. 2017;6(6): CD005987. [18] 杨勤玲,冯爱芳,鱼晓清.切口脂肪液化的诊治及护理研究综述[C]//第三届中华护理学会科技奖颁奖大会暨第三届护理学术年会论文集,2013:467-471. [19] 宋静.手术切口脂肪液化换药方法的研究进展[J].全科护理,2016,14(27): 2838-2840. [20] 纪光伟,吴远志,王旭,等.高频电刀对腹部切口愈合影响的实验和临床研究[J].中华医学杂志,2002,82(17):1199-1202. [21] 李巍,张艳秋.钨针电刀在耳内镜鼓膜修补手术中的应用[J].中华耳科学杂志,2021,19(2):248. [22] MUKESHKUMAR MM. A comparative study of colorado micro dissection needle versus scalpel in maxillofacial surgery. Rajiv Gandhi University of Health Sciences (India), 2016. [23] FURUDATE K, KATO J, HORIMOTO K, et al. Evaluation of degeneration of excision margin tissue using a Colorado MicroDissection Needle: A report of two cases. Australas J Dermatol. 2023;64(3):e301-e303. [24] RIDEOUT B, SHAW GY. Tonsillectomy using the Colorado microdissection needle: a prospective series and comparative technique review. South Med J. 2004;97(1):11-17. [25] ZHENG W, ZHU S, ZHANG Y, et al. Novel application of microdissection tungsten needle in total thyroidectomy with central neck dissection for papillary thyroid carcinoma. Front Surg. 2022;9:896275. [26] 陈懿,田武国,张刚,等.微创钨针在开放性甲状腺手术中喉返神经和甲状旁腺保护的临床应用[J].中华内分泌外科杂志,2020,14(1):23-27. [27] MISHRA P, BHOLA N, AGARWAL A, et al. To assess and compare the outcomes of the cutaneous neck dissection incision taken by colorado microdissection needle, surgical blade and cutting electrocautery in patients with oral squamous cell carcinoma: A prospective study. Natl J Maxillofac Surg. 2024;15(2):233-238. [28] AL-ZYOUD W, HADDADIN D, HASAN SA, et al. Biocompatibility testing for implants: A novel tool for selection and characterization. Materials. 2023;16(21):6881. [29] MIRI Z, FARÈ S, MA Q, et al. Updates on polyurethane and its multifunctional applications in biomedical engineering. Prog BiomedEng. 2023;5(4):042001. [30] XING M. Compatibility of Composite Biomaterials in Sports Injury Repair. Adv Mater Sci Eng. 2021;2021(1):4954325. [31] PRUKSAPONG C, JANKAJORN S, BURUSAPAT C, et al. Comparison of Colorado Needle Electrocautery and Traditional Scalpel for Lower Eyelid Blepharoplasty Incision: A Randomized Controlled Trial. Plast Reconstr Surg Glob Open. 2024; 12(11):e6325. |
| [1] | Yang Qiongqiong, Liu Wei. Comparison of performance and clinical effects of zirconia and titanium implants [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 2063-2071. |
| [2] | Wang Qisa, Lu Yuzheng, Han Xiufeng, Zhao Wenling, Shi Haitao, Xu Zhe. Cytocompatibility of 3D printed methyl acrylated hyaluronic acid/decellularized skin hydrogel scaffolds [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 1912-1920. |
| [3] | Zhou Hongli, Wang Xiaolong, Guo Rui, Yao Xuanxuan, Guo Ru, Zhou Xiongtao, He Xiangyi. Fabrication and characterization of nanohydroxyapatite/sodium alginate/polycaprolactone/alendronate scaffold [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 1962-1970. |
| [4] | Yang Guang, Yin Zhitao, Xu Yan. Three-dimensional printed isoniazid liposome photothermal composite scaffolds and their performance evaluation [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6701-6709. |
| [5] | Liu Weiwei, Gou Yuanbin, Cui Xiaoxue, Li Xin, Liu Dawei, Shi Mengrou, Chen Bin, Li Zhifeng. Comparison of different 3D-printed allogeneic bone and artificial polymer composite porous scaffold materials for repairing cranial bone defects in rats [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6744-6751. |
| [6] | Lyu Tianyang, Li Ning, Huang Shuo, Liu Changkui, Guo Yayuan, Hu Kaijin. Preparation and biocompatibility of odanacatib microspheres-gel composite sustained-release carrier [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6840-6848. |
| [7] | Yu Jinye, Jiang Nan, Zhao Yixun, Huang Mengjing, Yang Jie, Sun Rui, Feng Suolan, Jiang Hui, Yang Jun. Ready-to-use sodium alginate@paper material for three-dimensional cell culture [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6873-6879. |
| [8] | Song Muze, Liu Chuyi, , Tang Qingjuan, Dai Yuankun, , Song Wenshan, Li Bafang, Wang Yuanyuan. Biocompatibility evaluation of polylactic acid/collagen electrospinning bilayer guided tissue regeneration membrane [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6880-6891. |
| [9] | Zhang Mindi, Ma Teng, Su Qilong, Diao Kaixuan, Ren Guanghui, . Properties of boron nitride nanosheet-reinforced resin-matrix ceramics [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6899-6906. |
| [10] | Liu Xuemiao, Zhang Yuchang, Zhang Weiguo, Tian Kang, Wang Xing. Three-dimensional bioprinting and tendon repair: application advances and future directions [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6923-6929. |
| [11] | Li Shu, Zhao Zhengyi, Zeng Qin, Zhu Xiangdong. Nanohydroxyapatite induces immunogenic cell death in tumors [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(20): 5143-5151. |
| [12] | Tang Hao, Zhong Qian, Wu Honghan, Wu Hengpeng, Wu Xingkai, Wa Qingde. 3D-printed biodegradable polyester-based scaffolds in bone regeneration therapy [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(20): 5304-5311. |
| [13] | Wang Zitong, Wu Zijian, Yang Aofei, Mao Tian, Fang Nan, Wang Zhigang. Biomaterials regulate microenvironment imbalance for treating spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(20): 5321-5330. |
| [14] | Liu Shuting, Qiu Muen, Li Wei. Application and development trend of hydrogels in ophthalmic diseases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(20): 5349-5360. |
| [15] | Li Keyun, Yang Yuqi, Fei Yingying, Huang Shuai. Physicochemical properties and in vitro biological effects of resveratrol-eluting stents [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(20): 5243-5256. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||