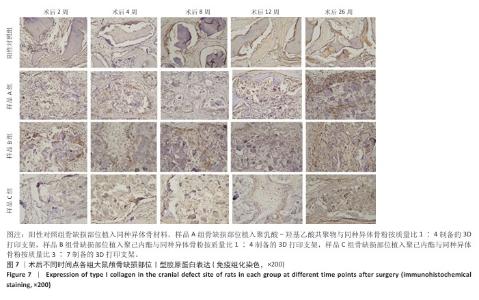
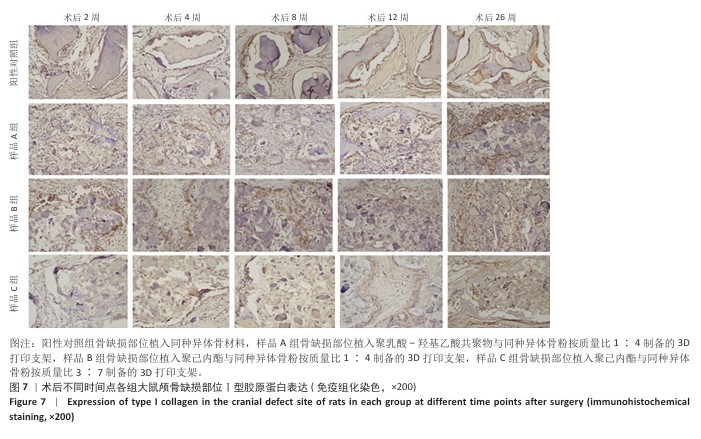
[1] KHAN DZ, AlI AHMAD MS, KOH CH, et al. Skull base repair following endonasal pituitary and skull base tumour resection: a systematic revie. Pituitary. 2021; 24(5):698-713.
[2] 刘冬,秦虎,汪永新,等.3D 打印羟基磷灰石/聚乳酸网状复合物修复颅骨缺损[J].中国组织工程研究,2019,23(6):833-837.
[3] WANG ZZ, YANG Y. Application of 3D Printing in Implantable Medical Devices. Biomed Res Int. 2021;2021:6653967.
[4] LEE SC, JUNG KA, NAM CH, et al. The Short-term Follow-up Results of Open Wedge High Tibial Osteotomy with Using an Aescula Open Wedge Plate and an Allogenic Bone Graft: The Minimum 1-Year Follow-up Results. Clin Orthop Surg. 2010;2(1):47-54.
[5] CHU CF, MAO SH, SHYU BH, et al. Allogeneic Bone-Marrow Mesenchymal Stem Cell with Moldable Cryogel for Craniofacial Bone Regeneration. J Pers Med. 2021;11(12):1326-1339.
[6] GHAYIBSHAIAN M, SALEHI M, BEHESHTIZADEH N, et al. Recent advances on 3D-printed PCL-based composite scaffolds for bone tissue engineering. Front Bioeng Biotechnol. 2023;11:1168504
[7] SUN Y, JIA XN, MENG Q. Characteristic Evaluation of Recombinant MiSp/Poly(lactic-co-glycolic) Acid (PLGA) Nanofiber Scaffolds as Potential Scaffolds for Bone Tissue Engineering. Int J Mol Sci. 2023;24(2):1219.
[8] 魏晨旭,何怡文,王聃,等.组织工程学中骨修复材料的研究热点与进展[J].中国组织工程研究,2020,24(10):1615-1621.
[9] 邓小磊,尹鹏开,魏巍,等.全杜仲胶囊对犬股骨头坏死骨组织病理变化及RUNX2、COL-1蛋白表达的影响[J].中华中医药学刊,2023,41(10):238-241, 后插38-后插39.
[10] 徐玉德,徐玉娥,周明旺,等.补肾中药通过Runt相关转录因子2 基因调控骨代谢相关研究进展[J].中国中医药信息杂志,2021,28(2):141-144.
[11] 孔令驰,孙兴亮,左荣台,等.早期移植生长分化因子11 表达沉默的骨髓基质干细胞促进大鼠激素性股骨头坏死区成骨的研究[J].中华创伤骨科杂志, 2019,21(9):802-809.
[12] XU YH, ZHANG FY, ZHAI WJ, et al. Unraveling of Advances in 3D-Printed Polymer-Based Bone Scaffolds. Polymers (Basel). 2022;14(3):566-596.
[13] HUANG D, LI ZH, LI GF, et al. Biomimetic structural design in 3D-printed scaffolds for bone tissue engineering. Mater Today Bio. 2025;32:101664.
[14] 张敏波.3D打印微小颗粒骨/聚乳酸-羟基乙酸支架的物理性能和生物相容性研究[D].遵义:遵义医科大学,2019.
[15] WU L, DING J. In vitro degradation of three-dimensional porous poly(D,Llactide-co-glycolide) scaffolds for tissue engineering. Biomaterials. 2004;25(27): 5821-5830.
[16] BABILOTTE J, MARTIN B, GUDURIC V, et al. Development and characterization of a PLGA-HA composite material to fabricate 3D-printed scaffolds for bone tissue engineering. Mater Sci Eng C Mater Biol Appl. 2021;118:111334.
[17] KADE JC, DALTON PD. Polymers for Melt Electrowriting. Adv Healthc Mater. 2021;10(1):e2001232.
[18] ILYAS K, AKHTAR MA, AMMAR EB, et al. Surface Modification of 3D-Printed PCL/BG Composite Scaffolds via Mussel-Inspired Polydopamine and Effective Antibacterial Coatings for Biomedical Applications. Materials (Basel, Switzerland). 2022;15(23):8289.
[19] HEWITT E, MROS S, MCCONNELL M, et al. Melt-electrowriting with novel milk protein/PCL biomaterials for skin regeneration. Biomed Mater. 2019;14(5): 055013.
[20] HAMADA S, MORI Y, SHIWAKU Y, et al. Octacalcium phosphate/gelatin composite(OCP/Gel) enhances bone repair in a criticalsized transcortical femoral defect rat model. Clin Orthop Relat Res. 2022;480(10):2043-2055.
[21] WANG Y. Acidic microenvironment induced by PLGA degradation accelerates scaffold degradation and bone formation. Biomater Sci. 2019;7(3):1073-1085.
[22] RASHID H,SMITH CM,CONVERS V, et al. RUNX2 deletion in hypertrophic chondrocytes impairs osteoclast mediated bone resorption. Bone. 2024;181: 117014-117014.
|