Chinese Journal of Tissue Engineering Research ›› 2015, Vol. 19 ›› Issue (7): 1094-1098.doi: 10.3969/j.issn.2095-4344.2015.07.020
Previous Articles Next Articles
Double effects of Chemerin on inflammation and metabolic syndrome
Li Xue-mei1, Zhai Li-dong2
- 1Key Laboratory of Hormones and Development (Ministry of Health), Metabolic Diseases Hospital & Tianjin Institute of Endocrinology, Tianjin Medical University, Tianjin 300070, China; 2Department of Human Anatomy Histology and Embryology, School of Basic Medicine, Tianjin Medical University, Tianjin 300070, China
-
Online:2015-02-12Published:2015-02-12 -
About author:Li Xue-mei, Master, Attending physician, Key Laboratory of Hormones and Development (Ministry of Health), Metabolic Diseases Hospital & Tianjin Institute of Endocrinology, Tianjin Medical University, Tianjin 300070, China
CLC Number:
Cite this article
Li Xue-mei, Zhai Li-dong. Double effects of Chemerin on inflammation and metabolic syndrome[J]. Chinese Journal of Tissue Engineering Research, 2015, 19(7): 1094-1098.
share this article
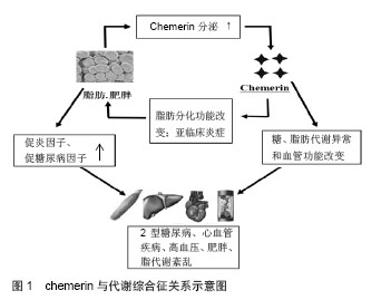
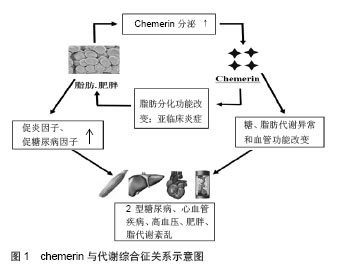
2.1 Chemerin的发现 Chemerin基因也称他佐罗汀诱导基因2(tazarotene-induced gene 2,TIG2)或视黄酸受体反应元件2(retinoic acid receptor responder 2,RARRES2),由Nag-pal等[1]于1997年培养银屑病患者的皮肤细胞时发现,他佐罗汀或维甲酸激活视黄酸受体后,Chemerin基因明显上调,曾被称为视黄酸受体应答基因2或者维甲酸受体反应元件2。2003年Wittamer等[2]在寻找G蛋白偶联受体chemerinR23(CMKLR1)的配体时,发现在炎症体液里TIGZ编码的蛋白是其配体,为了便于与chemerinR23对应,命名为Chemerin。2007年Bozaoglu等[3]用信号序列捕获技术首次鉴定出的Chemerin是一个新的脂肪因子,通过自分泌和旁分泌的方式分泌。 2.2 Chemerin的结构及生物活性 人类chemerin基因定位于E2DL3基因[4]。Chemerin基因编码一个含163个氨基酸残基的蛋白质.属无活性前体分泌蛋白,即prochemerin,相对分子质量为18 000,这种前体蛋白的生物活性很低,需要在凝血、纤溶、炎症级联反应过程中在细胞外进一步经过纤溶酶、羧肽酶或丝氨酸蛋白酶对C末端进行裂解才能成为有活性的蛋白[2,5-6]。prochemerin经胞外蛋白酶水解羧基端的一段序列后转变为相对分子质量为16 000有活性的chemerin,出现在血清、血浆和体液中[1,7]。chemerin及其受体chemerinR基因在白色脂肪组织高表达。目前认为,白色脂肪细胞是唯一同时高表达chemerin及chemerinR的细胞;白色脂肪细胞是chemerin-chemerinR自分泌/旁分泌信号的靶细胞[8]。 目前认为,内源性活化的chemerin之所以具有如此广泛而多样的生理学效应可能与其胞外存在多种蛋白酶对prochemerin产生不同的酶解方式相关。Chemerin 在羧基末端有多个蛋白酶切割位点。例如,来自于卵巢、血清和血液过滤的chemerin分别缺失6,8和9个C末端氨基酸残基[7,9]。这个发现与研究者观察到的多个酶都能将chemerin裂解成有活性的蛋白一致。在某些情况下需要多重裂解以激活chemerin。例如,最初由类胰蛋白酶裂解成含有158个氨基酸残基的chemerin活性非常低,然而它却可以作为羧肽酶N或B再次裂解的底物,形成具有完全生物活性的chemerin[10]。而蛋白水解过程被认为也参与了chemerin的灭活。因此,chemerin的蛋白酶解加工可能是其调节机制的关键,这可能决定全身和局部的有活性chemerin的浓度。 2.3 与炎症的关系 Chemerin最初是以炎症因子被发现的,研究发现chemerin通过其受体CMKLR1促进未成熟的树突细胞和巨噬细胞趋化[2]。目前发现CMKLR1在很多免疫细胞都有表达,包括未成熟的树突状细胞、髓样树突细胞、巨噬细胞和自然杀伤细胞[2,11-12],在先天性免疫和获得性免疫中都发挥着非常重要的作用。Chemerin血清浓度与炎症因子、肿瘤坏死因子α、白细胞介素6和C反应蛋白水平有关[13]。在病理情况下,chemerin表达于银屑病患者[1],一些先天免疫的效应细胞受其影响,包括树突细胞、单核细胞、巨噬细胞和自然杀伤细胞[14]。Chemerin的表达被认为是早期损害的一个标志,chemerin募集并激活类浆细胞树突细胞[15]。这意味着chemerin 及其受体CMKLR1参与了不同免疫细胞向损伤部位的募集,并可能影响炎症发生、发展[16]。Chemerin及其受体在很多炎症状态时升高,如ChemR23在吸烟诱导的炎症时在肺细胞系高表达[17],慢性阻塞性肺疾病患者血浆chemerin水平升高[18]。在对牙周炎患者的研究中发现牙周炎患者唾液中chemerin浓度升高,并且与牙周组织的破坏程度相关,唾液中chemerin浓度可能成为牙周疾病严重程度的预测因子[19]。 虽然许多实验支持chemerin/CMKLR1的促炎作用,但其他一些研究显示这一信号途径也可能具有抗炎作用。例如,Luangsay等[20]在脂多糖诱导的小鼠急性肺炎模型证实,chemerin处理后气道巨噬细胞动员增加,中性粒细胞的募集和活化减少,表明了chemerin/ CMKLR1的抗炎和促炎的双重作用。在CMKLR1缺失鼠并不能观测到这些chemerin诱导效应。另外,Cash等报道,在酵母多糖诱导的鼠腹膜炎模型中,chemerin处理后中性粒细胞和单核细胞募集明显减少,同时促炎因子表达减少[21]。CMKLR1敲除鼠的缺失效应支持了chemerin的抗炎作用通过CMKLR1发挥作用的理论。Chemerin在不同的炎症状态下作用不同,可能与其在不同的炎症环境中裂解方式的不同有关。 2.4 与代谢综合征的关系 肥胖、糖尿病、胰岛素抵抗与慢性低水平炎症之间存在着公认的联系[22],chemerin在炎症中的重要作用提示其可能与代谢综合征密切相关。大量临床数据也支持了chemerin与肥胖和代谢综合征之间的联系。例如,一项对高加索人研究发现,患有代谢综合征患者血清chemerin水平明显高于健康对照组,且与血糖、三酰甘油、收缩压、舒张压成正相关[23]。Sang Hui Chu等的研究也认为chemerin在代谢综合征的发病中发挥重要作用(图1) [24]。 2.4.1 与肥胖的关系 一项单样本研究显示,经过以减肥为目的的外科手术后,chemerin明显降低,并且与体质量指数和脂肪含量相关[24]。另外,从肥胖女性分离培养的脂肪组织分泌的chemerin明显高于消瘦女性[17]。最近的动物研究也得出相似结论,肥胖和糖尿病鼠血清chemerin增高[25]。这提示chemerin可能在肥胖的发生发展中发挥着非常重要的作用。 近年来,肥胖与慢性低水平全身炎症的相关性已经明确,慢性低度炎症是肥胖相关代谢及心血管疾病发生的关键因素。在对非糖尿病的肥胖患者的研究中发现血清chemerin水平与体质量指数相关[26]。病理状态下,chemerin大量存在于人体不同的炎性体液中,通过chemerin-CMKLR1轴实现对局部炎症反应的调节[27]。Sell等[28]研究发现,chemerin血清浓度与炎症介质超敏C反应蛋白、白细胞介素6、肿瘤坏死因子α呈正相关。促炎因子肿瘤坏死因子α、白细胞介素6等能显著增加脂肪组织Chemerin及CMKLR1的表达和chemerin的分泌。肥胖症患者的肿瘤坏死因子α、白细胞介素6、C-反应蛋白等炎症标记物的血清水平明显增加[29],脂肪组织是产生这些细胞因子的主要场所[30]。另外,肥胖者白色脂肪组织的巨噬细胞浸润明显增加[31]。这些巨噬细胞产生的前炎症细胞因子通过干扰胰岛素信号通路增加胰岛素抵抗[30]。白色脂肪组织是chemerin的主要表达器官,随着脂肪细胞的分化和肥胖程度的增加chemerin的分泌增加。目前尚无实验证实chemerin/CMKLR1信号途径与脂肪组织炎症之间的联系;然而,对chemerin免疫功能的一些研究资料提示了这一联系的可能。 Chemerin的另一项重要生物学作用是调节脂肪分化、成熟和代谢,chemerin的表达失衡可能会导致脂肪的生成和代谢的失衡,从而影响肥胖的病理过程;Chemerin增强脂肪组织胰岛素的敏感性[32],但却诱导外周组织的胰岛素抵抗[28],同时chemerin的促脂解作用会使游离脂肪酸的释放增加,而加重胰岛素抵抗。Chemerin的这些特性势必也会影响到肥胖的发生发展。文献报道,虽然chemerin基因的单核苷酸多态性与总体肥胖无关,但在偏瘦的个体中与内脏脂肪含量有关[33]。这些数据表明,chemerin影响局部脂肪分布,尤其是内脏脂肪,内脏脂肪的增多与伴随肥胖出现的代谢紊乱密切相关。 2.4.2 与脂代谢及心血管的关系 Herová等[34]研究发现,血浆chemerin水平与C-反应蛋白、体质量指数、和低密度脂蛋白相关,炎症和肥胖是冠心病发病的危险因素,进一步研究发现,服用低剂量阿司匹林的冠心病患者血浆chemerin与对照组无明显差别,而未服用低剂量阿司匹林的冠心病患者血浆chemerin水平明显升高,这提示阿司匹林可能通过chemerin降低心血管疾病的发病风险。Malin等[35]的一项前瞻性临床试验研究显示,30名老年肥胖患者经12周运动训练后血浆chemerin水平降低,并且可能与内脏脂肪、总体脂肪的减少及三酰甘油、胆固醇、葡萄糖刺激的胰岛素分泌降低有关,提示chemerin可能是肥胖和代谢调节的重要环节。 虽然证据表明血液循环中chemerin水平与代谢综合征的多个方面都有联系,但是局部浓度可能是决定心血管等疾病病理结果更重要的因素。例如在人类,血清chemerin水平仅与冠状动脉斑块及非钙化斑块数目成弱相关[36]。而且,在矫正已经明确的心血管危险因素后,这种相关性不再存在。然而,另外一项研究显示,在主动脉和冠状动脉粥样硬化时其周围的脂肪组织中chemerin表达升高[37]。这表明局部分泌的chemerin以旁分泌的方式影响动脉粥样硬化的发生发展。因此,虽然血清chemerin水平并不能预示动脉粥样硬化,但是局部chemerin浓度可能影响斑块的发生发展。动脉粥样硬化是一个进展性炎症性疾病,巨噬细胞在粥样斑块的聚集与疾病的进展明确相关。一个可能的解释是冠状动脉周围局部增加的chemerin促进了巨噬细胞的聚集,影响粥样斑块内的炎症反应。这需要进一步的研究阐明chemerin在炎症、斑块始发和进展中的作用。 2.4.3 与血压的关系 Yang等[38]研究报道新诊断的2型糖尿病合并高血压的患者血浆chemerin水平升高。Gu等[39]对237例新诊断的原发性高血压患者进行病例对照研究发现,高血压患者血清chemerin水平明显升高,相关分析发现chemerin水平升高是高血压发生的独立预测指标,在校正性别、年龄及其他代谢危险因素后仍与高血压独立相关。Meric等[40]在对60例高血压患者和30例健康对照者研究发现,与杓状高血压患者和正常血压者相比,非杓状高血压患者血浆chemerin水平明显升高。因此,许多研究都报道了chemerin水平与血压的相关性,但其机制还有待进一步研究。Chemerin也可能成为高血压治疗的新靶点。 2.4.4 与糖代谢关系 为了研究chemerin在糖尿病发生早期中的作用,Zhuang等[41]对74例糖耐量正常的2型糖尿病的一级亲属进行研究发现,其血浆chemerin水平明显升高,提示其可能与胰岛素抵抗的发生发展有关。雄性OLETF大鼠是理想的胰岛素抵抗和2型糖尿病模型,其不同阶段的病理生理变化与临床2型糖尿病患者的病理表现极为相似,有研究发现OLETF模型组网膜及皮下脂肪组织中chemerin和CMKLRl基因表达明显增加,说明脂肪因子chemerin及其受体基因表达的变化可能是肥胖、胰岛素抵抗和2型糖尿病发病的机制之一[42]。在3T3-Ll细胞,Chemerin以自分泌和旁分泌的方式释放,通过与ChemerinR结合,增强胰岛素样受体底物l酪氨酸的磷酸化水平,增强胰岛素刺激信号,从而调节脂肪组织胰岛素的敏感性[32]。也有学者在对3T3脂肪细胞的研究发现chemerin可下调胰岛素刺激的葡萄糖的吸收[43]。这些研究得出不同的结论,可能与其研究方法不同有关,不同的浓度,不同的处理过程和条件可能导致结论的相反,作用时间短、剂量低可能刺激葡萄糖吸收,而长时间高剂量的处理可能引起负反馈调节或者可能导致抵抗状态而产生降低葡萄糖摄取的网络。另外对人骨骼肌细胞研究显示,Chemerin能使骨骼肌细胞葡萄糖摄取减少,诱导骨骼肌细胞产生胰岛素抵抗[44]。Takahashi等[45]研究认为鼠胰岛β细胞同时表达chemrin及CMKLR1,chemrin也可能通过调节β细胞功能影响糖代谢。多项研究已经证实chemerin参与了血糖内环境的调节,chemerin通过何种机制改变血糖内环境的稳态还不十分清楚,还需要进一步实验来证实其在糖代谢中的作用。"
| [1]Nagpal S, Patel S, Jacobe H, et al. Tazarotene-induced gene 2(TIG2), a novel retinoid-responsive gene in skin.J Invest Dermatol.1997;109(1):91-95. [2]Wittamer V, Franssen JD, Vuleano M, et al. Specific recruitment of antigen-presenting cells by chemerin,a novel processed ligand from human inflammatory fluids. J Exp Med. 2003;198(7):977-985. [3]Bozaoglu K, Bolton K, McMillan J, et al. Chemerin is a novel adipokine associated with obesity and metabolic syndrome. Endocrinology. 2007;148(10):4687-4694. [4]Bozaoglu K, Curran JE, Stocker CJ, et al.Chemerin, a novel adipokine in the regulation of angiogenesis. J Clin Endocrinol Metab. 2010;95(5):2476-2485. [5]Zabel BA, Ohyama T, Zuniga L, et al.Chemokine-like receptor 1 expression by macrophages in vivo: regulation by TGF-beta and TLR ligands. Exp. Hematol. 2006;34(8):1106-1114. [6]Zabel BA, Nakae S, Zúñiga L, et al. Mast cell-expressed orphan receptor CCRL2 binds chemerin and is required for optimal induction of IgE-mediated passive cutaneous anaphylaxis. J Exp Med. 2008;205(10):2207-2220. [7]Zabelb A, Aliens J, Kulig P, et al. Chemerin activation by serine proteases of the coagulation, fibrinolytic,and inflammalory cascades. J Biol Chem.2005; 280(41):34661-34666. [8]Goralski KB, McCarthy TC, Hanniman EA, et al. Chemerin,a novel adipokine that regulates adipogenesis and adipocyte metabolism. J Biol Chem.2007;282(38):28175-28188. [9]Berg V, Sveinbj rnsson B, Bendiksen S, et al. Human articular chondrocytes express ChemR23 and chemerin; ChemR23 promotes inflammatory signaling upon binding the ligand chemerin (21-157). Arthritiis Res Ther.2010;12(6):R228. [10]Du XY, Zabel BA, Myles T, et al. Regulation of chemerin bioactivity by plasma carboxypeptidase N,carboxypeptidase B(activated thrombin-activable fibrinolysis inhibitor), and platelets. J Biol Chem. 2009;284(2):751-758. [11]Bondue B, Vosters O, de Nada?¨ P, et al. ChemR23 Dampens Lung Inflammation and Enhances Anti-viral Immunity in a Mouse Model of Acute Viral Pneumonia. PLoS Pathog. 2011; 7(11):e1002358. [12]Bondue B, Wittamer V, Parmentier M,et al. Chemerin and its receptors in the leukocyte trafficking, inflammation and metabolism. Cytokine Growth FactorRev.2011;22:(5-6): 331-338. [13]Weigert J, Neumeier M, Wanninger J, et al. Systemic chemerin is related to inflammation rather than obesity in type 2 diabetes.Clin Endocrinol (Oxf). 2010; 72(3):342-348 [14]Ottaviani C, Nasorri F, Bedini C, et al. CD56 bright CD16(-) NK cells accumulate in psoriatic skin in response to CXCL10 and CCL5 and exacerbate skin inflammation. Eur J Immunol. 2006;36(1):118-128. [15]Skrzeczyńska-Moncznik J, Wawro K, Stefańska A, et al. Potential role of chemerin in recruitment of plasmacytoid dendritic cells to diseased skin. Biochem Biophys Res Commun. 2009;380(2):323-327. [16]Moretta A, Marcenaro E, Parolini S, et al. NK cells at the interface between innate and adaptive immunity. Cell Death Differ. 2008;15(2):226-233. [17]Demoor T, Bracke KR, Dupond LL, et al. The role of ChemR23 in the induction and resolution of cigarette smoke-induced inflammation. J Immunol.2011;186(9):5457-5467. [18]Boyuk B, Guzel EC, Atalay H, et al. Relationship between plasma chemerin levels and disease severity in COPD patients. Clin Respir J. 2014 May 27. doi:10.1111/crj.12164. [Epub ahead of print] [19]Ozcan E, Saygun NI, Serdar MA, et al. Evaluation of the salivary levels of visfatin, chemerin, and progranulin in periodontal inflammation. Clin Oral Investig. 2014,28. [Epub ahead of print] [20]Luangsay S, Wittamer V, Bondue B, et al. Mouse ChemR23 Is Expressed in Dendritic Cell Subsets and Macrophages, and Mediates an Anti-Inflammatory Activity of Chemerin in a Lung Disease Model. J Immunol. 2009;183(10):6489-6499. [21]Cash JL, Hart R, Russ A, et al.Synthetic chemerin-derived peptides suppress inflammation through ChemR23. J. Exp. Med. 2008;205(4):767-775. [22]Olefsky JM, Glass CK. Macrophages, inflammation, and insulin resistance. Annu Rev Physiol. 2010;72: 219-246. [23]Stejskal D, Karpisek M, Hanulova Z, et al. Chemerin is an independent marker of the metabolic syndrome in a Caucasian population–a pilot study. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub.2008;152(2):217-221. [24]Sell H, Divoux A, Poitou C, et al. Chemerin correlates with markers for fatty liver in morbidly obese patients and strongly decreases after weight loss induced by bariatric surgery. J Clin Endocrinol Metab.2010;95(6):2892-2896. [25]Ernst MC, Issa M, Goralski KB, et al. Chemerin exacerbates glucose intolerance in mouse models of obesity and diabetes. Endocrinology.2010;151(5):1998-2007. [26]Sledzinski T, Korczynska J, Hallmann A, et al. The increase of serum chemerin concentration is mainly associated with the increase of body mass index in obese, non-diabetic subjects. J Endocrinol Invest. 2013;36(6):428-434. [27]Lehrke M, Becker A, Greif M, et al. Chemerin is associated with markers of inflammation and components of the metabolic syndrome but does not predict coronary atherosclerosis. Eur J Endocrinol.2009;161(2):339-344. [28]Sell H, Laurencikiene J, Taube A, et al. Chemerin is a novel adipocyte-derived factor inducing insulin resistance in primary human skeletal muscle cells. Diabetes. 2009;58(12): 2731-2740. [29]Cottam DR, Mattar SG, Barinas-Mitchell E, et al. The chronic inflammatory hypothesis for the morbidity associated with morbid obesity: implications and effects of weight loss. Obes Surg. 2004;14(5):589-600. [30]Trayhurn P, Wood IS. Signalling role of adipose tissue: adipokines and inflammation in obesity. Biochem Soc Trans. 2005;33(pt5):1078-1081. [31]Caspar-Bauguil S, Cousin B, Bour S, et al. Adipose tissue lymphocytes: types and roles. J Physiol Biochem. 2009;65(4): 423-436. [32]Takahashi M, Takahashi Y, Takahashi K, et al. Chemerin enhances insulin signaling and potentiates insulin-stimulated glucose uptake in 3T3-L1 adipocytes.FEBS Lett.2008; 582(5): 573-578. [33]Müssig K, Staiger H, Machicao F, et al. RARRES2, encoding the novel adipokine chemerin, is a genetic determinant of disproportionate regional body fat distribution: a comparative magnetic resonance imaging study. Metabolism.2009; 58(14): 519-524. [34]Herová M, Schmid M, Gemperle C, et al. Low dose aspirin is associated with plasma chemerin levels and may reduce adipose tissue inflammation. Atherosclerosis. 2014;235(2): 256-262. [35]Malin SK, Navaneethan SD, Mulya A, et al. Exercise-induced lowering of chemerin is associated with reduced cardiometabolic risk and glucose-stimulated insulin secretion in older adults. J Nutr Health Aging. 2014;18(6):608-615. [36]Graham KL, Zabel BA, Loghavi S, et al. Chemokine-like receptor-1 expression by central nervous system-infiltrating leukocytes and involvement in a model of autoimmune demyelinating disease. Immunol. 2009;183(10):6717-6723. [37]Spiroglou SG, Kostopoulos CG, Varakis JN, et al. Adipokines in periaortic and epicardial adipose tissue: differential expression and relation to atherosclerosis. Atheroscler Thromb. 2010;17(2):115-130. [38]Yang M, Yang G, Dong J, et al. Elevated plasma levels of chemerin in newly diagnosed type 2 diabetes mellitus with hypertension. J Investig Med. 2010;58(7):883-886. [39]Gu P, Jiang W, Lu B, et al. Chemerin is associated with inflammatory markers and metabolic syndrome phenotypes in hypertension patients. Clin Exp Hypertens. 2014;36(5):326-332. [40]Meric M, Soylu K, Avci B, et al. Evaluation of plasma chemerin levels in patients with non-dipper blood pressure patterns. Med Sci Monit. 2014;28;20:698-705. [41]Zhuang XH, Sun FD, Chen SH, et al. Circulating chemerin levels are increased in first-degree relatives of type 2 diabetic patients.Clin Lab. 2014;60(6):983-988. [42]李冰,杨菊红,王楠,等.OLETF大鼠脂肪组织chemerin及其受体1的基因表达[J].中华内分泌代谢杂志,2010,26(5):413-415. [43]Kralisch S, Weise S, Sommer G,et al. Interleukin-1beta induces the novel adipokine chemerin in adipocytes in vitro. Regul Pept. 2009;154 (1-3):102-106. [44]Becker M, Rabe K, Lebherz C, et al.Expression of human chemerin Induces insulin resistance in the skeletal muscle but does not affect weight, lipid levels, and atherosclerosis in lDL receptor knockout mice on high-fat diet. Diabetes.2010; 59(11):2898-2903. [45]Takahashi M, Okimura Y, Iguchi G, et al. Chemerin regulates b-cell function in mice.Sci Rep.2011;1:123. |
| [1] | Geng Qiudong, Ge Haiya, Wang Heming, Li Nan. Role and mechanism of Guilu Erxianjiao in treatment of osteoarthritis based on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1229-1236. |
| [2] | Gu Xia, Zhao Min, Wang Pingyi, Li Yimei, Li Wenhua. Relationship between hypoxia inducible factor 1 alpha and hypoxia signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1284-1289. |
| [3] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [4] | Wang Zhengdong, Huang Na, Chen Jingxian, Zheng Zuobing, Hu Xinyu, Li Mei, Su Xiao, Su Xuesen, Yan Nan. Inhibitory effects of sodium butyrate on microglial activation and expression of inflammatory factors induced by fluorosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1075-1080. |
| [5] | Xie Wenjia, Xia Tianjiao, Zhou Qingyun, Liu Yujia, Gu Xiaoping. Role of microglia-mediated neuronal injury in neurodegenerative diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1109-1115. |
| [6] | Yang Yang, Yao Yu, Shen Xiaotian, Liu Jiajia, Xue Jianhua. Expression and significance of interleukin-21 in intervertebral disc degeneration [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 690-694. |
| [7] | Ma Binxiang, He Wanqing, Zhou Guangchao, Guan Yonglin. Triptolide improves motor dysfunction in rats following spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 701-706. |
| [8] | Song Shan, Hu Fangyuan, Qiao Jun, Wang Jia, Zhang Shengxiao, Li Xiaofeng. An insight into biomarkers of osteoarthritis synovium based on bioinformatics [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 785-790. |
| [9] | Zhao Xiang, Wei Cuilan, Zhang Yeting. Neurogenesis and neuroinflammation under exercise: alteration and regulation [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 813-820. |
| [10] | Chen Ziyang, Pu Rui, Deng Shuang, Yuan Lingyan. Regulatory effect of exosomes on exercise-mediated insulin resistance diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 4089-4094. |
| [11] | Xu Xiaoming, Chen Yan, Song Qian, Yuan Lu, Gu Jiaming, Zhang Lijuan, Geng Jie, Dong Jian. Human placenta derived mesenchymal stem cell gel promotes the healing of radiation skin damage in SD rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 3976-3980. |
| [12] | Bi Qingwei, Liu Chengpu, Li Yan, Zhao Wenwen, Han Mei. Structure analysis of platelet-rich fibrin derived from two centrifugation procedures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3534-3539. |
| [13] | Wang Chaoge, Weng Xiquan, Lin Baoxuan, Chen Lina, Xu Guoqin. Exercises under cold exposure change fat type and function in obese rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(20): 3162-3167. |
| [14] | Cui Tiantian, Yi Lan, Ouyang Hougan, Wu Huiting, Ouyang Yanchu, Chen Chu. Effect of thermosensitive moxibustion in a rat model of pelvic inflammation based on trifocal focal membrane theory [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(20): 3168-3172. |
| [15] | Song Shilei, Chen Yueping, Zhang Xiaoyun, Li Shibin, Lai Yu, Zhou Yi. Potential molecular mechanism of Wuling powder in treating osteoarthritis based on network pharmacology and molecular docking [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(20): 3185-3193. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||