Chinese Journal of Tissue Engineering Research ›› 2013, Vol. 17 ›› Issue (28): 5184-5190.doi: 10.3969/j.issn.2095-4344.2013.28.014
Previous Articles Next Articles
Telomere-associated factor expression in replicative senescence of human embryonic lung fibroblasts
Du Hua1, Xu Xiao-yan1, Hai Ling1, Shi Ying-xu2, 3
- 1Department of Pathological Anatomy, School of Basic Medical Sciences, Inner Mongolia Medical University, Hohhot 010059, Inner Mongolia Autonomous Region, China;
2Clinical Research Center, Affiliated Hospital of Inner Mongolia Medical University, Hohhot 010050, Inner Mongolia Autonomous Region, China;
3Institute of Hematology, Chinese Academy of Medical Sciences & Peking Union Medical College, Tianjin 300020, China
-
Online:2013-07-09Published:2013-07-09 -
About author:Du Hua, Master, Lecturer, Department of Pathological Anatomy, School of Basic Medical Sciences, Inner Mongolia Medical University, Hohhot 010059, Inner Mongolia Autonomous Region, China duhua20042007@sina.com Shi Ying-xu, M.D., Assistant researcher, Clinical Research Center, Affiliated Hospital of Inner Mongolia Medical University, Hohhot 010050, Inner Mongolia Autonomous Region, China; Institute of Hematology, Chinese Academy of Medical Sciences & Peking Union Medical College, Tianjin 300020 China shi.yingxu.wolf@gmail.com Du Hua and Shi Ying-xu contributed equally to this paper. -
Supported by:Funds of Inner Mongolia Medical University, No. YKD2012KJBW006*
CLC Number:
Cite this article
Du Hua, Xu Xiao-yan, Hai Ling, Shi Ying-xu. Telomere-associated factor expression in replicative senescence of human embryonic lung fibroblasts[J]. Chinese Journal of Tissue Engineering Research, 2013, 17(28): 5184-5190.
share this article
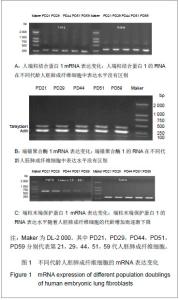
2.1 Western blot检测结检测人端粒结合蛋白1、端锚聚合酶1与端粒末端保护蛋白1 3种蛋白在细胞复制性衰老过程中的变化情况 见图1。 通过检测发现衰老过程中不同代龄的人胚肺成纤维细胞中人端粒结合蛋白1的mRNA表达水平没有明显的变化,见图1A。表明人端粒结合蛋白1的转录水平在细胞复制性衰老过程中没有发生变化,说明细胞正常衰老的发生不会影响人端粒结合蛋白1基因的表达。端锚聚合酶1调节人端粒结合蛋白1与端粒的结合,通过检测衰老过程中不同代龄的人胚肺成纤维细胞发现端锚聚合酶1的mRNA表达水平没有明显变化,见图1B。表明端锚聚合酶1的转录水平在细胞复制性衰老过程中没有发生变化,说明细胞正常衰老的发生不会影响端锚聚合酶1基因的表达。端粒末端保护蛋白1是维护端粒突出端的重要蛋白。端粒末端保护蛋白1的mRNA在所有组织中都存在,缺少端粒末端保护蛋白1的细胞会由于染色体末端的融合而死亡,这说明端粒末端保护蛋白1是确保染色体末端完整的管家基因。端粒末端保护蛋白1有5种剪接体,4种是普遍表达的,其中剪接体1,2,3,4在各种正常组织与肿瘤组织中都有大量表达,而剪接体5只在外周血白细胞中表达。端粒末端保护蛋白1剪接体2缺失exons 12a,剪接体5缺失exons 15a,如存在剪接体2与5,通过RT-PCR扩增exons 12-16应得到3条带,由上至下依次为端粒末端保护蛋白1、剪接体5、剪接体2。"
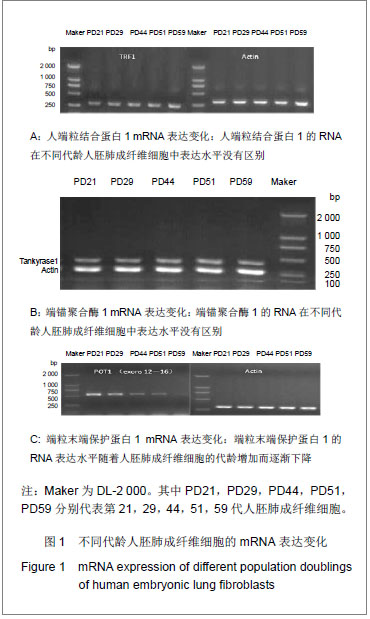
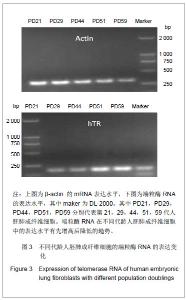
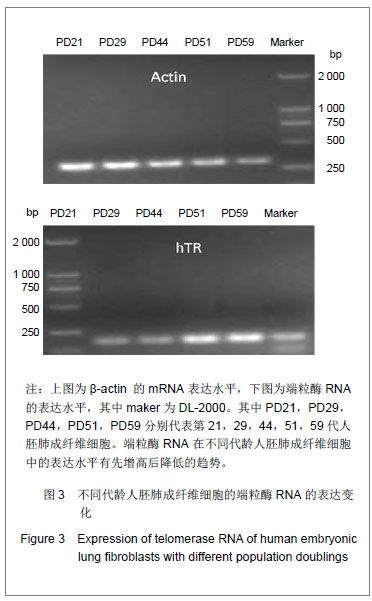
研究发现细胞的衰老伴随着端粒末端保护蛋白1的低表达,端粒末端保护蛋白1低表达可能是细胞复制性衰老的一个检测标志。人端粒结合蛋白1与端锚聚合酶1的在基因水平未发生变化,进一步检测了它们的蛋白表达情况。通过检测衰老过程中不同代龄人胚肺成纤维细胞发现人端粒结合蛋白1的蛋白表达水平随细胞的衰老呈增加的趋势,见图2A。表明人端粒结合蛋白1的蛋白表达量与衰老正相关。图2A中PD59的人端粒结合蛋白1低表达,推测是因为此时细胞开始进入细胞死亡阶段。前期细胞想通过高表达人端粒结合蛋白1来阻止细胞的衰老进程,但在细胞端粒不断缩短后,接近“Hayflick”极限,细胞启动凋亡机制,便不再高表达人端粒结合蛋白1的蛋白。通过检测衰老过程中不同代龄人胚肺成纤维细胞发现端锚聚合酶1的蛋白表达水平没有明显的变化,见图2B。表明端锚聚合酶1的蛋白水平在细胞复制性衰老过程中没有发生变化。端锚聚合酶1调节人端粒结合蛋白1与端粒DNA的结合,在细胞复制性衰老过程中未发生改变,说明端锚聚合酶1并未参与正常的衰老过程,可能只是在延长端粒或细胞复制过程中起作用。 2.3 端粒酶RNA在细胞衰老中的表达 端粒酶RNA在1995年被发现,在各种细胞中普遍存在,且含量恒定。正常细胞无端粒酶活性但表达端粒酶RNA,端粒酶RNA会对细胞的复制性衰老起作用吗?实验通过检测衰老过程中不同代龄的人胚肺成纤维细胞端粒酶RNA的表达水平发现其mRNA表达水平随着细胞的衰老呈递增趋势,见图3。表明端粒酶RNA可能也参与细胞的复制衰老过程。说明衰老过程对于细胞造成巨大的生存压力,细胞为了缓解由于端粒丢失造成的应激,会激活促进端粒酶RNA的表达。"
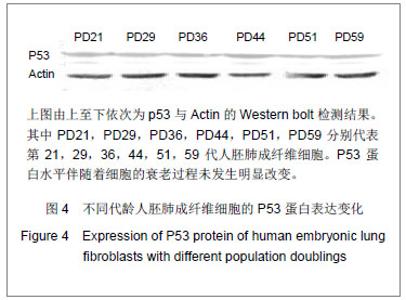
2.4 P53蛋白在细胞衰老中的表达 最近大量研究涉及P53诱导细胞衰老作用,人们积极探讨P53诱导细胞衰老的基因机制。有研究表明P53能够加速衰老,因此作者检测衰老过程中不同代龄人胚肺成纤维细胞P53的表达发现其蛋白表达水平随着细胞的衰老并未发生变化,见图4。表明P53的蛋白表达水平在细胞复制性衰老过程中没有变化,但其活性是否增高还有待进一步的研究。 实验研究了与端粒稳定性密切相关的3个端粒蛋 白——人端粒结合蛋白1、端锚聚合酶1与端粒末端保护蛋白1基因与蛋白表达水平与细胞复制性衰老的关系,通过研究发现,细胞的衰老会导致端粒末端保护蛋白1的表达减少,可能是由于细胞的不断衰老端粒的3’悬突变短,端粒末端保护蛋白1结合位点减少,导致端粒末端保护蛋白1的转录下降,从而加剧端粒的不稳定性,细胞衰老过程加快。 人端粒结合蛋白1与端锚聚合酶1的基因表达水平未见改变,但人端粒结合蛋白1的蛋白表达明显提高,说明可能是细胞为了提高端粒DNA的稳定性促进了人端粒结合蛋白1的翻译。 端粒酶RNA在衰老过程中也高表达,可能与人端粒结合蛋白1蛋白的高表达相类似,是细胞为了阻止衰老带来的应激而产生的应答。 有研究发现P53可能促进衰老,但本文没有发现,可能与P53的磷酸化状态有关,仍需要进一步的研究证实。"

| [1]Xie Y, Yang D, He Q, et al. Zebrafish as a model system to study the physiological function of telomeric protein TPP1. PLoS One. 2011;6(2):e16440. [2]Takai KK, Hooper S, Blackwood S, et al. In vivo stoichiometry of shelterin components. J Biol Chem. 2010;285(2):1457-1467. [3]Takai KK, Kibe T, Donigian JR, et al. Telomere Protection by TPP1/POT1 Requires Tethering to TIN2. Molecular Cell. 2011;4 4: 647-659.[4]Abreu E, Aritonovska E, Reichenbach P, et al. TIN2-tethered TPP1 recruits human telomerase to telomeres in vivo. Mol Cell Biol. 2010;30:2971-2982.[5]Lundblad V. Taking the measuree. Nature. 2003;423:926-927.[6]Blasco MA. Mammalian telomeres and telomerase:why they matter for cancer and aging. Eur J Cell Biol. 2003;82:441-446.[7]Giannone RJ, McDonald HW, Hurst GB, et al. The Protein Network Surrounding the Human Telomere Repeat Binding Factors TRF1, TRF2, and POT1. PLoS ONE. 2010;5(8): e12407.[8]Miller AS, Balakrishnan L, Buncher NA, et al. Telomere proteins POT1, TRF1 and TRF2 augment long-patch base excision repair in vitro. Cell Cycle. 2012;11(5):998-1007.[9]Choi KH, Farrell AS, Lakamp AS, et al. Characterization of the DNA binding specificity of Shelterin complexes. Nucleic Acids Res. 2011;39(21):9206-9223.[10]Martínez P, Blasco MA. Role of shelterin in cancer and aging. Aging Cell. 2010;9:653-666.[11]Opresko PL, Kobbe Cv, Laine JP, et al. Telomere-binding Protein TRF2 Binds to and Stimulates the Werner and Bloom Syndrome Helicases. J Biol Chem. 2000;277(43):41110- 41119.[12]Chiang YJ, Hsiao SJ, Yver D, et al. Tankyrase 1 and Tankyrase 2 Are Essential but Redundant for Mouse Embryonic Development. PLoS ONE. 2008;3(7): e2639.[13]Ha GH, Kim HS, Go H, et al. Tankyrase-1 function at telomeres and during mitosis is regulated by Polo-like kinase-1-mediated phosphorylation. Cell Death Differ. 2012;19(2):321-332.[14]Donigian JR, de Lange T. The role of the poly(ADP-ribose) polymerase tankyrase1 in telomere length control by the TRF1 component of the shelterin complex. J Biol Chem. 2007;282:22662-22667.[15]Chang P, Coughlin M, Mitchison TJ. Tankyrase-1 polymerization of poly(ADP-ribose) is required for spindle structure and function. Nat Cell Biol. 2005;7:1133-1139.[16]Gelmini S, Poggesi M, Distante V, et al. Tankyrase, a positive regulator of telomere elongation, is overexpressed in human breast cancer. Cancer Lett.2004;216:81-87.[17]Flynn RL, Centore RC, O’Sullivan RJ, et al. TERRA and hnRNPA1 orchestrate an RPA-to-POT1 switch on telomeric single-stranded DNA. Nature. 2011;471:532-536.[18]Krüger AC, Raarup MK, Nielsen MM, et al. Interaction of hnRNP A1 with telomere DNA G-quadruplex structures studied at the single molecule level. Eur Biophys J. 2010; 39(9):1343-1350.[19]Choi YH, Lim JK, Jeong MW, et al. HnRNP A1 phosphorylated by VRK1 stimulates telomerase and its binding to telomeric DNA sequence. Nucleic Acids Res. 2012;40(17):8499-8518[20]Ford LP, Wright WE, Shay JW. A model for heterogeneous nuclear ribonucleoproteins in telomere and telomerase regulation. Oncogene. 2002;21(4):580-583.[21]Cairney CJ, Keith WN. Telomerase redefined: integrated regulation of hTR and hTERT for telomere maintenance and telomerase activity. Biochimie. 2008;90(1):13-23. [22]Brault ME, Autexier C. Telomeric recombination induced by dysfunctional telomeres. Mol Biol Cell. 2011;22(2): 179-188.[23]Ting NS, Pohorelic B, Yu Y, et al. The human telomerase RNA component, hTR, activates the DNA-dependent protein kinase to phosphorylate heterogeneous nuclear ribonucleoprotein A1. Nucleic Acids Res. 2009;37(18): 6105-6115.[24]Fakhoury J, Marie-Egyptienne DT, Londoño-Vallejo JA, et al. Telomeric function of mammalian telomerases at short telomeres. J Cell Sci. 2010;123:1693-1704.[25]Sexton AN, Youmans DT, Collins K. Specificity Requirements for Human Telomere Protein Interaction with Telomerase Holoenzyme. J Biol Chem. 2012;287(41):34455-34464.[26]Cairney CJ, Keith WN. Telomerase redefined: integrated regulation of hTR and hTERT for telomere maintenance and telomerase activity. Biochimie. 2008;90(1):13-23. [27]Itahana K, Dimri G, Campisi J. Regulation of cellular senescence by P53. Eur J Biochem. 2001;268:2784-2791.[28]Feng Z, Lin M, Wu R. The Regulation of Aging and Longevity A New and Complex Role of p53. Genes Cancer. 2011; 2(4): 443-452.[29]Harly CB, Futcher AB, Greider CW. Telomeres shorten during aging of human fibroblasts. Nature. 1990;345:4458-4604.[30]Smogorzewska A , de-Lange T. Regulation of telomerase by telomeric proteins. Annu Rev Biochem. 2004;73: 177-208.[31]Martínez P, Blasco MA. Telomeric and extra-telomeric roles for telomerase and the telomere-binding proteins. Nat Rev Cancer. 2011;11(3):161-176.[32]van Steensel B, de Lange T. Control of telomere length by the human telomeric protein TRF1. Nature. 1997;385(6618): 740-743.[33]Lee TH, Tun-Kyi A, Shi R, et al. Essential role of Pin1 in regulation of TRF1 stability and telomere maintenance. Nat Cell Biol. 2009;11(1): 97-105. [34]Sfeir A, Kosiyatrakul ST, Hockemeyer D, et al. Mammalian telomeres resemble fragile sites and require TRF1 for efficient replication. Cell. 2009;138:90-103.[35]Smith S, Giriat I, Schmitt A, et al. Tankyrase, a poly(ADP-ribose) polymerase at human telomeres. Science. 1998;282:1484-1487.[36]Smith S, de Lange T. Tankyrase promotes telomere elongation in human cells. Curr Biol. 2000;10:1299-1302.[37]Loayza D, De Lange T. POT1 as a terminal transducer of TRF1 telomere length control. Nature. 2003;423:1013-1018.[38]Nandakumar J, Podell ER, Cech TR. How telomeric protein POT1 avoids RNA to achieve specificity for single-stranded DNA. Proc Natl Acad Sci U S A. 2010;107(2):651-656.[39]Palm W, Hockemeyer D, Kibe T, et al. Functional dissection of human and mouse POT1 proteins. Mol Cell Biol. 2009;29: 471-482.[40]Zhang DY, Wang HJ, Tan YZ. Wnt/β-catenin signaling induces the aging of mesenchymal stem cells through the DNA damage response and the p53/p21 pathway. PLoS One. 2011; 6(6):e21397. [41]Demidenko ZN, Korotchkina LG, Gudkov AV, et al. Paradoxical suppression of cellular senescence by p53. Proc Natl Acad Sci U S A. 2010;107:9660-9664.[42]Itahana K, Dimri G, Campisi J. Regulation of cellular senescence by P53. Eur J Biochem. 2001;268:2784-2791. |
| [1] | Lai Shuaiwei, Zhang Shasha, Liu Xiaoyun, Haniya Mazhar, Amber Naz, He Lin, Zhu Hongxin. Ultraviolet resistance-associated gene (Uvrag) deficiency promotes cellular senescence in the heart [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(14): 2241-2246. |
| [2] | Xu Guofeng, Li Xuebin, Tang Yifan, Zhao Yin, Zhou Shengyuan, Chen Xiongsheng, Jia Lianshun. The role of autophagy in ossification of the human ligamentum flavum [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(8): 1174-1181. |
| [3] | Zhang Fangxin, Kang Peng, Wang Qiteng, Zhang Xiao, Liu Wei, Yang Hongtao, Aierken•Amudong. Apoptosis and expression of apoptotic factors caspase-3, fas and p53 in lumbar ligamentum flavum [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(8): 1195-1199. |
| [4] | Song Xudong, He Yunwu, Li Yonglin, Chen Jing, Hu Junlan. Ultrasound-guided paravertebral nerve block for zoster-associated pain: a Meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(11): 1797-1804. |
| [5] | Zhong Qiusheng1, Xia Weichao1, Guo Meizhen1, Zhu Haiqing1, Zhong Cuiqiong1, Shao Jieqi1, He Xiaohong2, Chen Xiumin2. Sandwiched Moxibustion plus Bushen Quhan recipe for treating knee osteoarthritis: a randomized controlled trial [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(35): 5670-5675. |
| [6] | Xiong Fei1, Wei Yishan2. Reduction for developmental dysplasia of the hip in rabbits: expression levels of Caspase-3 and Bcl-2 in acetabular chondrocytes [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(31): 4974-4978. |
| [7] | Li Xianan, Tian Shaoqi, Wang Yuanhe, Liu Jiangjun, Ding Tao, Chu Guoqing, Sun Kang. Hydrogen sulfide protects articular cartilage in rabbit models of knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(31): 4992-4997. |
| [8] | Ding Huan1, Chen Lihong1, Chen Yujing1, Yue Rongzhao1, Yang Min1, Xu Guihua2 . Efficacy of different scraping protocols in the intervention of rat models of lumbar disc herniation based on urine metabolomics [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(23): 3654-3659. |
| [9] | Fan Chaoqun1, Xu Kai2, Nie Mingjian1, Xu Wenfeng1, Wang Mei1. Evaluation of the cardiopulmonary endurance: cardiopulmonary exercise test versus 6-minute two-step test [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(23): 3686-3691. |
| [10] | Yang Na1, Bao Pingping2, Lei Tao2. Adiponectin levels in plasma and periodontal tissue of mouse models of diabetic periodontitis treated by adiponectin [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(23): 3692-3697. |
| [11] | Xu Qing, Fang Haolin, Liu Yang, Zhang Cunxin, Tian Baofang. Proanthocyanidins inhibit high glucose-induced apoptosis in nucleus pulposus cells in a rabbit [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(21): 3426-3431. |
| [12] | Li Xiaoju, Song Guangbao, Yang Jianzhen, Wei Bo, Wu Bin. Effect of concentrated growth factors on proliferation and differentiation of human gingival fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(21): 3342-3348. |
| [13] | Liu Yan, Yu Xi, He Hongchen, He Chengqi, He Jing. Biological therapy in rotator cuff injury [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(20): 3248-3254. |
| [14] | Zhang Shiwei, Ma Xiulin, Qi Yusen, Luo Jinwei, Jia Tianyang, Xu Cong, Lü Yongming. Relationship between the improvement of muscle atrophy and functional outcomes after arthroscopic rotator cuff repair [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(19): 2996-3001. |
| [15] | Yuan Tao1, Wang Yongzhuo1, Huang Yuanzhang1, Xi Gang1, Wei Lei1, 2, Zhang Min1. Establishment of Indian hedgehog protein conditional knockout mouse models based on Cre/LoxP system [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(19): 3013-3018. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

