Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (24): 6259-6266.doi: 10.12307/2026.198
Previous Articles Next Articles
Roles of pregnane X receptor in sodium arsenite-induced oxidative stress and inflammatory injury in human normal hepatocytes
Zhang Xiaoxu1, Tian Zhenli2, Xie Tingting1
- 1Center for Clinical Laboratory, Affiliated Hospital of Guizhou Medical University, School of Clinical Laboratory Science of Guizhou Medical University, Guiyang 550004, Guizhou Province, China; 2Liupanshui Maternal and Child Health Hospital, Liupanshui 553001, Guizhou Province, China
-
Received:2025-06-16Revised:2025-09-11Online:2026-08-28Published:2026-02-02 -
Contact:Xie Tingting, MD, Associate professor, Associate chief technician, Center for Clinical Laboratory, Affiliated Hospital of Guizhou Medical University, School of Clinical Laboratory Science of Guizhou Medical University, Guiyang 550004, Guizhou Province, China -
About author:Zhang Xiaoxu, MS candidate, Junior technician, Center for Clinical Laboratory, Affiliated Hospital of Guizhou Medical University, School of Clinical Laboratory Science of Guizhou Medical University, Guiyang 550004, Guizhou Province, China -
Supported by:National Natural Science Foundation of China, No. 81560514 (to XTT); 2022 Doctoral Research Initiation Fund Project of Affiliated Hospital of Guizhou Medical University, No. gyfybsky-2022-33 (to XTT)
CLC Number:
Cite this article
Zhang Xiaoxu, Tian Zhenli, Xie Tingting. Roles of pregnane X receptor in sodium arsenite-induced oxidative stress and inflammatory injury in human normal hepatocytes[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6259-6266.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
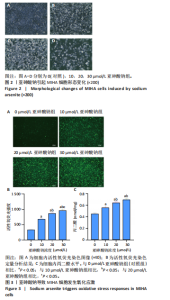
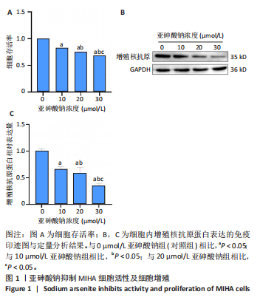
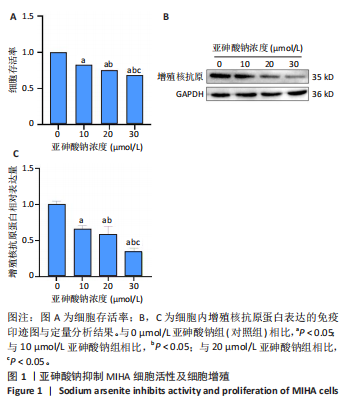
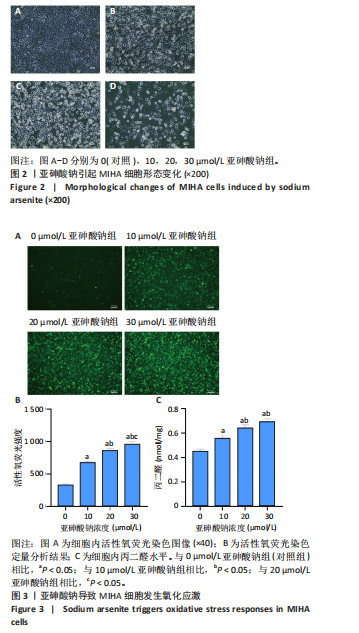
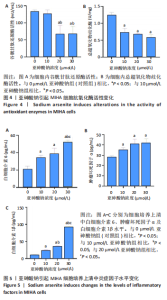
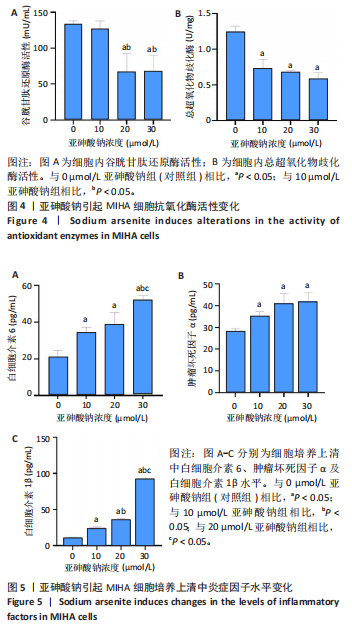
2.2 亚砷酸钠引起MIHA细胞形态变化 倒置显微镜观察下可见对照组细胞状态良好,细胞间呈多边形排列紧密;与对照组相比,各浓度亚砷酸钠组细胞胞膜边界不清,胞质减少,细胞失去正常形态,细胞融合率降低,变圆浮起,间隙增宽,见图2。 2.3 亚砷酸钠引起MIHA细胞发生氧化应激 荧光显微镜下可见对照组活性氧荧光强度较弱,10,20,30 μmol/L亚砷酸钠组活性氧荧光强度逐渐增强,见图3A,B。与对照组相比,10,20,30 μmol/L亚砷酸钠组细胞内丙二醛水平升高;20,30 μmol/L亚砷酸钠组细胞内丙二醛水平高于10 μmol/L亚砷酸钠组(P < 0.05),见图3C。 2.4 亚砷酸钠导致MIHA细胞内抗氧化酶活性降低 与对照组相比,20,30 μmol/L 亚砷酸钠组细胞内谷胱甘肽还原酶活性显著降低(P < 0.05),10,20,30 μmol/L 亚砷酸钠组细胞内总超氧化物歧化酶活性降低(P < 0.05);20,30 μmol/L 亚砷酸钠组细胞内谷胱甘肽还原酶活性低于10 μmol/L 亚砷酸钠组(P < 0.05),见图4。 2.5 亚砷酸钠导致MIHA细胞培养液上清中炎症因子水平升高 与对照组相比,10,20,30 μmol/L亚砷酸钠组白细胞介素6、肿瘤坏死因子α、白细胞介素1β水平均升高(P < 0.05);"
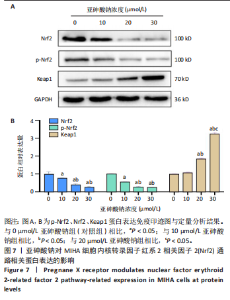
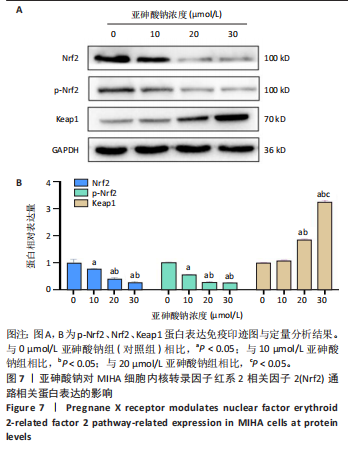
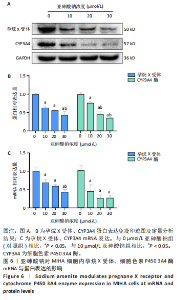
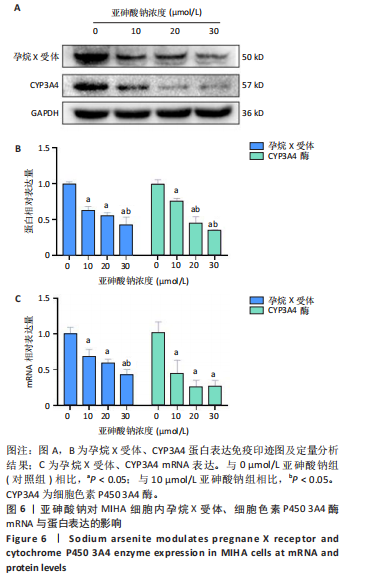
30 μmol/L亚砷酸钠组白细胞介素6、白细胞介素1β水平高于10,20 μmol/L亚砷酸钠组(P < 0.05),20 μmol/L亚砷酸钠组白细胞介素1β水平高于10 μmol/L亚砷酸钠组(P < 0.05),见图5。 2.6 亚砷酸钠对MIHA细胞内孕烷X受体、CYP3A4酶mRNA及蛋白表达的影响 qRT-PCR与Western blot检测结果显示,与对照组相比,10,20,30 μmol/L亚砷酸钠组细胞内孕烷X受体、CYP3A4酶mRNA及蛋白表达均降低(P < 0.05);30 μmol/L亚砷酸钠组孕烷X受体mRNA及蛋白表达均低于10 μmol/L亚砷酸钠组(P < 0.05),20,30 μmol/L亚砷酸钠组CYP3A4酶蛋白表达低于10 μmol/L亚砷酸钠组(P < 0.05),见图6。 2.7 亚砷酸钠对MIHA细胞内Nrf2、p-Nrf2、Keap1蛋白表达的影响 Western blot检测结果显示,与对照组相比,10,20,"
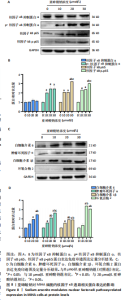
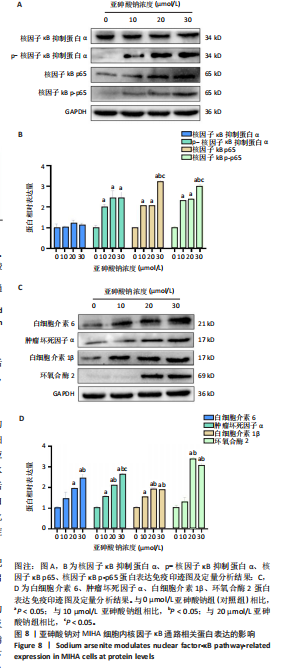
30 μmol/L亚砷酸钠组p-Nrf2、Nrf2蛋白表达降低(P < 0.05),20,30 μmol/L亚砷酸钠组Keap1蛋白表达升高(P < 0.05);20,30 μmol/L亚砷酸钠组p-Nrf2、Nrf2蛋白表达低于10 μmol/L亚砷酸钠组,Keap1蛋白表达高于10 μmol/L亚砷酸钠组(P < 0.05);30 μmol/L亚砷酸钠组Keap1蛋白表达高于20 μmol/L亚砷酸钠组(P < 0.05),见图7。 2.8 亚砷酸钠对MIHA细胞内核因子κB通路相关蛋白表达的影响 Western blot 检测结果显示,与对照组相比,10,20,30 μmol/L亚砷酸钠组p-核因子κB抑制蛋白α、核因子κB p-p65、核因子κB p65、肿瘤坏死因子α、白细胞介素1β蛋白表达均升高(P < 0.05),20,30 μmol/L亚砷酸钠组白细胞介素6、环氧合酶2蛋白表达升高(P < 0.05);30 μmol/L亚砷酸钠组核因子κB p65、核因子κB p-p65、肿瘤坏死因子α蛋白表达高于10,20 μmol/L亚砷酸钠组(P < 0.05),白细胞介素6蛋白表达高于10 μmol/L亚砷酸钠组(P < 0.05);20,30 μmol/L亚砷酸钠组白细胞介素"
| [1] PALMA-LARA I, MARTÍNEZ-CASTILLO M, QUINTANA-PÉREZ JC, et al. Arsenic exposure: A public health problem leading to several cancers. Regul Toxicol Pharmacol. 2020;110:104539. [2] UPADHYAY MK, SHUKLA A, YADAV P, et al. A review of arsenic in crops, vegetables, animals and food products. Food Chem. 2019;276:608-618. [3] KHAN MI, AHMAD MF, AHMAD I, et al. Arsenic Exposure through Dietary Intake and Associated Health Hazards in the Middle East. Nutrients. 2022;14(10):2136. [4] ISLAM MS, MUSTAFA RA, PHOUNGTHONG K, et al. Arsenic in the foodstuffs: potential health appraisals in a developing country, Bangladesh. Environ Sci Pollut Res Int. 2023;30(10):26938-26951. [5] HIRANO S. Biotransformation of arsenic and toxicological implication of arsenic metabolites. Arch Toxicol. 2020;94(8):2587-2601. [6] MA G, YAN X, WANG C, et al. Mechanism of arsenic-induced liver injury in rats revealed by metabolomics and ionomics based approach. Ecotoxicol Environ Saf. 2025;293:118038. [7] XING Y, YAN J, NIU Y. PXR: a center of transcriptional regulation in cancer. Acta Pharm Sin B. 2020;10(2):197-206. [8] LV Y, LUO YY, REN HW, et al. The role of pregnane X receptor (PXR) in substance metabolism. Front Endocrinol (Lausanne). 2022;13:959902. [9] ZHANG J, HUANG Y, LI H, et al. B3galt5 functions as a PXR target gene and regulates obesity and insulin resistance by maintaining intestinal integrity. Nat Commun. 2024;15(1):5919. [10] WAHLI W. A gut feeling of the PXR, PPAR and NF-kappaB connection. J Intern Med. 2008;263(6):613-619. [11] FENG Y, SHEN J, LIN Z, et al. PXR Activation Relieves Deoxynivalenol-Induced Liver Oxidative Stress Via Malat1 LncRNA m6A Demethylation. Adv Sci (Weinh). 2024;11(25): e2308742. [12] 王甜,赵哲仪,穆银贵,等.亚砷酸钠所致L-02人肝细胞损伤与p14ARF表达下调及MDM2、 p53表达增加有关[J].细胞与分子免疫学杂志,2020, 36(6):507-512. [13] 田侦丽,张小旭,方兴艳,等.亚砷酸钠对人正常肝细胞脂质代谢及因子调控的作用[J].中国组织工程研究,2025,29(23):4956-4964. [14] BEINSTEINER B, BILLAS IML, MORAS D. Structural insights into the HNF4 biology. Front Endocrinol (Lausanne). 2023;14:1197063. [15] RADI SH, VEMURI K, MARTINEZ-LOMELI J, et al. HNF4α isoforms: the fraternal twin master regulators of liver function. Front Endocrinol (Lausanne). 2023;14: 1226173. [16] FREDIANI JK, NAIOTI EA, VOS MB, et al. Arsenic exposure and risk of nonalcoholic fatty liver disease (NAFLD) among U.S. adolescents and adults: an association modified by race/ethnicity, NHANES 2005-2014. Environ Health. 2018;17(1):6. [17] ABU EL-SAAD AM, AL-KAHTANI MA, ABDEL-MONEIM AM. N-acetylcysteine and meso-2,3-dimercaptosuccinic acid alleviate oxidative stress and hepatic dysfunction induced by sodium arsenite in male rats. Drug Des Devel Ther. 2016;10: 3425-3434. [18] MOLAVINIA S, MOOSAVI M, HEJAZI S, et al. Metformin alleviates sodium arsenite-induced hepatotoxicity and glucose intolerance in mice by suppressing oxidative stress, inflammation, and apoptosis. J Trace Elem Med Biol. 2023;80: 127299. [19] PHULL AR, NASIR B, HAQ IU, et al. Oxidative stress, consequences and ROS mediated cellular signaling in rheumatoid arthritis. Chem Biol Interact. 2018; 281:121-36. [20] ZHANG B, PAN C, FENG C, et al. Role of mitochondrial reactive oxygen species in homeostasis regulation. Redox Rep. 2022;27(1): 45-52. [21] SENA LA, CHANDEL NS. Physiological roles of mitochondrial reactive oxygen species. Mol Cell. 2012;48(2):158-167. [22] LI Y, MA Q, LIU G, et al. Effects of donkey milk on oxidative stress and inflammatory response. J Food Biochem. 2022;46(4):e13935. [23] WU KC, LIU J, KLAASSEN CD. Role of Nrf2 in preventing ethanol-induced oxidative stress and lipid accumulation. Toxicol Appl Pharmacol. 2012;262(3):321-329. [24] LEE YH, KIM JH, SONG CH, et al. Ethanol Extract of Ganoderma lucidum Augments Cellular Anti-oxidant Defense through Activation of Nrf2/HO-1. J Pharmacopuncture. 2016;19(1): 59-69. [25] ZINATIZADEH MR, SCHOCK B, CHALBATANI GM, et al. The Nuclear Factor Kappa B (NF-kB) signaling in cancer development and immune diseases. Genes Dis. 2021;8(3):287-297. [26] LIU P, LI Y, WANG W, et al. Role and mechanisms of the NF-ĸB signaling pathway in various developmental processes. Biomed Pharmacother. 2022;153:113513. [27] MOTOLANI A, MARTIN M, SUN M, et al. Phosphorylation of the Regulators, a Complex Facet of NF-κB Signaling in Cancer. Biomolecules. 2020;11(1):15. [28] LIU F, ZHAO Y, PEI Y, et al. Role of the NF-kB signalling pathway in heterotopic ossification: biological and therapeutic significance. Cell Commun Signal. 2024;22(1):159. [29] EBRAHIMI N, ABDULWAHID ARR, MANSOURI A, et al. Targeting the NF-κB pathway as a potential regulator of immune checkpoints in cancer immunotherapy. Cell Mol Life Sci. 2024;81(1):106. [30] GUO Q, JIN Y, CHEN X, et al. NF-κB in biology and targeted therapy: new insights and translational implications. Signal Transduct Target Ther. 2024;9(1):53. [31] KARPALE M, HUKKANEN J, HAKKOLA J. Nuclear Receptor PXR in Drug-Induced Hypercholesterolemia. Cells. 2022;11(3):313. [32] YUAN T, LV S, ZHANG W, et al. PF-PLC micelles ameliorate cholestatic liver injury via regulating TLR4/MyD88/NF-κB and PXR/CAR/UGT1A1 signaling pathways in EE-induced rats. Int J Pharm. 2022;615:121480. [33] LIANG HF, YANG X, LI HL, et al. Activation of pregnane X receptor protects against cholestatic liver injury by inhibiting hepatocyte pyroptosis. Acta Pharmacol Sin. 2025;46(1):147-158. [34] DOU JY, ZHANG M, CEN H, et al. Salvia miltiorrhiza Bunge (Danshen) and Bioactive Compound Tanshinone IIA Alleviates Cisplatin-Induced Acute Kidney Injury Through Regulating PXR/NF-κB Signaling. Front Pharmacol. 2022;13:860383. [35] OKAMURA M, SHIZU R, HOSAKA T, et al. Possible involvement of the competition for the transcriptional coactivator glucocorticoid receptor-interacting protein 1 in the inflammatory signal-dependent suppression of PXR-mediated CYP3A induction in vitro. Drug Metab Pharmacokinet. 2019;34(4):272-279. [36] SHAO YY, GUO Y, FENG XJ, et al. Oridonin Attenuates TNBS-induced Post-inflammatory Irritable Bowel Syndrome via PXR/NF-κB Signaling . Inflammation. 2021;44(2):645-658. [37] LI H, FU Y, GONG W, et al. Remission of copper-induced liver injury through the PXR/NF-kB signaling pathway: The effects of dietary curcumin supplementation in largemouth bass (Micropterus salmoides). Ecotoxicol Environ Saf. 2024;285: 117070. [38] ZHANG X, MA Z, LIANG Q, et al. Tanshinone IIA exerts protective effects in a LCA-induced cholestatic liver model associated with participation of pregnane X receptor. J Ethnopharmacol. 2015;164: 357-367. [39] NIU H, ZHOU X, LIU P, et al. Lactobacillus rhamnosus MN-431 Metabolic Tryptophan Alleviates Complementary Food-Induced Diarrhea through PXR-NF-κB Pathway and AHR-Th17 Cell Response Pathways. Mol Nutr Food Res. 2023;67(13):e2200530. [40] DU L, JIANG W, ZHU X, et al. Rifaximin alleviates intestinal barrier disruption and systemic inflammation via the PXR/NFκB/MLCK pathway and modulates intestinal Lachnospiraceae abundance in heat-stroke mice. Int Immunopharmacol. 2024;143(Pt 2):113462. [41] YU X, XU M, MENG X, et al. Nuclear receptor PXR targets AKR1B7 to protect mitochondrial metabolism and renal function in AKI. Sci Transl Med. 2020; 12(543):eaay7591. [42] SUN M, CUI W, WOODY SK, et al. Pregnane X receptor modulates the inflammatory response in primary cultures of hepatocytes. Drug Metab Dispos. 2015;43(3):335-343. [43] MULDER TAM, VAN EERDEN RAG, DE WITH M, et al. CYP3A4∗22 Genotyping in Clinical Practice: Ready for Implementation? Front Genet. 2021;12:711943. [44] KONDŽA M, BRIZIĆ I, JOKIĆ S. Flavonoids as CYP3A4 Inhibitors In Vitro. Biomedicines. 2024;12(3):644. [45] ZHAO M, MA J, LI M, et al. Cytochrome P450 Enzymes and Drug Metabolism in Humans. Int J Mol Sci. 2021;22(23):12808. [46] BI G, LIANG F, WU T, et al. Pregnane X receptor activation induces liver enlargement and regeneration and simultaneously promotes the metabolic activity of CYP3A1/2 and CYP2C6/11 in rats. Basic Clin Pharmacol Toxicol. 2024;135(2):148-163. [47] NOREAULT TL, KOSTRUBSKY VE, WOOD SG, et al. Arsenite decreases CYP3A4 and RXRalpha in primary human hepatocytes. Drug Metab Dispos. 2005;33(7): 993-1003. [48] EL-GHIATY MA, EL-KADI AOS. Arsenic: Various species with different effects on cytochrome P450 regulation in humans. EXCLI J. 2021;20:1184-1242. [49] JIANG W, SANG R, ZHANG C, et al. Application of small interfering RNA technology in cytochrome P450 gene modulation. Drug Metab Dispos. 2025;53(3):100040. [50] YANG X, WEBER AA, MENNILLO E, et al. Oral arsenic administration to humanizedUDP-glucuronosyltransferase1 neonatal mice induces UGT1A1 through a dependence on Nrf2 and PXR. J Biol Chem. 2023;299(3):102955. |
| [1] | Yang Xuetao, Zhu Menghan, Zhang Chenxi, Sun Yimin, Ye Ling. Applications and limitations of antioxidant nanomaterials in oral cavity [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 2044-2053. |
| [2] |
Dong Chunyang, Zhou Tianen, Mo Mengxue, Lyu Wenquan, Gao Ming, Zhu Ruikai, Gao Zhiwei.
Action mechanism of metformin combined with Eomecon chionantha Hance dressing in treatment of deep second-degree burn wounds#br#
#br#
[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 2001-2013.
|
| [3] | Chen Yulin, He Yingying, Hu Kai, Chen Zhifan, Nie Sha Meng Yanhui, Li Runzhen, Zhang Xiaoduo , Li Yuxi, Tang Yaoping. Effect and mechanism of exosome-like vesicles derived from Trichosanthes kirilowii Maxim. in preventing and treating atherosclerosis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1768-1781. |
| [4] | Liu Anting, Lu Jiangtao, Zhang Wenjie, He Ling, Tang Zongsheng, Chen Xiaoling. Regulation of AMP-activated protein kinase by platelet lysate inhibits cadmium-induced neuronal apoptosis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1800-1807. |
| [5] | Cao Xinyan, Yu Zifu, Leng Xiaoxuan, Gao Shiai, Chen Jinhui, Liu Xihua. Effect of repetitive transcranial magnetic stimulation and transcranial direct current stimulation on motor function and gait in children with cerebral palsy: a network meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1539-1548. |
| [6] | Guo Ying, Tian Feng, Wang Chunfang. Potential drug targets for the treatment of rheumatoid arthritis: large sample analysis from European databases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1549-1557. |
| [7] | Peng Zhiwei, Chen Lei, Tong Lei. Luteolin promotes wound healing in diabetic mice: roles and mechanisms [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1398-1406. |
| [8] | Yang Zhijie, Zhao Rui, Yang Haolin, Li Xiaoyun, Li Yangbo, Huang Jiachun, Lin Yanping, Wan Lei, HuangHongxing. Postmenopausal osteoporosis: predictive values of muscle mass, grip strength, and appendicular skeletal muscle index [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1073-1080. |
| [9] | Chen Yixian, Chen Chen, Lu Liheng, Tang Jinpeng, Yu Xiaowei. Triptolide in the treatment of osteoarthritis: network pharmacology analysis and animal model validation [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 805-815. |
| [10] | Yang Xiao, Bai Yuehui, Zhao Tiantian, Wang Donghao, Zhao Chen, Yuan Shuo. Cartilage degeneration in temporomandibular joint osteoarthritis: mechanisms and regenerative challenges [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 926-935. |
| [11] | Yuan Min, Han Yu, Liu Jinhong, Zhang Jingyu, Cao Jimin, Sun Teng . Role and mechanism of ABL1 in myocardial necroptosis and cardiac ischemia/reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6247-6258. |
| [12] | Li Huaying, Li Hao, Peng Wuxun, Dong Wentao. Mechanism of cuproptosis in the diagnosis and treatment of orthopedic-related diseases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6275-6281. |
| [13] | Xia Caigui, Li Wei, Su Yuying, Shi Yu, Yang Zhonghe. Effects of antioxidant pretreatment on skeletal muscle damage and oxidative stress following acute high-intensity exercise: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6345-6353. |
| [14] | Jiang Chao, Che Yanjun. Biological mechanisms and future research trends of cartilaginous endplate degeneration [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(23): 5915-5924. |
| [15] | Gao Jiabin, Li Tianqi, Xu Kun, Zhu Hanmin, Zhou Xi, Li Wei. Mitophagy regulates osteoclasts: a new perspective for osteoporosis treatment [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(23): 5982-5991. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||