Chinese Journal of Tissue Engineering Research ›› 2015, Vol. 19 ›› Issue (5): 697-703.doi: 10.3969/j.issn.2095-4344.2015.05.008
Previous Articles Next Articles
Activation of Akt/mTOR/p70S6K signaling pathway limits excessive astrocytic responses after spinal cord injury in rats
Hu Ling-yun1, Zhang Jian-ying2, Lin Hong1, Gou Lin1, Lin Tao1, Li Wei1, Liu Kang3, Sun Zhen-gang4
- 1Department of Orthopaedics, 2Imaging Center, Nanchong Central Hospital, Second Clinical Medical College of North Sichuan Medical College, Nanchong 637000, Sichuan Province, China; 3Institute of Tissue Engineering and Stem Cells, North Sichuan Medical College, Nanchong 637000, Sichuan Province, China; 4Department of Orthopaedics, the First People’s Hospital of Qingdao Economic Development District, Qingdao 266555, Shandong Province, China
-
Revised:2014-11-25Online:2015-01-30Published:2015-03-02 -
About author:Hu Ling-yun, Master, Attending physician, Second Clinical Medical College of North Sichuan Medical College, Department of Orthopaedics, Nanchong Central Hospital, Nanchong 637000, Sichuan Province, China -
Supported by:the National Natural Science Foundation of China, No. 81100929
CLC Number:
Cite this article
Hu Ling-yun, Zhang Jian-ying, Lin Hong, Gou Lin, Lin Tao, Li Wei, Liu Kang, Sun Zhen-gang . Activation of Akt/mTOR/p70S6K signaling pathway limits excessive astrocytic responses after spinal cord injury in rats[J]. Chinese Journal of Tissue Engineering Research, 2015, 19(5): 697-703.
share this article
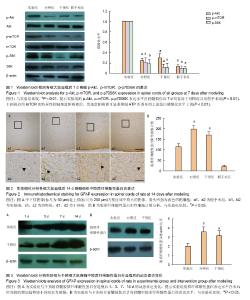
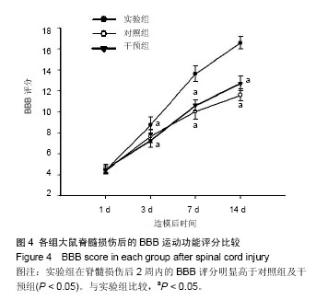
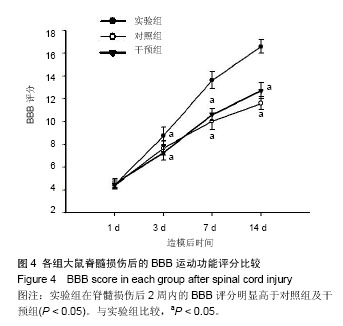
2.1 实验动物数量分析 实验分批进行,脱落动物在下一批随机分组补充。总共饲养104只SD大鼠,96只纳入实验,8只不纳入实验统计:3只死于造模过程中,5只死于后期膀胱破裂或泌尿系感染。 2.2 ATP激活脊髓损伤后脊髓组织中的Akt/mTOR/ p70S6K信号通路 Western blot检测结果发现,实验组的p-Akt、p-mTOR、p-p70S6K表达水平在脊髓损伤后7 d明显高于对照组以及假手术组(P < 0.01,图1)。应用mTOR的特异性抑制剂雷帕霉素后,发现雷帕霉素可显著抑制ATP所诱导的上述蛋白磷酸化水平上调(P < 0.01,图1)。mTOR的Ser2448位点磷酸化是体内、体外依赖Akt磷酸化的主要位点[5],Thr389位点是mTOR在体内与体外激活p70S6K的主要位点[29],且p70S6K的磷酸化依赖于mTOR,提示ATP诱导了受损脊髓组织中Akt/mTOR/ p70S6K信号通路的激活。 2.3 激活的Akt/mTOR/p70S6K信号对脊髓损伤后星形胶质细胞增生反应的影响 免疫组织化学检测发现,在脊髓损伤后脊髓组织中的星形胶质细胞显著增生,胶质纤维酸性蛋白的表达水平明显高于假手术组(图2A)。在脊髓损伤后14 d,实验组的胶质纤维酸性蛋白表达水平低于对照组,而雷帕霉素阻碍了由ATP所诱导的此种效应。细胞计数发现实验组的胶质纤维酸性蛋白阳性细胞明显少于对照组与干预组(P < 0.05,图2B),但实验组星形胶质细胞的增殖反应及胶质纤维酸性蛋白表达并未完全受到抑制。与实验组相比,对照组与干预组中的脊髓组织损伤附近可见粗厚与多支链的星形胶质细胞分布。 Western blot检测证实了免疫组化的发现,实验组的胶质纤维酸性蛋白表达水平明显低于对照组和干预组(P < 0.05,图3B)。与干预组相比,实验组胶质纤维酸性蛋白表达水平在术后时间段的增加趋势更为缓慢(图3A)。"

| [1] Young W. Spinal cord regeneration. Cell Transplant. 2014;23: 573-611. [2] Fitch MT, Silver J. CNS injury, glial scars, and inflammation: Inhibitory extracellular matrices and regeneration failure. Exp Neurol. 2008;209:294-301. [3] Neary JT. Protein kinase signaling cascades in CNS trauma. IUBMB Life. 2005;57: 711-718. [4] Swiech L, Perycz M, Malik A, et al. Role of mTOR in physiology and pathology of the nervous system. Biochim Biophys Acta. 2008; 1784:116-132. [5] Inoki K, Ouyang H, Li Y, et al. Signaling by target of rapamycin proteins in cell growth control. Microbiol Mol Biol Rev. 2005; 69:79-100. [6] Chung H, Li E, Kim Y, et al. Multiple signaling pathways mediate ghrelin-induced proliferation of hippocampal neural stem cells. J Endocrinol. 2013;218: 49-59. [7] Ryu JK, Choi HB, Hatori K, et al. Adenosine triphosphate induces proliferation of human neural stem cells: Role of calcium and p70 ribosomal protein S6 kinase, J Neurosci Res. 2003;72:352-362. [8] Denis JA, Gauthier M, Rachdi L, et al. mTOR-dependent proliferation defect in human ES-derived neural stem cells affected by myotonic dystrophy type 1. J Cell Sci. 2013; 126: 1763-1772. [9] Jiang LH, Yang NY, Yuan XL, et al. Daucosterol promotes the proliferation of neural stem cells, J Steroid Biochem Mol Biol. 2013;140 :90-99. [10] Wyatt LA, Filbin MT, Keirstead HS. PTEN inhibition enhances neurite outgrowth in human embryonic stem cell-derived neuronal progenitor cells. J Comp Neurol. 2014;522:2741-2755. [11] Jaworski J, Sheng M. The growing role of mTOR in neuronal development and plasticity. Mol Neurobiol. 2006;34:205-219. [12] Chen YN, Liu H, Zhao HB, et al. [Salidroside via ERK1/2 and PI3K/AKT/mTOR signal pathway induces mouse bone marrow mesenchymal stem cells differentiation into neural cells]. Yao Xue Xue Bao. 2013;48 :1247-1252. [13] Otaegi G, Yusta-Boyo MJ, Vergano-Vera E, et al. Modulation of the PI 3-kinase-Akt signalling pathway by IGF-I and PTEN regulates the differentiation of neural stem/precursor cells. J Cell Sci. 2006;119:2739-2748. [14] Han J, Wang B, Xiao Z, et al. Mammalian target of rapamycin (mTOR) is involved in the neuronal differentiation of neural progenitors induced by insulin. Mol Cell Neurosci. 2008;39: 118-124. [15] Magri L, Cambiaghi M, Cominelli M, et al. Sustained activation of mTOR pathway in embryonic neural stem cells leads to development of tuberous sclerosis complex- associated lesions. Cell Stem Cell. 2011;9: 447-462. [16] Lu Y, Belin S, He Z. Signaling regulations of neuronal regenerative ability. Curr Opin Neurobiol. 2014;27C :135-142. [17] Morgan-Warren PJ, Berry M, Ahmed Z, et al. Exploiting mTOR signaling: a novel translatable treatment strategy for traumatic optic neuropathy? Invest Ophthalmol Vis Sci. 2013; 54:6903-6916. [18] Wahl SE, McLane LE, Bercury KK, et al. Mammalian target of rapamycin promotes oligodendrocyte differentiation, initiation and extent of CNS myelination. J Neurosci. 2014;34: 4453-4465. [19] Narayanan SP, Flores AI, Wang F, et al. Akt signals through the mammalian target of rapamycin pathway to regulate CNS myelination. J Neurosci. 2009;29: 6860-6870. [20] Wang M, Li P, Liu M, et al. Potential protective effect of biphasic electrical stimulation against growth factor-deprived apoptosis on olfactory bulb neural progenitor cells through the brain-derived neurotrophic factor-phosphatidylinositol 3'-kinase/Akt pathway. Exp Biol Med (Maywood). 2013; 238: 951-959. [21] Horwood JM, Dufour F, Laroche S, et al. Signalling mechanisms mediated by the phosphoinositide 3-kinase/Akt cascade in synaptic plasticity and memory in the rat. Eur J Neurosci. 2006;23:3375-3384. [22] Sperlagh B, Csolle C, Ando RD, et al. The role of purinergic signaling in depressive disorders, Neuropsychopharmacol Hung. 2012;14: 231-238. [23] Tozaki-Saitoh H, Tsuda M, Inoue K. Role of purinergic receptors in CNS function and neuroprotection, Adv Pharmacol. 2011;61:495-528. [24] Franke H, Illes P. Involvement of P2 receptors in the growth and survival of neurons in the CNS. Pharmacol Ther. 2006; 109:297-324. [25] Ahmad S, Ahmad A, Ghosh M, et al. Extracellular ATP-mediated signaling for survival in hyperoxia-induced oxidative stress. J Biol Chem. 2004;279:16317-16325. [26] Mothe AJ, Tator CH. Proliferation, migration, and differentiation of endogenous ependymal region stem/progenitor cells following minimal spinal cord injury in the adult rat. Neuroscience. 2005;131:177-187. [27] Hentges KE, Sirry B, Gingeras AC, et al. FRAP/mTOR is required for proliferation and patterning during embryonic development in the mouse. Proc Natl Acad Sci U S A. 2001; 98:13796-13801. [28] Basso DM, Beattie MS, Bresnahan JC. A sensitive and reliable locomotor rating scale for open field testing in rats. J Neurotrauma. 1995;12: 1-21. [29] Burnett PE, Barrow RK, Cohen NA, et al. RAFT1 phosphorylation of the translational regulators p70 S6 kinase and 4E-BP1. Proc Natl Acad Sci U S A. 1998;95:1432-1437. [30] White RE, Jakeman LB. Don't fence me in: harnessing the beneficial roles of astrocytes for spinal cord repair. Restor Neurol Neurosci. 2008;26:197-214. [31] Pekny M, Wilhelmsson U, Pekna M. The dual role of astrocyte activation and reactive gliosis, Neurosci Lett. 2014;565:30-38. [32] Bramanti V, Tomassoni D, Avitabile M, et al. Biomarkers of glial cell proliferation and differentiation in culture, Front Biosci (Schol Ed). 2009;2: 558-570. [33] Gris P, Tighe A, Levin D, S, et al. Transcriptional regulation of scar gene expression in primary astrocytes. Glia. 2007;55: 1145-1155. [34] Drew KL, McGee RC, Wells MS, et al. Growth and differentiation of adult hippocampal arctic ground squirrel neural stem cells. J Vis Exp. 2011;(47). pii: 2199. [35] Bai Y, Cui M, Meng Z, et al. Ectopic expression of angiopoietin-1 promotes neuronal differentiation in neural progenitor cells through the Akt pathway, Biochem Biophys Res Commun. 2009:378:296-301. [36] Shioda N, Han F, Morioka M, et al. Bis(1-oxy-2- pyridinethiolato)oxovanadium(IV) enhances neurogenesis via phosphatidylinositol 3-kinase/Akt and extracellular signal regulated kinase activation in the hippocampal subgranular zone after mouse focal cerebral ischemia. Neuroscience. 2008;155:876-887. [37] Don AS, Tsang CK, Kazdoba TM, et al. Targeting mTOR as a novel therapeutic strategy for traumatic CNS injuries. Drug Discov Today. 2012;17: 861-868. [38] Liu G, Detloff MR, Miller KN, et al. Exercise modulates microRNAs that affect the PTEN/mTOR pathway in rats after spinal cord injury. Exp Neurol. 2011;233:447-456. [39] Schachtrup C, Ryu JK, Helmrick MJ, et al. Fibrinogen triggers astrocyte scar formation by promoting the availability of active TGF-beta after vascular damage. J Neurosci. 2010;30: 5843-5854. [40] O'Callaghan JP, Kelly KA, VanGilder RL, et al. Early Activation of STAT3 Regulates Reactive Astrogliosis Induced by Diverse Forms of Neurotoxicity. PLoS One. 2014;9:e102003. [41] Okada S, Nakamura M, Katoh H, et al. Conditional ablation of Stat3 or Socs3 discloses a dual role for reactive astrocytes after spinal cord injury. Nat Med. 2006;12:829-834. [42] Oki K, Kaneko N, Kanki H,et al. Musashi1 as a marker of reactive astrocytes after transient focal brain ischemia, Neurosci Res. 2009;66:390-395. [43] Wu L, Li JJ, Chen L, et al. Combined transplantation of GDAsBMP and hr-decorin in spinal cord contusion repair. Neural Regen Res. 2013;8(24): 2236-2248. [44] Okano H, Kaneko S, Okada S, et al. Regeneration-based therapies for spinal cord injuries. Neurochem Int. 2007;51: 68-73. [45] Busch SA, Horn KP, Cuascut FX, et al. Adult NG2+ cells are permissive to neurite outgrowth and stabilize sensory axons during macrophage-induced axonal dieback after spinal cord injury. J Neurosci. 2010;30: 255-265. [46] Pastor MD, Garcia-Yebenes I, Fradejas N, et al. mTOR/S6 Kinase Pathway Contributes to Astrocyte Survival during Ischemia. J Biol Chem. 2009;284:22067-22078. [47] Shih CH, Lacagnina M, Leuer-Bisciotti K, et al. Astroglial- derived periostin promotes axonal regeneration after spinal cord injury. J Neurosci. 2014;34:2438-2443. |
| [1] | You Zhengqiu, Zhang Zhongzu, Wang Qunbo. Early symptomatic intervertebral disc pseudocysts after discectomy detected on MRI [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1403-1409. |
| [2] | Liu Qinghua, Cai Yongqiang, Jin Feng, Yu Jinghong, Wang Haiyan, Zhang Yunfeng, Wang Lidong, Li Jiawei, Wang Xing, He Yujie, Dai Lina, Wang Jianzhong, Wu Chao, Tong Ling, Kang Zhijie, Li Zhijun, Li Xiaohe. Finite element model of the 12-year-old child whole cervical spine: establishment and validity verification based on CT data [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(4): 500-504. |
| [3] | Tao Xin, Xu Yi, Song Zhiwen, Liu Jinbo. Hippo signaling pathway in the regulation of spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(4): 619-625. |
| [4] | Zhan Yi, Wang Biao, Ma Yuli, He Simin, Sun Honghui, Hao Dingjun. Biomechanical comparison between a novel bone cement screw system and common surgical methods for the treatment of Kummell’s disease [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 385-390. |
| [5] | Hao Yunteng, Shi Jun, Liu Yuhang, Li Kun, Ma Yuan, Zhang Shaojie, Wang Chaoqun, Chen Jie, Zhang Zhifeng, Zheng Leigang, Wang Xing, Li Zhijun. Comparative finite element analysis of the cervical articular process after resection of different ranges of the uncinate processes [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(18): 2789-2796. |
| [6] | Gao Xu, Xing Wenhua. Application of finite element analysis in spine surgery [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(18): 2921-2927. |
| [7] | . Chinese expert consensus on neurorestorative therapy with cerebrospinal fluid administration (2022) [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(17): 2780-2788. |
| [8] | Yan Laijun, Ge Haiya, Zhou Bin, Mi Daguo, Wang Pengxiang, Li Nan. Finite element analysis of lumbar vertebra biomechanics after oblique lateral interbody fusion combined with bilateral cortical bone trajectory screw fixation [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(13): 1969-1974. |
| [9] | Yu Chengxiang, Liu Lehong, Li Wenbo, Chen Jinshi, Ran Chunlei, Wang Zhongping. Correlation between spine-pelvic sagittal parameters and prognosis of vertebroplasty in the treatment of thoracolumbar osteoporotic vertebral compression fractures [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1412-1417. |
| [10] | Liu Yuhang, Zhou Jianqiang, Xu Xuebin, Qu Xingyue, Li Ziyu, Li Kun, Wang Xing, Li Zhijun, Li Xiaohe, Zhang Shaojie. Establishment and validation of finite element model of lower cervical spine in 6-year-old children [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(6): 870-874. |
| [11] | Li Yuqiao, Sun Tianwei, Ma Bin, Zhou Zhaohong, Dong Runbei, Wu Haiyang. A comparative study of imaging parameters and quality of life scores between subtypes of lumbar spondylolisthesis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(6): 943-948. |
| [12] | Li Jian, Bao Zhengqi, Zhou Pinghui, Zhu Ruizhi, Li Zhixiang, Wang Jinzi. Effects of posterior single open-door laminoplasty and anterior cervical corpectomy fusion on cervical sagittal balance parameters in the treatment of multilevel cervical spondylotic myelopathy [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(6): 949-953. |
| [13] | Yi Xinrong, Jia Fuquan, He Xin, Zhang Shaojie, Ren Xiaoyan, Li Zhijun. Establishment of cervical bone age equation for male adolescents aged 8-16 years old in Hohhot based on thin-slice CT [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(6): 954-958. |
| [14] | Kou Bowen, Han Ye, Hao Yiguang, Xu Haoxiang, Wen Wangqiang, Zhang Zepei, Lan Jie, Miao Jun. Morphological changes and strain characteristics of lumbar intervertebral disc during sitting in forward flexion [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(36): 5792-5797. |
| [15] | Xiong Hengheng, Nie Weizhi. Accurate simulation of stress state in bone joint and related soft tissue injury by three-dimensional finite element analysis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(36): 5875-5880. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||