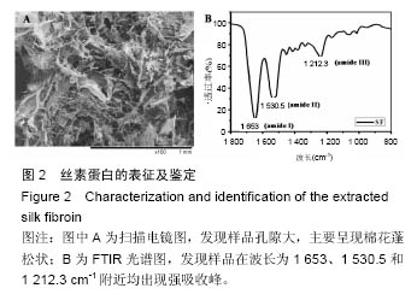
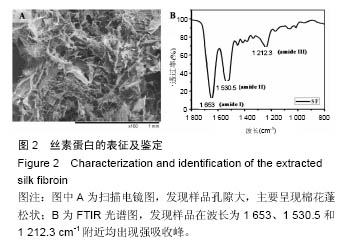
Chinese Journal of Tissue Engineering Research ›› 2016, Vol. 20 ›› Issue (52): 7788-7795.doi: 10.3969/j.issn.2095-4344.2016.52.005
Previous Articles Next Articles
Osteoinductivity and performance of silk fibroin solution
- College of Chemistry, Chemical Engineering & Biotechnology, Donghua University, Shanghai 201620, China
-
Received:2016-09-25Online:2016-12-16Published:2016-12-16 -
Contact:Zhang Yan-zhong, Ph.D., Professor, College of Chemistry, Chemical Engineering & Biotechnology, Donghua University, Shanghai 201620, China -
About author:Yi Bing-cheng, Ph.D. candidate, College of Chemistry, Chemical Engineering & Biotechnology, Donghua University, Shanghai 201620, China -
Supported by:the National Natural Science Foundation of China, No. 51073032, 31570969; the Key Project of Basic Research of Shanghai Science and Technology Commission, No. 14JC1490100
CLC Number:
Cite this article
Yi Bing-cheng, Zhang Hui-lan, Yu Zhe-pao, Yuan Hui-hua, Wang Xian-liu, Shen Yan-bing, Bao Jia-yu, Lou Xiang-xin, Zhang Yan-zhong.
share this article
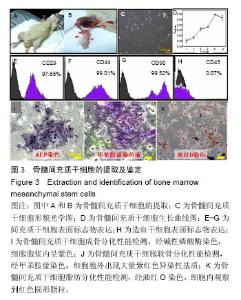
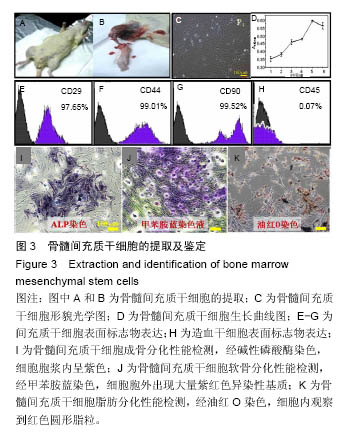
2.2 骨髓间充质干细胞的提取、培养及鉴定 实验从SD大鼠胫骨中取出骨髓细胞后进行培养并鉴定(图3)。原代细胞贴壁后,大部分细胞呈成纤维细胞样或多边形,小部分杂细胞呈圆形。当细胞传至第4代时,细胞生长最为活跃,细胞形貌一致,呈长梭形或星芒状(图3C)。然后对该细胞不同培养时间点的细胞活力进行鉴定(图3D):培养5 d内细胞一直处于增殖阶段,培养至 6 d时,细胞活力有所下降。 流式细胞仪鉴定发现(图3E-H),该细胞表面抗原标记物CD29,CD44,CD90和CD45表达率分别为97.65%,99.01%,99.52%和0.07%。 三系分化鉴定,细胞培养至14 d经碱性磷酸酶染色,细胞胞浆内呈紫色(图3I);经甲苯胺蓝染色,细胞胞外出现大量紫红色异染性基质(图3J);经油红O染色,细胞内观察到红色圆形脂粒(图3K)。"
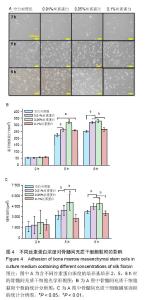
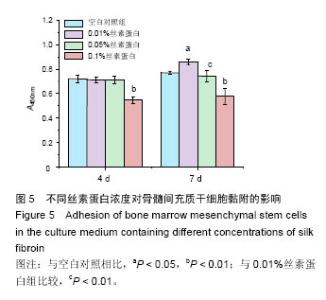
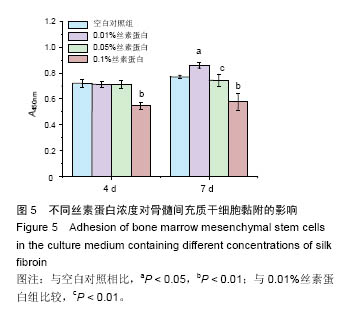
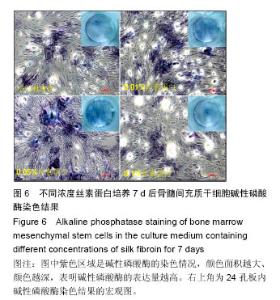
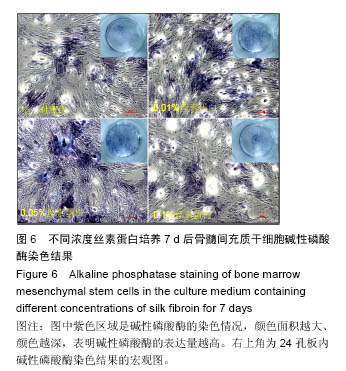
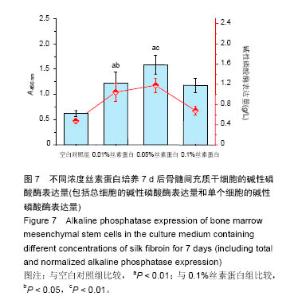
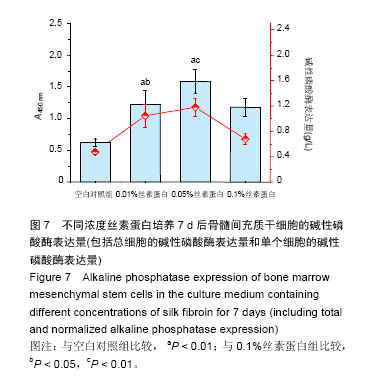
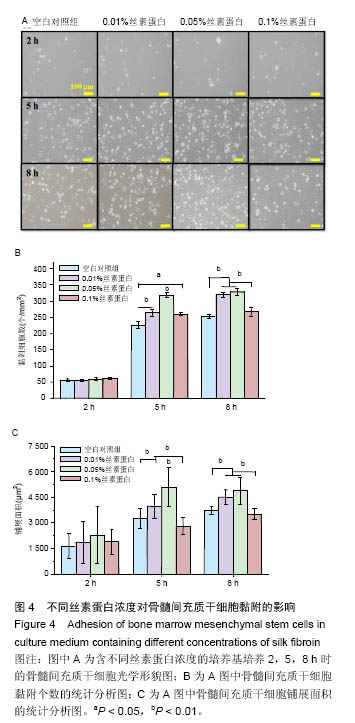
2.3 丝素蛋白溶液对骨髓间充质干细胞黏附及增殖能力的影响 实验首先选用0.01%、0.05%和0.1%这3种不同的丝素蛋白浓度,探索丝素蛋白溶液对骨髓间充质干细胞黏附行为的影响(图4A):细胞培养2 h时,仅有小部分细胞贴壁,大部分细胞仍处于悬浮状态;培养至5 h后,绝大部分细胞已沉积并贴壁,但仍有部分细胞悬浮(图中白点);继续培养至8 h时细胞形貌与5 h时基本无差异。 接着对细胞黏附情况进行定量评估,包括细胞黏附个数(图4B)及铺展面积(图4C)。结果表明低浓度丝素蛋白有利于促进骨髓间充质干细胞的黏附和铺展,且促进效果随着丝素蛋白浓度的提高而增强,但丝素蛋白浓度过高时(0.1%)对骨髓间充质干细胞黏附行为的促进作用会产生抑制作用,但仍高于空白对照(用不含丝素蛋白的纯培养基培养)。"
| [1]Setzen G, Williams EF 3rd. Tissue response to suture materials implanted subcutaneously in a rabbit model. Plast Reconstr Surg. 1997;100(7):1788-1795.[2]Altman GH, Diaz F, Jakuba C, et al. Silk-based biomaterials. Biomaterials. 2003;24(3):401-416.[3]Wang Y, Kim HJ, Vunjak-Novakovic G, et al. Stem cell-based tissue engineering with silk biomaterials. Biomaterials. 2006;27(36):6064-6082.[4]Kundu B, Rajkhowa R, Kundu SC, et al. Silk fibroin biomaterials for tissue regenerations. Adv Drug Deliv Rev. 2013;65(4):457-470.[5]Bray LJ, George KA, Ainscough SL,et al. Human corneal epithelial equivalents constructed on Bombyx mori silk fibroin membranes. Biomaterials. 2011;32(22): 5086-5091.[6]薛豪杰,胡丹丹.低溶胀壳聚糖/丝素蛋白复合膜的制备及性能测试[J].蚕业科学, 2011,37(6): 1073-1078.[7]Mottaghitalab F, Hosseinkhani H, Shokrgozar MA, et al. Silk as a potential candidate for bone tissue engineering. J Control Release. 2015;215:112-128.[8]Teimouri A,Azadi M,Emadi R,et al. Preparation, characterization, degradation and biocompatibility of different silk fibroin based composite scaffolds prepared by freeze-drying method for tissue engineering application. Polymer Degradation & Stability. 2015; 121:18-29.[9]Wenk E, Merkle HP, Meinel L. Silk fibroin as a vehicle for drug delivery applications. J Control Release. 2011; 150(2):128-141.[10]孙春光,张芃.纳米丝素颗粒药物缓释剂的研制及在治疗小鼠溃疡性结肠炎中的药物控制释放作用[J].蚕业科学, 2011,37(4):706-712.[11]Wang X, Wenk E, Zhang X, et al. Growth factor gradients via microsphere delivery in biopolymer scaffolds for osteochondral tissue engineering. J Control Release. 2009;134(2):81-90.[12]Hyun CK, Kim IY, Frost SC. Soluble fibroin enhances insulin sensitivity and glucose metabolism in 3T3-L1 adipocytes. J Nutr. 2004;134(12):3257-3263.[13]Park JH, Nam Y, Park SY, et al. Silk fibroin has a protective effect against high glucose induced apoptosis in HIT-T15 cells. J Biochem Mol Toxicol. 2011;25(4):238-243.[14]黄国平,陈克平.家蚕丝素水解物治疗糖尿病的研究进展[J].安徽农业科学,2010, 38(25): 13577-13579.[15]Byun EB, Sung NY, Kwon SK, et al. In vitro and in vivo studies on the cytotoxicity of irradiated silk fibroin against mouse melanoma tumor cell. Radiation Physics & Chemistry.2009; 78(7-8):429-431.[16]Cheema SK, Gobin AS, Rhea R, et al. Silk fibroin mediated delivery of liposomal emodin to breast cancer cells. Int J Pharm. 2007;341(1-2):221-229.[17]Gupta V, Mun GH, Choi B, et al. Repair and reconstruction of a resected tumor defect using a composite of tissue flap-nanotherapeutic-silk fibroin and chitosan scaffold. Ann Biomed Eng. 2011;39(9): 2374-2387.[18]Inouye K, Kurokawa M, Nishikawa S, et al. Use of Bombyx mori silk fibroin as a substratum for cultivation of animal cells. J Biochem Biophys Methods. 1998; 37(3):159-164.[19]Altman GH, Horan RL, Lu HH, et al. Silk matrix for tissue engineered anterior cruciate ligaments. Biomaterials. 2002;23(20):4131-4141.[20]吴海涛,钟翠平,顾云娣. 蚕丝在软骨细胞立体培养中的应用[J].中国修复重建外科杂志, 2000,14(5): 301-304.[21]Gu Y, Chen L, Yang HL, et al. Evaluation of an injectable silk fibroin enhanced calcium phosphate cement loaded with human recombinant bone morphogenetic protein-2 in ovine lumbar interbody fusion. J Biomed Mater Res A. 2011;97(2):177-185.[22]Zhang W, Wang X, Wang S, et al. The use of injectable sonication-induced silk hydrogel for VEGF(165) and BMP-2 delivery for elevation of the maxillary sinus floor. Biomaterials. 2011;32(35):9415-9424.[23]Mirahmadi F, Tafazzoli-Shadpour M, Shokrgozar MA, et al. Enhanced mechanical properties of thermosensitive chitosan hydrogel by silk fibers for cartilage tissue engineering. Mater Sci Eng C Mater Biol Appl. 2013;33(8):4786-4794.[24]Song JY, Kim SG, Lee JW, et al. Accelerated healing with the use of a silk fibroin membrane for the guided bone regeneration technique. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2011;112(6):e26-33.[25]Yang L, Yaseen M, Zhao X, et al. Gelatin modified ultrathin silk fibroin films for enhanced proliferation of cells. Biomed Mater. 2015;10(2):025003.[26]Rodríguez-Lozano FJ, García-Bernal D, Aznar-Cervantes S, et al. Effects of composite films of silk fibroin and graphene oxide on the proliferation, cell viability and mesenchymal phenotype of periodontal ligament stem cells. J Mater Sci Mater Med. 2014; 25(12):2731-2741.[27]Dhyani V, Singh N. Controlling the cell adhesion property of silk films by graft polymerization. ACS Appl Mater Interfaces. 2014;6(7):5005-5011.[28]Meechaisue C, Wutticharoenmongkol P, Waraput R, et al. Preparation of electrospun silk fibroin fiber mats as bone scaffolds: a preliminary study. Biomed Mater. 2007;2(3):181-188.[29]Li C, Vepari C, Jin HJ, et al. Electrospun silk-BMP-2 scaffolds for bone tissue engineering. Biomaterials. 2006;27(16):3115-3124.[30]Zhang K, Mo X, Huang C, et al. Electrospun scaffolds from silk fibroin and their cellular compatibility. J Biomed Mater Res A. 2010;93(3):976-983.[31]Sofia S, McCarthy MB, Gronowicz G, et al. Functionalized silk-based biomaterials for bone formation. J Biomed Mater Res. 2001;54(1):139-148.[32]蔡鹏,朱绍兴,苏一鸣,等.全骨髓贴壁法分离培养大鼠骨髓间充质干细胞及其诱导分化[J].中国组织工程研究与临床康复,2009,13(36):7073-7077.[33]Um IC, Kweon HY, Park YH, et al. Structural characteristics and properties of the regenerated silk fibroin prepared from formic acid. Int J Biol Macromol. 2001;29(2):91-97.[34]黄雷. 酶法水解丝素蛋白工艺的优化及丝素肽抗氧化活性功能的研究[D]. 沈阳:沈阳农业大学, 2006.[35]程蕾,辛伟彪,刘祉序,等.碱脱胶法优化提取蚕丝素蛋白及其构象光谱[J].广西师范大学学报:自然科学版, 2014, 32(2): 88-94.[36]Jeong L, Lee KY, Liu JW, et al. Time-resolved structural investigation of regenerated silk fibroin nanofibers treated with solvent vapor. Int J Biol Macromol. 2006;38(2):140-144.[37]Zhou J, Cao C, Ma X. A novel three-dimensional tubular scaffold prepared from silk fibroin by electrospinning. Int J Biol Macromol. 2009;45(5):504-510.[38]Chen C, Cao C, Ma X, et al. Preparation of non-woven mats from all-aqueous silk fibroin solution with electrospinning method. Polymer. 2006; 47(18): 6322-6327.[39]Zur Nieden NI, Kempka G, Ahr HJ. In vitro differentiation of embryonic stem cells into mineralized osteoblasts. Differentiation. 2003;71(1):18-27. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [3] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [4] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [5] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [6] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [7] | Fan Quanbao, Luo Huina, Wang Bingyun, Chen Shengfeng, Cui Lianxu, Jiang Wenkang, Zhao Mingming, Wang Jingjing, Luo Dongzhang, Chen Zhisheng, Bai Yinshan, Liu Canying, Zhang Hui. Biological characteristics of canine adipose-derived mesenchymal stem cells cultured in hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1002-1007. |
| [8] | Geng Yao, Yin Zhiliang, Li Xingping, Xiao Dongqin, Hou Weiguang. Role of hsa-miRNA-223-3p in regulating osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1008-1013. |
| [9] | Lun Zhigang, Jin Jing, Wang Tianyan, Li Aimin. Effect of peroxiredoxin 6 on proliferation and differentiation of bone marrow mesenchymal stem cells into neural lineage in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1014-1018. |
| [10] | Zhu Xuefen, Huang Cheng, Ding Jian, Dai Yongping, Liu Yuanbing, Le Lixiang, Wang Liangliang, Yang Jiandong. Mechanism of bone marrow mesenchymal stem cells differentiation into functional neurons induced by glial cell line derived neurotrophic factor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1019-1025. |
| [11] | Duan Liyun, Cao Xiaocang. Human placenta mesenchymal stem cells-derived extracellular vesicles regulate collagen deposition in intestinal mucosa of mice with colitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1026-1031. |
| [12] | Pei Lili, Sun Guicai, Wang Di. Salvianolic acid B inhibits oxidative damage of bone marrow mesenchymal stem cells and promotes differentiation into cardiomyocytes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1032-1036. |
| [13] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [14] | Wang Shiqi, Zhang Jinsheng. Effects of Chinese medicine on proliferation, differentiation and aging of bone marrow mesenchymal stem cells regulating ischemia-hypoxia microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1129-1134. |
| [15] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||