Chinese Journal of Tissue Engineering Research ›› 2025, Vol. 29 ›› Issue (8): 1548-1555.doi: 10.12307/2025.326
Previous Articles Next Articles
Apelin-13 alleviates systemic inflammatory bone loss by inhibiting macrophage M1 polarization
Wang Wentao1, Hou Zhenyang2, Wang Yijun1, Xu Yaozeng1
- 1Department of Orthopedics, First Affiliated Hospital of Suzhou University, Suzhou 215006, Jiangsu Province, China; 2Department of Joint Sports Medicine, Tengzhou Central People’s Hospital, Tengzhou 277500, Shandong Province, China
-
Received:2024-03-20Accepted:2024-04-19Online:2025-03-18Published:2024-07-05 -
Contact:Xu Yaozeng, MD, Chief physician, Department of Orthopedics, First Affiliated Hospital of Suzhou University, Suzhou 215006, Jiangsu Province, China -
About author:Wang Wentao, Master, Department of Orthopedics, First Affiliated Hospital of Suzhou University, Suzhou 215006, Jiangsu Province, China -
Supported by:the National Natural Science Foundation of China, No. 82072498 (to XYZ)
CLC Number:
Cite this article
Wang Wentao, Hou Zhenyang, Wang Yijun, Xu Yaozeng. Apelin-13 alleviates systemic inflammatory bone loss by inhibiting macrophage M1 polarization[J]. Chinese Journal of Tissue Engineering Research, 2025, 29(8): 1548-1555.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
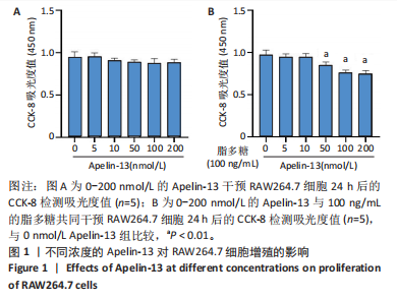
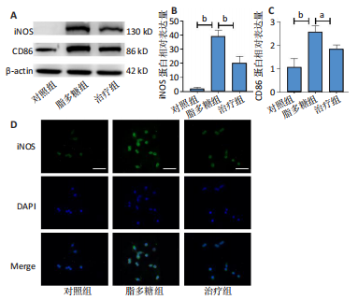
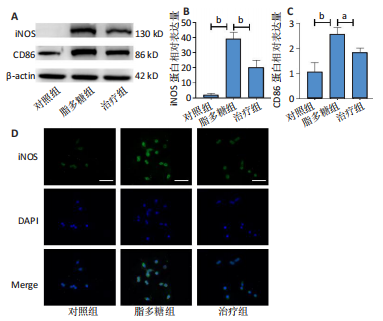
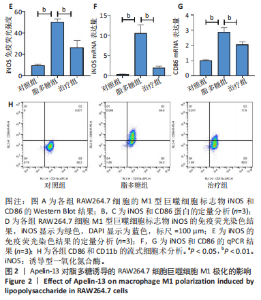
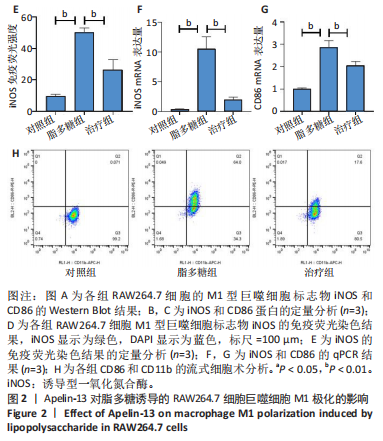
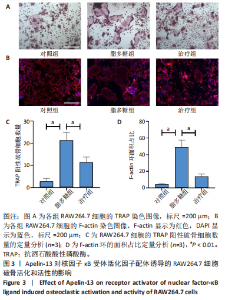
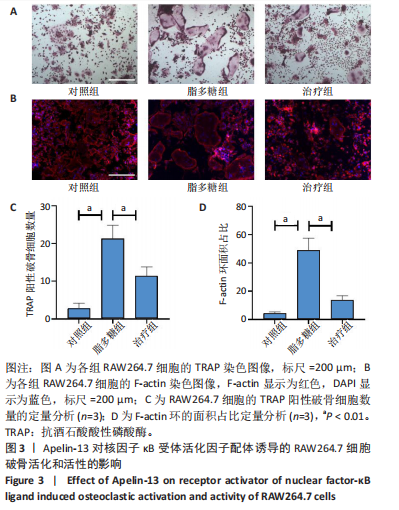
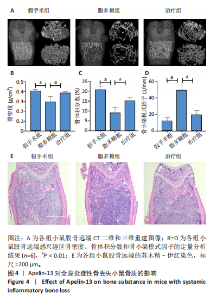
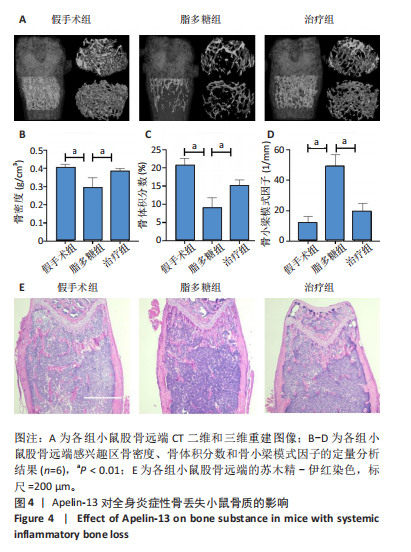
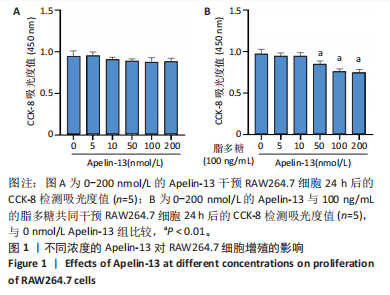
2.1 Apelin-13对巨噬细胞极化的影响 2.1.1 Apelin-13浓度的筛选 Apelin-13是一种内源性的配体,首先使用CCK-8确定Apelin-13干预RAW264.7细胞的最佳给药剂量。检测结果显示见图1A,各个浓度(0,5,10,50,100,200 nmol/L)下的Apelin-13对吸光度值均无显著影响,这也表明Apelin-13作为内源性配体的用药安全性。而当使用100 ng/mL的脂多糖与不同浓度(0,5,10,50,100,200 nmol/L)的Apelin-13共同干预RAW264.7细胞时,高于10 nmol/L药物浓度的Apelin-13即会抑制RAW264.7细胞的增殖。因此最终选择药物浓度为10 nmol/L的Apelin-13处理RAW264.7细胞,见图1B。 2.1.2 Western Blot分析结果 将RAW264.7细胞分成3组,即对照组,脂多糖组,治疗组。使用脂多糖(100 ng/mL)干预脂多糖组细胞来模拟炎性环境,并使用脂多糖(100 ng/mL)+Apelin-13(10 nmol/L)干预治疗组细胞来观察Apelin-13的治疗效果,24 h后对各组的M1型巨噬细胞型巨噬细胞标志物iNOS和CD86的表达情况进行Western Blot分析,结果显示,脂多糖组iNOS和CD86表达较对照组显著增加,而治疗组的iNOS和CD86表达则较脂多糖组出现明显降低,见图2A-C。这表明脂多糖能够显著促进巨噬细胞M1极化,而Apelin-13则能够有效抑制脂多糖诱导的巨噬细胞M1极化。 2.1.3 免疫荧光染色分析结果 各组细胞免疫荧光染色iNOS的表达结果显示,对照组iNOS的荧光强度非常低,而脂多糖组的荧光强度显著增强,这表明脂多糖显著促进了RAW264.7细胞的M1极化,而Apelin-13治疗组的荧光强度较脂多糖组显著降低,表现出与Western Blot结果相同的趋势,见图2D-E。 2.1.4 qPCR和细胞流式分析结果 各组M1型巨噬细胞标志物iNOS和CD86的mRNA表达结果显示,脂多糖组iNOS和CD86的mRNA表达较对照组显著升高,而治疗组M1型巨噬细胞标志物inos和cd86的mRNA的表达水平较脂多糖组出现显著降低,这与Western Blot的结果相一致,见图2F-G。CD11b和CD86分别是巨噬细胞和M1型巨噬细胞特异性表面抗原,各组流式结果显示,脂多糖组M1型巨噬细胞的比例较治疗组明显升高(0.071∶64.0),而Apelin-13治疗组M1型巨噬细胞的比例较脂多糖组显著减少(64.0∶17.6)。这表明Apelin-13能够显著减少脂多糖诱导的M1型巨噬细胞的比例,见图2H。 综上所述,结果发现Apelin-13能够有效抑制脂多糖诱导的巨噬细胞M1极化。 2.2 Apelin-13对炎症诱导的破骨细胞活化的影响 为了进一步探究Apelin-13调控巨噬细胞M1极化是否会对破骨细胞的活化和活性产生影响,在对照组、脂多糖组和治疗组完成干预后,加入RANKL(50 ng/mL)诱导破骨细胞,诱导6 d后进行TRAP和F-actin染色,来进一步探究Apelin-13能否通过调控巨噬细胞极化影响破骨细胞的活化。 2.2.1 TRAP染色结果 见图3A和C。结果表明,有更多M1极化巨噬细胞的脂多糖组较对照组诱导出更多的TRAP染色阳性的破骨细胞,在镜下呈现融合的多核细胞(细胞核数量≥3个),而治疗组TRAP阳性的破骨细胞数量较脂多糖组显著减少,结果与治疗组的M1型巨噬细胞减少趋势相同。这一结果表明,脂多糖诱导的炎性环境引起的M1型巨噬细胞极化容易导致破骨细胞的激活,而Apelin-13能够通过抑制巨噬细胞M1极化来抑制破骨细胞的异常激活。 2.2.2 F-actin染色结果 见图3B和D。脂多糖组经破骨诱导后出现较对照组明显更大的F-actin环,而F-actin环的大小与破骨细胞的破骨活性相关。经Apelin-13干预后,治疗组的F-actin环较脂多糖组显著缩小,结果表明Apelin-13能够显著抑制脂多糖诱导的炎性环境下巨噬细胞M1极化促进的破骨细胞的破骨活性。 综上所述,结果发现Apelin-13能够有效抑制脂多糖炎症诱导的巨噬细胞M1极化促进的破骨细胞的活化和破骨活性。 2.3 Apelin-13对全身炎症性骨丢失小鼠的影响 在上述实验中,发现Apelin-13能够抑制巨噬细胞M1极化,并抑制了体外炎症环境下破骨细胞的活化和破骨活性,基于此,拟通过体内实验来进一步验证Apelin-13对全身炎症性骨丢失小鼠中是否具有与体外实验相似的治疗效果。 2.3.1 实验动物数量分析 实验选用的18只雄性C57BL/6小鼠,实验过程均无脱失,全部进入结果分析。 2.3.2 Micro-CT的二维和三维重建结果 脂多糖诱导全身炎症性骨丢失小鼠模型获得成功,脂多糖组全身炎症小鼠的股骨远端骨质较假手术组出现严重的流失,骨小梁数量和密度显著减少,见图4A。骨参数分析显示,脂多糖组骨密度较假手术组出现显著降低,同样降低的还有骨体积分数,脂多糖组的骨小梁模式因子则较假手术组升高,而Apelin-13治疗组则较脂多糖组有着明显升高的骨密度和骨体积分数,治疗组的骨小梁较脂多糖组也出现增加,见图4B-D。 2.3.3 苏木精-伊红染色结果 对小鼠股骨远端的苏木精-伊红染色可以看出(图4E),脂多糖组的骨小梁较假手术组明显减少,而Apelin-13治疗组的骨小梁数量稍有恢复。 鉴于上述重建、骨参数分析及苏木精-伊红染色的结果,发现全身炎症性小鼠骨质出现明显丢失,而Apelin-13能够减轻全身炎症性骨丢失。"
| [1] FISCHER V, HAFFNER-LUNTZER M. Interaction between bone and immune cells: Implications for postmenopausal osteoporosis. Semin Cell Dev Biol. 2022;123:14-21. [2] MICHALSKI MN, MCCAULEY LK. Macrophages and skeletal health. Pharmacol Ther. 2017;174:43-54. [3] KIM HJ.Glucocorticoids suppress bone formation via the osteoclast. J Clin Invest. 2006;116:2152-2160. [4] DURU N, VAN DER GOES MC, JACOBS JWG, et al. EULAR evidence-based and consensus-based recommendations on the management of medium to high-dose glucocorticoid therapy in rheumatic diseases. Ann Rheum Dis. 2013;72:1905-1913. [5] HARDY RS, ZHOU H, SEIBEL MJ, et al. Glucocorticoids and Bone: Consequences of Endogenous and Exogenous Excess and Replacement Therapy. Endocr Rev. 2018;39:519-548. [6] GADO M, BASCHANT U, HOFBAUER LC, et al. Bad to the Bone: The Effects of Therapeutic Glucocorticoids on Osteoblasts and Osteocytes. Front Endocrinol. 2022;13:835720. [7] YAO Y, CAI X, REN F, et al. The Macrophage-Osteoclast Axis in Osteoimmunity and Osteo-Related Diseases. Front Immunol. 2021;12: 664871. [8] MUÑOZ J, AKHAVAN NS, MULLINS AP, et al. Macrophage Polarization and Osteoporosis: A Review. Nutrients. 2020;12:2999. [9] NAKAO Y, FUKUDA T, ZHANG Q, et al. Exosomes from TNF-α-treated human gingiva-derived MSCs enhance M2 macrophage polarization and inhibit periodontal bone loss. Acta Biomater. 2021;122:306-324. [10] LOCATI M, CURTALE G, MANTOVANI A. Diversity, Mechanisms, and Significance of Macrophage Plasticity. Annu Rev Pathol Mech Dis. 2020;15:123-147. [11] VAN DEN BOSSCHE J, O’NEILL LA, MENON D. Macrophage Immunometabolism: Where Are We (Going)? Trends Immunol. 2017; 38:395-406. [12] WANG Q, BU Q, LIU M, et al. XBP1-mediated activation of the STING signalling pathway in macrophages contributes to liver fibrosis progression. JHEP Rep. 2022;4:100555. [13] TSAI ML, TSAI YG, LIN YC, et al. IL-25 Induced ROS-Mediated M2 Macrophage Polarization via AMPK-Associated Mitophagy. Int J Mol Sci. 2021;23:3. [14] ARABABADI MK, ASADIKARAM P, ASADIKARAM G. APLN/APJ pathway: The key regulator of macrophage functions. Life Sci. 2019;232:116645. [15] HE L, ZHOU Q, HUANG Z, et al. PINK1/Parkin‐mediated mitophagy promotes apelin‐13‐induced vascular smooth muscle cell proliferation by AMPKα and exacerbates atherosclerotic lesions.J Cell Physiol. 2019;234:8668-8682. [16] SHAO Z, DOU S, ZHU J, et al. Apelin-36 Protects HT22 Cells Against Oxygen-Glucose Deprivation/Reperfusion-Induced Oxidative Stress and Mitochondrial Dysfunction by Promoting SIRT1-Mediated PINK1/Parkin-Dependent Mitophagy. Neurotox Res. 2021;39:740-753. [17] CHEN L, SHI X, XIE J, et al. Apelin-13 induces mitophagy in bone marrow mesenchymal stem cells to suppress intracellular oxidative stress and ameliorate osteoporosis by activation of AMPK signaling pathway. Free Radic Biol Med. 2021;163:356-368. [18] HANG K, YE C, XU J, et al. Apelin enhances the osteogenic differentiation of human bone marrow mesenchymal stem cells partly through Wnt/β-catenin signaling pathway. Stem Cell Res Ther. 2019;10:189. [19] YIN Z, CHENG Q, WANG C, et al. Apelin‐13 alleviates osteoclast formation and osteolysis through Nrf2‐pyroptosis pathway. Microsc Res Tech. 2024;87(6):1348-1358. [20] REDLICH K, SMOLEN JS. Inflammatory bone loss: pathogenesis and therapeutic intervention. Nat Rev Drug Discov. 2012;11:234-250. [21] ADAMI G. Regulation of bone mass in inflammatory diseases. Best Pract Res Clin Endocrinol Metab. 2022;36:101611. [22] CHEN Z, BOZEC A, RAMMING A, et al. Anti-inflammatory and immune-regulatory cytokines in rheumatoid arthritis. Nat Rev Rheumatol. 2019;15:9-17. [23] LIU P, GAO Y, LUO P, et al. Glucocorticoid-induced expansion of classical monocytes contributes to bone loss. Exp Mol Med. 2022;54:765-776. [24] CHEN M, FU W, XU H, et al. Pathogenic mechanisms of glucocorticoid-induced osteoporosis. Cytokine Growth Factor Rev. 2023;70:54-66. [25] ULUÇKAN Ö, JIMENEZ M, KARBACH S, et al. Chronic skin inflammation leads to bone loss by IL-17–mediated inhibition of Wnt signaling in osteoblasts. Sci Transl Med. 2016;8(330):330ra37. [26] VIEIRA LV, DE SOUSA LM, MAIA TAC, et al. Milk Kefir therapy reduces inflammation and alveolar bone loss on periodontitis in rats. Biomed Pharmacother. 2021;139:111677. [27] YANG D, WAN Y. Molecular determinants for the polarization of macrophage and osteoclast. Semin Immunopathol. 2019;41:551-563. [28] FENG X, ZHU S, QIAO J, et al. CX3CL1 promotes M1 macrophage polarization and osteoclast differentiation through NF-κB signaling pathway in ankylosing spondylitis in vitro. J Transl Med. 2023;21(1):573. [29] CHEN M, WANG Y, SUN B, et al. HIF-1α activator DMOG inhibits alveolar bone resorption in murine periodontitis by regulating macrophage polarization.Int Immunopharmacol. 2021:99:107901. [30] MAHON OR, BROWE DC, GONZALEZ-FERNANDEZ T, et al. Nano-particle mediated M2 macrophage polarization enhances bone formation and MSC osteogenesis in an IL-10 dependent manner. Biomaterials. 2020;239:119833. [31] TOITA R, KANG JH, TSUCHIYA A. Phosphatidylserine liposome multilayers mediate the M1-to-M2 macrophage polarization to enhance bone tissue regeneration. Acta Biomater. 2022;154:583-596. [32] SCHLUNDT C, FISCHER H, BUCHER CH, et al. The multifaceted roles of macrophages in bone regeneration: A story of polarization, activation and time. Acta Biomater. 2021;133:46-57. [33] YAN J, WANG A, CAO J, et al. Apelin/APJ system: an emerging therapeutic target for respiratory diseases. Cell Mol Life Sci. 2020;77: 2919-2930. [34] XU W, LI T, GAO L, et al. Apelin-13/APJ system attenuates early brain injury via suppression of endoplasmic reticulum stress-associated TXNIP/NLRP3 inflammasome activation and oxidative stress in a AMPK-dependent manner after subarachnoid hemorrhage in rats. J Neuroinflammation. 2019;16(1):247. [35] İZGÜT-UYSAL VN, GEMICI B, BIRSEN İ, et al. The Effect of Apelin on the Functions of Peritoneal Macrophages. Physiol Res. 2017;66(3):489-496. [36] HAN S, ENGLANDER EW, GOMEZ GA, et al. Apelin Regulates Nuclear Factor-κB’s Involvement in the Inflammatory Response of Pancreatitis. Pancreas. 2017;46:64-70. [37] GARCIA‐DIAZ DF, CAMPION J, QUINTERO P, et al. Vitamin C modulates the interaction between adipocytes and macrophages. Mol Nutr Food Res. 2011;55 Suppl 2:S257-63. [38] ZHOU H, YANG R, WANG W, et al. Fc-apelin fusion protein attenuates lipopolysaccharide-induced liver injury in mice. Sci Rep. 2018;8(1): 11428. [39] TRANG NTN, LAI CY, TSAI HC, et al. Apelin promotes osteosarcoma metastasis by upregulating PLOD2 expression via the Hippo signaling pathway and hsa_circ_0000004/miR-1303 axis. Int J Biol Sci. 2023; 19(2):412-425. [40] CHEN JC, SHIH HC, LIN CY, et al. MicroRNA-631 Resensitizes Doxorubicin-Resistant Chondrosarcoma Cells by Targeting Apelin. Int J Mol Sci. 2023;24(1):839. [41] CHAE SA, DU M, SON JS, et al. Exercise improves homeostasis of the intestinal epithelium by activation of apelin receptor–AMP‐activated protein kinase signalling. J Physiol. 2023;601(12):2371-2389. |
| [1] | You Huijuan, Wu Shuzhen, Rong Rong, Chen Liyuan, Zhao Yuqing, Wang Qinglu, Ou Xiaowei, Yang Fengying. Macrophage autophagy in lung diseases: two-sided effects [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1516-1526. |
| [2] | Cao Wenqi, Feng Xiuzhi, Zhao Yi, Wang Zhimin, Chen Yiran, Yang Xiao, Ren Yanling. Effect of macrophage polarization on osteogenesis-angiogenesis coupling in type 2 diabetic osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 917-925. |
| [3] | Xie Peisen, Guan Zhenpeng, Wei Xianjie, Zhang Keshi, Kang Qingyuan, Xiao Wentao, Guo Xiaoshuai. Cerium dioxide nanoparticles regulate expression of inflammatory factors in M1 macrophages and affect fibroblast co-culture system [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(2): 375-383. |
| [4] | Chang Jinxia, Liu Yufei, Niu Shaohui, Wang Chang, Cao Jianchun. Visualization analysis of macrophage polarization in tissue repair process [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(7): 1486-1496. |
| [5] | Wang Sifan, He Huiyu, Yang Quan, Han Xiangzhen. miRNA-378a overexpression of macrophage cell line composite collagen sponge: anti-inflammation and tissue repair promotion [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(4): 789-799. |
| [6] | Zhao Jianwei, Li Xunsheng, Lyu Jinpeng, Zhou Jue, Jiang Yidi, Yue Zhigang, Sun Hongmei. Deer antler stem cell exosome composite hydrogel promotes the repair of burned skin [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(34): 7344-7352. |
| [7] | Yao Lanxuan, Wang Xuefei, Liu Yang, Yang Yujia, Zhao Yi, Qi Fangfang, Li Yinghui . Mesenchymal stem cells and their derived extracellular vesicles target macrophages to intervene in autoimmune diseases [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(31): 6772-6781. |
| [8] | Chen Yixin, Lu Yan, Zhang Xuan, Chen Xiaoli, Tan Liangyuan, Xu Zhangjie, Chen Wanglong, Su Shaoting, Liang Jiyao, Zhou Honghai. Mechanism by which Tongan Decoction regulates synovial macrophage polarization in rats with knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(26): 5621-5631. |
| [9] | Zou Shunyi, Chai yuan, Li Kunjian. Involvement of macrophage polarization in osteoarticular diseases: a visual analysis based on SCI-Expanded information [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(24): 5245-5253. |
| [10] | Cao Panxia, Peng Zining, Liu Shanshan, Fei Tiantian, Liang Tengyun, Zhang Mengwen, Wu Hong. AMP-activated protein kinase mediates macrophage fatty acid oxidation: an approach to prevent and treat atherosclerosis with traditional Chinese medicine [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(18): 3906-3914. |
| [11] | Zhao Yuqing, Wang Wei, You Huijuan, Chen Liyuan, Chen Yan, Wang Qinglu, Yang Fengying. Relationship between macrophage subtypes in obese adipose tissue and metabolic diseases [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(13): 2832-2841. |
| [12] | Yang Peng, Zhang Wei, Li Wenming, Li Wenhao, Wu Zebin, Zhou Jun, Geng Dechun. Linagliptin alleviates wear particle-induced inflammatory osteolysis by regulating macrophage polarization and osteoclast formation [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(12): 2421-2428. |
| [13] | Ge Ruiyang, Ni Can, Yang Kun, Yan Fuhua. The role of macrophage polarization in the pathogenesis and treatment of periodontitis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(20): 3246-3251. |
| [14] | Dong Hongfei, Huang Xi, Li Xianhui, Zhang Yanbiao, Wang Xuyang, Wang Bing, Sun Hongyu. Placenta-derived mesenchymal stem cells in promoting acute skin wound healing in rats [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(13): 2047-2053. |
| [15] | Zhang Wei, Yu Lei, Yang Peng, Geng Dechun. Tabersonine alleviates wear particle-induced inflammatory osteolysis by inhibiting osteoclast activation [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(10): 1519-1525. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||