Chinese Journal of Tissue Engineering Research ›› 2020, Vol. 24 ›› Issue (22): 3566-3572.doi: 10.3969/j.issn.2095-4344.2273
Previous Articles Next Articles
Research progress of intervertebral disc repair with decellularized matrix materials
Jin Xiaoyu1, 2, Deng Rongrong1, 2, Rong Weihao3, Xie Lin1, 2, Kang Ran1, 2, 3
- 1Affiliated Hospital of Integrated Traditional Chinese and Western Medicine, Nanjing University of Chinese Medicine, Nanjing 210028, Jiangsu Province, China; 2Department of Orthopedics, Jiangsu Province Academy of Traditional Chinese Medicine, Nanjing 210028, Jiangsu Province, China; 3Department of Orthopedics, Nanjing Lishui Hospital of Traditional Chinese Medicine, Nanjing 211200, Jiangsu Province, China
-
Received:2019-09-28Revised:2019-10-26Accepted:2019-12-05Online:2020-08-08Published:2020-04-26 -
Contact:Kang Ran, MD, Chief physician, Researcher, Affiliated Hospital of Integrated Traditional Chinese and Western Medicine, Nanjing University of Chinese Medicine, Nanjing 210028, Jiangsu Province, China; Department of Orthopedics, Jiangsu Province Academy of Traditional Chinese Medicine, Nanjing 210028, Jiangsu Province, China; Department of Orthopedics, Nanjing Lishui Hospital of Traditional Chinese Medicine, Nanjing 211200, Jiangsu Province, China -
About author:Jin Xiaoyu, Master candidate, Affiliated Hospital of Integrated Traditional Chinese and Western Medicine, Nanjing University of Chinese Medicine, Nanjing 210028, Jiangsu Province, China; Department of Orthopedics, Jiangsu Province Academy of Traditional Chinese Medicine, Nanjing 210028, Jiangsu Province, China -
Supported by:the National Natural Science Foundation of China (General Project), No. 81772356
CLC Number:
Cite this article
Jin Xiaoyu, Deng Rongrong, Rong Weihao, Xie Lin, Kang Ran. Research progress of intervertebral disc repair with decellularized matrix materials[J]. Chinese Journal of Tissue Engineering Research, 2020, 24(22): 3566-3572.
share this article

2.1 去细胞化基质材料制备工艺 目前,去细胞化材料的制备工艺主要包括物理、化学和酶学方法,其基本原理均是裂解细胞,去除细胞内容物,保留细胞外基质和天然构架。物理方法中最常用的是冲击,直接外力冲击可以裂解细胞[25],也有尝试脉冲式电场破坏细胞膜裂解细胞[26],但存在冲击损伤细胞外基质的弊端。冻融循环是另一种常用的物理方法,通过液体冷冻膨胀破坏细胞膜,导致细胞裂解[27]。化学方法通过化学试剂崩解细胞膜,将相关组织和器官脱细胞,获得器官特异性的细胞外基质[28]。常用试剂有Triton X-100、SDS、EDTA,其中SDS为离子洗涤剂,存在一定的细胞毒性[29],并且会改变胶原微结构[30];Triton X-100是非离子洗涤剂,通常和氢氧化铵合用,脱细胞的同时对组织结构的损伤小[31];EDTA是一种可结合金属离子的螯合剂,常与其他脱细胞试剂联用,从而抑制细胞黏附于细胞外基质。酶裂解方法中的DNA酶能够有效裂解DNA片段,通常与脱氧胆酸钠联用[29],可溶解细胞质和核膜;胰蛋白酶用于破坏细胞基质的黏附,常和EDTA联用,以减少盐溶性和酸溶性细胞外基质蛋白[32];核酶(DNA酶和RNA酶)通过水解可以将抗原物质降到最低。实际应用中多联合以上方法制备椎间盘去细胞化组织工程支架,通过去除同种异体或异种细胞成分,理论上可利用这些细胞外基质制造出具备合适生物力学和生物特性的最低免疫原性支架,见表1。 "

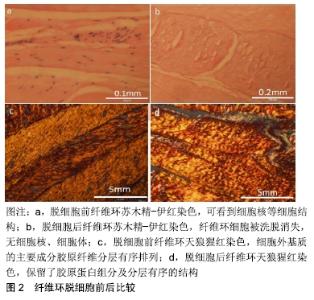
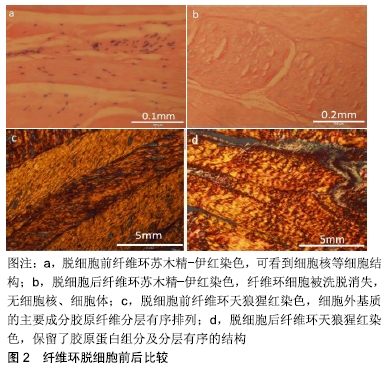
2.1.1 全椎间盘去细胞化 CHAN等[33]于2013年较早尝试了将整个椎间盘去细胞化的方法,通过液氮冻融循环和SDS、EDTA化学洗涤剂脱细胞,成功去除了70%的内源性细胞,并能够较好地保留椎间盘的糖胺聚糖含量、胶原纤维结构和力学性能。随后,LIN等[34]在物理冻融和化学洗脱的基础上加入了酶学方法,优化了椎间盘去细胞方案,通过延长反应时间和降低脱细胞试剂浓度(2%Triton X-100和1%SDS)明显提升了脱细胞效率。而且,椎间盘细胞外基质的主要生物活性成分(糖胺聚糖、蛋白聚糖、Ⅰ型胶原、Ⅱ型胶原)保存良好,去细胞化的椎间盘支架具备良好的力学性能和体外相容性,免疫原性低。通过对椎间盘细胞经典标记基因(Ⅱ型胶原、蛋白聚糖、SOX-9)和新标记基因GPC3的检测,更是发现了此脱细胞基质诱导间充质干细胞向椎间盘细胞分化的潜能。目前已经能够制备完全脱细胞的全椎间盘支架。全椎间盘包含髓核、纤维环、终板等多种复杂结构,体积较大,特别是对于人或大型动物,实现整个椎间盘去细胞化的技术难度大,质量控制不易。 此外,如何制造出具备合适孔隙率的全椎间盘支架以利于细胞接种,以及如何使得植入体内的支架能够在生理力学环境下保持稳定、与周围组织紧密贴合也是需要解决的问题。 2.1.2 纤维环去细胞化 与全椎间盘相比,纤维环结构单一,体积相对较小,对单纯的纤维环组织进行脱细胞处理能够更好的控制质量。XU等[35]于2014年报道应用Triton X-100、SDS和胰蛋白酶对猪纤维环脱细胞,结果显示Triton X-100的脱细胞效果相对最佳,在脱细胞的同时基本保留了纤维环原有的结构和生物力学性能,但有部分基质丢失。WU等[36]于2017年报道通过对比不同的物理冻融温度(-80,-196 ℃)、脱细胞时间(24,48,72 h)、脱细胞试剂(0.1%SDS、1%Triton X-100)对纤维环去细胞化的影响,建议选择液氮冻融、0.1%SDS脱细胞效果最佳,且脱细胞时间宜控制在24 h。这种去细胞方法不仅保留了纤维环基质的主要成分、细胞外基质超微结构和纤维环的生物力学属性,还显著降低了纤维环的免疫原性。作者的研究利用液氮冻融处理,0.1%SDS、0.1%EDTA、10 kIU/mL抑肽酶和50 U/mL Dnase、1 U/mL Rnase洗脱纤维环,可以得到完全去细胞的细胞外基质支架,基质中排列有序的胶原纤维结构能够被很好的保留(图2);但也面临与既往研究相似的难点,纤维环基质致密紧凑,孔隙不足,细胞接种困难。如何制备、产生合适的孔隙,利于细胞的接种及长入,还有待于进一步的研究。 "

2.1.3 髓核去细胞化 较全椎间盘和纤维环而言,髓核组织疏松、细胞稀少,有关髓核去细胞化的研究较多,在提升脱细胞效果的同时,进一步研究了如何最大程度的降低免疫原性,并从去细胞髓核基质中成功分离出抗原α-Gal,给出了有效去除抗原的方法。YUAN等[37]通过对比不同浓度脱细胞试剂对组织结构、糖胺聚糖、DNA、羟脯氨酸含量的影响,发现2%Triton X-100和1%脱氧胆酸钠均能保留80%以上的糖胺聚糖,为选择合适种类和浓度的脱细胞试剂提供了良好参考。FEMANDEZ等[38]更是评估了13对牛尾椎间盘髓核组织去细胞化方法的效果,确立了一种有效去除牛DNA的最佳方法:3次声波降解;0.4%EDTA、1.2%Triton X-100、0.02%叠氮化钠、720 U/L DNase、720 U/L Rnase试剂组合脱细胞3次。去细胞后的DNA低至9.07 μg/g,糖胺聚糖、Ⅱ型胶原的含量及力学性能接近人类髓核的报道值。在脱细胞的同时有效去除与组织器官细胞内成分相关的抗原表位,这对于减少或避免髓核支架材料的不良免疫反应是必要的。ILLIEN-JUNGER等[39]采用物理联合化学、酶学(5次冻融循环+2%脱氧胆酸钠、Dnase)方法有效去除了DNA,降低了免疫原性,同时保留了最好的胶原微结构和最高的胶原蛋白、糖胺聚糖含量,间充质干细胞在髓核支架中的高度生长活性也进一步表明了该脱细胞基质材料的良好生物相容性。MERCURI等[40]采用化学清洗、超声处理、核酶消化等方法制备出了具有良好膨胀率、生物相容性和生物力学性能的去细胞髓核支架,并成功从中分离出抗原α-Gal,极大降低了支架的免疫原性。 2.2 去细胞化基质材料质量控制 2.2.1 组织结构形态评估 要求在脱细胞的同时保留组织结构。要通过病理切片进行组织学观察观察组织脱细胞后基质结构的完整性,髓核内是纵横交错的纤维网状结构,纤维环内的胶原纤维是排列有序的同心层状结构,去细胞后的髓核、纤维环应尽量接近其原始结构。常利用苏木精-伊红染色、天狼猩红染色[35]、阿利新蓝染色对组织切片进行处理[34],再通过扫描电镜/透射电镜[34]、偏振光显微镜观察组织脱细胞前后微观结构的变化。 2.2.2 特定基质成分测定 要求脱细胞后的组织不损关键细胞外基质。免疫组化用以评价椎间盘特征性组分在基质中的大体分布[41],纤维环以Ⅰ型胶原为主,髓核以蛋白多糖、Ⅱ型胶原为主。通常进行定量评价,要求去细胞后关键基质的含量以接近组织脱细胞前为目标。胶原蛋白含量可采用羟脯氨酸检测试剂盒检测[42],因为羟脯氨酸含量约占胶原蛋白α链的10%,故可得出胶原蛋白总含量。糖胺聚糖含量多采用二甲基亚甲基蓝法进行分析。糖胺聚糖是髓核的主要成分之一,正常人髓核糖胺聚糖的平均含量为300-600 mg/g组织干质量[43]。如HENSLEY等[44]对脱细胞后的纤维环和髓核中的糖胺聚糖含量进行质控,发现含量分别降低了56.84%和45.00%。最大程度地保留糖胺聚糖,使其尽可能地接近天然椎间盘的含量是各种脱细胞方法追求的目标。可采用1,9-二甲基亚甲基蓝染料结合实验和分光光度法测定经木瓜蛋白酶消化的组织中硫酸糖胺聚糖含量,从而测定组织蛋白聚糖含量[45]。目前对于脱细胞基质中蛋白聚糖的含量要求同样没有标准,以接近脱细胞前的组织为佳。脱细胞基质的成分越接近天然基质成分效果越佳[37]。实际上,在脱细胞过程中或多或少都会损失部分细胞外基质,细胞外基质保留到多少程度可以接受,目前还没有量化的标准,仍然需要进一步的研究。 2.2.3 生物力学与机械性能评估 衡量去细胞支架力学性的几个重要参数包括弹性模量、压缩模量、极限荷载和应力值等。已有研究证实力学特性(如弹性模量)还会影响支架内植入种子细胞的行为,如黏附、增殖、分化等[46]。因此,构建良好生物力学性能的支架对于后期诱导干细胞定向分化具有重要意义。目前常采用的力学检测方式有:压缩测试,应用万能力学试验机进行压缩实验,构建应力-应变压缩曲线;动态黏弹性测试[47]。早有研究表明,生理情况下整个椎间盘需要承受1 500 N的轴向压力[48],用于体内修复的支架至少应达到此要求。目前还没有详细、精准的力学要求标准。 2.2.4 免疫原性评估 降低细胞外基质支架的免疫原性是脱细胞技术的关键要求之一,能够诱导免疫排斥反应的成分主要包括遗留的遗传物质(DNA、RNA)和抗原。CRAPO等[28]提出去细胞化后的dsDNA应小于50 μg/g 细胞外基质,DNA片段长度<200 bp。常用琼脂糖凝胶电泳、乙锭染色法等定量分析,通过DAPI或苏木精-伊红染色观察组织切片中核酸物质是否被清除而进行定性验证。同种异体和异种基因的细胞抗原会被宿主识别为外来抗原,产生不良的炎症反应或免疫介导明显的排斥反应[49-51]。有效去除与细胞膜和组织器官细胞内成分相关的抗原表位,对于减少或避免细胞外基质支架材料的同种异体和异种受体不良免疫反应是必要的。WU等[36]和MERCURI等[40]已成功从纤维环/髓核去细胞化基质中分离出α-Gal抗原,极大降低了细胞外基质支架的免疫原性。SDS被认为可有效去除这些异种抗原[52],但目前的技术仍无法完全去除同种异体/异种抗原,最大限度地减少免疫排斥反应仍然在研究中。 2.2.5 生物相容性评估 去细胞基质材料的应用必须以良好的生物相容性为前提,通常将料与组织细胞共同培养,通过评判细胞的增殖、分化能力反映脱细胞材料的生物相容性。可以采用含去细胞细胞外基质的培养基与标准培养基分别培养间充质干细胞的方法,并用活-死细胞染色法对比细胞活性、细胞活动情况;用CCK-8法检测对比间充质干细胞代谢活性,从而推断脱细胞支架的生物相容性[53-54]。 目前已有诸多体外实验用于评估细胞外基质支架的生物相容性。CHAN等[33]通过将髓核细胞植入去细胞化的牛椎间盘支架中,发现细胞能够顺利的向材料中渗透,而且髓核细胞存活率很高,从而判定其所制备的去细胞支架具有良好的生物相容性,适合椎间盘的组织工程修复。FEMANDEZ等[38]在去细胞髓核支架上培养人类间充质干细胞,结果表明细胞能够长期存活,并表现出产生细胞外基质修复椎间盘的能力。 2.3 去细胞化基质材料在椎间盘修复中的应用 2.3.1 全椎间盘 将全椎间盘脱细胞作为支架再植体内修复的研究目前还鲜报道。LIN等[34]将椎间盘的终板去除后保留纤维环和髓核,脱细胞后进行了一系列的体外实验,包括组织结构、生物相容性、力学性能等;体内实验只是将去细胞基质切碎注入损伤的椎间盘中,考察免疫排斥反应及其对干细胞分化的影响,没有从真正意义上利用完整的脱细胞支架修复椎间盘。李浩等[55]报道了同种异体腰椎间盘移植的手术技术,选择合适大小的供体,术中注意保留前、后纵韧带及对侧纤维环的连续性,可以实现在无固定条件下椎间盘的移植,共20例手术,虽有2例出现椎间盘的滑移脱出,但仍可为全椎间盘脱细胞支架植入体内提供了技术上的借鉴。全椎间盘脱细胞支架植入体内如何维持稳定可能是巨大的挑战。 2.3.2 纤维环 纤维环是由胶原纤维(主要Ⅰ型胶原)组成的多层纤维软骨环。多种高分子材料能够模仿纤维环的有序排列和层状结构,但由于纤维环结构复杂、成分分布不均,很难制造出与纤维环组成和结构相似的人工支架。因而有人尝试用天然的去细胞化纤维环支架修复椎间盘,选取了6只大鼠不相邻的2个尾椎间盘造模,沿皮肤纵向切开暴露后经纤维环至髓核作1 mm×1 mm缺口,在修复组中植入去细胞化纤维环支架,结果表明14 d后炎症反应较前减少,修复组中糖胺聚糖和胶原含量明显增加,但该研究数据有限,没有评估椎间盘修复后的力学性能[36]。 2.3.3 髓核 髓核再生是修复椎间盘的重要环节[56]。髓核是由蛋白聚糖和Ⅱ型胶原组成的集中高度水化纤维胶状核,因而对于髓核的修复多采用可注射材料,如壳聚糖[57]、透明质酸等[58],体内移植便捷。但是以上高分子合成物的生物力学性能差,而去细胞化髓核支架材料无论在结构还是力学性能上都具备明显的优势。YUAN等[37]通过腹膜后入路方式暴露兔腰椎椎间盘(L2-L5),髓核抽吸造模,应用脱细胞髓核基质作为间充质干细胞载体进行椎间盘注射,结果表明与对照组胶原微球载体相比,脱细胞髓核基质能更好地修复髓核,保持髓核的含水量。 用于修复椎间盘的去细胞组织工程材料主要包括以几种,见表2。 "

| [1] WISMER N, GRAD S, FORTUNATO G, et al. Biodegradable electrospun scaffolds for annulus fibrosus tissue engineering:effect of scaffold structure and composition on annulus fibrosus cells in vitro. Tissue Eng Part A.2012;20(3-4):672. [2] URBAN JP, ROBERTS S. Degeneration of the intervertebral disc. Arthritis Res Ther. 2003;5(3):120-130. [3] DOWDELL J, ERWIN M, CHOMA T, et al. Intervertebral Disk Degeneration and Repair.Neurosurgery.2017;80(3S):S46. [4] NAKAMICHI R, ITO Y, INUI M, et al. Mohawk promotes the maintenance and regeneration of the outer annulus fibrosus of intervertebral discs.Nat Commun.2016;7:12503. [5] LIKHITPANICHKUL M, KIM Y, TORRE OM, et al. Fibrin-genipin annulus fibrosus sealant as a delivery system for anti-TNFαdrug.Spine J.2015;15(9):2045-2054. [6] DRISCOLL TP, NAKASONE RH, SZCZESNY SE, et al. Biaxial mechanics and inter-lamellar shearing of stem-cell seeded electrospun angle-ply laminates for annulus fibrosus tissue engineering.J Orthop Res.2013;31(6):864-870. [7] KANG R, LI H, RINGGAARD S, et al. Interference in the endplate nutritional pathway causes intervertebral disc degeneration in an immature porcine model.Int Orthop.2014;38(5):1011-1017. [8] HOLM S, MAROUDAS A, URBAN JP, et al. Nutrition of the intervertebral disc:solute transport and metabolism.Connect Tissue Res.1981;8(2):101-119. [9] BARTLETT A,WALES L, HOUFBURG R, et al. Optimizing the effectiveness of a mechanical suture-based anulus fibrosus repair construct in an acute failure laboratory simulation.J Spinal Disord Tech.2013;26(7):393-399. [10] GUTERL CC, SEE EY, BLANQUER SB, et al. Challenges and strategies in the repair of ruptured annulus fibrosus.Eur Cell Mater. 2013;25:1-21. [11] KANG R, LI H, XI Z, et al. Surgical repair of annulus defect with biomimetic multilamellar nano/microfibrous scaffold in a porcine model. J Tissue Eng Regen Med.2018;12(1):164-174. [12] SILVA-CORRIA J, CORREIA SI, OLIVEIRA JM, et al. Tissue engineering strategies applied in the regeneration of the human intervertebral disk.Biotechnol Adv.2013;31(8):1514-1531. [13] XU B, XU, WU Y, et al. Intervertebral Disc Tissue Engineering with Natural Extracellular Matrix-Derived Biphasic Composite Scaffolds. PLoS One.2015;10(4):e124774. [14] ZHANG H, ZHOU L, ZHANG W. Control of scaffold degradation in tissue engineering:a review.Tissue Eng Part B Rev. 2014;20(5):492. [15] RAN KANG, DANG QUANG SVEND LE, HAISHENG LI, et al. Engineered three-dimensional nanofibrous multi-lamellar structure for annulus fibrosus repair.J Mater Chem B. 2013;1(40):5462-5468. [16] CHOI Y, PARK MH, LEE K. Tissue Engineering Strategies for Intervertebral Disc Treatment Using Functional Polymers. Polymers (Basel).2019;11(5).pii:E872.doi:10.3390/polym11050872. [17] ROTHRUFF BB, SHIMOMURA K, GOTTARDI R, et al. Anatomical region-dependent enhancement of 3-dimensional chondrogenic differentiation of human mesenchymal stem cells by soluble meniscus extracellular matrix.Acta Biomater.2017;49:140-151. [18] SCHNIDER C, LEHMANN J, VAN OSCH GJ, et al. Systematic Comparison of Protocols for the Preparation of Human Articular Cartilage for Use as Scaffold Material in Cartilage Tissue Engineering. Tissue Eng Part C Methods.2016;22(12):1095-1107. [19] KIYTAKE EA, BECK EC, DETAMORE MS. Cartilage extracellular matrix as a biomaterial for cartilage regeneration.Ann N Y Acad Sci. 2016;1383(1):139-159. [20] OT HC, CLIPPINGER B, CONRAD C, et al. Regeneration and orthotopic transplantation of a bioartificial lung.Nat Med. 2010;16(8): 927-933. [21] ITRIDIS JC, NICOLL SB, MICHALEK AJ, et al. Role of biomechanics in intervertebral disc degeneration and regenerative therapies:what needs repairing in the disc and what are promising biomaterials for its repair?Spine J. 2013;13(3):243-262. [22] KIM S H, YOON SJ, CHOI B, et al. Evaluation of various types of scaffold for tissue engineered intervertebral disc.Adv Exp Med Biol. 2006;585:167-181. [23] XU J, LIU S, WANG S, et al. Decellularised nucleus pulposus as a potential biologic scaffold for disc tissue engineering.Mater Sci Eng C Mater Biol Appl.2019;99:1213-1225. [24] SHAN Z, LIN X, WANG S, et al. An injectable nucleus pulposus cell-modified decellularized scaffold:biocompatible material for prevention of disc degeneration.Oncotarget. 2017;8(25):40276-40288. [25] YANG B, ZHANG Y, ZHOU L, et al. Development of a porcine bladder acellular matrix with well-preserved extracellular bioactive factors for tissue engineering.Tissue Eng Part C Methods. 2010;16(5):1201-1211. [26] PHILLIPS M, MAOR E, RUBINSKY B. Nonthermal irreversible electroporation for tissue decellularization.J Biomech Eng. 2010; 132(9): 91003. [27] XING Q, YATES K, TAHTINEN M, et al. Decellularization of fibroblast cell sheets for natural extracellular matrix scaffold preparation.Tissue Eng Part C Methods.2015;21(1):77-87. [28] CRAPO PM, GILBERT TW, BADYLAK SF. An overview of tissue and whole organ decellularization processes.Biomaterials. 2011;32(12): 3233-3243 [29] SYED O, WALTERS NJ, DAY RM, et al. Evaluation of decellularization protocols for production of tubular small intestine submucosa scaffolds for use in oesophageal tissue engineering. Acta Biomater.2014;10(12): 5043-505. [30] O'NEILL JD, ANFANG R, ANANDAPPA A, et al. Decellularization of human and porcine lung tissues for pulmonary tissue engineering.Ann Thorac Surg.2013;96(3):1046-1055,1055-1106. [31] SULLIVAN DC, MIRMALEK-SANI SH, DEEGAN DB, et al. Decellularization methods of porcine kidneys for whole organ engineering using a high-throughput system.Biomaterials. 2012;33(31): 7756-7764. [32] ZHOU J, FRITZE O, SCHLEICHER M, et al. Impact of heart valve decellularization on 3-D ultrastructure,immunogenicity and thrombogenicity. Biomaterials.2010;31(9):2549-2554. [33] CHAN LK, LEUNG VY, TAM V, et al. Decellularized bovine intervertebral disc as a natural scaffold for xenogenic cell studies.Acta Biomater.2013;9(2):52625272. [34] LIN X, FANG X, WANG Q, et al. Decellularized allogeneic intervertebral disc:natural biomaterials for regenerating disc degeneration. Oncotarget.2016;7(11):1212112136. [35] XU H, XU B, YANG Q, et al. Comparison of decellularization protocols for preparing a decellularized porcine annulus fibrosus scaffold.PLoS One.2014;9(1):e86723. [36] WU LC, KUO YJ, SUN FW, et al. Optimized decellularization protocol includingα-Gal epitope reduction for fabrication of an acellular porcine annulus fibrosus scaffold.Cell Tissue Bank. 2017;18(3)383-396. [37] YUAN M, YEUNG CW, LI YY, et al. Effects of nucleus pulposus cell-derived acellular matrix on the differentiation of mesenchymal stem cells.Biomaterials.2013;34(16):948-3961. [38] FERNANDEZ C, MARIONNEAUX A, GILL S, et al. Biomimetic nucleus pulposus scaffold created from bovine caudal intervertebral disc tissue utilizing an optimal decellularization procedure.J Biomed Mater Res A. 2016;104(12)3093-3106. [39] ILLIEN-JUNGER S, SEDAGHATPOUR DD, LAUDIER DM, et al. Development of a bovine decellularized extracellular matrix-biomaterial for nucleus pulposus regeneration.J Orthop Res.2016;345):876-888. [40] MERCURI JJ, GILL SS, SIMIONESCU DT. Novel tissue-derived biomimetic scaffold for regenerating the human nucleus pulposus. J Biomed Mater Res A.2011;9(2):422-435. [41] CHEN S, HUANG Y, ZHOU ZJ, et al. Upregulation of tumor necrosis factor alpha and ADAMTS-5,but not ADAMTS-4,in human intervertebral cartilage endplate with modic changes.Spine(Phila Pa 1976).2014;39(4):E817-E825. [42] ZHANG L, DONG Y, DONG Y, et al. Role of integrin-beta3 protein in macrophage polarization and regeneration of injured muscle.J Biol Chem.2012;28(9):6177-6186. [43] IATRIDIS JC, MACLEAN JJ, O'BRIEN M, et al. Measurements of proteoglycan and water content distribution in human lumbar intervertebral discs.Spine(Phila Pa 1976).2007;3(14):1493-1497. [44] HENSLEY A, RAMES J, CASLER V, et al. Decellularization and characterization of a whole intervertebral disk xenograft scaffold.J Biomed Mater Res A.2018;06(9):2412-2423. [45] FARNDALE RW, BUTTLE DJ, BARRETT AJ. Improved quantitation and discrimination of sulphated glycosaminoglycans by use of dimethylmethylene blue.Biochim Biophys Acta. 1986;883(2):173-177. [46] ENGLER AJ, SEN S, SWEENEY HL, et al. Matrix elasticity directs stem cell lineage specification.Cell.2006;126(4):677-689. [47] MIYAMOTO K, MASUDA K, KIM JG, et al. Intradiscal injections of osteogenic protein-1 restore the viscoelastic properties of degenerated intervertebral discs.Spine(Phila Pa 176). 2006;6(6):692-703. [48] NACHEMSON AL.Disc pressure measurements.Spine(Phila Pa 176). 1981;6(1):93-97. [49] ERDAG G, MORGAN JR. Allogeneic versus xenogeneic immune reaction to bioengineered skin grafts.Cell Transplant. 2004;13(6): 701-712. [50] GOCK H, MURRAY-SEGAL L, SALVARIS E, et al. Allogeneic sensitization is more effective than xenogeneic sensitization in eliciting Gal-mediated skin graft rejection.Transplantaton. 2004;77(5):751-753. [51] ROSS JR, KIRK AD, IBRAHIM SE, et al. Characterization of human anti-porcine"natural antibodies"recovered from ex vivo perfused hearts--predominance of IgM and IgG2.Transplantation. 1993;55(5): 1144-1150. [52] GONCALVES AC, GRIFFITHS LG, ANTHONY RV, et al. Decellularization of bovine pericardium for tissue-engineering by targeted removal of xenoantigens.J Heart Valv Dis. 2005;14(2): 212-217. [53] CHEN K, LIN X, ZHANG Q, et al. Decellularized periosteum as a potential biologic scaffold for bone tissue engineering.Aca Biomater. 2015;19:46-55. [54] CHEN S, JIN G, HUANG KM, et al. Lycorine suppresses RANKL-induced osteoclastogenesis in vitro and prevents ovariectomy- induced osteoporosis and titanium particle-induced osteolysis in viv.Sci Rep.2015;5:12853. [55] 李浩,侯黎升,伍耀宏,等.犬同种异体腰椎间盘移植的手术技术[J].中国矫形外科杂志,2015,23(9):840-844. [56] DING F, SHAO ZW, XIONG LM. Cell death in intervertebral disc degeneration. Poptosis.2013;18(7):777-785. [57] 田海泉,李放.体外三维支架诱导脂肪干细胞向髓核样细胞的分化[J].中国组织工程究与临床康复,2009,13(38):7417-7421. [58] BENZ K, STIPPICH C, OSSWALD C, et al. Rheological and biological properties of a hydrogel support for cells intended for intervertebral disc repair.BMC Musculoskelet Disord. 2012;13:54. |
| [1] | Yao Xiaoling, Peng Jiancheng, Xu Yuerong, Yang Zhidong, Zhang Shuncong. Variable-angle zero-notch anterior interbody fusion system in the treatment of cervical spondylotic myelopathy: 30-month follow-up [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1377-1382. |
| [2] | Lu Pan, Zhang Chunlin, Wang Yongkui, Yan Xu, Dong Chao, Yue Yisen, Li Long, Zhu Andi. Volume changes of cervical herniated discs after open-door laminoplasty and conservative treatment as assessed by three-dimensional volume method [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1395-1401. |
| [3] | Zhang Haobo, Zhao Yunan, Yang Xuejun. Role and therapeutic implications of pyroptosis in intervertebral disc degeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1445-1451. |
| [4] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [5] | Zhang Jinglin, Leng Min, Zhu Boheng, Wang Hong. Mechanism and application of stem cell-derived exosomes in promoting diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1113-1118. |
| [6] | Chen Xiaoxu, Luo Yaxin, Bi Haoran, Yang Kun. Preparation and application of acellular scaffold in tissue engineering and regenerative medicine [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 591-596. |
| [7] | Kang Kunlong, Wang Xintao. Research hotspot of biological scaffold materials promoting osteogenic differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 597-603. |
| [8] | Shen Jiahua, Fu Yong. Application of graphene-based nanomaterials in stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 604-609. |
| [9] | Wang Ruanbin, Cheng Liqian, Chen Kai. Application and value of polymer materials in three-dimensional printing biological bones and scaffolds [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 610-616. |
| [10] | Zhang Tong, Cai Jinchi, Yuan Zhifa, Zhao Haiyan, Han Xingwen, Wang Wenji. Hyaluronic acid-based composite hydrogel in cartilage injury caused by osteoarthritis: application and mechanism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 617-625. |
| [11] | Li Hui, Chen Lianglong. Application and characteristics of bone graft materials in the treatment of spinal tuberculosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 626-630. |
| [12] | Gao Cangjian, Yang Zhen, Liu Shuyun, Li Hao, Fu Liwei, Zhao Tianyuan, Chen Wei, Liao Zhiyao, Li Pinxue, Sui Xiang, Guo Quanyi. Electrospinning for rotator cuff repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 637-642. |
| [13] | Tan Guozhong, Tu Xinran, Guo Liyang, Zhong Jialin, Zhang Yang, Jiang Qianzhou. Biosafety evaluation of three-dimensional printed gelatin/sodium alginate/58S bioactive glass scaffolds for bone defect repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 521-527. |
| [14] | He Yunying, Li Lingjie, Zhang Shuqi, Li Yuzhou, Yang Sheng, Ji Ping. Method of constructing cell spheroids based on agarose and polyacrylic molds [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 553-559. |
| [15] | He Guanyu, Xu Baoshan, Du Lilong, Zhang Tongxing, Huo Zhenxin, Shen Li. Biomimetic orientated microchannel annulus fibrosus scaffold constructed by silk fibroin [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 560-566. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||