Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (19): 4990-4999.doi: 10.12307/2026.791
Previous Articles Next Articles
Action mechanism of mesenchymal stem cells and their derivatives in the treatment of liver fibrosis
Fan Longyu1, Yuan Xiao1, Xie Yanan1, Yin Xiaoxuan2
- 1Shandong University of Traditional Chinese Medicine, Jinan 250355, Shandong Province, China; 2Yankuang New Journey General Hospital, Jining 273599, Shandong Province, China
-
Received:2025-08-14Accepted:2025-12-08Online:2026-07-08Published:2026-02-24 -
Contact:Yin Xiaoxuan, MD, Chief physician, Yankuang New Journey General Hospital, Jining 273599, Shandong Province, China -
About author:Fan Longyu, Master candidate, Physician, Shandong University of Traditional Chinese Medicine, Jinan 250355, Shandong Province, China -
Supported by:Shandong Province Traditional Chinese Medicine Science and Technology Project, No. 2020Z41 (to YXX)
CLC Number:
Cite this article
Fan Longyu, Yuan Xiao, Xie Yanan, Yin Xiaoxuan . Action mechanism of mesenchymal stem cells and their derivatives in the treatment of liver fibrosis[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(19): 4990-4999.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
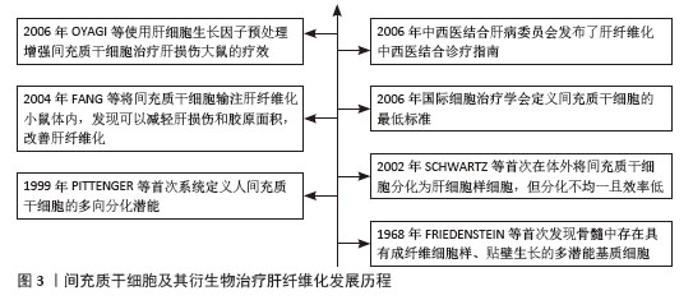
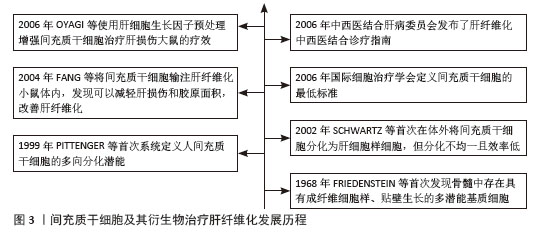
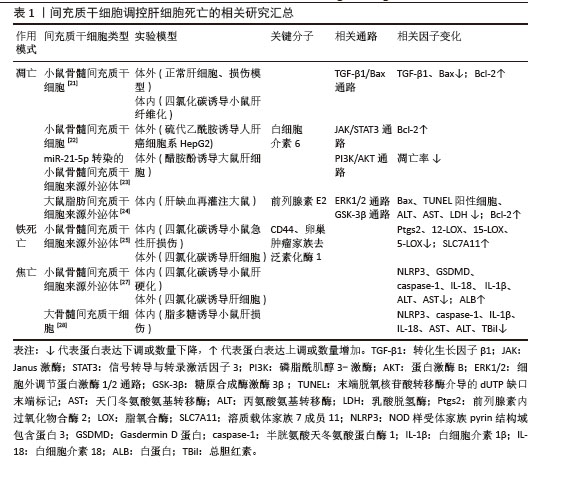
2.2 间充质干细胞 间充质干细胞具有显著的自我更新特性及多向分化潜能,能够向成骨细胞、软骨细胞和脂肪细胞等多谱系分化。随着研究的深入,研究人员发现这些细胞的分化能力有限,但在免疫调节、抗炎和分泌生物活性因子方面的功能更为显著。由于“干细胞”的定义逐渐严格(需要明确的自我更新和多向分化能力),许多研究者开始使用“间充质基质细胞”对这些细胞进行描述,以更准确地反映功能特性[5]。为了使全文术语统一,使用“间充质干细胞”这一术语,探讨细胞分化潜能以及分泌和免疫调节功能。 2.2.1 间充质干细胞的来源 间充质干细胞最初是从骨髓中被识别并分离出来的。然而由于间充质干细胞在骨髓中的数量有限(仅占骨髓细胞总数的0.001%-0.01%),且从骨髓中分离的过程具有侵入性,研究人员开始探寻其他来源。随后的研究中成功从不同组织中分离出在体外具有相似特性的间充质干细胞,这些组织主要包括滑膜、脂肪、脐血、脐带、羊水、胎盘、胎儿/新生儿组织、牙髓、月经血、扁桃体和诱导多能干细胞等[4,6]。尽管不同组织来源的间充质干细胞均符合国际细胞治疗学会的表型标准(CD73+、CD90+、CD105+),但分化潜能、分泌组及临床应用特性存在显著差异。骨髓来源间充质干细胞在成骨和软骨分化中表现突出,而脐血间充质干细胞是唯一体内可形成软骨的群体;脐带和月经血来源间充质干细胞因高表达血管生成相关基因(如血管内皮生长因子、转化生长因子β),从而展现出更强的血管生成和伤口修复能力;脂肪来源间充质干细胞则具有高吲哚胺-2,3-双加氧酶活性及低免疫原性在免疫调节中占优势[6]。虽然间充质干细胞具有组织来源多样性且在肝纤维化治疗中可能呈现不同的作用机制,该研究主要探讨间充质干细胞抗纤维化的共性机制,不区分组织来源差异。 2.2.2 间充质干细胞的通讯方式 (1)旁分泌可溶性因子:间充质干细胞通过旁分泌生长因子、抗炎因子、趋化因子等直接调控靶细胞功能,这是间充质干细胞最早被确认的通讯方式,具有抗炎与免疫调节、促组织修复和促进间充质干细胞归巢的功能。常见的可溶性因子包括转化生长因子β、肝细胞生长因子、前列腺素E2、吲哚胺-2,3-双加氧酶、一氧化氮和血管内皮生长因子等。房星星等[7]发现间充质干细胞与活化的CD4+ T细胞共培养后,培养上清液中转化生长因子β1、白细胞介素6、白细胞介素10水平显著升高,同时活化的CD4+ T细胞增殖受到抑制,这表明间充质干细胞可通过分泌转化生长因子β1、白细胞介素6、白细胞介素10等可溶性因子构建局部免疫微环境,发挥免疫抑制作用。另一项研究表明,间充质干细胞分泌的转化生长因子β通过激活蛋白激酶B/叉头框蛋白O1通路,诱导脂多糖刺激的巨噬细胞向抗炎M2样表型极化,显著降低白细胞介素6、白细胞介素1β等促炎因子水平,提高白细胞介素10等抗炎因子水平及巨噬细胞吞噬能力。实验证实,阻断转化生长因子β受体或抑制蛋白激酶B/叉头框蛋白O1通路均能逆转间充质干细胞对巨噬细胞极化的调控作用[8]。此外,间充质干细胞分泌的基质衍生因子1在组织修复和间充质干细胞向受损组织归巢中发挥关键作用。通过建立骨缺损诱导膜和骨移植模型,研究发现基质衍生因子1过表达可使两种模型大鼠骨髓、外周血和诱导膜组织中的间充质干细胞数量增加,并驱动间充质干细胞向受损的骨髓组织迁移;同时,基质衍生因子1过表达能激活基质衍生因子1/CXC趋化因子受体4信号通路,上调成骨相关基因和蛋白的表达[9]。 (2)细胞外囊泡:细胞外囊泡是存在于所有体液中的双层囊泡纳米颗粒。细胞外囊泡可以划分为3大类:外泌体、微泡以及凋亡小体。外泌体(30-200 nm)源于细胞内多泡体与细胞膜融合释放过程,富含CD63、CD81等特异性膜蛋白,荷载蛋白质、环状RNA、miRNA等多种生物活性物质,通过旁分泌作用参与细胞间信息交流,影响多种生理及病理过程。微泡(100-1 000 nm)由细胞膜直接出芽形成,参与病理生理过程中的信号传递。凋亡小体(500-2 000 nm)作为凋亡细胞终末产物,包含DNA片段、细胞器及膜结合蛋白,凋亡小体表面暴露的磷脂酰丝氨酸可被吞噬细胞识别,在免疫调节和组织再生中发挥独特作用。尽管细胞外囊泡的形成方式不同,但它们都富含蛋白质、RNA、脂质等多种生物活性分子,发挥免疫调节、靶向递送等作用[10]。一项对野百合碱诱导的肺动脉高压大鼠模型的研究表明,间充质干细胞来源细胞外囊泡通过递送miR-200b靶向调控磷酸二酯酶A/蛋白激酶A轴,促进巨噬细胞向抗炎的M2表型极化,而M2型巨噬细胞通过抑制炎症因子(如白细胞介素6、肿瘤坏死因子α)释放、下调肺动脉平滑肌细胞增殖并诱导其凋亡,从而改善肺动脉高压[11]。COSTA-FERRO等[12]研究表明,间充质干细胞来源细胞外囊泡的蛋白质组成具有显著的时间依赖性变化。通过质谱分析发现,随着条件培养基收集时间延长(24 h→72 h),细胞外囊泡的蛋白质异质性逐渐降低:早期(24 h)细胞外囊泡高表达代谢相关蛋白(如糖酵解酶相关丙酮酸激酶M型、氧化磷酸化相关苹果酸脱氢酶1);而后期(48-72 h)细胞外囊泡显著富集抗炎(膜联蛋白A1、乳脂肪球表皮生长因子8)、免疫调节(补体C3、转化生长因子β通路蛋白)及组织修复相关蛋白(Ⅰ型胶原蛋白/Ⅲ型胶原蛋白、纤连蛋白1)。功能富集分析进一步表明,后期细胞外囊泡的蛋白质组更集中于“细胞外基质组织”和“免疫调节”等通路。 研究发现,直接应用间充质干细胞存在异质性和致瘤性的风险。间充质干细胞来源和制备过程中存在高度可变性,例如供体的健康状况、组织来源、培养环境和分离技术等,这些因素均会影响最终的纯度和细胞亚群构成。另外,体外的长期扩增可诱发细胞衰老和癌变倾向,体内移植后可能自我更新失控,从而增加肿瘤形成的风险[13]。相比之下,间充质干细胞来源细胞外囊泡作为无细胞疗法具有很大优势。细胞外囊泡作为纳米颗粒,既携带生物活性分子又无自我复制能力,从而大幅降低致瘤风险;同时具有生物相容性和低免疫原性,能够跨越保护屏障,如血脑屏障;还具备包封功能,可加载特定药物靶向输送至细胞。此外,它们还可以由永生化细胞持续分泌以获得足够的数量[14]。通过高氨血症大鼠的体内外实验证明,间充质干细胞来源细胞外囊泡可穿过血脑屏障并靶向递送转化生长因子β至小脑,调控相关通路,下调白细胞介素1β、白细胞介素6、白细胞介素18等促炎因子,上调白细胞介素4、白细胞介素10及精氨酸酶1等抗炎标志物,减少小脑中小胶质细胞和星形胶质细胞的激活,恢复运动协调能力[15]。 (3)线粒体转移:线粒体作为真核细胞的重要细胞器,可为细胞活动提供必需的能量。过去人们认为细胞只在细胞分裂或线粒体生物发生过程中通过垂直遗传获得线粒体。后来的研究表明线粒体可以在细胞之间进行转移,转移途径分为形成短暂细胞连接(如隧道纳米管和间隙连接通道)、通过细胞外囊泡包裹释放、释放游离线粒体再被捕获。线粒体转移不仅能支持受体细胞代谢,还在支持供体细胞进行线粒体质量控制、调节伤口愈合、调节免疫系统和维持代谢稳态中起重要作用[16-17]。在动物实验中,将间充质干细胞或其分离的线粒体直接移植到脊髓损伤大鼠体内,均能减少神经元凋亡,促进轴突再生和髓鞘修复,最终显著改善大鼠运动功能恢复。体外实验表明,间充质干细胞线粒体显著改善氧糖剥夺损伤神经元的能量代谢、恢复线粒体膜电位并减少细胞凋亡,此过程可被缝隙连接细胞间通讯增强剂视黄酸促进,并被缝隙连接细胞间通讯抑制剂18β-甘草次酸抑制,这是首次证明间充质干细胞可通过间隙连接通道将线粒体转移到受伤神经元来预防脊髓损伤[18]。有研究共培养间充质干细胞与单核细胞来源巨噬细胞,结合共聚焦显微镜和流式细胞术证实,间充质干细胞通过隧道纳米管向巨噬细胞转移线粒体,显著增强巨噬细胞的氧化磷酸化能力和细菌吞噬功能,且这一效应可被隧道纳米管抑制剂细胞松弛素B完全阻断,证实间充质干细胞可通过隧道纳米管介导的线粒体转移作为调控先天免疫细胞功能的新途径[19]。特别值得注意的是,线粒体转移在某些疾病中可能呈现双重作用:如在代谢性疾病(如肥胖)和器官损伤中可通过维持组织稳态发挥保护性效应[16];而在癌症中,一方面可促进肿瘤侵袭和免疫逃逸,另一方面癌相关间充质干细胞能将线粒体转移至线粒体含量低的卵巢癌细胞,增强细胞增殖、化疗抵抗能力,恢复代谢功能[20]。 2.3 间充质干细胞及其衍生物治疗肝纤维化的作用机制 2.3.1 调控肝细胞死亡 在肝纤维化进程中,凋亡、铁死亡与焦亡等程序性死亡方式通过驱动肝细胞损伤与炎症级联反应,加速纤维化进展。研究表明,间充质干细胞能显著降低肝纤维化大鼠的转氨酶水平,明显改善肝功能,这种改善是通过下调促凋亡因子Bax和转化生长因子β1的表达,上调抗凋亡蛋白Bcl-2的表达,从而调控Bcl-2/Bax凋亡平衡实现的,该过程依赖转化生长因子β1/Bax相关通路,但具体机制尚未明确[21]。另一项关于间充质干细胞的实验也证实调控凋亡相关信号可进一步调控肝细胞死亡,其作用机制是分泌高浓度的白细胞介素6结合肝细胞膜受体,从而激活Janus激酶/信号转导和转录激活因3通路,最终上调下游抗凋亡信号(如Bcl-2)的释放[22]。另外,细胞外囊泡能靶向调控肝细胞中凋亡基因表达并激活相关通路。郑嵘炅等[23]研究证明间充质干细胞分泌的外泌体可以直接靶向磷脂酰肌醇3-激酶调节亚基1激活磷脂酰肌醇3-激酶/蛋白激酶B 信号通路抑制大鼠肝细胞凋亡,在加入蛋白激酶B抑制剂后可显著增加大鼠肝细胞凋亡率。使用间充质干细胞来源外泌体可升高肝损伤小鼠体内前列腺素E2水平,激活细胞外信号调节激酶1/2磷酸化,并抑制糖原合成酶激酶3β磷酸化,从而上调抗凋亡蛋白Bcl-2表达,下调促凋亡蛋白Bax表达,同时抑制线粒体通透性转换孔的开放,最终阻断线粒体介导的凋亡途径[24]。 铁死亡作为一种以铁过载和铁依赖性脂质过氧化积累为特征的程序性细胞死亡形式,在肝纤维化进展中扮演关键角色,而间充质干细胞可通过调控谷胱甘肽代谢、抑制脂质过氧化,显著减轻肝细胞铁死亡,为缓解肝纤维化提供了潜在治疗靶点。溶质载体家族7成员11作为系统XC-关键亚基,可转运半胱氨酸用于合成谷胱甘肽,并与谷胱甘肽过氧化物酶4协同抑制铁死亡。LIN等[25]研究发现间充质干细胞衍生外泌体可以恢复四氯化碳诱导肝损伤小鼠体内溶质载体家族7成员11蛋白水平。在肝纤维化中,类似机制可能同样存在,间充质干细胞迁移至受损肝脏部位,它分泌的细胞外囊泡可能通过上调CD44和卵巢肿瘤家族去泛素化酶1,稳定溶质载体家族7成员11蛋白,抑制铁死亡的发生,进而保护受伤肝细胞。 肝细胞在各种病因刺激下会激活NOD样受体家族pyrin结构域包含蛋白3炎性小体,诱导半胱氨酸天冬氨酸蛋白酶1切割GSDMD形成膜孔,导致肝细胞肿胀破裂发生焦亡。焦亡肝细胞释放白细胞介素1β、白细胞介素18及炎症小体颗粒(如NOD样受体家族pyrin结构域包含蛋白3、ASC和半胱氨酸天冬氨酸蛋白酶1),这些释放的炎症小体成分通过内吞作用进入肝星状细胞,触发分泌白细胞介素1β并表达α-平滑肌肌动蛋白,促进胶原沉积和纤维化进程[26]。多项研究证明间充质干细胞及其外泌体可显著降低肝损伤大鼠体内NOD样受体家族pyrin结构域包含蛋白3、GSDMD、半胱氨酸天冬氨酸蛋白酶1的表达,减少白细胞介素1β和白细胞介素18分泌,阻断了焦亡级联反应,并且作用效果优于单一使用NOD样受体家族pyrin结构域包含蛋白3抑制剂MCC950。此外间充质干细胞抗焦亡疗效呈剂量依赖性,且高剂量疗效最佳[27-28]。相关研究汇总见表1。 "
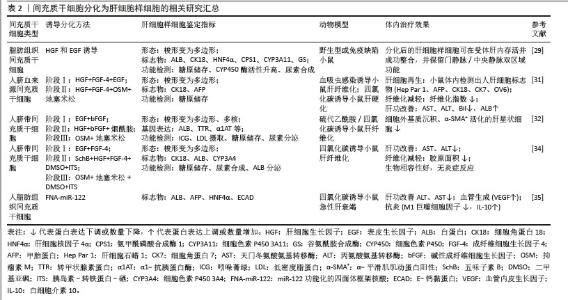
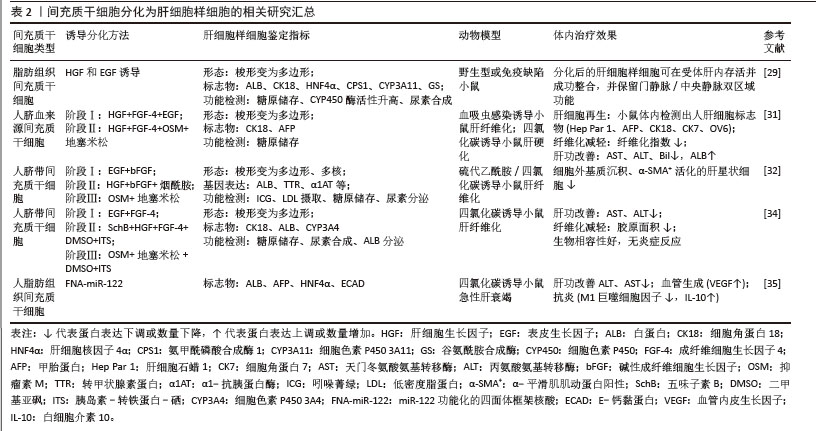
2.3.2 分化为肝细胞样细胞 间充质干细胞具有多向分化潜能,在特定条件下可以在体内外分化为肝细胞样细胞,在逆转肝纤维化病理进程中发挥关键作用。通过模拟肝胚胎发育的时序性细胞因子暴露(添加成纤维细胞生长因子4、肝细胞生长因子、表皮生长因子、胰岛素-转铁蛋白硒和地塞米松等)实验发现间充质干细胞可在体外分化为成熟肝细胞样细胞,这些细胞高度表达甲胎蛋白、肝细胞核因子4α、细胞角蛋白18、白蛋白及细胞色素P450等肝细胞标志物,并具备白蛋白分泌、尿素代谢、糖原储存及细胞色素P450活性的肝细胞功能[29-30]。上述研究证实间充质干细胞在体外诱导体系中可获得功能性肝细胞样表型,为了进一步探究间充质干细胞在体内微环境中的肝向转分化潜能及对肝纤维化病理进程的干预机制,研究人员将间充质干细胞通过静脉或肝内途径移植到血吸虫感染小鼠和四氯化碳诱导肝纤维化仓鼠体内,移植12周后,两种模型均观察到显著纤维化面积减少,肝功能指标(天门冬氨酸氨基转移酶、丙氨酸氨基转移酶、白蛋白、胆红素)明显改善,免疫组化阳性表达甲胎蛋白、细胞角蛋白18、细胞角蛋白7和OV6等肝细胞标志物,这表明间充质干细胞已在小鼠体内分化为具有人源特征的肝细胞样细胞促进肝脏修复[31]。AN等[32]通过三阶段分化方案将间充质干细胞定向分化为肝细胞样细胞,通过体内实验证明移植间充质干细胞分泌组和肝细胞样细胞分泌组可显著减少硫代乙酰胺/四氯化碳诱导的肝纤维化小鼠的纤维化面积,并且后者效果优于前者。体外预处理方法对调控间充质干细胞向肝细胞分化的效率和功能成熟度至关重要,AFSHARI等[33]总结了3种预处理方法:①添加与肝分化相关的细胞因子和生长因子,例如早期添加成纤维细胞生长因子、肝细胞生长因子,成熟阶段添加抑瘤素M等基因改造;②联合使用7种miRNA (miR-1246、miR-1290、miR-148a、miR-30a、miR-424、miR-542-5p与miR-122);③微环境和物理参数的调整,例如3D支架(提供结构支持)、微流控设备(模拟动态微环境)和生物反应器(规模化生产)等,这些预处理方法均可提高间充质干细胞的分化效率。另外,中药单体优化分化也具有良好的应用前景,JIN等[34]在分化的关键第二阶段添加10 μmol/L五味子素B,发现肝细胞的成熟标志物(白蛋白、细胞色素P450等)表达提升2.3-6.5倍,同时白蛋白分泌、尿素合成及糖原储存等肝脏功能得到增强,机制研究表明该效应与五味子素B特异性激活Janus激酶2/信号转导及转录激活因子3信号通路有关。在肝纤维化小鼠体内,移植五味子素B诱导的肝细胞样细胞组相较于未分化间充质干细胞组,疗效更显著(血清转氨酶水平下降幅度更大、肝组织胶原沉积面积明显减少)。此外,可以采取联合策略进行预处理,WEI等[35]开发了基于四面体框架核酸的miR-122递送系统,高效诱导间充质干细胞在无外源因子条件下分化为肝细胞样细胞(白蛋白表达提升22.5倍并具备尿素合成、糖原储存等成熟肝功能),进一步与脐静脉内皮细胞按9∶1比例构建肝球体,显著增强细胞极性及血管化潜能,机制分析表明分化过程与血管内皮生长因子、转化生长因子β、丝裂原活化蛋白激酶通路密切相关。在体内治疗中,移植的肝细胞样细胞还可促进M2巨噬细胞极化、抑制炎症及凋亡等协同抑制纤维化进程。相关研究汇总见表2。 "
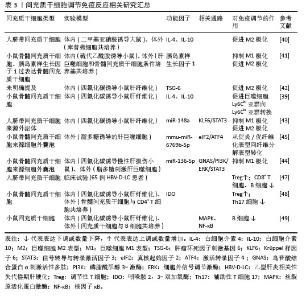
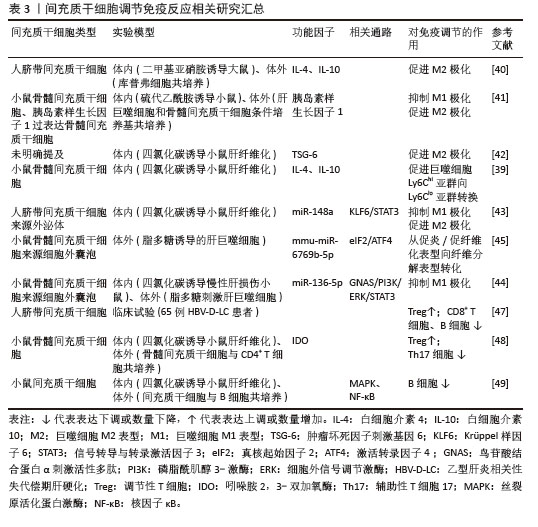
2.3.3 调节免疫反应 间充质干细胞通过协同调控肝脏固有免疫和适应性免疫反应,纠正纤维化微环境中过度激活的免疫应答,以减轻炎症损伤并抑制纤维化进程。受损伤的肝细胞迅速产生炎症递质并刺激肝巨噬细胞极化成促炎/促纤维化表型,分泌促炎细胞因子、参与抗原呈递、产生诱导型一氧化氮合酶、活化肝星状细胞。而间充质干细胞可以诱导肝巨噬细胞极化为抗炎/抗纤维化型,分泌抗炎因子,还能促进细胞外基质的降解[36]。其中促炎M1型和抗炎M2型是经典表型,研究人员通过体外非接触Transwell共培养模型,证实间充质干细胞可通过旁分泌作用显著抑制脂多糖和干扰素γ诱导的巨噬细胞M1型极化,同时促进巨噬细胞向M2表型转化,并伴随促炎因子(肿瘤坏死因子α、白细胞介素23a)的mRNA表达显著下调及抗炎标志物(精氨酸酶1、CD163)的mRNA表达上调[37]。体内实验进一步验证了上述研究结果,将间充质干细胞移植到四氯化碳诱导肝纤维化小鼠体内,发现M1型活化标志物(一氧化氮合酶)的显著抑制及M2型表型(CD206?/基质金属蛋白酶13?)的显著富集,同时促炎因子如肿瘤坏死因子α、白细胞介素6、干扰素γ转录水平下调及抗炎因子白细胞介素10表达上调[38]。LI等[39]提出这些过程中存在新的促炎亚群Ly6Chi和抗炎亚群Ly6Clo,前者高表达趋化因子受体1和受体2响应趋化因子(如趋化因子配体2),快速迁移至炎症部位;后者高表达CX3C趋化因子受体1,其与CX3C趋化因子配体1结合可抑制炎症反应。通过实验证明间充质干细胞旁分泌白细胞介素4和白细胞介素10调节肝巨噬细胞Ly6Chi亚群转换为 Ly6Clo亚群,此外Ly6Clo亚群可吞噬间充质干细胞凋亡小体通过PtdSer-MerTK-细胞外信号调节激酶信号通路上调基质金属蛋白酶12表达促进细胞外基质降解。进一步研究显示,间充质干细胞介导的巨噬细胞极化可能涉及旁分泌作用及细胞外囊泡。研究表明,间充质干细胞通过分泌白细胞介素4和胰岛素样生长因子1诱导巨噬细胞向M2表型极化(白细胞介素4抗体可阻断该过程),它分泌的肿瘤坏死因子刺激基因6则通过调控M2极化及上调基质金属蛋白酶12表达发挥抗纤维化作用。值得注意的是,M1巨噬细胞可通过激活Janus激酶/信号转导和转录激活因子信号通路增强间充质干细胞的肿瘤坏死因子刺激基因6表达[40-42]。间充质干细胞衍生的外泌体通过递送miR-148a靶向Kruppel样因子6抑制信号转导和转录激活因子3通路以抑制促炎巨噬细胞并促进抗炎表型,miR-136-5p经鸟嘌呤核苷酸结合蛋白α亚基/磷脂酰肌醇3-激酶/细胞外信号调节激酶/信号转导和转录激活因子3信号轴抑制M1极化,而miR-6769b-5p通过调控真核起始因子2/激活转录因子4轴驱动巨噬细胞从促纤维化向促纤维溶解表型转化[43-45]。 除调控巨噬细胞极化外,间充质干细胞还可通过调节适应性免疫中T/B淋巴细胞的增殖分化及细胞因子分泌,重塑失衡的免疫环境。肝脏中的T淋巴细胞主要包括CD4+辅助T细胞、CD8+细胞毒性T细胞、调节性T细胞等。Th17、Th1、Th2是CD4+ T细胞的主要亚型,纤维化肝脏中Th17产生白细胞介素17和白细胞介素22刺激肝星状细胞驱动纤维化,而调节性T细胞分泌白细胞介素10抑制M1巨噬细胞激活并限制白细胞介素17分泌发挥免疫抑制作用[46]。Th17/调节性T细胞失衡在肝纤维化中起到关键作用。FANG等[47]将间充质干细胞输注到肝硬化患者体内发现调节性T细胞数量明显上升,CD8+ T细胞、B细胞明显下降,血清白细胞介素6、肿瘤坏死因子α水平明显下降,白细胞介素10、转化生长因子β水平明显升高。在另一项四氯化碳诱导的动物研究中发现间充质干细胞显著改善了纤维化小鼠的肝脏质地、肝组织胶原沉积以及血清肝酶水平,流式细胞术分析肝浸润免疫细胞发现治疗组分泌白细胞介素10的调节性T细胞百分比升高,产生白细胞介素17的Th17细胞比例降低。进一步体外非接触细胞培养发现间充质干细胞以吲哚胺2,3-双加氧酶依赖的方式抑制Th17细胞的产生,而添加抑制剂1-甲基色氨酸会消除间充质干细胞对Th17/调节性T细胞平衡的调节作用,证实吲哚胺2,3-双加氧酶是间充质干细胞发挥效应的关键因子[48]。FENG等[49]发现B细胞在纤维化肝脏中活化,并显著富集幼稚B细胞亚型(B-Ⅱ),该亚型高表达增殖和炎症相关基因,而间充质干细胞通过外泌体下调丝裂原活化蛋白激酶/核因子κB 通路关键磷酸化蛋白,抑制B细胞增殖活化及促炎因子(肿瘤坏死因子α、白细胞介素6)分泌。相关研究汇总见表3。 "

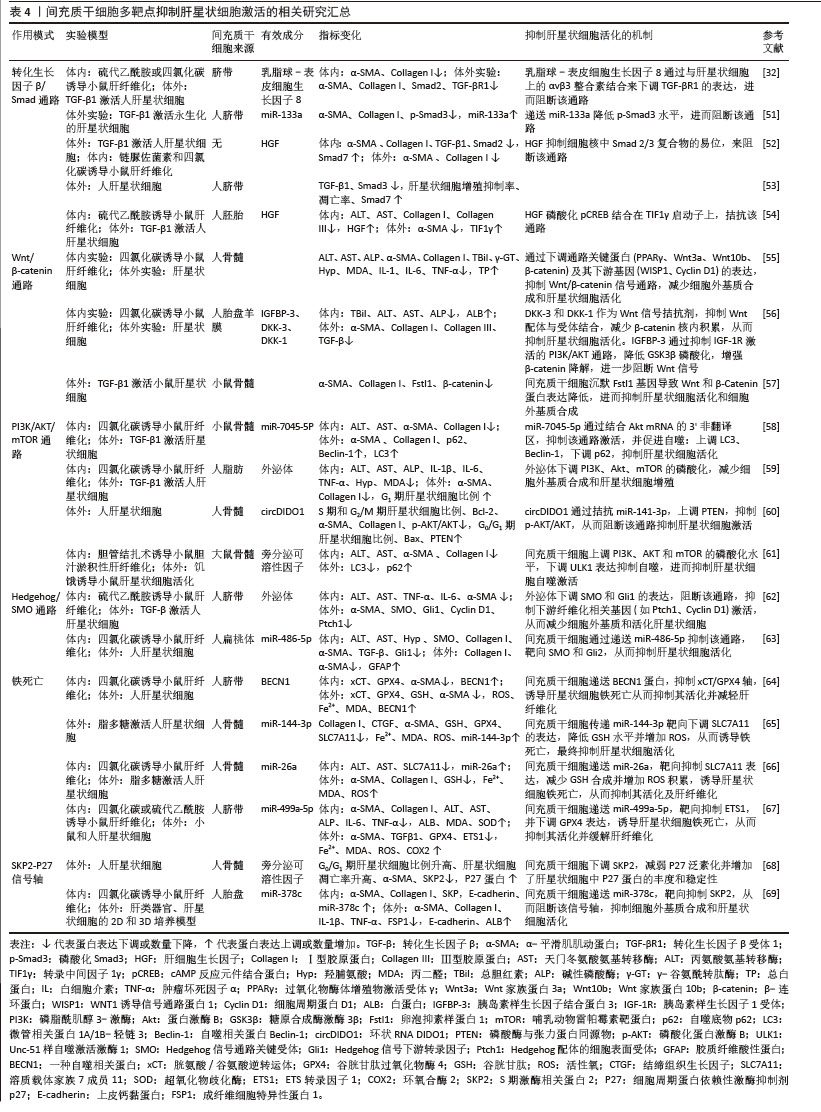
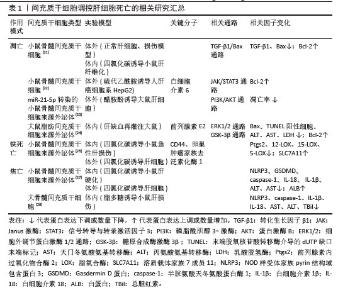
(1)通路类 转化生长因子β/Smad通路:该通路是激活肝星状细胞并驱动肝纤维化的核心通路。转化生长因子β通过结合细胞膜上的转化生长因子βⅡ型受体,诱导Ⅰ型受体磷酸化并激活Smad3,后者与Smad2形成复合体转入细胞核,直接调控促纤维化基因的转录,导致肝星状细胞的活化。虽然抑制型Smad7可阻断该通路,但它表达不足反而加剧了Smad3介导的促纤维化信号持续活化,最终促使细胞外基质过度沉积及肝纤维化进展[51-53]。已知间充质干细胞可旁分泌肝细胞生长因子,GHARBIA等[52]用肝细胞生长因子处理激活的肝星状细胞发现,α-平滑肌肌动蛋白和Ⅰ型胶原蛋白的表达明显降低,并且细胞核中Smad2/3复合物的易位被抑制,这提示转化生长因子β/Smad通路受到抑制。另一项间充质干细胞和肝星状细胞共培养研究揭示肝细胞生长因子通过调控转录中间因子1γ的表达来拮抗转化生长因子β/Smad信号通路[54]。此外,间充质干细胞还通过旁分泌乳脂球表皮细胞生因子8与肝星状细胞表面的αvβ3整合素结合,下调转化生长因子βⅠ型受体的表达,从而阻断转化生长因子β/Smad信号传导[32]。另外,间充质干细胞衍生外泌体可递送miR-133a抑制转化生长因子β/Smad3通路,逆转了活化肝星状细胞中上调的α-平滑肌肌动蛋白和胶原蛋白水平[51],从而达到治疗肝纤维化的目的。 Wnt/β-catenin通路:研究发现间充质干细胞衍生的外泌体可以下调Wnt信号通路相关蛋白(Wnt3a、Wnt10b、β-catenin)的表达,并抑制下游基因表达来抑制肝星状细胞激活[55]。进一步机制研究表明间充质干细胞分泌的DKK-1和DKK-3结合Wnt受体LRP,阻断了Wnt信号传导;此外,分泌的胰岛素样生长因子结合蛋白3通过抑制磷脂酰肌醇3-激酶/蛋白激酶B通路,减少蛋白激酶B介导的糖原合成酶激酶3β磷酸化,从而增强糖原合成酶激酶3β对β-catenin的降解作用[56]。ZHANGDI等[57]用间充质干细胞处理活化肝星状细胞后,发现α-平滑肌肌动蛋白和Ⅰ型胶原明显减少,卵泡抑素样蛋白1表达下调,且上调的β-Catenin和Wnt蛋白表达水平被逆转,而过表达卵泡抑素样蛋白1可逆转间充质干细胞的治疗作用,这证明间充质干细胞可靶向调节卵泡抑素样蛋白1基因抑制Wnt/β-catenin通路传导,对受伤的肝细胞起到保护作用。 磷脂酰肌醇3-激酶/蛋白激酶B/哺乳动物雷帕霉素靶蛋白通路:该通路是自噬的关键负调控通路,抑制该通路可解除对自噬的抑制,从而清除活化的肝星状细胞中受损的细胞器和异常蛋白聚集物,恢复细胞稳态,促使肝星状细胞从活化状态逆转为静息状态[58]。ZHANG等[59]研究表明间充质干细胞衍生的外泌体可降低磷脂酰肌醇3-激酶、蛋白激酶B和哺乳动物雷帕霉素靶蛋白的磷酸化水平,阻断该通路的激活,从而抑制肝星状细胞的增殖、诱导细胞周期停滞(G1期)并促进凋亡。机制研究发现间充质干细胞衍生细胞外囊泡可递送miR-7045-5P[58]、circDIDO1等抑制该通路[60],促进肝星状细胞自噬,减轻肝星状细胞活化。但也有研究表明自噬有利于肝星状细胞的激活。ULK1被确定为肝星状细胞自噬中受间充质干细胞调控的主要自噬相关基因,研究人员用间充质干细胞处理激活后的肝星状细胞发现磷脂酰肌醇3-激酶/蛋白激酶B/哺乳动物雷帕霉素靶蛋白信号通路被激活,并通过靶向ULK1抑制肝星状细胞自噬,最终抑制肝星状细胞的活化[61]。 Hedgehog/SMO通路:Hedgehog信号通路参与调控细胞增殖、分化及活化状态等多种生物学过程,因此,针对该通路的干预策略有望成为肝纤维化治疗的潜在靶点。ZONG等[62]用间充质干细胞衍生外泌体干预活化的肝星状细胞,检测到α-平滑肌肌动蛋白、SMO及通路转录因子Gli1的表达降低,且该作用可被SMO激动剂SAG逆转,这表明间充质干细胞可下调Hedgehog/SMO通路抑制肝星状细胞的活化,进一步研究表明miR-486-5p在间充质干细胞衍生细胞外囊泡中高度富集,并靶向Hedgehog/SMO通路下调SMO和Gli1的表达,从使肝星状细胞失活和肝纤维化减轻[63]。 (2)其他 铁死亡:研究表明间充质干细胞通过抑制肝细胞铁死亡从而促进肝细胞存活,有趣的是,间充质干细胞经多靶点诱导肝星状细胞脂质过氧化与铁死亡进而抑制肝星状细胞活化并促进纤维化消退。作为系统XC-的核心成分,溶质载体家族7成员11介导半胱氨酸转运至细胞内,促进谷胱甘肽生物合成,进而激活谷胱甘肽过氧化物酶4清除脂质过氧化物,最终抑制氧化应激及铁死亡[64]。研究人员发现间充质干细胞衍生外泌体递送miR-144-3p[65]、miR-26a靶向降低溶质载体家族7成员11表达[66],致使肝星状细胞内谷胱甘肽水平降低、活性氧和铁离子蓄积,从而触发铁死亡和肝星状细胞活化的抑制,且这一过程伴随胶原蛋白Ⅰ和α-平滑肌肌动蛋白显著减少。另一项研究表明间充质干细胞衍生外泌体还递送关键蛋白BECN1抑制胱氨酸/谷氨酸逆转运体驱动的谷胱甘肽过氧化物酶4表达,诱导铁死亡并抑制肝星状细胞活化,从而缓解肝纤维化[64]。WANG等[67]的研究发现间充质干细胞来源外泌体递送miR-499a-5p,靶向抑制肝纤维化组织中高表达的ETS转录因子1,并阻断其激活谷胱甘肽过氧化物酶4而导致肝星状细胞铁死亡。 SKP2-P27信号轴:P27蛋白是一种细胞周期蛋白依赖性激酶抑制剂,可以阻滞肝星状细胞细胞周期从G0/G1到S的进展,但易受到E3泛素化连接酶SKP2介导的多泛素化和26S蛋白酶体依赖性降解。体外非接触共培养间充质干细胞与青霉素/链霉素培养的肝星状细胞发现,间充质干细胞促进肝星状细胞周期停滞和细胞凋亡抑制细胞增殖,并呈时间依赖性。进一步机制研究表明间充质干细胞下调E3泛素连接酶SKP2,从而减弱了它介导的P27泛素化,并增加了肝星状细胞中P27蛋白的丰度和稳定性[68]。有研究表明这有可能是间充质干细胞衍生细胞外囊泡中miR-378C介导的[69]。 2.4 间充质干细胞治疗肝纤维化的临床研究 目前国内外临床研究更多聚焦于失代偿期肝硬化、慢加急性肝衰竭和肝移植后等终末期肝病,这可能归因于肝纤维化早期临床表现隐匿或与原发慢性肝病表现重叠,导致受试者不易被募集;而终末期肝病患者病情严重,对间充质干细胞治疗的临床转化需求更为急迫[2,70]。在疗效评估方面,肝纤维化研究主要关注肝组织病理学改善和血清纤维化标志物(如透明质酸、Ⅲ型前胶原等)的变化;终末期肝病研究更着重于肝功能、MELD评分、生存率及病死率等指标,因此在肝纤维化这一可逆转阶段进行早期干预,对减少终末期肝病的发生具有重要意义。张东锋等[71]招募了60例肝豆状核变性合并肝纤维化患者,随机分为青霉胺组、间充质干细胞组及联合组。患者每10 d经静脉输注自体骨髓来源间充质干细胞,3次为1个疗程,12周后分析血清学指标和肝组织病理,发现联合治疗组改善程度最优,其次为间充质干细胞组和青霉胺组,且研究期间未报告相关不良反应,这表明间充质干细胞治疗肝纤维化临床安全有效,与药物联用可增强疗效,然而该试验规模相对较小,并且缺乏后期随访数据,因此长期治疗的有效性和安全性尚待评估。此外,间充质干细胞的临床应用仍面临诸多亟待解决的问题,如最佳细胞类型选择、最适移植细胞数量、给药途径优化以及相关临床应用规范与标准的建立等[72]。因此,在实现大规模临床应用之前,亟需通过大规模、设计严谨的临床试验来确定应用标准,相关临床研究见表5。 "

| [1] BOGLIOTTI Y, VANDER ROEST M, MATTIS AN, et al. Clinical Application of Induced Hepatocyte-like Cells Produced from Mesenchymal Stromal Cells: A Literature Review. Cells. 2022;11(13):1998. [2] 徐列明,刘平,沈锡中,等.肝纤维化中西医结合诊疗指南(2019年版)[J].中国中西医结合杂志,2019,39(11):1286-1295. [3] FRIEDMAN S L, PINZANI M. Hepatic fibrosis 2022: Unmet needs and a blueprint for the future. Hepatology. 2022;75(2):473-488. [4] ALFAIFI M, EOM YW, NEWSOME PN, et al. Mesenchymal stromal cell therapy for liver diseases. J Hepatol. 2018;68(6):1272-1285. [5] VISWANATHAN S, SHI Y, GALIPEAU J, et al. Mesenchymal stem versus stromal cells: International Society for Cell & Gene Therapy (ISCT®) Mesenchymal Stromal Cell committee position statement on nomenclature. Cytotherapy. 2019;21(10):1019-1024. [6] PARK S, JUNG SC. New Sources, Differentiation, and Therapeutic Uses of Mesenchymal Stem Cells. Int J Mol Sci. 2021;22(10):5288. [7] 房星星,路臻豪,汤建平,等.MSCs通过促分泌TGF-β1、IL-6和IL-10抑制干燥综合征患者CD4+ T细胞的活化增殖[J].同济大学学报(医学版),2016,37(4):31-35. [8] LIU F, QIU H, XUE M, et al. MSC-secreted TGF-β regulates lipopolysaccharide-stimulated macrophage M2-like polarization via the Akt/FoxO1 pathway. Stem Cell Res Ther. 2019;10(1): 345. [9] ZHANG H, LI X, LI J, et al. SDF-1 mediates mesenchymal stem cell recruitment and migration via the SDF-1/CXCR4 axis in bone defect. J Bone Miner Metab. 2021;39(2):126-138. [10] TANG H, LUO H, ZHANG Z, et al. Mesenchymal Stem Cell-Derived Apoptotic Bodies: Biological Functions and Therapeutic Potential. Cells. 2022; 11(23):3879. [11] WAN M, LU C, LIU Y, et al. Mesenchymal stem cell-derived extracellular vesicles prevent the formation of pulmonary arterial hypertension through a microRNA-200b-dependent mechanism. Respir Res. 2023;24(1):233. [12] COSTA-FERRO ZSM, ROCHA GV, DA SILVA KN, et al. GMP-compliant extracellular vesicles derived from umbilical cord mesenchymal stromal cells: manufacturing and pre-clinical evaluation in ARDS treatment. Cytotherapy. 2024;26(9):1013-1025. [13] ZHOU T, YUAN Z, WENG J, et al. Challenges and advances in clinical applications of mesenchymal stromal cells. J Hematol Oncol. 2021;14(1):24. [14] KOU M, HUANG L, YANG J, et al. Mesenchymal stem cell-derived extracellular vesicles for immunomodulation and regeneration: a next generation therapeutic tool? Cell Death Dis. 2022; 13(7):580. [15] IZQUIERDO-ALTAREJOS P, MARTÍNEZ-GARCÍA M, ATIENZA-PÉREZ I, et al. Extracellular Vesicles from Mesenchymal Stem Cells Reverse Neuroinflammation and Restore Motor Coordination in Hyperammonemic Rats. J Neuroimmune Pharmacol. 2024;19(1):52. [16] BORCHERDING N, BRESTOFF JR. The power and potential of mitochondria transfer. Nature. 2023;623(7986):283-291. [17] LIU D, GAO Y, LIU J, et al. Intercellular mitochondrial transfer as a means of tissue revitalization. Signal Transduct Target Ther. 2021; 6(1):65. [18] LI H, WANG C, HE T, et al. Mitochondrial Transfer from Bone Marrow Mesenchymal Stem Cells to Motor Neurons in Spinal Cord Injury Rats via Gap Junction. Theranostics. 2019;9(7):2017-2035. [19] JACKSON MV, KRASNODEMBSKAYA AD. Analysis of Mitochondrial Transfer in Direct Co-cultures of Human Monocyte-derived Macrophages (MDM) and Mesenchymal Stem Cells (MSC). Bio Protoc. 2017;7(9):e2255. [20] FRISBIE L, PRESSIMONE C, DYER E, et al. Carcinoma-associated mesenchymal stem cells promote ovarian cancer heterogeneity and metastasis through mitochondrial transfer. Cell Rep. 2024;43(8):114551. [21] XU TB, LI L, LUO XD, et al. BMSCs protect against liver injury via suppressing hepatocyte apoptosis and activating TGF-β1/Bax singling pathway. Biomed Pharmacother. 2017;96:1395-1402. [22] CHEN HC, AWALE S, WU CP, et al. Co-cultured bone marrow mesenchymal stem cells repair thioacetamide-induced hepatocyte damage. Cell Biol Int. 2020;44(12):2459-2472. [23] 郑嵘炅,邓泽润,韩丹,等.骨髓间充质干细胞来源外泌体调节大鼠肝细胞凋亡的机制[J].中国组织工程研究,2024,28(1):44-49. [24] ZHANG Y, LI Y, WANG Q, et al. Attenuation of hepatic ischemia‑reperfusion injury by adipose stem cell‑derived exosome treatment via ERK1/2 and GSK‑3β signaling pathways. Int J Mol Med. 2022;49(2):13. [25] LIN F, CHEN W, ZHOU J, et al. Mesenchymal stem cells protect against ferroptosis via exosome-mediated stabilization of SLC7A11 in acute liver injury. Cell Death Dis. 2022;13(3):271. [26] GAUL S, LESZCZYNSKA A, ALEGRE F, et al. Hepatocyte pyroptosis and release of inflammasome particles induce stellate cell activation and liver fibrosis. J Hepatol. 2021;74(1): 156-167. [27] ZHANG Y, ZHANGDI H, NIE X, et al. Exosomes Derived from BMMSCs Mitigate the Hepatic Fibrosis via Anti-Pyroptosis Pathway in a Cirrhosis Model. Cells. 2022;11(24):4004. [28] YIN Y, TANG L, LIU K, et al. Attenuation of Lipopolysaccharide-induced Liver Injury by Bone Marrow Mesenchymal Stem Cells via Inhibiting the NLRP3 Inflammasome and Hepatocyte Pyroptosis. Curr Stem Cell Res Ther. 2022;17(4):361-369. [29] WINKLER S, HEMPEL M, BRÜCKNER S, et al. Mouse white adipose tissue-derived mesenchymal stem cells gain pericentral and periportal hepatocyte features after differentiation in vitro, which are preserved in vivo after hepatic transplantation. Acta Physiol (Oxf). 2015;215(2):89-104. [30] CHEN L, ZHANG N, HUANG Y, et al. Multiple Dimensions of using Mesenchymal Stem Cells for Treating Liver Diseases: From Bench to Beside. Stem Cell Rev Rep. 2023;19(7):2192-2224. [31] KAMEL MM, BAZ HGE, DEMERDASH Z, et al. Cord blood-derived mesenchymal stem cells with hepatogenic differentiation potential ameliorate chronic liver affection in experimental models. Adv Clin Exp Med. 2018;27(10):1329-1339. [32] AN SY, JANG YJ, LIM HJ, et al. Milk Fat Globule-EGF Factor 8, Secreted by Mesenchymal Stem Cells, Protects Against Liver Fibrosis in Mice. Gastroenterology. 2017;152(5):1174-1186. [33] AFSHARI A, SHAMDANI S, UZAN G, et al. Different approaches for transformation of mesenchymal stem cells into hepatocyte-like cells. Stem Cell Res Ther. 2020;11(1):54. [34] JIN M, YI X, ZHU X, et al. Schisandrin B promotes hepatic differentiation from human umbilical cord mesenchymal stem cells. iScience. 2024;27(2): 108912. [35] WEI H, XUE T, LI F, et al. Framework nucleic Acid-MicroRNA mediated hepatic differentiation and functional hepatic spheroid development for treating acute liver failure. Bioact Mater. 2024; 41:611-626. [36] FAN B, XIE H, TAN Q, et al. Role of Exosomal Modulation of Macrophages in Liver Fibrosis. J Clin Transl Hepatol. 2024;12(2):201-209. [37] HOU Y, ZHOU X, CAI WL, et al. Regulatory effect of bone marrow mesenchymal stem cells on polarization of macrophages. Zhonghua Gan Zang Bing Za Zhi. 2017;25(4):273-278. [38] LUO XY, MENG XJ, CAO DC, et al. Transplantation of bone marrow mesenchymal stromal cells attenuates liver fibrosis in mice by regulating macrophage subtypes. Stem Cell Res Ther. 2019; 10(1):16. [39] LI YH, SHEN S, SHAO T, et al. Mesenchymal stem cells attenuate liver fibrosis by targeting Ly6Chi/lo macrophages through activating the cytokine-paracrine and apoptotic pathways. Cell Death Discov. 2021;7(1):239. [40] CHAI NL, ZHANG XB, CHEN SW, et al. Umbilical cord-derived mesenchymal stem cells alleviate liver fibrosis in rats. World J Gastroenterol. 2016; 22(26):6036-6048. [41] FIORE E, MALVICINI M, BAYO J, et al. Involvement of hepatic macrophages in the antifibrotic effect of IGF-I-overexpressing mesenchymal stromal cells. Stem Cell Res Ther. 2016;7(1):172. [42] WANG M, ZHANG M, FU L, et al. Liver-targeted delivery of TSG-6 by calcium phosphate nanoparticles for the management of liver fibrosis. Theranostics. 2020;10(1):36-49. [43] TIAN S, ZHOU X, ZHANG M, et al. Mesenchymal stem cell-derived exosomes protect against liver fibrosis via delivering miR-148a to target KLF6/STAT3 pathway in macrophages. Stem Cell Res Ther. 2022;13(1):330. [44] JIANG X, LIU Z, YOU H, et al. Quercetin-primed BMSC-derived extracellular vesicles ameliorate chronic liver damage through miR-136-5p and GNAS/STAT3 signaling pathways. Int Immunopharmacol. 2024;142(Pt B):113162. [45] NISHI M, MATSUMOTO T, FUJISAWA K, et al. Mesenchymal Stem Cells Induce a Fibrolytic Phenotype By Regulating mmu-miR-6769b-5p Expression in Macrophages. Stem Cells Dev. 2020;29(22):1457-1466. [46] HAMMERICH L, TACKE F. Hepatic inflammatory responses in liver fibrosis. Nat Rev Gastroenterol Hepatol. 2023;20(10):633-646. [47] FANG XQ, ZHANG JF, SONG HY, et al. Effect of umbilical cord mesenchymal stem cell transplantation on immune function and prognosis of patients with decompensated hepatitis B cirrhosis. Zhonghua Gan Zang Bing Za Zhi. 2016;24(12):907-910. [48] MILOSAVLJEVIC N, GAZDIC M, SIMOVIC MARKOVIC B, et al. Mesenchymal stem cells attenuate liver fibrosis by suppressing Th17 cells - an experimental study. Transpl Int. 2018;31(1):102-115. [49] FENG X, FENG B, ZHOU J, et al. Mesenchymal stem cells alleviate mouse liver fibrosis by inhibiting pathogenic function of intrahepatic B cells. Hepatology. 2025;81(4):1211-1227. [50] LEE C, KIM M, HAN J, et al. Mesenchymal Stem Cells Influence Activation of Hepatic Stellate Cells, and Constitute a Promising Therapy for Liver Fibrosis. Biomedicines. 2021;9(11):1598. [51] SAKI S, MONJEZI S, GHAFFARI F, et al. Unraveling the Exosome-miR-133a Axis: Targeting TGF-β Signaling via WJ-MSC-Derived Exosomes for Anti-Fibrotic Therapy in Liver Fibrosis. Iran Biomed J. 2024;28(5 & 6):235-244. [52] GHARBIA S, NAZARIE SR, DINESCU S, et al. Adipose-Derived Stem Cells (ADSCs) Supplemented with Hepatocyte Growth Factor (HGF) Attenuate Hepatic Stellate Cell Activation and Liver Fibrosis by Inhibiting the TGF-β/Smad Signaling Pathway in Chemical-Induced Liver Fibrosis Associated with Diabetes. Cells. 2022;11(21):3338. [53] ZHANG LT, PENG XB, FANG XQ, et al. Human umbilical cord mesenchymal stem cells inhibit proliferation of hepatic stellate cells in vitro. Int J Mol Med. 2018;41(5):2545-2552. [54] LEE EJ, HWANG I, LEE JY, et al. Hepatic stellate cell-specific knockout of transcriptional intermediary factor 1γ aggravates liver fibrosis. J Exp Med. 2020;217(6):e20190402. [55] RONG X, LIU J, YAO X, et al. Human bone marrow mesenchymal stem cells-derived exosomes alleviate liver fibrosis through the Wnt/β-catenin pathway. Stem Cell Res Ther. 2019;10(1):98. [56] LIU QW, YING YM, ZHOU JX, et al. Human amniotic mesenchymal stem cells-derived IGFBP-3, DKK-3, and DKK-1 attenuate liver fibrosis through inhibiting hepatic stellate cell activation by blocking Wnt/β-catenin signaling pathway in mice. Stem Cell Res Ther. 2022;13(1):224. [57] ZHANGDI H, GENG X, LI N, et al. BMSCs alleviate liver cirrhosis by regulating Fstl1/Wnt/β-Catenin signaling pathway. Heliyon. 2023;9(11):e21010. [58] LIU Z, JIANG X, YOU H, et al. Extracellular vesicles derived from bone marrow mesenchymal stem cells ameliorate liver fibrosis via micro-7045-5p. Mol Cell Biochem. 2025;480(5):2903-2921. [59] ZHANG Z, SHANG J, YANG Q, et al. Exosomes derived from human adipose mesenchymal stem cells ameliorate hepatic fibrosis by inhibiting PI3K/Akt/mTOR pathway and remodeling choline metabolism. J Nanobiotechnology. 2023;21(1):29. [60] MA L, WEI J, ZENG Y, et al. Mesenchymal stem cell-originated exosomal circDIDO1 suppresses hepatic stellate cell activation by miR-141-3p/PTEN/AKT pathway in human liver fibrosis. Drug Deliv. 2022;29(1):440-453. [61] HUANG T, ZHANG C, SHANG Z, et al. Bone mesenchymal stem cells improve cholestatic liver fibrosis by targeting ULK1 to regulate autophagy through PI3K/AKT/mTOR pathway. Stem Cells Transl Med. 2024;13(7):648-660. [62] ZONG R, ZHENG Y, YAN Y, et al. Mesenchymal stem cells-derived exosomes alleviate liver fibrosis by targeting Hedgehog/SMO signaling. Hepatol Int. 2024;18(6):1781-1791. [63] KIM J, LEE C, SHIN Y, et al. sEVs from tonsil-derived mesenchymal stromal cells alleviate activation of hepatic stellate cells and liver fibrosis through miR-486-5p. Mol Ther. 2021;29(4):1471-1486. [64] TAN Y, HUANG Y, MEI R, et al. HucMSC-derived exosomes delivered BECN1 induces ferroptosis of hepatic stellate cells via regulating the xCT/GPX4 axis. Cell Death Dis. 2022;13(4):319. [65] HAO Y, WANG R, ZHOU Q, et al. Bone marrow mesenchymal stem cell-originated exosomes suppress activation of hepatic stellate cells through the miR-144-3p/SLC7A11 axis. Clin Exp Hepatol. 2024;10(3):197-210. [66] CAO Y, YANG H, HUANG Y, et al. Mesenchymal stem cell-derived exosomal miR-26a induces ferroptosis, suppresses hepatic stellate cell activation, and ameliorates liver fibrosis by modulating SLC7A11. Open Med (Wars). 2024;19(1):20240945. [67] WANG Z, YUAN M, YAO L, et al. Exosomal miR-499a-5p from human umbilical cord mesenchymal stem cells attenuates liver fibrosis via targeting ETS1/GPX4-mediated ferroptosis in hepatic stellate cells. J Nanobiotechnology. 2025;23(1):222. [68] WANG L, BAI G, CHEN F. Human bone marrow mesenchymal stem cells suppress the proliferation of hepatic stellate cells by inhibiting the ubiquitination of p27. Biochem Cell Biol. 2017;95(6):628-633. [69] ZHENG W, BIAN S, QIU S, et al. Placenta mesenchymal stem cell-derived extracellular vesicles alleviate liver fibrosis by inactivating hepatic stellate cells through a miR-378c/SKP2 axis. Inflamm Regen. 2023;43(1):47. [70] 徐小元,丁惠国,李文刚,等.肝硬化诊治指南[J].临床肝胆病杂志,2019,35(11):2408-2425. [71] 张东锋,王煜姝,腾军放.骨髓间充质干细胞联合青霉胺治疗肝豆状核变性肝纤维化的效果[J].实用医学杂志,2015,31(19):3195-3199. [72] TSUCHIYA A, TAKEUCHI S, WATANABE T, et al. Mesenchymal stem cell therapies for liver cirrhosis: MSCs as “conducting cells” for improvement of liver fibrosis and regeneration. Inflamm Regen. 2019;39:18. [73] SUK KT, YOON JH, KIM MY, et al. Transplantation with autologous bone marrow-derived mesenchymal stem cells for alcoholic cirrhosis: Phase 2 trial. Hepatology. 2016;64(6):2185-2197. [74] YU SJ, CHEN LM, LYU S, et al. Safety and efficacy of human umbilical cord derived-mesenchymal stem cell transplantation for treating patients with HBV-related decompensated cirrhosis. Zhonghua Gan Zang Bing Za Zhi. 2016;24(1):51-55. [75] LIN BL, CHEN JF, QIU WH, et al. Allogeneic bone marrow-derived mesenchymal stromal cells for hepatitis B virus-related acute-on-chronic liver failure: A randomized controlled trial. Hepatology. 2017;66(1):209-219. [76] ZHANG D. A clinical study of bone mesenchymal stem cells for the treatment of hepatic fibrosis induced by hepatolenticular degeneration. Genet Mol Res. 2017;16(1). doi: 10.4238/gmr16019352. [77] DETRY O, VANDERMEULEN M, DELBOUILLE MH, et al. Infusion of mesenchymal stromal cells after deceased liver transplantation: A phase I-II, open-label, clinical study. J Hepatol. 2017;67(1):47-55. [78] HUANG KC, CHUANG MH, LIN ZS, et al. Transplantation with GXHPC1 for Liver Cirrhosis: Phase 1 Trial. Cell Transplant. 2019;28(1_suppl): 100S-111S. [79] ZHANG K, SUN H, CAO H, et al. The impact of recipient age on the effects of umbilical cord mesenchymal stem cells on HBV-related acute-on-chronic liver failure and liver cirrhosis. Stem Cell Res Ther. 2021;12(1):466. [80] SHI M, LI YY, XU RN, et al. Mesenchymal stem cell therapy in decompensated liver cirrhosis: a long-term follow-up analysis of the randomized controlled clinical trial. Hepatol Int. 2021;15(6):1431-1441. [81] ZHANG K, JIA Y, SHU X, et al. Relationship Between Platelets and the Clinical Efficacy of Umbilical Cord Mesenchymal Stem Cells for HBV-Related Acute-on-Chronic Liver Failure and Liver Cirrhosis: A Preliminary Clinical Study. Stem Cells Transl Med. 2023;12(6):325-333. |
| [1] | Jiang Xianglong, Li Zhongshan, Che Tongtong. Application effects and mechanisms of low-frequency pulsed electromagnetic fields in muscle repair and growth [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(9): 2350-2360. |
| [2] | Zeng Xuan, Weng Rui, Ye Shicheng, Tang Jiadong, Mo Ling, Li Wenchao. Two lumbar rotary manipulation techniques in treating lumbar disc herniation: a finite element analysis of biomechanical differences [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(9): 2153-2161. |
| [3] | Jiang Xinghai, Song Yulin, Li Dejin, Shao Jianmin, Xu Junzhi, Liu Huakai, Wu Yingguo, Shen Yuehui, Feng Sicheng. Vascular endothelial growth factor 165 genes transfected into bone marrow mesenchymal stem cells to construct a vascularized amphiphilic peptide gel module [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 1903-1911. |
| [4] | Wu Yanting, Li Yu, Liao Jinfeng. Magnesium oxide nanoparticles regulate osteogenesis- and angiogenesis-related gene expressions to promote bone defect healing [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 1885-1895. |
| [5] | Hu Xiongke, Liu Shaohua, Tan Qian, Liu Kun, Zhu Guanghui. Shikonin intervention with bone marrow mesenchymal stem cells improves microstructure of femur in aged mice [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1609-1615. |
| [6] | Cai Ziming, Yu Qinghe, Ma Pengfei, Zhang Xin, Zhou Longqian, Zhang Chongyang, Lin Wenping. Heme oxygenase-1 alleviates lipopolysaccharide-induced inflammatory response in nucleus pulposus mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1624-1631. |
| [7] | Yuan Xiaoshuang, Yang Xu, Yang Bo, Chen Xiaoxu, Tian Ting, Wang Feiqing, Li Yanju, Liu Yang, Yang Wenxiu. Effect of conditioned medium of diffuse large B-cell lymphoma cells on proliferation and apoptosis of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1632-1640. |
| [8] | Li Zhenyu, Zhang Siming, Bai Jiaxiang, Zhu Chen. Osthole improves osteogenic differentiation function of bone marrow mesenchymal stem cells under high-glucose conditions [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1641-1648. |
| [9] | Han Nianrong, Huang Yifei, Akram · Osman, Liu Yanlu, Hu Wei . Programmed cell death receptor-1 suppresses osteogenic differentiation of rat bone marrow mesenchymal stem cells in a high-glucose microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1649-1657. |
| [10] | Jin Dongsheng, Zhao Zhanghong, Zhu Ziyin, Zhang Sen, Sun Zuyan, Deng Jiang. Effects of icariin-loaded microsphere-three-dimensional scaffold on osteogenic differentiation of rabbit bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1658-1668. |
| [11] | Zou Yulian, Chen Chaopei, Huang Haixia, Lan Yuyan, Liu Min, Huang Ting. Resveratrol promotes osteogenic differentiation of bone marrow mesenchymal stem cells in an inflammatory microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1669-1678. |
| [12] | Wang Qiuhua, Du Ziwei, Wang Wenshuang, Zhao Dongmei, Zhang Xiaoqing. Differences in metabolism, proliferation, differentiation of adipose-derived mesenchymal stem cells, and differentiation into vascular smooth muscle cells between male and female rats [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1687-1698. |
| [13] | He Jiale, Huang Xi, Dong Hongfei, Chen Lang, Zhong Fangyu, Li Xianhui. Acellular dermal matrix combined with adipose-derived stem cell exosomes promotes burn wound healing [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1699-1710. |
| [14] | Xia Linfeng, Wang Lu, Long Qianfa, Tang Rongwu, Luo Haodong, Tang Yi, Zhong Jun, Liu Yang. Human umbilical cord mesenchymal stem cell-derived exosomes alleviate blood-brain barrier damage in mice with septic encephalopathy [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1711-1719. |
| [15] | Ye Qianqian, Pan Hang, Tian Chuan, Zhu Xiangqing, Ye Li, Zhao Xiaojuan, Shu Liping, Pan Xinghua. Effects of highly active umbilical cord mesenchymal stem cells on structure and function of thymus in elderly tree shrews [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1720-1729. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||