Chinese Journal of Tissue Engineering Research ›› 2017, Vol. 21 ›› Issue (1): 54-59.doi: 10.3969/j.issn.2095-4344.2017.01.010
Previous Articles Next Articles
Interleukin-1beta regulates stem cell gene and chemokine receptor gene expression of myeloma cell lines via pretreatment on bone marrow mesenchymal stem cells
Wu Yu-jiao1, Fei Xiao-ming1, Ye Wei1, Tang Yu2, Lei Fang1, Zhu Yan1
- 1Department of Hematology, 2Department of Rheumatism, Affiliated Hospital of Jiangsu University, Zhenjiang 212001, Jiangsu Province, China
-
Revised:2016-11-04Online:2017-01-08Published:2017-03-15 -
Contact:Fei Xiao-ming, M.D., Associate chief physician, Department of Hematology, Affiliated Hospital of Jiangsu University, Zhenjiang 212001, Jiangsu Province, China -
About author:Wu Yu-jiao, Studying for master’s degree, Department of Hematology, Affiliated Hospital of Jiangsu University, Zhenjiang 212001, Jiangsu Province, China -
Supported by:the National Natural Science Foundation of China, No. 81202358, 81571582; Jiangsu Provincial Health Planning and Research Project, No. Z201512
CLC Number:
Cite this article
Wu Yu-jiao, Fei Xiao-ming, Ye Wei, Tang Yu, Lei Fang, Zhu Yan . Interleukin-1beta regulates stem cell gene and chemokine receptor gene expression of myeloma cell lines via pretreatment on bone marrow mesenchymal stem cells[J]. Chinese Journal of Tissue Engineering Research, 2017, 21(1): 54-59.
share this article
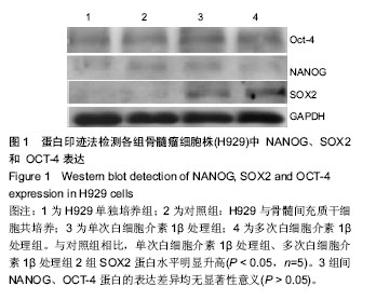
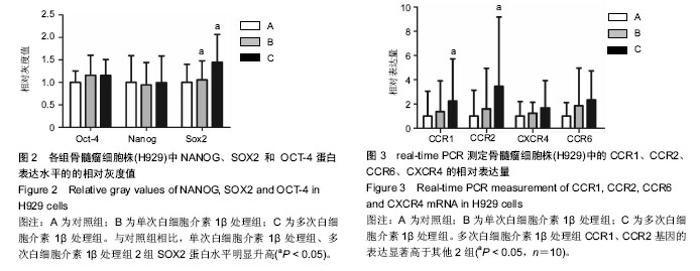
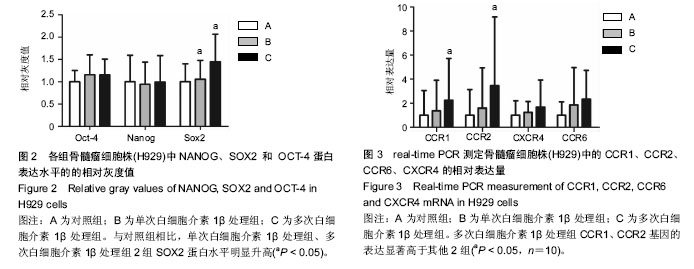
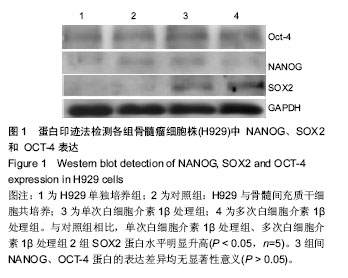
2.1 体外培养的骨髓间充质干细胞形态 各组骨髓间充质干细胞均能在体外贴壁生长。经白细胞介素1β预处理1 d 和7 d后的骨髓间充质干细胞,其形态与对照组均无明显区别,均呈梭形。 2.2 蛋白印迹法检测H929中NANOG、SOX2和OCT-4的表达 与对照组相比,单次白细胞介素1β处理组、多次白细胞介素1β处理组2组SOX2蛋白水平明显升高,相对灰度值比较差异有显著性意义(P < 0.05,n=5)。3组间NANOG、OCT-4蛋白的表达差异均无显著性意义(P > 0.05),见图1,2。 2.3 real-time RT-PCR检测H929中趋化因子受体CCR1、CCR2、CCR6、CXCR4的表达 首先对照组、单次白细胞介素1β处理组、多次白细胞介素1β处理组3组的H929 细胞均有CCR1、CCR2、CCR6、和CXCR4基因的检出。以GAPDH为内参,按 2-??Ct 法求出各基因在各标本中的相对表达量。结果提示H929 细胞 CCR1、CCR2、CCR6、和CXCR4基因表达水平均是在对照组中最低,多次白细胞介素1β处理组中最高(图3)。经统计学处理后发现:与对照组、单次白细胞介素1β处理组比较,多次白细胞介素1β处理组CCR1、CCR2基因的表达水平差异均有显著性意义 (P < 0.05,n=10),但3组间CCR6、CXCR4基因的表达差异无显著性意义(P > 0.05)。"
| [1] Garcia-Gomez A, De Las Rivas J, Ocio EM, et al. Transcriptomic profile induced in bone marrow mesenchymal stromal cells after interaction with multiple myeloma cells:implications in myeloma progression and myeloma bone disease. Oncotarget. 2014;5(18):8284-8305.[2] Bataille R, Harousseau JL.Multiple myeloma. N Eng J Med 1997;336(23):1657-1664. [3] Barillé-Nion S, Bataille R. New insights in myeloma induced osteolysis. Leuk Lymphoma. 2003;44(9): 1463-1467.[4] Noll JE, Williams SA, Tong CM,et al.Myeloma plasma cells alter the bone marrow microenvironment by stimulating the proliferation of mesenchymal stromal cells. Haematologica. 2014;99(1):163-171.[5] Xu X, Yang J, Tang Y, et al.In vitro migratory aberrancies of mesenchymal stem cells derived from mutiple myeloma patients only partially modulated by Bortezomib. Int J Clin Exp Pathol.2014;7(10):6705-6715. [6] Podar K,Richardson PG,Hideshima T,et al.The malignant clone and the bone-marrow environment. Best Pract Res Clin Haematol.2007;20(4):597-612.[7] Vidriales MB, Anderson KC. Adhesion of multiple myeloma cells to the bone marrow microenvironment: implications for future therapeutic strategies. Molecular Medicine Today. 1996; 2(10):425-431.[8] Witzig TE. The role of adhesion receptors in the pathogenesis of multiple myeloma. Hematology Oncology Clinics of North America.1999;13(6):1127-1143.[9] Grigorieva I, Thomas X, Epstein J,et al.The bone marrow stromal environment is a major factor in myeloma cell resistance to dexamethasone. Experimental Hematology. 1998;26(7):597-603.[10] Damiano JS, Cress AE, Hazlehurst LA,et al. Cell adhesion mediated drug resistance (CAM-DR): role of integrins and resistance to apoptosis in human myeloma cell lines. Blood. 1999;93(5):1658-1667.[11] Wang X, Zhang Z, Yao C. Survivin is upregulated in myeloma cell lines cocultured with mesenchymal stem cells. Leuk Res. 2010;34(10):1325-1329. [12] Skotnicki AB, Majka M. The Analysis of the Relationship between Multiple Myeloma Cells and Their Microenvironment. J Cancer.2015; 6(2):160-168.[13] Reagan, MR; Ghobrial, IM, et al. Multiple Myeloma Mesenchymal Stem Cells:Characterization, Origin, and Tumor-Promoting Effects.Clinical Cancer Research. Clin Cancer Res. 2012;18(2):342-349. [14] Wang X, Zhang Z, Yao C. Angiogenic activity of mesenchymal stem cells in multiple myeloma. Cancer Invest.2011;29(1): 37-41.[15] Garayoa M, Garcia JL, Santamaria C, et al . Mesenchymal stem cells from multiple myeloma patients display distinct genomic profile as compared with those from normal donors. Leukemia. 2009;23(8):1515-1527.[16] Garderet L, Mazurier C, Chapel A,et al.Mesenchymal stem cell abnormalities in patients with multiple myeloma. Leuk- ymphoma.2007; 48(10):2032-2041. [17] Corre J, Mahtouk K, Attal M,et al.Bone marrow mesenchymal stem cells are abnormal in multiple myeloma. Leukemia.2007; 21(5):1079-1088.[18] Maffini MV, Soto AM, Calabro JM, et al. The stroma as a crucial target in rat mammary gland carcinogenesis.J Cell Sci 2004;117(Pt 8):1495-1502. [19] Hideshima T, Mitsiades C, Tonon G, et al.Understanding multiple myeloma pathogenesis in the bone marrow to identify new therapeutic targets.Nat Rev Cancer.2007;7(8):585-98.[20] Mitsiades CS,Mitsiades NS,Munshi NC,et al.The role of the bone microenvironment in the pathophysiology and therapeutic management of multiple myeloma: interplay of growth factors, their receptors and stromal interactions. Eur J Cancer.2006; 42(11):1564-1573.[21] Podar K, Chauhan D, Anderson KC,et al. Bone marrow microenvironment and the identification of new targets for myeloma therapy. Leukemia .2009; 23(1):10-24[22] Basak GW, Srivastava AS, Malhotra R, et al. Multiple myeloma bone marrow niche. Curr Pharm Biotechnol. 2009; 10(3):345-6.[23] 夏雷,杨姣,陈诗婧,等.FCFR2,MMP1,CCL7在多发性骨髓瘤患者的骨髓微环境中的表达及其临床意义[J].江苏医药,2014; 40(12):1404-1406.[24] Bergfeld SA,DeClerck YA.Bone marrow-derived mesenchymal stem cells and the tumor microenvironment. Cancer Metastasis Rev.2010; 29(2):249-261.[25] Roccaro AM, Sacco A, Maiso P, et al.BM mesenchymal stromal cell-derived exosomes facilitate multiple myeloma progression.J Clin Invest.2013;123(4):1542-1555.[26] Nakajima S, Fujiwara T, Ohguchi H, et al.Induction of thymic stromal lymphopoietin in mesenchymal stem cells by interaction with myeloma cells.Leukemia & Lymphoma.2014; 55(11): 2605-2613.[27] Augello A, De Bari C.The regulation of differentiation in mesenchymal stem cells.Hum Gene Ther.2010;21(10): 1226-1238.[28] 杨姣,夏雷,雷芳,等.多次肿瘤坏死因子预处理骨髓间充质干细胞促进其成骨分化潜能[J].南京医科大学学报:自然科学版,2014, 34(3):275-280. [29] Pittenger MF. Mesenchymal stem cells from adult bone marrow. Methods Mol Biol. 2008; 449:27-44.[30] Giuliani N,Mangoni M,Rizzoli V,et al.Osteogenic differentiation of mesenchymal stem cells in multiple myeloma: identification of potential therapeutic targets. Exp Hematol. 2009;37(8):879-886. [31] Arnulf B,Lecourt S,Soulier J, et al. Phenotypic and functional characterization of bone marrow mesenchymal stem cells derived from patients with multiple myeloma. Leukemia.2007; 21(1):158-163. [32] Wallace SR,Oken MM,Lunetta KL, et al.Abnormalities of bone marrow mesenchymal cells in multiple myeloma patients. Cancer.2001; 91(7):1219-1230.[33] Li B, Fu J, Chen P,et al. Impairment in immunomodulatory function of mesenchymal stem cells from multiple myeloma patients. Arch Med Res. 2010;41(8):623-633.[34] Kawano M, Hirano T, Matsuda T, et al. Autocrine generation and requirement of BSF-2/IL-6 for human multiple myelomas. Nature. 1988;332(6159):83-85.[35] Schwab G,Siegall CB,Aarden LA,et al.Characterization of an interleukin-6-mediated autocrine growth loop in the human multiple myeloma cell line, U266. Blood.1991;77(3):587-593.[36] Klein B, Zhang XG, Jourdan M, et al. Paracrine rather than autocrine regulation of myeloma-cell growth and differentiation by interleukin-6.Blood. 1989;73(2):517-526.[37] Portier M, Rajzbaum G, Zhang XG, et al. In vivo interleukin 6 gene expression in the tumoral environment in multiple myeloma.Eur J Immunol.1991;21(7):1759-1762.[38] Xiong Y, Donovan KA, Kline MP, et al. Identification of two groups of smoldering multiple myeloma patients who are either high or low producers of interleukin-1. J Interferon Cytokine Res.2006;26(2):83-95.[39] Cozzolino F, Torcia M, Aldinucci D, et al. Production of interleukin-1 by bone marrow myeloma cells. Blood.1989; 74(1):380-387.[40] Torcia M, Lucibello M, Vannier E,et al. Modulation of osteoclast-activating factor activity of multiple myeloma bone marrow cells by different interleukin-1 inhibitors. Exp Hematol. 1996;24(8):868-874.[41] Ferreira E, Porter RM, Wehling N, et al.Inflammatory Cytokines Induce a Unique Mineralizing Phenotype in Mesenchymal Stem Cells Derived from Human Bone Marrow. The Journal Of Biological Chemistry.2013;288(41):29494- 29505.[42] Masui S,Nakatake Y,Toyooka Y,et al.Pluripotency governed by Sox2 via regulation of Oct3/4 expression in mouse embryonic stem cells.Nat Cell Biol.2007;9(6):625-635.[43] Nichols J,Zevnik B, Anastassiadis K, et al. Formation of pluripotent stem cells in the mammalian embryo depends on the POU transcription factor Oct4.Cell. 1998;95(3):379-391.[44] Rodda DJ, Chew JL, Lim LH,et al.Transcriptional regulation of Nanog by OCT4 and SOX2. J Biol Chem. 2005;280(26): 24731-24737.[45] Riekstina U, Cakstina I, Parfejevs V, et al. Embryonic stem cell marker expression pattern in human mesenchymal stem cells derived from bone marrow, adipose tissue, heart and dermis. Stem Cell Rev.2009;5(4):378-386.[46] Takahashi K, Yamanaka S.Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors.Cell.2006;126(4):663-676.[47] Avilion AA, Nicolis SK, Pevny LH,et al.Multipotent cell lineages in early mouse development depend on SOX2 function. Genes Dev.2003;17(1):126-140.[48] Kallas A,Pook M,Trei A,et al.SOX2 Is Regulated Differently from NANOG and OCT4 in Human Embryonic Stem Cells during Early Differentiation Initiated with Sodium Butyrate. Stem Cells International.2014;2014:298163.[49] Peltier J,Conway A,Keung AJ,et al.Akt increases sox2 expression in adult hippocampal neural progenitor cells, but increased sox2 does not promote proliferation. Stem Cells Dev.2011; 20(7):1153-1161.[50] Spisek R,Kukreja A,Chen LC,et al.Frequent and specific immunity to the embryonal stem cell-associated antigen SOX2 in patients with monoclonal gammopathy. J Exp Med. 2007; 204(4):831-840.[51] Tanno T, Lim Y, Wang Q,et al.Growth differentiating factor 15 enhances the tumor-initiating and self-renewal potential of multiple myeloma cells.Blood.2014;123(5):725-733.[52] Baggiolini M.Chemokines and leukocyte traffic. Nature.1998; 392(6676):565-568.[53] Mackay C.Chemokines: immunology’s high impact factors. Nat Immunol .2001;2(2):95-101.[54] Wright N, de Lera TL, Garcia-Moruja C, et al. Transforming growth factor-betal down-regulates expression of chemokine stromal cell-derived factor-1: functional consequences in cell migration and adhesion.Blood.2003;102(6):1978-1984.[55] Colombo M, Mirandola L, Platonova N,et al. Notch-directed micro -environment reprogramming in myeloma: a single path to multiple outcomes. Leukemia.2013;27(5):1009-1018. [56] Möller C, Strömberg T, Juremalm M, et al. Expression and function of chemokine receptors in human multiple myeloma. Leukemia . 2003;17(1):203-210.[57] Oba Y,Lee JW,Ehrlich LA ,et al.MIP-1 alpha utilizes both CCR 1 and CCR 5 to induce osteoclast formation and increase adhesion of myeloma cells to marrow stromal cells Experimental Hematology.2005;33(3):272-278.[58] Vallet S, Raje N, Ishitsuka K, et al.MLN3897, a novel CCR1 inhibitor, impairs osteoclastogenesis and inhibits the interaction of multiple myeloma cells and osteoclasts. Blood. 2007;110(10):3744-3752. [59] Moreaux J, Hose D, Kassambara A,et al.Osteoclast-gene expression profiling reveals osteoclast-derived CCR2 chemokines promoting myeloma cell migration.Blood. 2011; 117(4):1280-1290. |
| [1] | Wang Jing, Xiong Shan, Cao Jin, Feng Linwei, Wang Xin. Role and mechanism of interleukin-3 in bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1260-1265. |
| [2] | Xiao Hao, Liu Jing, Zhou Jun. Research progress of pulsed electromagnetic field in the treatment of postmenopausal osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1266-1271. |
| [3] | Tian Chuan, Zhu Xiangqing, Yang Zailing, Yan Donghai, Li Ye, Wang Yanying, Yang Yukun, He Jie, Lü Guanke, Cai Xuemin, Shu Liping, He Zhixu, Pan Xinghua. Bone marrow mesenchymal stem cells regulate ovarian aging in macaques [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 985-991. |
| [4] | Hou Jingying, Guo Tianzhu, Yu Menglei, Long Huibao, Wu Hao. Hypoxia preconditioning targets and downregulates miR-195 and promotes bone marrow mesenchymal stem cell survival and pro-angiogenic potential by activating MALAT1 [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1005-1011. |
| [5] | Liang Xuezhen, Yang Xi, Li Jiacheng, Luo Di, Xu Bo, Li Gang. Bushen Huoxue capsule regulates osteogenic and adipogenic differentiation of rat bone marrow mesenchymal stem cells via Hedgehog signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1020-1026. |
| [6] | Wen Dandan, Li Qiang, Shen Caiqi, Ji Zhe, Jin Peisheng. Nocardia rubra cell wall skeleton for extemal use improves the viability of adipogenic mesenchymal stem cells and promotes diabetes wound repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1038-1044. |
| [7] | Zhu Bingbing, Deng Jianghua, Chen Jingjing, Mu Xiaoling. Interleukin-8 receptor enhances the migration and adhesion of umbilical cord mesenchymal stem cells to injured endothelium [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1045-1050. |
| [8] | Fang Xiaolei, Leng Jun, Zhang Chen, Liu Huimin, Guo Wen. Systematic evaluation of different therapeutic effects of mesenchymal stem cell transplantation in the treatment of ischemic stroke [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1085-1092. |
| [9] | Guo Jia, Ding Qionghua, Liu Ze, Lü Siyi, Zhou Quancheng, Gao Yuhua, Bai Chunyu. Biological characteristics and immunoregulation of exosomes derived from mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1093-1101. |
| [10] | Huang Chuanjun, Zou Yu, Zhou Xiaoting, Zhu Yangqing, Qian Wei, Zhang Wei, Liu Xing. Transplantation of umbilical cord mesenchymal stem cells encapsulated in RADA16-BDNF hydrogel promotes neurological recovery in an intracerebral hemorrhage rat model [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 510-515. |
| [11] | Kang Kunlong, Wang Xintao. Research hotspot of biological scaffold materials promoting osteogenic differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 597-603. |
| [12] | Cao Wei, Mao Furong, Hu Xiaohua, Yang Xiaohong. N-6 methyladenosine RNA methylation regulates osteogenic and adipogenic differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(2): 266-270. |
| [13] | Fan Danyang, Fu Runze, Mi Jiajing, Liu Chunyan. Expression and role of cannabinoid receptors during bone remodeling [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(2): 283-288. |
| [14] | Zhou Hongbo, Yu Zhengwen, Liu Jianguo. Molecular mechanism of bone regeneration promoted by medical metal implant materials [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(10): 1588-1596. |
| [15] | Song Huifang, Tan Jiayin, Kang Yi, Li Bin, Bi Zhifei, Long Nü, Xia Zhongnian, Guo Rui. Hypoxic pretreatment enhances the protective effect of aged human bone marrow mesenchymal stem cells conditioned medium against H9C2 oxidative stress damage [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 1-6. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||