Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (2): 283-288.doi: 10.12307/2022.046
Previous Articles Next Articles
Expression and role of cannabinoid receptors during bone remodeling
Fan Danyang1, Fu Runze1, Mi Jiajing1, Liu Chunyan2
- 1Hebei Medical University, Shijiazhuang 050017, Hebei Province, China; 2Department of Orthodontics, School of Stomatology, Hebei Medical University, Hebei Key Laboratory of Stomatology, Hebei Provincial Oral Disease Research Center, Shijiazhuang 050017, Hebei Province, China
-
Received:2020-11-12Revised:2020-11-14Accepted:2020-12-25Online:2022-01-18Published:2021-10-28 -
Contact:Liu Chunyan, MD, Associate chief physician, Associate professor, Department of Orthodontics, School of Stomatology, Hebei Medical University, Hebei Key Laboratory of Stomatology, Hebei Provincial Oral Disease Research Center, Shijiazhuang 050017, Hebei Province, China -
About author:Fan Danyang, Hebei Medical University, Shijiazhuang 050017, Hebei Province, China -
Supported by:Talent Support Plan of Hebei Provincial Department of Human Resources and Social Security, No. A201901036 (to LCY); Geriatrics Prevention and Control Research Project of Hebei Provincial Department of Finance, No. 361029 (to LCY)
CLC Number:
Cite this article
Fan Danyang, Fu Runze, Mi Jiajing, Liu Chunyan. Expression and role of cannabinoid receptors during bone remodeling[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(2): 283-288.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
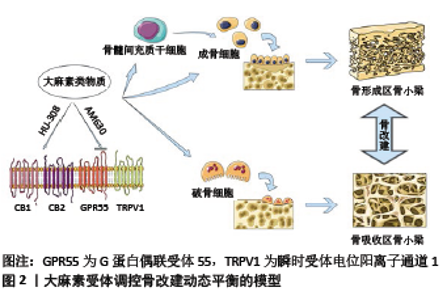
2.1 大麻素受体与骨改建的关系 2.1.1 CB1受体与骨改建的关系 交感神经纤维在骨组织,特别是在骨小梁上密集分布,这些神经纤维与成骨细胞形成突触样结构[15-18]。TAM等[18-19]研究发现,CB1受体在交感神经纤维上存在免疫反应性,在CB1缺失的小鼠中则无此效应,这提示CB1受体存在于支配骨小梁的交感神经纤维中。在交感神经末梢,当突触前膜分泌的去甲肾上腺素激活成骨细胞上的β-2肾上腺素能受体时,成骨细胞的数量和活性就会受到抑制,导致骨形成减少[20]。CB1受体可以与内源性大麻素2-花生四烯酸甘油结合,抑制去甲肾上腺素的释放,这提示骨交感神经末梢的CB1受体可能通过抑制去甲肾上腺素的释放来刺激成骨细胞活性,从而减轻去甲肾上腺素对骨形成的抑制作用[21]。同一研究发现,在创伤性脑损伤模型中,CB1信号能促进外周成骨和骨折愈合,强烈刺激股骨远端部位的骨形成,这主要是由于合成2-花生四烯酸甘油必需的二酰甘油脂肪酶在股骨远端部位表达增强,使2-花生四烯酸甘油水平升高,激活并结合CB1受体,抑制骨交感神经末梢去甲肾上腺素释放和去甲肾上腺素与β2-肾上腺素能受体的结合,从而促进骨形成过程。 另一项研究发现,在C57BL/6J小鼠中,CB1受体的整体缺失会导致骨小梁数量减少[18],表现为低骨量表型,但上述研究结果的获得基于小鼠CB1受体的整体缺失,故其低骨量表型除与骨交感神经末梢CB1受体的单独缺失有关外,还可能与成骨细胞、破骨细胞以及中枢神经元等处的CB1受体缺失相关。在最近的一项研究中,BUSQUETS-GARCIA等[21]利用一种在肾上腺素能/去甲肾上腺素能细胞中缺乏CB1受体的小鼠(DBH-CB1-KO小鼠),研究骨中交感神经末梢CB1受体单独缺失对骨的影响,结果显示这种CB1受体的条件性缺失上调了老年小鼠的骨形成和骨量,表明交感神经CB1受体结构的特异性缺失扰乱了骨组织中的2-花生四烯酸甘油-去甲肾上腺素负反馈回路,并可能通过一种与骨微环境中交感神经纤维去甲肾上腺素释放无关的机制来调节小鼠衰老过程中的骨重建过程。 CB1受体也可通过中枢神经系统调节骨改建过程。CB1受体表达于下丘脑腹内侧核,中枢脂联素可通过下调CB1的表达,增加小鼠骨小梁的数量,脑室注射CB1受体激动剂和拮抗剂能分别减弱和增强中枢脂联素对骨形成的诱导。同时,球状脂联素能增强各种组蛋白去乙酰化酶,尤其是组蛋白去乙酰化酶5的表达水平,从而增强组蛋白去乙酰化酶5与CB1启动子的转录起始位点的结合,这表明可能存在一种中枢APN-HDAC5-CB1信号机制,它能通过对下丘脑CB1表达的表观遗传调控来促进周围骨的形成[22]。 除上述两种途径外,CB1受体还可表达在骨细胞中。在成骨前体细胞,如骨髓间充质干细胞中检测到了CB1 mRNA的表达,同时特异性CB1受体激动剂ACEA对骨髓间充质干细胞表现出显著的归巢反应,而CB2受体选择性激动剂JWH133对骨髓间充质干细胞的归巢则无影响[23]。在人单核细胞、成熟破骨细胞能检测到CB1受体的表达,并且CB1 mRNA的表达在破骨细胞分化过程中保持不变[24]。小鼠成骨细胞和破骨细胞均可表达CB1受体[25-26],但目前的技术并未在人类成骨细胞当中检测到CB1受体的表达[13,27-29]。CB1受体在小鼠中可同时调节成骨细胞和破骨细胞的活性[25,30-31]。具体而言,CB1受体可以通过调节破骨细胞和骨髓间充质干细胞向成骨细胞和脂肪细胞的分化对骨改建产生影响。表达在破骨细胞上的CB1受体在骨改建中发挥着重要的作用。IDRIS等[29]研究发现,人工合成的CB1受体拮抗剂AM251在体外可抑制破骨细胞的形成,AM251在体内可通过抑制破骨细胞性骨吸收来保护卵巢切除所致的骨丢失。与之相反,内源性大麻素激动剂anadamide在体外促进破骨细胞形成,逆转了AM251对于破骨细胞形成的抑制作用。同时,与野生型小鼠相比,CB1基因敲除小鼠产生的破骨细胞能够耐受AM251的抑制作用。SAMIR等[32]展示了抑制CB1受体后幼鼠RANKL基因表达减少和骨保护素表达增加。IDRIS 等[29]的研究提示,CB1基因缺陷小鼠破骨细胞分化缺陷是由于RANKL表达降低,从而削弱了成骨细胞支持破骨细胞分化的能力。以上研究证明CB1受体的激活可以促进破骨细胞形成和骨吸收。 大量研究发现,CB1受体缺失小鼠的骨骼表型不尽相同,这可能与小鼠品系、性别和年龄等因素有关。TAM等[18]研究发现,与野生型小鼠相比,雄性和雌性C57CB1-/-小鼠均表现为低骨量表型;而CD1CB1-/-小鼠骨骼表型则表现出明显的性别差异:雄性CD1CB1-/-小鼠具有明显的高骨量表型,并伴有骨小梁厚度增加;雌性CD1CB1-/-小鼠则表现为骨干轻度异常,皮质骨扩张以及骨干和髓腔直径的增加。IDRIS等[29]研究发现,CD1CB1-/-雌性幼鼠表现出高骨量表型,并可防止卵巢切除引起的骨丢失;同一研究小组发现,CD1CB1-/-小鼠出现了年龄相关性骨丢失增加[32]。这表明CB1受体的激活在小鼠体内会引起幼年动物的骨质丢失,但可以预防晚年与年龄相关的骨质疏松症[26],而年轻的C57BL/6JCB1-/-小鼠中出现了低骨量表型[18],提示在C57遗传背景下的CB1受体似乎发挥着与年龄无关的骨调节作用。SAMIR等[32]通过研究发现,CB1受体拮抗剂Rimonabant能减轻青年大鼠糖皮质激素应用后的骨质疏松症,但却会加重老年大鼠的骨质疏松程度,这些数据表明CB1与骨改建的关系可能与年龄有关[25-26,32]。 CB1受体对机体不同部位的骨改建调节也存在差异。有研究表明,轻微创伤性脑损伤对股骨产生的成骨效应持续时间短暂[19],而对颅骨则相对较长[33]。WASSERMAN等[34]报道了CB1受体在骨骼生长中的作用,CB1受体表达于促进脊椎动物骨骼生长的骨骺生长软骨的肥大软骨细胞中,研究发现,Δ-9-四氢大麻酚对野生型小鼠和CB2-/-小鼠的股骨和腰椎体骨骼生长有抑制作用,但对CB1-/-小鼠则无明显影响,这说明四氢大麻酚对软骨内骨骼生长的抑制是由CB1受体介导的。 综上所述,CB1受体对骨改建的调节可能受多种机制和途径的影响,其具体作用机制仍有待进一步研究。 2.1.2 CB2受体与骨改建的关系 MUNRO等[9]在1993年首次克隆出CB2受体,该受体主要分布在外周的免疫细胞表面。随后有研究在中枢神经系统的不同区域如脊髓、背根神经节和小胶质细胞中检测到了CB2受体的表达[7,35]。与CB1受体相比,CB2受体在成骨细胞、破骨细胞、骨细胞中表达较高。OFEK等[28]的研究报道了CB2 mRNA在小鼠骨髓基质细胞、MC3T3-E1成骨细胞、骨髓来源的破骨细胞及其前体细胞、RAW264.7来源破骨细胞样细胞中的表达,以及CB2受体在野生型小鼠成骨细胞、破骨细胞、骨细胞中的显著表达。此外,CB2受体在人破骨细胞中的表达水平要明显低于单核细胞,并且在破骨细胞分化过程中显著降低[24]。钱红等[36-37]研究发现,人牙周膜细胞也表达CB2受体,且其激活能够促进人牙周膜细胞的成骨分化。 CB2受体主要通过影响成骨细胞分化和活性来影响骨形成。SOPHOCLEOUS等[38]的研究表明,CB2选择性激动剂HU-308可促进野生型小鼠成骨细胞的骨结节形成,但对 CB2-/-小鼠的成骨细胞无效。进一步研究表明,在MC3T3-E1成骨样细胞中,HU-308促进细胞迁移并激活ERK磷酸化,而这些作用可被CB2选择性反向激动剂AM630阻断。在野生型小鼠体内,HU-308可通过刺激骨形成减少卵巢切除引起的骨丢失。与之不同的是,OFEK等[28]的研究显示,这种作用主要归因于骨吸收被抑制。成骨细胞是来源于骨髓间充质干细胞并由RUNX2诱导而来的单核细胞,ZHANG等[39-40]通过研究缺氧条件下CB2受体在骨髓间充质干细胞成骨分化中的作用发现,CoCl2诱导的缺氧使骨钙素、RUNX2等表达水平明显增加,而CB2拮抗剂AM630可部分抑制缺氧诱导的p38和ERK通路,降低RUNX2转录水平,这提示在缺氧条件下,CB2受体参与大鼠骨髓间充质干细胞的成骨分化过程,胡旭治等[41]的研究也得出了相似的结论;CB2受体在恢复骨质疏松症患者骨髓间充质干细胞成骨分化和矿化中也起着重要作用。慢病毒可以诱导CB2受体在骨质疏松患者骨髓间充质干细胞中过表达,CB2过表达能提高碱性磷酸酶的活性,促进成骨基因表达和细胞外基质的矿化沉积,也能增加p38丝裂原活化蛋白激酶的磷酸化,促进骨髓间充质干细胞的成骨分化[42]。有研究发现,CB2的活化能抑制钛粒诱导下MC3T3-E1成骨细胞RANKL的表达,使骨保护素/RANKL比值上升,促进碱性磷酸酶和骨钙素的表达,提高成骨细胞活性,从而促进骨形成[43]。此外,大麻素可能通过CB2介导辅助细胞,间接地刺激骨髓间充质干细胞从骨髓中募集[44]。近来在基因水平的研究发现,miR-187-3p可以通过靶向CB2受体基因的3’非翻译区(UTR)来抑制CB2受体基因的表达,而上调CB2受体基因表达可逆转miR-187-3p对hFOB1.19成骨分化的抑制作用[45]。综上所述,激活CB2受体可以提高成骨细胞活性、促进分化以及相关酶和细胞因子的表达,调节骨形成过程。 CB2受体可以通过调节破骨细胞活性影响骨吸收,但到目前为止,该领域的研究仍存在着争议。OFEK等[28]的研究表明,由于骨转换增加,CB2-/-小鼠会随着年龄的增长而出现骨质疏松的症状,CB2选择性激动剂HU-308既可以抑制骨髓中RANKL诱导的破骨细胞的产生,也可抑制体外RAW264.7培养中RANKL诱导的破骨细胞的形成。另外,ZHU等[46]研究发现,CB2受体激动剂JWH133可以预防类风湿性关节炎局部和全身炎症性骨破坏;ROSSI等[47]利用17-雌二醇对人破骨细胞作用的体外实验发现雌激素可以增加大麻素CB2受体的表达抑制破骨细胞活性。与上述结果相反,TAM等[25]发现,在1-1 000 nmol/L的浓度范围内,花生四烯乙醇胺、2-花生四烯酸甘油、HU-308以及JWH133可促进巨噬细胞集落刺激因子和RANKL诱导的破骨细胞形成,CB2反向激动剂AM630则对破骨细胞的形成具有抑制作 用[25,48]。进一步研究表明,与野生型小鼠相比,从CB2缺陷小鼠中分离的骨髓细胞对RANKL的反应较弱,产生的破骨细胞较少,可减少卵巢切除引起的骨丢失的影响。SCHUEHLY等[49]发现一种新的具有高度选择性的CB2配体,在体外培养过程中,这种配体可以强烈抑制RANKL诱导的小鼠和人类破骨细胞生成,同时证明内源性大麻素可以刺激破骨细胞的形成。在体内,CB2受体拮抗剂可以抑制成年小鼠破骨细胞的形成,减少骨丢失。IDRIS等[48]还发现,AM630可以预防卵巢切除引起的骨丢失,并且这一反应依赖于给药剂量。此外,LUNN等[50]报告称新型CB2选择性拮抗剂Sch.036可以预防关节炎小鼠的骨损伤。 ROSS[51]报道了CB2选择性拮抗剂AM630在高浓度 (10 μmol/L)时刺激人破骨细胞形成,这与IDRIS等[48]报道的AM630对小鼠培养的破骨细胞形成的抑制作用完全相反。其原因尚不清楚,可能与AM630作用的物种差异性、所使用的高浓度AM630的非靶点效应、以及不同的血清选择等因素有关[52]。 除此之外,CB2受体缺失小鼠的骨骼表型不尽相同,这可能与小鼠品系、年龄和性别等因素有关。SOPHOCLEOUS等[53]将Cnr2-/-CD1小鼠与Cnr2-/-C57BL/6小鼠进行了比较,研究发现,Cnr2-/-C57BL/6小鼠的骨小梁量与野生型相似,而年轻雌性Cnr2-/-CD1小鼠较野生型小鼠具有较低的骨转换率和较高的骨小梁量。在雄性小鼠中,Cnr2-/-和野生型的骨骼表型没有显著差异,但两种性别的Cnr2-/-和野生型小鼠的皮质骨表型相似。随着年龄的增长,C57BL/6Cnr2-/-小鼠和Cnr2-/-CD1小鼠较野生型小鼠骨量减少,而在12个月时,Cnr2-/-CD1小鼠和野生型小鼠之间的松质骨体积却没有差异。 2.1.3 GPR55受体与骨改建的关系 许多实验发现CB1和CB2基因敲除动物中仍然保留着大麻素类效应,这提示体内存在着除上述两种受体以外的大麻素受体。目前已经确定了的其他大麻素受体包括GPR55和瞬时受体电位阳离子通道1 [54-55]。 GPR55受体基因定位于人类2q37号染色体上,其首次发现于中枢神经系统[56],随着研究的不断深入,GPR55也被证明在骨代谢中发挥相关作用[57]。WHYTE等[57]发现在依赖巨噬细胞集落刺激因子单核细胞来源的人破骨细胞、人和小鼠多核破骨细胞和原代成骨细胞以及人TE85成骨样细胞中均能检测到GPR55 mRNA的表达,GPR55 mRNA在人破骨细胞中的表达高于人破骨前体细胞。进一步研究发现,GPR55受体激动剂O-1602对体外培养小鼠破骨细胞的形成有抑制作用,而对人破骨细胞的形成无影响;GPR55受体拮抗剂CBD却能显著促进人破骨细胞的形成[57]。OSSOLA等[58]研究证实CBD能以时间和浓度依赖的方式促进脂肪来源间充质干细胞的募集,而GPR55受体激动剂O-1602可抑制CBD诱导的募集,阻碍间充质干细胞的成骨分化,这提示GPR55受体可能在成骨细胞分化和破骨细胞形成过程中发挥着不同的作用。 2.1.4 瞬时受体电位阳离子通道1受体与骨改建的关系 研究发现瞬时受体电位阳离子通道1在来源于人外周血单核细胞的破骨细胞中表达,并且通过提高花生四烯乙醇胺水平,可以引起瞬时受体电位阳离子通道1介导的对破骨细胞形成的刺激作用[59]。此外,瞬时受体电位阳离子通道1也存在于人骨髓间充质干细胞上,与CB2受体在体外对人成骨细胞活性的促进作用相反,瞬时受体电位阳离子通道1受体激活后会抑制成骨细胞活性,阻碍骨形成。 CB1、CB2、GPR55以及瞬时受体电位阳离子通道1受体均属于7次跨膜蛋白,在体内外可以与不同配体结合参与骨改建过程。大麻素受体与其配体的亲和力与其种类密切相关,不同配受体间的结合以及受体间的联合作用可对成骨细胞、破骨细胞和骨髓间充质干细胞的分化和活性产生不同的生物学效应,见图2。成骨、破骨细胞将这种复杂的效应转换成对骨形成和骨吸收过程的精密调节,使大麻素系统在骨改建的动态平衡中起到了重要的调控作用,为临床骨相关疾病的治疗和研究提供了新的思路。 2.2 大麻素受体与骨质疏松 骨质疏松症是一种因骨骼强度丧失导致脆性骨折的疾病,其主要特征为骨形成缺陷和骨吸收过多,使得骨量丢失和骨骼结构破坏。骨质疏松症的常见病因为缺乏雌激素和应用糖皮质激素治疗,这两者都与骨吸收增强和骨形成减少有关[60]。 雌激素缺乏会使妇女绝经后立即进入加速的骨丢失阶段,从而导致骨吸收和骨形成的失衡[61]。近些年大麻素受体被认为对雌激素缺乏性骨质疏松症有调节作用。研究发现CB1选择性拮抗剂AM251和CB2选择性拮抗剂AM630能减少体内破骨细胞的生成,从而减轻成年小鼠卵巢切除引起的骨丢失[25-26,48]。此外,CB1-/-小鼠能免受卵巢切除引起的骨丢失的影响,CB2受体缺陷小鼠的骨丢失仅比野生型小鼠略有减少[41,48]。最近研究证实CB2受体基因的多态性与绝经后妇女骨质疏松症具有相关性[62],这表明CB2受体基因在未来可能成为治疗骨质疏松症的靶点。 糖皮质激素诱导的骨质疏松症是最常见的继发性骨质疏松症。临床研究发现,慢性糖皮质激素治疗与低骨密度和高骨折易感性密切相关[63-67]。应用糖皮质激素会使大鼠患上骨质疏松症,而应用CB1拮抗剂AM251后,骨质疏松大鼠成骨细胞凋亡减少、活性增强,减轻了糖皮质激素对骨形成的负面影响[68-69]。但有研究发现,CB1受体拮抗剂利莫那班(Rimonabant)虽然能减轻年轻大鼠糖皮质激素应用后的骨质疏松症,但会加重老年大鼠的骨质疏松程度,这提示临床应用的拮抗剂药物可能具有年龄相关性。BELLINI等[70]发现激活CB2受体可以抑制甲基强的松龙引起的破骨细胞过度活化,这提示CB2受体在防治糖皮质激素诱导的骨量丢失方面有一定的应用价值。 2.3 大麻素受体与牙周炎 牙周炎是一种由细菌等多种因素引起的慢性炎症破坏性疾病,与炎症免疫和骨代谢密切相 关[63-64,71]。牙周炎病变过程中局部组织产生多种炎症递质以及细菌及其毒素进入血液,可能会增加很多系统性疾病的风险[72]。最近有研究证实大麻素受体在牙周组织骨代谢和炎症愈合等方面发挥着重要作用。YAN等[73]发现炎症环境下CB1受体激活能通过p38丝裂原活化蛋白激酶和JNK信号通路增强牙周膜干细胞的成骨/牙本质分化能力,从而为成骨分化营造良好的微环境。同时有研究发现,CB2受体的激活可以调节脂多糖诱导的人牙周韧带细胞促炎因子的产生和破骨细胞基因的表达[36]。激活CB2受体对脂多糖刺激的人牙周膜细胞具有抗炎和抗吸收作用,这提示大麻素受体活化可能是治疗牙周炎症、减缓牙槽骨吸收的有效策略。 "
| [1] SALAZAR M, CARRACEDO A, SALANUEVA IJ, et al. Cannabinoid action induces autophagy-mediated cell death through stimulation of ER stress in human glioma cells. J Clin Invest. 2009;119(5):1359-1372. [2] CAFFAREL MM, SARRIÓ D, PALACIOS J, et al. Delta9-tetrahydrocannabinol inhibits cell cycle progression in human breast cancer cells through Cdc2 regulation. Cancer Res. 2006;66(13):6615-6621. [3] GOWRAN A, CAMPBELL VA. A role for p53 in the regulation of lysosomal permeability by delta 9-tetrahydrocannabinol in rat cortical neurones: implications for neurodegeneration. J Neurochem. 2008;105(4):1513-1524. [4] GREENHOUGH A, PATSOS HA, WILLIAMS AC, et al. The cannabinoid delta(9)-tetrahydrocannabinol inhibits RAS-MAPK and PI3K-AKT survival signalling and induces BAD-mediated apoptosis in colorectal cancer cells. Int J Cancer. 2007;121(10):2172-2180. [5] DEVANE WA, HANUS L, BREUER A, et al. Isolation and structure of a brain constituent that binds to the cannabinoid receptor. Science (New York, NY). 1992;258(5090):1946-1949. [6] KUNOS G, BÁTKAI S, OFFERTÁLER L, et al. The quest for a vascular endothelial cannabinoid receptor. Chem Phys Lipids. 2002;121(1-2):45-56. [7] PERTWEE RG. Cannabinoid receptors and pain. Prog Neurobiol. 2001; 63(5):569-611. [8] MATSUDA LA, LOLAIT SJ, BROWNSTEIN MJ, et al. Structure of a cannabinoid receptor and functional expression of the cloned cDNA. Nature. 1990;346(6284):561-564. [9] MUNRO S, THOMAS KL, ABU-SHAAR M. Molecular characterization of a peripheral receptor for cannabinoids. Nature. 1993;365(6441):61-65. [10] NAGARKATTI P, PANDEY R, RIEDER SA, et al. Cannabinoids as novel anti-inflammatory drugs. Future Med Chem. 2009;1(7):1333-1349. [11] KUPCZYK P, REICH A, SZEPIETOWSKI JC. Cannabinoid system in the skin- a possible target for future therapies in dermatology. Exp Dermatol. 2009;18(8):669-679. [12] YATES ML, BARKER EL. Inactivation and biotransformation of the endogenous cannabinoids anandamide and 2-arachidonoylglycerol. Mol Pharmacol. 2009;76(1):11-17. [13] BAB I, OFEK O, TAM J, et al. Endocannabinoids and the regulation of bone metabolism. J Neuroendocrinol. 2008;20 Suppl 1:69-74. [14] BAB I. Themed issue on cannabinoids in biology and medicine. Br J Pharmacol. 2011;163(7):1327-1328. [15] MACH DB, ROGERS SD, SABINO MC, et al. Origins of skeletal pain: sensory and sympathetic innervation of the mouse femur. Neuroscience. 2002;113(1):155-166. [16] IDRIS AI, SOPHOCLEOUS A, LANDAO-BASSONGA E, et al. Cannabinoid receptor type 1 protects against age-related osteoporosis by regulating osteoblast and adipocyte differentiation in marrow stromal cells. Cell Metab. 2009;10(2):139-147. [17] TAKEDA S, ELEFTERIOU F, LEVASSEUR R, et al. Leptin regulates bone formation via the sympathetic nervous system. Cell. 2002;111(3):305-317. [18] TAM J, OFEK O, FRIDE E, et al. Involvement of neuronal cannabinoid receptor CB1 in regulation of bone mass and bone remodeling. Mol Pharmacol. 2006;70(3):786-792. [19] IDRIS AI, VAN’T HOF RJ, GREIG IR, et al. Regulation of bone mass, bone loss and osteoclast activity by cannabinoid receptors. Nature Med. 2005;11(7):774-779. [20] ELEFTERIOU F, AHN JD, TAKEDA S, et al. Leptin regulation of bone resorption by the sympathetic nervous system and CART. Nature. 2005; 434(7032):514-520. [21] BUSQUETS-GARCIA A, GOMIS-GONZÁLEZ M, SRIVASTAVA RK, et al. Peripheral and central CB1 cannabinoid receptors control stress-induced impairment of memory consolidation. Proc Natl Acad Sci U S A. 2016;113(35):9904-9909. [22] JIANG H, WU Y, VALVERDE P, et al. Central adiponectin induces trabecular bone mass partly through epigenetic downregulation of cannabinoid receptor CB1. J Cell Physiol. 2019;234(5):7062-7069. [23] WANG L, YANG L, TIAN L, et al. Cannabinoid Receptor 1 Mediates Homing of Bone Marrow-Derived Mesenchymal Stem Cells Triggered by Chronic Liver Injury. J Cell Physiol. 2017;232(1):110-121. [24] WHYTE LS, FORD L, RIDGE SA, et al. Cannabinoids and bone: endocannabinoids modulate human osteoclast function in vitro. Br J Pharmacol. 2012;165(8):2584-2597. [25] TAM J, TREMBOVLER V, DI MARZO V, et al. The cannabinoid CB1 receptor regulates bone formation by modulating adrenergic signaling. FASEB J. 2008;22(1):285-294. [26] SERRE CM, FARLAY D, DELMAS PD, et al. Evidence for a dense and intimate innervation of the bone tissue, including glutamate-containing fibers. Bone. 1999;25(6):623-629. [27] SMITH M, WILSON R, O’BRIEN S, et al. The Effects of the Endocannabinoids Anandamide and 2-Arachidonoylglycerol on Human Osteoblast Proliferation and Differentiation. PloS One. 2015;10(9): e0136546. [28] OFEK O, KARSAK M, LECLERC N, et al. Peripheral cannabinoid receptor, CB2, regulates bone mass. Proc Natl Acad Sci U S A. 2006;103(3):696-701. [29] IDRIS AI, LANDAO-BASSONGA E, RALSTON SH. The TRPV1 ion channel antagonist capsazepine inhibits osteoclast and osteoblast differentiation in vitro and ovariectomy induced bone loss in vivo. Bone. 2010;46(4):1089-1099. [30] BAB I, ZIMMER A. Cannabinoid receptors and the regulation of bone mass. Br J Pharmacol. 2008;153(2):182-188. [31] BAB IA. Regulation of skeletal remodeling by the endocannabinoid system. Ann N Y Acad Sci. 2007;1116:414-422. [32] SAMIR SM, MALEK HA. Effect of cannabinoid receptors 1 modulation on osteoporosis in a rat model of different ages. J Physiol Pharmacol. 2014;65(5):687-694. [33] EGER M, BADER M, BREE D, et al. Bone Anabolic Response in the Calvaria Following Mild Traumatic Brain Injury is Mediated by the Cannabinoid-1 Receptor. Sci Rep. 2019;9(1):16196. [34] WASSERMAN E, TAM J, MECHOULAM R, et al. CB1 cannabinoid receptors mediate endochondral skeletal growth attenuation by Δ9-tetrahydrocannabinol. Ann N Y Acad Sci. 2015;1335:110-119. [35] ASHTON JC, FRIBERG D, DARLINGTON CL, et al. Expression of the cannabinoid CB2 receptor in the rat cerebellum: an immunohistochemical study. Neurosci Lett. 2006;396(2):113-116. [36] 钱红,赵亚,胡静,等.大麻素受体CB2在机械牵张力介导的人牙周膜细胞成骨分化中的作用[J].中国美容医学,2012,21(13):1765-1767. [37] QIAN H, ZHAO Y, PENG Y, et al. Activation of cannabinoid receptor CB2 regulates osteogenic and osteoclastogenic gene expression in human periodontal ligament cells. J Periodontal Res. 2010;45(4):504-511. [38] SOPHOCLEOUS A, LANDAO-BASSONGA E, VAN’T HOF RJ, et al. The type 2 cannabinoid receptor regulates bone mass and ovariectomy-induced bone loss by affecting osteoblast differentiation and bone formation. Endocrinology. 2011;152(6):2141-2149. [39] ZHANG M, SHI X, WU J, et al. CoCl(2) induced hypoxia enhances osteogenesis of rat bone marrow mesenchymal stem cells through cannabinoid receptor 2. Arch Oral Biol. 2019;108:104525. [40] ZHENG W, LIU C, LEI M, et al. Evaluation of common variants in the CNR2 gene and its interaction with abdominal obesity for osteoporosis susceptibility in Chinese post-menopausal females. Bone Joint Res. 2019;8(11):544-549. [41] 胡旭治,史新连,邓辉.大麻素Ⅱ型受体参与调控低氧微环境下大鼠骨髓间充质干细胞的骨向分化[J].温州医科大学学报,2019, 49(8):563-567. [42] WANG B, LIAN K, LI J, et al. Restoration of osteogenic differentiation by overexpression of cannabinoid receptor 2 in bone marrow mesenchymal stem cells isolated from osteoporotic patients. Exp Ther Med. 2018;15(1):357-364. [43] QIU S, ZHAO F, TANG X, et al. Type-2 cannabinoid receptor regulates proliferation, apoptosis, differentiation, and OPG/RANKL ratio of MC3T3-E1 cells exposed to Titanium particles. Mol Cell Biochem. 2015; 399(1-2):131-141. [44] SCUTT A, WILLIAMSON EM. Cannabinoids stimulate fibroblastic colony formation by bone marrow cells indirectly via CB2 receptors. Calcif Tissue Int. 2007;80(1):50-59. [45] XU A, YANG Y, SHAO Y, et al Inhibiting effect of microRNA-187-3p on osteogenic differentiation of osteoblast precursor cells by suppressing cannabinoid receptor type 2. Differentiation. 2019;109:9-15. [46] ZHU M, YU B, BAI J, et al. Cannabinoid Receptor 2 Agonist Prevents Local and Systemic Inflammatory Bone Destruction in Rheumatoid Arthritis. J Bone Miner Res. 2019;34(4):739-751. [47] ROSSI F, BELLINI G, LUONGO L, et al. The 17-β-oestradiol inhibits osteoclast activity by increasing the cannabinoid CB2 receptor expression. Pharmacol Res. 2013;68(1):7-15. [48] IDRIS AI, SOPHOCLEOUS A, LANDAO-BASSONGA E, et al. Regulation of bone mass, osteoclast function, and ovariectomy-induced bone loss by the type 2 cannabinoid receptor. Endocrinology. 2008;149(11):5619-5626. [49] SCHUEHLY W, PAREDES JM, KLEYER J, et al. Mechanisms of osteoclastogenesis inhibition by a novel class of biphenyl-type cannabinoid CB(2) receptor inverse agonists. Chem Biol. 2011;18(8):1053-1064. [50] LUNN CA, FINE J, ROJAS-TRIANA A, et al. Cannabinoid CB(2)-selective inverse agonist protects against antigen-induced bone loss. Immunopharmacol Immunotoxicol. 2007;29(3-4):387-401. [51] ROSS RA. The enigmatic pharmacology of GPR55. Trends Pharmacol Sci. 2009;30(3):156-163. [52] MARAZZI J, KLEYER J, PAREDES JM, et al. Endocannabinoid content in fetal bovine sera-unexpected effects on mononuclear cells and osteoclastogenesis. J Immunol Methods. 2011;373(1-2):219-228. [53] SOPHOCLEOUS A, IDRIS AI, RALSTON SH. Genetic background modifies the effects of type 2 cannabinoid receptor deficiency on bone mass and bone turnover. Calcif Tissue Int. 2014;94(3):259-268. [54] BAKER D, PRYCE G, DAVIES WL, et al. In silico patent searching reveals a new cannabinoid receptor. Trends Pharmacol Sci. 2006;27(1):1-4. [55] SMART D, GUNTHORPE MJ, JERMAN JC, et al. The endogenous lipid anandamide is a full agonist at the human vanilloid receptor (hVR1). Br J Pharmacol. 2000;129(2):227-230. [56] SAWZDARGO M, NGUYEN T, LEE DK, et al. Identification and cloning of three novel human G protein-coupled receptor genes GPR52, PsiGPR53 and GPR55: GPR55 is extensively expressed in human brain. Brain Res Mol Brain Res. 1999;64(2):193-198. [57] WHYTE LS, RYBERG E, SIMS NA, et al. The putative cannabinoid receptor GPR55 affects osteoclast function in vitro and bone mass in vivo. Proc Natl Acad Sci U S A. 2009;106(38):16511-16516. [58] OSSOLA CA, BALCARCEL NB, ASTRAUSKAS JI, et al. A new target to ameliorate the damage of periodontal disease: The role of transient receptor potential vanilloid type-1 in contrast to that of specific cannabinoid receptors in rats. J Periodontol. 2019;90(11):1325-1335. [59] GROTENHERMEN F, MÜLLER-VAHL K. The therapeutic potential of cannabis and cannabinoids. Deutsch Arztebl Int. 2012;109(29-30):495-501. [60] Raisz LG. Pathogenesis of osteoporosis: concepts, conflicts, and prospects. J Clin Invest. 2005;115(12):3318-3325. [61] RIGGS BL, WAHNER HW, SEEMAN E, et al. Changes in bone mineral density of the proximal femur and spine with aging. Differences between the postmenopausal and senile osteoporosis syndromes. J Clin Invest. 1982;70(4):716-723. [62] WOO JH, KIM H, KIM JH, et al. Cannabinoid receptor gene polymorphisms and bone mineral density in Korean postmenopausal women. Menopause (New York, NY). 2015;22(5):512-519. [63] GÜLER-YÜKSEL M, HOES JN, BULTINK IEM, et al. Glucocorticoids, Inflammation and Bone. Calcif Tissue Int. 2018;102(5):592-606. [64] WHITTIER X, SAAG KG. Glucocorticoid-induced Osteoporosis. Rheum Dis Clin North Am. 2016;42(1):177-189, x. [65] NATSUI K, TANAKA K, SUDA M, et al. High-dose glucocorticoid treatment induces rapid loss of trabecular bone mineral density and lean body mass. Osteoporos Int. 2006;17(1):105-108. [66] SOSA M, JÓDAR E, SAAVEDRA P, et al. Postmenopausal Canarian women receiving oral glucocorticoids have an increased prevalence of vertebral fractures and low values of bone mineral density measured by quantitative computer tomography and dual X-ray absorptiometry, without significant changes in parathyroid hormone. Eur J Int Med. 2008;19(1):51-56. [67] MIGLIACCIO S, BRAMA M, FORNARI R, et al. Glucocorticoid-induced osteoporosis: an osteoblastic disease. Aging Clin Exp Res. 2007;19(3 Suppl):5-10. [68] KO JY, WU RW, KUO SJ, et al. Cannabinoid receptor 1 mediates glucocorticoid-induced bone loss in rats by perturbing bone mineral acquisition and marrow adipogenesis. Arthritis Rheum. 2012;64(4): 1204-1214. [69] WU RW, LIN TP, KO JY, et al. Cannabinoid receptor 1 regulates ERK and GSK-3β-dependent glucocorticoid inhibition of osteoblast differentiation in murine MC3T3-E1 cells. Bone. 2011;49(6):1255-1263. [70] BELLINI G, TORELLA M, MANZO I, et al. PKCβII-mediated cross-talk of TRPV1/CB2 modulates the glucocorticoid-induced osteoclast overactivity. Pharmacol Res. 2017;115:267-274. [71] PIHLSTROM BL, MICHALOWICZ BS, JOHNSON NW. Periodontal diseases. Lancet (London, England). 2005;366(9499):1809-1820. [72] 张力木,林晓萍.C反应蛋白介导的牙周炎与全身系统性疾病相关机制研究进展[J].口腔疾病防治,2020,28(3):184-188. [73] YAN W, CAO Y, YANG H, et al. CB1 enhanced the osteo/dentinogenic differentiation ability of periodontal ligament stem cells via p38 MAPK and JNK in an inflammatory environment. Cell Prolif. 2019;52(6):e12691. [74] SCHMUHL E, RAMER R, SALAMON A, et al. Increase of mesenchymal stem cell migration by cannabidiol via activation of p42/44 MAPK. Biochem Pharmacol. 2014;87(3):489-501. [75] SOPHOCLEOUS A, MARINO S, KABIR D, et al. Combined deficiency of the Cnr1 and Cnr2 receptors protects against age-related bone loss by osteoclast inhibition. Aging Cell. 2017;16(5):1051-1061. [76] ROSSI F, BELLINI G, TORTORA C, et al. CB(2) and TRPV(1) receptors oppositely modulate in vitro human osteoblast activity. Pharmacol Res. 2015;99:194-201. [77] ROSSI F, BELLINI G, LUONGO L, et al. The endovanilloid/endocannabinoid system: a new potential target for osteoporosis therapy. Bone. 2011;48(5):997-1007. |
| [1] | Xue Yadong, Zhou Xinshe, Pei Lijia, Meng Fanyu, Li Jian, Wang Jinzi . Reconstruction of Paprosky III type acetabular defect by autogenous iliac bone block combined with titanium plate: providing a strong initial fixation for the prosthesis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1424-1428. |
| [2] | Zhu Chan, Han Xuke, Yao Chengjiao, Zhou Qian, Zhang Qiang, Chen Qiu. Human salivary components and osteoporosis/osteopenia [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1439-1444. |
| [3] | Jiang Huanchang, Zhang Zhaofei, Liang De, Jiang Xiaobing, Yang Xiaodong, Liu Zhixiang. Comparison of advantages between unilateral multidirectional curved and straight vertebroplasty in the treatment of thoracolumbar osteoporotic vertebral compression fracture [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1407-1411. |
| [4] | Li Wei, Zhu Hanmin, Wang Xin, Gao Xue, Cui Jing, Liu Yuxin, Huang Shuming. Effect of Zuogui Wan on bone morphogenetic protein 2 signaling pathway in ovariectomized osteoporosis mice [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1173-1179. |
| [5] | Wang Jing, Xiong Shan, Cao Jin, Feng Linwei, Wang Xin. Role and mechanism of interleukin-3 in bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1260-1265. |
| [6] | Xiao Hao, Liu Jing, Zhou Jun. Research progress of pulsed electromagnetic field in the treatment of postmenopausal osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1266-1271. |
| [7] | Gao Yujin, Peng Shuanglin, Ma Zhichao, Lu Shi, Cao Huayue, Wang Lang, Xiao Jingang. Osteogenic ability of adipose stem cells in diabetic osteoporosis mice [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 999-1004. |
| [8] | Tian Chuan, Zhu Xiangqing, Yang Zailing, Yan Donghai, Li Ye, Wang Yanying, Yang Yukun, He Jie, Lü Guanke, Cai Xuemin, Shu Liping, He Zhixu, Pan Xinghua. Bone marrow mesenchymal stem cells regulate ovarian aging in macaques [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 985-991. |
| [9] | Hou Jingying, Guo Tianzhu, Yu Menglei, Long Huibao, Wu Hao. Hypoxia preconditioning targets and downregulates miR-195 and promotes bone marrow mesenchymal stem cell survival and pro-angiogenic potential by activating MALAT1 [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1005-1011. |
| [10] | Liang Xuezhen, Yang Xi, Li Jiacheng, Luo Di, Xu Bo, Li Gang. Bushen Huoxue capsule regulates osteogenic and adipogenic differentiation of rat bone marrow mesenchymal stem cells via Hedgehog signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1020-1026. |
| [11] | Fang Xiaolei, Leng Jun, Zhang Chen, Liu Huimin, Guo Wen. Systematic evaluation of different therapeutic effects of mesenchymal stem cell transplantation in the treatment of ischemic stroke [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1085-1092. |
| [12] | Peng Kun. Improvement of the treatment effect of osteoporotic fractures: research status and strategy analysis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(6): 980-984. |
| [13] | Hu Weifan, Zheng Li, Li Dadi, Sun Yang, Zhao Fengchao. Overexpression of miR-25 downregulates titanium particle-induced osteoclast differentiation through the NFATc1 signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 682-687. |
| [14] | Shen Song, Xu Bin. Diffuse distribution of bone cement in percutaneous vertebroplasty reduces the incidence of refracture of adjacent vertebral bodies [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 499-503. |
| [15] | Yang Sidi, Wang Qian, Xu Nuo, Wang Ronghan, Jin Chuanqi, Lu Ying, Dong Ming. Biodentine enhances the proliferation and differentiation of osteoblasts through upregulating bone morphogenetic protein-2 [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 516-520. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||