Chinese Journal of Tissue Engineering Research
Expression of nuclear factor-kappa B p65 in acute rejection of liver transplantation in rhesus monkey
Zhang Sheng-ning, Ran Jiang-hua, Liu Jing, Li Zhu, Li Lai-bang, Gao Yang, Zhang Xi-bing, Wu Shu-yuan, Li Li
- First Department of Hepatopancreatobiliary Surgery, the Affiliated Ganmei Hospital of Kunming Medical University and the First People’s Hospital of Kunming City, Liver Transplantation Center of Organ Transplantation Institute of Yunnan Province, Kunming 650011, Yunnan Province, China
-
Received:2013-03-22Revised:2013-03-27Online:2013-07-30Published:2013-07-30 -
Contact:Ran Jiang-hua, M.D., Professor, Chief physician, First Department of Hepatopancreatobiliary Surgery, the Affiliated Ganmei Hospital of Kunming Medical University and the First People’s Hospital of Kunming City, Liver Transplantation Center of Organ Transplantation Institute of Yunnan Province, Kunming 650011, Yunnan Province, China rjh2u@163.com -
About author:Zhang Sheng-ning☆, M.D., Associate chief physician, Master’s supervisor, First Department of Hepatopancreatobiliary Surgery, the Affiliated Ganmei Hospital of Kunming Medical University and the First People’s Hospital of Kunming City, Liver Transplantation Center of Organ Transplantation Institute of Yunnan Province, Kunming 650011, Yunnan Province, China zsn813@163.com
CLC Number:
Cite this article
Zhang Sheng-ning, Ran Jiang-hua, Liu Jing, Li Zhu, Li Lai-bang, Gao Yang, Zhang Xi-bing, Wu Shu-yuan, Li Li. Expression of nuclear factor-kappa B p65 in acute rejection of liver transplantation in rhesus monkey[J]. Chinese Journal of Tissue Engineering Research, doi: 10.3969/j.issn.2095-4344.2013.31.001.
share this article

2.1 实验动物数量分析 肝移植后受体动物共8只,急性排斥反应组和对照组各4只,急性排斥反应组中途丢失2只,对照组中途丢失1只,丢失原因为移植后腹腔出血及呼吸循环功能衰竭,丢失动物均及时补齐,计4个时点,每个时点获取标本4个,每组共获取肝脏标本16个。 2.2 肝移植后血清丙氨酸氨基转移酶活性 2组恒河猴肝移植后血清丙氨酸氨基转移酶活性同期比较,急性排斥反应组由于没有使用抗免疫排斥药物,移植后未使用激素等原因导致肝细胞受到机体炎性细胞的攻击损伤较对照组严重。2组移植后6 h和12 h的丙氨酸氨基转移酶活性比较接近,组间比较差异无显著性意义(P > 0.05)。恒河猴肝移植后,随着时间的延长,肝移植后6,12,24和72 h的肝功能丙氨酸氨基转移酶活性呈逐渐上升趋势,因而2组组内比较,差异有显著性意义(P < 0.05);并且急性排斥反应组比对照组变化大,肝移植后24 h到72 h的变化趋势最明显,见表1。"
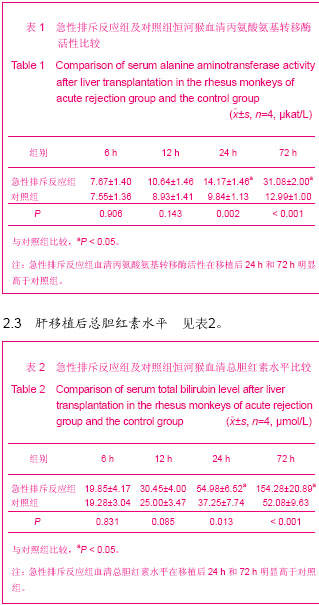
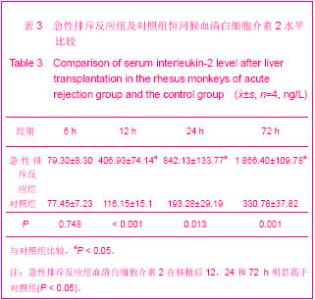
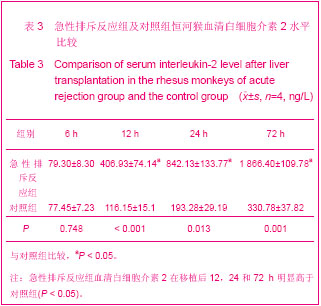
表2可见,2组恒河猴肝移植后血清总胆红素同期比较,急性排斥反应组由于没有使用抗免疫排斥药物,移植后未使用激素等原因导致肝细胞受到机体炎性细胞的攻击损伤较对照组严重。2组随着移植后时间的延长总胆红素呈逐渐上升的趋势,2组移植后6 h和12 h的总胆红素水平比较接近,组间比较差异无显著性意义(P > 0.05)。恒河猴肝移植后,随着时间的延长,肝移植后6,12,24和72 h的肝功能总胆红素逐渐上升趋势,因而2组组内比较差异有显著性意义(P < 0.05);并且急性排斥反应组比对照组变化大,肝移植后24 h到72 h的变化趋势最明显。 2.4 肝移植后白细胞介素2水平 2组随着移植后时间延的长白细胞介素2呈逐渐上升的趋势,2组移植后6 h白细胞介素2水平比较接近,组间比较差异无显著性意义(P > 0.05)。恒河猴肝移植后,随着时间的延长,肝移植后6,12,24和72 h的血清白细胞介素2呈逐渐上升趋势,因而2组组内比较,差异有显著性意义(P < 0.05);并且急性排斥反应组比对照组变化大,肝移植后24 h到72 h的变化趋势最明显,见表3。"
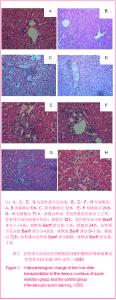
2.5 肝移植后肝脏组织病理学变化 见图1。 移植后6 h:2组的肝脏组织基本上正常,没有排斥反应的镜下特点,可见肝小叶结构正常,汇管区有少量以中性粒细胞为主的炎性细胞浸润,未见淋巴细胞浸润或者仅有极少量的淋巴细胞,小叶间静脉内膜基本正常,但小叶间静脉内膜水肿,内膜下可见少量中性粒细胞侵润,汇管区可见有少量的混合炎症细胞侵润,胆管、血管均存在,但胆管上皮未受损伤。 移植后12 h:急性排斥反应组肝脏苏木精-伊红染色显示,肝小叶结构基本正常,汇管区有中等量以中性粒细胞为主的炎性细胞侵润,其中可以看到较多的淋巴细胞侵润,小叶间静脉内膜水肿明显增厚,内膜下可见中等量中性粒细胞侵润(淋巴细胞占的比例增多),胆管、血管均存在,但胆管上皮未明显受损伤,Banff评分Ⅰ-Ⅱ级。 对照组可见肝脏实质内有中等量的中性粒细胞侵润,汇管区仅见有少量中性粒细胞侵润(其中的淋巴细胞极少),排斥反应的分级不明确,Banff评分0-Ⅰ级。 移植后24 h:急性排斥反应组肝脏苏木精-伊红染色,中到重度排斥反应,汇管区大量单个核细胞浸润,并累及胆管、血管,肝小叶结构消失,伴有局灶性的出血、坏死。Banff评分Ⅱ-Ⅲ级。 对照组汇管区出现少量的单核细胞浸润,但侵及肝实质、胆管和血管的炎细胞浸润程度达到轻度急性排斥的诊断标准,Banff评分0-Ⅰ级。"
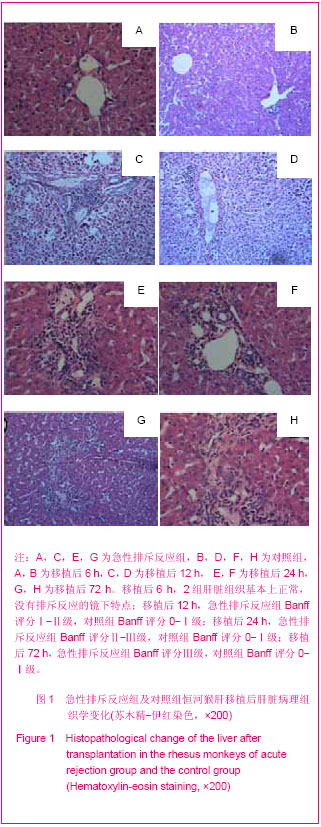

移植后72 h:急性排斥反应组肝脏苏木精-伊红染色,重度排斥反应,汇管区大量单个核细胞浸润,并累及胆管、血管,肝小叶结构消失明显,且伴有明显的局灶性出血、坏死,Banff评分Ⅲ级。 对照组汇管区出现少到中等量的单核细胞浸润,侵及肝实质程度较急性排斥反应组轻,Banff评分0-Ⅰ级。 2.6 肝移植后核因子κB p65蛋白表达变化 核因子κB p65条带(Mr 65 000)与相应β-actin(Mr 42 000)做比值后得到的数据进行统计学分析,对不同组采用单因素方差分析,组间对比采用t 检验。 整体变化趋势:急性排斥反应组6 h开始明显增高;对照组呈现下降趋势,12,24和72 h 3个时间点组急性排斥反应组明显高于对照组,差异有显著性意义(P=0.023,0.002,0.001,均< 0.05);6 h 2组之间差异无显著性意义(P=0.364,> 0.05),见表4及图2。"

| [1] Molinero LL, Alegre ML.Role of T cell-nuclear factor κB in ransplantation.Transplant Rev (Orlando). 2012 ;26(3): 189-200. [2] Tsoulfas G, Geller DA. NF-kappaB in transplantation: friend or foe? Transpl Infect Dis. 2001;3(4):212-219.[3] 冉江华,张升宁,刘静,等. 恒河猴同种异体原位肝移模型的改进[J].中国普外基础与临床杂志,2010;17(7):694-798.[4] Demetris A, Adams D, Bellamy C,et al.Update of the International Banff Schema for Liver Allograft Rejection: working recommendations for the histopathologic staging and reporting of chronic rejection. An International Panel. Hepatology. 2000;31(3):792-799.[5] Sánchez-Fueyo A, Strom TB.Immunologic basis of graft rejection and tolerance following transplantation of liver or other solid organs. Gastroenterology. 2011;140(1):51-64. [6] Uemura T, Ikegami T, Sanchez EQ,et al. Late acute rejection after liver transplantation impacts patient survival.Clin Transplant. 2008;22(3):316-323. [7] Finn PW, Stone JR, Boothby MR,et al. Inhibition of NF-kappaB-dependent T cell activation abrogates acute allograft rejection.J Immunol.2001;167(10): 5994-6001.[8] Ding X, Wang X, Xue W,et al. Blockade of the nuclear factor kappa B pathway prolonged islet allograft survival.Artif Organs. 2012;36(3):E21-27. [9] Wei JF, Zheng SS.NF-kappa B in allograft rejection. Hepatobiliary Pancreat Dis Int. 2003;2(2):180-183.[10] May MJ, Ghosh S. Rel/NF-kappa B and I kappa B proteins: an overview. Semin Cancer Biol. 1997;8(2):63-73.[11] Oeckinghaus A, Hayden MS, Ghosh S. Crosstalk in NF-κB signaling pathways.Nat Immunol. 2011;12(8):695-708.[12] Pasparakis M. Role of NF-κB in epithelial biology. Immunol Rev. 2012;246(1):346-458.[13] Vallabhapurapu S, Karin M.Regulation and function of NF-kappaB transcription factors in the immune system. Annu Rev Immunol. 2009;27:693-733.[14] Gilmore TD, Wolenski FS.NF-κB: where did it come from and why? Immunol Rev. 2012;246(1):14-35. [15] Urbanova M, Brabcova I, Girmanova E,et al. Differential regulation of the nuclear factor-κB pathway by rabbit antithymocyte globulins in kidney transplantation. Transplantation. 2012;93(6):589-556. [16] Hayden MS, West AP, Ghosh S.NF-kappaB and the immune response. Oncogene. 2006;25(51):6758-6780.[17] Mason NJ, Artis D, Hunter CA.New lessons from old pathogens: what parasitic infections have taught us about the role of nuclear factor-kappaB in the regulation of immunity. Immunol Rev. 2004;201:48-56.[18] Lan W, Petznick A, Heryati S, et al.Nuclear Factor-κB: central regulator in ocular surface inflammation and diseases.Ocul Surf. 2012;10(3):137-148.[19] Shih VF, Davis-Turak J, Macal M,et al.Control of RelB during dendritic cell activation integrates canonical and noncanonical NF-κB pathways.Nat Immunol. 2012;13(12):1162-1170. [20] Xie J, Wang Y, Bao J,et al.Immune tolerance induced by RelB short-hairpin RNA interference dendritic cells in liver transplantation. J Surg Res. 2013;180(1):169-175.[21] Hayden MS. A less-canonical, canonical NF-κB pathway in DCs.Nat Immunol. 2012;13(12):1139-1141. [22] Eldor R, Abel R, Sever D,et al. Inhibition of nuclear factor-κB activation in pancreatic β-cells has a protective effect on allogeneic pancreatic islet graft survival. PLoS One. 2013; 8(2):e56924. [23] Kaileh M, Sen R.NF-κB function in B lymphocytes. Immunol Rev. 2012 ;246(1):254-271. [24] Gerondakis S, Siebenlist U. Roles of the NF-kappaB pathway in lymphocyte development and function.Cold Spring Harb Perspect Biol. 2010;2(5):a000182.[25] Porras DL, Wang Y, Zhou P,et al.Role of T-cell-specific nuclear factor κB in islet allograft rejection. Transplantation. 2012;93(10):976-982.[26] Goebeler M, Gillitzer R, Kilian K,et al.Multiple signaling pathways regulate NF-kappaB-dependent transcription of the monocyte chemoattractant protein-1 gene in primary endothelial cells. Blood. 2001;97(1):46-55.[27] Visekruna A, Volkov A, Steinhoff U. A key role for NF-κB transcription factor c-Rel in T-lymphocyte-differentiation and effector functions.Clin Dev Immunol. 2012;2012:239368.[28] van den Hoogen MW, Hoitsma AJ. Anti-T-cell antibodies for the treatment of acute rejection after renal transplantation.Expert Opin Biol Ther. 2012;12(8):1031-1042. [29] Wang YL, Tang ZQ, Gao W, et al. Influence of Th1, Th2, and Th3 cytokines during the early phase after liver transplantation. Transplant Proc. 2003;35(8):3024-3025.[30] Millán O, Rafael-Valdivia L, Torrademé E,et al. Intracellular IFN-γ and IL-2 expression monitoring as surrogate markers of the risk of acute rejection and personal drug response in de novo liver transplant recipients.Cytokine. 2013;61(2):556-564.[31] Brunet M.Cytokines as predictive biomarkers of alloreactivity. Clin Chim Acta. 2012;413(17-18):1354-1358. [32] Millán O, Rafael-Valdivia L, Torrademé E,et al. Intracellular IFN-γ and IL-2 expression monitoring as surrogate markers of the risk of acute rejection and personal drug response in de novo liver transplant recipients. Cytokine. 2013;61(2): 556-564.[33] Maguire O, Collins C, O'Loughlin K,et al.Quantifying nuclear p65 as a parameter for NF-κB activation: Correlation between ImageStream cytometry, microscopy, and Western blot. Cytometry A. 2011;79(6):461-469.[34] Giardino Torchia ML, Conze DB, Jankovic D,et al.Balance between NF-κB p100 and p52 regulates T cell costimulation dependence. J Immunol. 2013;190(2):549-555. [35] Koch L, Fritzsching B, Frommhold D,et al.Lipopolysaccharide-induced expression of Th1/Th2 cytokines in whole neonatal cord and adult blood: role of nuclear factor-kappa B and p38 MAPK. Neonatology. 2011; 99(2):140-145. [36] Schmitz ML, Krappmann D.Controlling NF-kappaB activation in T cells by costimulatory receptors. Cell Death Differ. 2006;13(5):834-842.[37] Scheinman RI, Cogswell PC, Lofquist AK,et al.Role of transcriptional activation of I kappa B alpha in mediation of immunosuppression by glucocorticoids. Science. 1995; 270 (5234):283-286.[38] 万云乐,吴丽花,赵志成,等.他克莫司对移植免疫反应中核因子κB活性和淋巴细胞趋化因子表达的影响[J]. 中华移植杂志: 电子版, 2010,4(1):38-42.[39] Ouk S, Liou ML, Liou HC. Direct Rel/NF-κB inhibitors: structural basis for mechanism of action.Future Med Chem. 2009;1(9):1683-1707. [40] Ueki S, Yamashita K, Aoyagi T, et al. Control of allograft rejection by applying a novel nuclear factor-kappaB inhibitor, dehydroxymethylepoxyquinomicin. Transplantation. 2006; 82(12):1720-1727. |
| [1] | Jiang Xin, Qiao Liangwei, Sun Dong, Li Ming, Fang Jun, Qu Qingshan. Expression of long chain non-coding RNA PGM5-AS1 in serum of renal transplant patients and its regulation of human glomerular endothelial cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 741-745. |
| [2] | Yang Xin, Jin Zhe, Feng Xu, Lu Bing. The current situation of knowledge and attitudes towards organ, eye tissue, body donation of residents in Shenyang [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 779-784. |
| [3] | Li Liqiang, Jiao Longxing, Zhang Wu, Yan Wentao, Li Jian, Li Minghao. Effect of immature dendritic cells derived from bone marrow on rejection of orthotopic liver transplantation in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(13): 2025-2029. |
| [4] | Yang Feng, Chang Lipu, Huang Changshan, Gong Xiaoguang, Chang Shunwu. Macrolide antibiotics protects against ischemia-reperfusion injury after liver transplantation in rats [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(26): 4176-4182. |
| [5] | Liu Mengyuan1, Fang Fang2. Risk factors for multi-drug resistant organisms infection after liver transplantation [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(7): 1109-1114. |
| [6] | Ma Qiuhua, Wang Yunwen, Ma Hui, Qi Jie, Zhang Qingtan. Association of polymorphisms of klotho gene with primary osteoporosis in the elderly aging above 60 years old [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(35): 5587-5591. |
| [7] | Gao Hongqiang, Liu Jing, Li Zhiqiang, Wang Hailei, Zhao Xiongqi, Zhang Shengning, Ran Jianghua, Li Li . Ulinastatin improves rat liver metabolism after reduced-size liver transplantation [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(3): 435-440. |
| [8] | Wu Guangwen1, Liu Shuru2, Chen Jun3, Lin Jie1, Zhao Zhongsheng1, Ye Jinxia1. Mechanism of Duhuo Jisheng Decoction for treating knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(19): 2965-2971. |
| [9] | Li Qin, Li Yan, Gao Shanshan, Zheng Jintao. Pathways of astragalus polysaccharide inhibiting angiotensin II-induced cardiac hypertrophy [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(19): 3086-3091. |
| [10] | Cai Qiucheng, Fan Hongkai, Xiong Rihui, Jiang Yi. Intravenous administration of bone marrow mesenchymal stem cells protects liver function following fatty liver transplantation from donors after cardiac death [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(17): 2625-2629. |
| [11] | Huang Xiaolin, Liao Jian, Hong Wei, Li Fang, Cheng Yuting, Zhou Qian, Dong Qiang. Zoledronic acid in osteoclastogenesis: roles and mechanisms [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(17): 2716-2721. |
| [12] | Jiang Shanshan, Wang Feng, Yu Limei. Immunomodulatory properties of mesenchymal stem cells and their application in organ transplantation [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(1): 103-109. |
| [13] | Zhao Ying-peng, Li Li, Chen Gang, Bai Jian-hua, Liu Qi-yu. Reduced-size liver transplantation with fatty liver donors in a rat model [J]. Chinese Journal of Tissue Engineering Research, 2018, 22(4): 582-586. |
| [14] | Ding Chen1, Jiang Yi2, Pan Fan2, Cai Qiucheng2. Lipid metabolism after autologous orthotopic liver transplantation in rat models of fatty liver [J]. Chinese Journal of Tissue Engineering Research, 2018, 22(36): 5821-5827. |
| [15] | Liu Li-ye1, Zhao De-fang2, Gao Fei1, Zhang Tong2, Dong Qin2. Schisandra extract regulates Th17 cells and regulatory T cell imbalance in dogs undergoing liver transplantation [J]. Chinese Journal of Tissue Engineering Research, 2018, 22(20): 3213-3217. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||