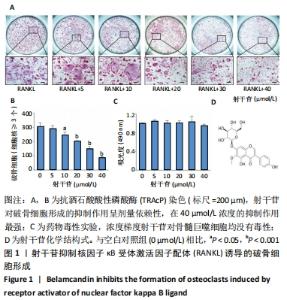
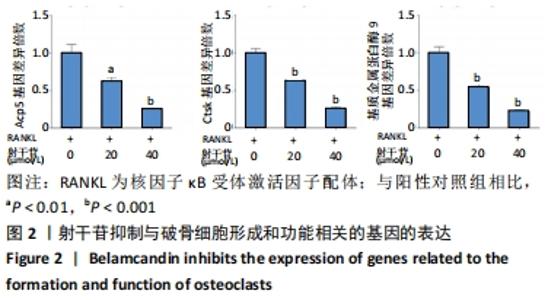
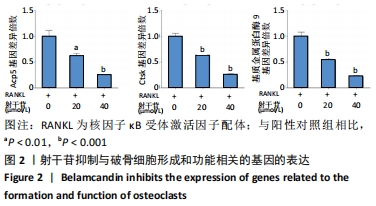
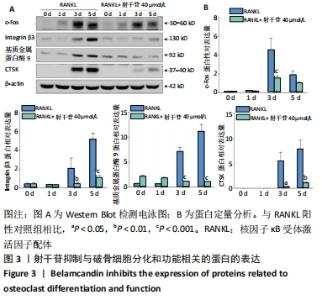
[1] BOYLE WJ, SIMONET WS, LACEY DL. Osteoclast differentiation and activation. Nature. 2003;423(6937):337-342.
[2] SOYSA NS, ALLES N. Osteoclast function and bone-resorbing activity: An overview. Biochem Biophys Res Commun. 2016;476(3):115-120.
[3] SIMS NA, VRAHNAS C. Regulation of cortical and trabecular bone mass by communication between osteoblasts, osteocytes and osteoclasts. Arch Biochem Biophys. 2014;561:22-28.
[4] WADA T, NAKASHIMA T, HIROSHI N, et al. RANKL-RANK signaling in osteoclastogenesis and bone disease. Trends Mol Med. 2006;12(1):17-25.
[5] 李啸群, 徐凯航, 纪方. 补骨脂异黄酮抑制破骨细胞分化缓解小鼠去卵巢骨质疏松[J].中国组织工程研究,2021,25(2):186-190.
[6] LIU Y, WANG Z, MA C, et al. Dracorhodin perchlorate inhibits osteoclastogenesis through repressing RANKL-stimulated NFATc1 activity. J Cell Mol Med. 2020;24(6):3303-3313.
[7] CHEN RS, ZHANG XB, ZHU XT, et al. LncRNA Bmncr alleviates the progression of osteoporosis by inhibiting RANML-induced osteoclast differentiation. Eur Rev Med Pharmacol Sci. 2019;23(21):9199-9206.
[8] ROBLING AG, BONEWALD LF. The Osteocyte: New Insights. Annu Rev Physiol. 2020;82:485-506.
[9] TAKAYANAGI H, KIM S, KOGA T, et al. Induction and activation of the transcription factor NFATc1 (NFAT2) integrate RANKL signaling in terminal differentiation of osteoclasts. Dev Cell. 2002;3(6):889-901.
[10] VAANANEN HK, HARKONEN PL. Estrogen and bone metabolism. Maturitas. 1996;23 Suppl:S65-S69.
[11] LI L, WANG Z. Ovarian Aging and Osteoporosis. Adv Exp Med Biol. 2018; 1086:199-215.
[12] GAVALI S, GUPTA MK, DASWANI B, et al. LYN, a key mediator in estrogen-dependent suppression of osteoclast differentiation, survival, and function. Biochim Biophys Acta Mol Basis Dis. 2019;1865(3):547-557.
[13] CHEN LR, KO NY, CHEN KH. Isoflavone Supplements for Menopausal Women: A Systematic Review. Nutrients. 2019;11(11):2649.
[14] LIU Y, WANG C, WANG G, et al. Loureirin B suppresses RANKL-induced osteoclastogenesis and ovariectomized osteoporosis via attenuating NFATc1 and ROS activities. Theranostics. 2019;9(16):4648-4662.
[15] CHEN K, QIU P, YUAN Y, et al. Pseurotin A Inhibits Osteoclastogenesis and Prevents Ovariectomized-Induced Bone Loss by Suppressing Reactive Oxygen Species. Theranostics. 2019;9(6):1634-1650.
[16] KANG K, LEE SB, JUNG SH, et al. Tectoridin, a poor ligand of estrogen receptor alpha, exerts its estrogenic effects via an ERK-dependent pathway. Mol Cells. 2009;27(3):351-357.
[17] 张明发, 沈雅琴. 射干药理研究进展[J]. 中国执业药师,2010,7(1): 14-19.
[18] CHEN Y, WU CM, DAI RJ, et al. Combination of HPLC chromatogram and hypoglycemic effect identifies isoflavones as the principal active fraction of Belamcanda chinensis leaf extract in diabetes treatment. J Chromatogr B Analyt Technol Biomed Life Sci. 2011;879(5-6):371-378.
[19] JUNG S H, LEE Y S, LEE S, et al. Isoflavonoids from the rhizomes of Belamcanda chinensis and their effects on aldose reductase and sorbitol accumulation in streptozotocin induced diabetic rat tissues. Arch Pharm Res. 2002;25(3):306-312.
[20] 熊玲凡. 中药射干提取物射干苷抑制结肠癌细胞增殖及迁移作用的研究[D]. 武汉:湖北中医药大学,2016.
[21] 汪天青,张颖,王健,等.射干中异黄酮成分对花生四烯酸代谢酶的分子对接研究[J]. 辽宁中医杂志,2019,46(8):1709-1713.
[22] PAN CH, KIM ES, JUNG SH, et al. Tectorigenin inhibits IFN-gamma/LPS-induced inflammatory responses in murine macrophage RAW 264.7 cells. Arch Pharm Res. 2008;31(11):1447-1456.
[23] MYKHAILENKO O, KORINEK M, IVANAUSKAS L, et al. Qualitative and Quantitative Analysis of Ukrainian Iris Species: A Fresh Look on Their Antioxidant Content and Biological Activities. Molecules. 2020; 25(19):4588.
[24] WOZNIAK D, JANDA B, KAPUSTA I, et al. Antimutagenic and anti-oxidant activities of isoflavonoids from Belamcanda chinensis (L.) DC. Mutat Res. 2010;696(2):148-153.
[25] AKTHER N, ANDRABI K, NISSAR A, et al. Hepatoprotective activity of LC-ESI-MS standardized Iris spuria rhizome extract on its main bioactive constituents. Phytomedicine. 2014;21(10):1202-1207.
[26] LEE HU, BAE EA, KIM DH. Hepatoprotective effect of tectoridin and tectorigenin on tert-butyl hyperoxide-induced liver injury. J Pharmacol Sci. 2005;97(4):541-544.
[27] KIM YP, YAMADA M, LIM SS, et al. Inhibition by tectorigenin and tectoridin of prostaglandin E2 production and cyclooxygenase-2 induction in rat peritoneal macrophages. Biochim Biophys Acta. 1999; 1438(3):399-407.
[28] 王丽娟,史惠蓉. 射干苷对卵巢癌SK-OV-3细胞增殖迁移和侵袭的影响[J].中华肿瘤杂志,2020,42(7):570-574.
[29] NOVACK DV, TEITELBAUM SL. The osteoclast: friend or foe?. Annu Rev Pathol. 2008;3:457-484.
[30] 王艳娇,马厚勋. 骨代谢调控因素与骨质疏松症[J]. 中国骨质疏松杂志,2010,16(11):876-881.
[31] MIYAUCHI Y, NINOMIYA K, MIYAMOTO H, et al. The Blimp1-Bcl6 axis is critical to regulate osteoclast differentiation and bone homeostasis. J Exp Med. 2010;207(4):751-762.
[32] SHIM M, BAE JY, LEE YJ, et al.Tectoridin from Maackia amurensis modulates both estrogen and thyroid receptors. Phytomedicine. 2014; 21(5):602-606.
[33] 张旋, 黄宁, 郑永唐. 我国中药来源的抗HIV天然化合物研究进展[J]. 药学学报,2010,45(2):141-153.
[34] WANG J, TANG Y, LV X, et al. Tectoridin inhibits osteoclastogenesis and bone loss in a murine model of ovariectomy-induced osteoporosis. Exp Gerontol. 2020;140:111057.
[35] CONG Q, JIA H, LI P, et al. p38alpha MAPK regulates proliferation and differentiation of osteoclast progenitors and bone remodeling in an aging-dependent manner. Sci Rep. 2017;7:45964.
[36] JIMI E, TAKAKURA N, HIURA F, et al. The Role of NF-kappaB in Physiological Bone Development and Inflammatory Bone Diseases: Is NF-kappaB Inhibition “Killing Two Birds with One Stone”?. Cells. 2019; 8(12):1636.
[37] OKADA H, OKABE K, TANAKA S. Finely-Tuned Calcium Oscillations in Osteoclast Differentiation and Bone Resorption. Int J Mol Sci. 2020; 22(1):180.
[38] TAO H, GE G, LIANG X, et al. ROS signaling cascades: dual regulations for osteoclast and osteoblast. Acta Biochim Biophys Sin (Shanghai). 2020;52(10):1055-1062.
[39] KUROTAKI D, YOSHIDA H, TAMURA T. Epigenetic and transcriptional regulation of osteoclast differentiation. Bone. 2020;138:115471.
[40] KIM JH, KIM N. Regulation of NFATc1 in Osteoclast Differentiation. J Bone Metab. 2014;21(4):233-241.
[41] ONO T, NAKASHIMA T. Recent advances in osteoclast biology. Histochem Cell Biol. 2018;149(4):325-341.
[42] KANG JY, KANG N, YANG Y, et al. The Role of Ca2+-NFATc1 Signaling and Its Modulation on Osteoclastogenesis. Int J Mol Sci. 2020;21(10):3646.
[43] CAO B, DAI X, WANG W. Knockdown of TRPV4 suppresses osteoclast differentiation and osteoporosis by inhibiting autophagy through Ca(2+) -calcineurin-NFATc1 pathway. J Cell Physiol. 2019;234(5):6831-6841.
[44] FENG H, CHENG T, STEER JH, et al. Myocyte enhancer factor 2 and microphthalmia-associated transcription factor cooperate with NFATc1 to transactivate the V-ATPase d2 promoter during RANKL-induced osteoclastogenesis. J Biol Chem. 2009;284(21):14667-14676.
|