Chinese Journal of Tissue Engineering Research ›› 2014, Vol. 18 ›› Issue (30): 4878-4888.doi: 10.3969/j.issn.2095-4344.2014.30.020
Previous Articles Next Articles
Design and application of biodegradable coronary stents: what will be brought by further innovations in materials science?
Chen Jia-hui, Shen Li, Wang Qi-bing, Ge Jun-bo
- Department of Cardiology, Zhongshan Hospital, Shanghai Institute of Cardiovascular Diseases, Fudan University, Shanghai 200032, China
-
Revised:2014-05-12Online:2014-07-16Published:2014-08-08 -
Contact:Wang Qi-bing, Chief physician, Professor, Master’s supervisor, Department of Cardiology, Zhongshan Hospital, Shanghai Institute of Cardiovascular Diseases, Fudan University, Shanghai 200032, China -
About author:Chen Jia-hui, Studying for master’s degree, Department of Cardiology, Zhongshan Hospital, Shanghai Institute of Cardiovascular Diseases, Fudan University, Shanghai 200032, China -
Supported by:the National Natural Science Foundation of China, No. 81370323; the Research Project of the Science and Technology Commission of Shanghai Municipality, No. 12DZ1940604
CLC Number:
Cite this article
Chen Jia-hui, Shen Li, Wang Qi-bing, Ge Jun-bo. Design and application of biodegradable coronary stents: what will be brought by further innovations in materials science?[J]. Chinese Journal of Tissue Engineering Research, 2014, 18(30): 4878-4888.
share this article
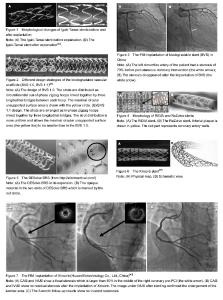
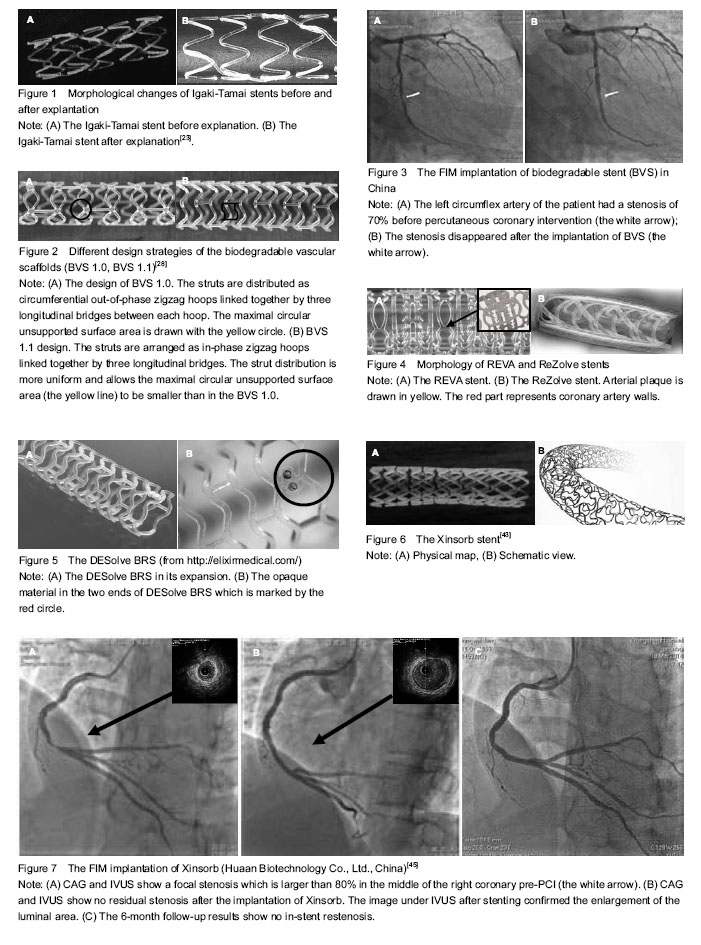
The history of percutaneous coronary intervention (PCI) Percutaneous transluminal coronary angioplasty (PTCA), bare metal stents and drug-eluting stents are three revolutions in the PCI therapy. PTCA Repeat bypass surgery is typically associated with a high rate of mortality[1]. In 1977, Andreas Gruentzig introduced a new concept, which was a huge leap forward in cardiovascular intervention therapy[2]. PTCA has emerged as the preferred treatment which IS thought to be effective in treating angina pectoris. However, acute occlusion and chronic restenosis constitute the most common reasons that PTCA may fail to provide long-term benefits and underlies the majority of ischemic complications[3], which sometimes could be aggravated by aortic dissection and sometimes create a need for potential emergency coronary artery bypass surgery. Bare metal stents Bare metal stents are conceived as the second revolution in the practice of interventional cardiology[4], which provide an attractive solution to abrupt vessel occlusion by sealing the dissection flaps and preventing acute recoil. Reducing the rate of late-stage restenosis when compared with balloon angioplasty, increasing the rate of procedural success as well, bare metal stent has a similar rate of clinical events after 6 months, but a less frequent need for revascularization of the original coronary lesion[5]. Nevertheless, metal stents have a thrombogenic milieu, the implantation of which is considered as a double-edged sword[6]. The two main concerns, consisting of the emergency of stent thrombosis and late restenosis, hamper the widely clinical application of bare metal stents. Drug-eluting stents Even though the early effects are thought to be beneficial, acute and sub-acute occlusion, late restenosis remains to be a limitation in broadening the indications of bare metal stents[7]. However, large meta-analysis has suggested that these concerns are no longer an issue with the new generation of drug-eluting stents. In 1999, drug-eluting stents coating and eluting cytostatic and cytotoxic drugs from the stent surface set a new and beneficial revolution in cardiac intervention therapy by permitting the percutaneous treatment used in increasingly complex artery lesions[8]. Cypher and Taxus, as representatives of the first-generation drug-eluting stents, are composed of a metallic platform, anti-proliferation drug coated with permanent polymers. Consequently, drug-eluting stents are being used in complex patients and lesion types. Clinical data and guideline recommendations support these results. However, a higher risk of late and very late stent thrombosis appears which were thought to be a nuisance in the application of drug-eluting stents[9]. Mechanisms for this include high chronic inflammations reaction of vascular walls to the stimulation of permanent stent coatings, incomplete endothelialization or delayed arterial healing and stent malapposition. Furthermore, extended dual-antiplatelet treatment is expensive and may cause bleeding risk. Bioresorbable stents (BRS) Current developments of biodegradable stents BRS are supposed to be the fourth revolution, as they are “fulfilling the mission and stepping away”[10]. It represents a novel stent concept which can prevent vessel recoil in the early stage and then fully disappear in certain future. Since the first bioresorbable stent which used a polymer of poly-L-lactide (PLA) invented by Stack et al [11] at Duke University in 1988, biodegradable stents have achieved a great improvement. Intra-coronary stent implantation obviously reduced the frequency of major adverse cardiac events such as restenosis and acute complications of coronary angioplasty. The main reason is potentially not to trigger thrombosis after full biodegradation, such as from uncovered stent struts, and durable polymers, or remnant drugs[12]. However, bioresorbable stent also provides a considerable advantage for patients who have to receive further operations such as PCI, coronary artery bypass surgery[13]. Besides, this unique property of degradable vascular implants can reduce the risk of late complications by adapting to vessel growth and late positive remodeling[14], allowing the vessel to maintain its integrity and vasomotor function[15]. In addition, there is no requirement for long-term dual anti-platelet therapy (clopidogrel/aspirin), and is associated with a lower risk of bleeding. Furthermore, BRS can also be used in child vessel intervention. Therefore, the indications of stents implantation are extended. BRS assessed in both preclinical and clinical setting Current evidence of significant developments has been achieved in the field of bioresorbable stents. Recently, BRSs are considered as the next frontier of stents for coronary artery disease[16]. Biodegradable polymeric intravascular stents: Increasing attention is being paid to polymeric compounds that can be bioresorbable. The potential of these degradable polymers is discussed with respect to a special attention to polymers of the poly (alpha-hydroxy acid) type[17]. Polymers highly recommended include polyglycolic acid (PGA), poly (D,L-lactide/glycolide) copolymer (PDLA), and polycaprolactone[18]. In 1990, Zidar et al [19] implanted PLLA BRS into the canine femoral arteries and published the promising outcomes of the preclinical experiments. The study claimed that the stents degraded significantly, possessed low-grade inflammation reactions which contributed to lower stent thrombosis and neointimal hyperplasia when compared with bare metal stents. In 1992, the experimental studies which tested the feasibility, safety, and effectiveness of fully biodegradable stents in vitro and in the porcine model were published[20]. This opened up a prosperous new period of research on bioresorbable stent. However, in 1996, Van der Giessen et al [21] reported marked inflammatory sequelae, giving rise to neointimal hyperplasia and/or thrombus formation, after the implantation of the Wiktor stent coated with five different types of biodegradable polymers (PGA/PLA copolymer, polycaprolactone, polyhydroxy-butyrate/-valerate copolymer, polyorthoester, and polyethyleneoxide/ polybutylene terephthalate) in the porcine coronary artery. With the deficiency of the former stents shown above, innovations of new scaffolds emerge as the times require. Currently, many types of BRSs are under preclinical and clinical evaluation. Of note, three kinds of BRSs have achieved CONFORMITE EUROPENDE (CE) mark approval and are used in clinical practice: the Igaki-Tamai stent for the treatment of peripheral vascular disease, the Absorb BVS and DESolve stent for coronary artery disease. Igaki-Tamai stents (Igaki Medical Planning Co., Ltd.): In 1999, Igaki-Tamai stent, made of a PLLA mono-polymer, was the first fully bioresorbable but non-drug-eluting polymeric stent examined in diseased human coronary arteries. The attractive innovations of the first revision are the zigzag helical coil pattern, the self-expanding radius stent system and the using of a balloon expandable covered sheath system with contrast heated (up to 70 °C) through an 8 French guiding catheter. The stent has two radiopaque gold markers to facilitate the identification of both ends of the prosthesis[22]. Igaki-Tamai scaffold completely disappeared at an average of 3 years[23]. The clinical trials showed less to no inflammations and no unexpected, adverse angiographic, intravascular, and clinical findings, which reassured the biocompatibility of the PLLA. In 2000, Tamai et al [22] reported the initial and 6 months follow-up results. In 2009, Onuma et al [24] published the 10-year follow-up in human. Nishio et al [23] continued the study and published the long-term (> 10 years) clinical outcomes in 2012. The attractive finding of this research was able to restore the artery capability responding to positive remodeling once the stents degraded, which is unique to PLLA. Although current preliminary experimental and clinical studies have shown the longevity of Igaki-Tamai stents, while demonstrating the limitations as well, the major concern was the relatively high rate of late restenosis which is mainly related to the late-stage in-stent thrombosis. Therefore, stents carrying anti-proliferation drugs were required as the time increased. Meanwhile, new design of Igaki-Tamai stents is undergoing preclinical testing in Germany[25] (Figure 1). Bioresorbable vascular scaffold system (Abbott Vascular, Santa Clara, CA, USA): The first revision of bioresorbable vascular scaffold (BioresorbableVascular Scaffold) 1.0 has a bioresorbable PLA backbone with a bioresorbable PDLA coating that contains and controls the release of the anti-proliferative drug, everolimus (Novartis, Basel, Switzerland). The design of the bioresorbable vascular scaffold with a crossing profile of 1.4 mm is circumferential hoops of PLLA with struts 150 μm thick either joined directly or linked by straight bridges. Both ends of the scaffold have two adjacent radio-opaque metal markers[26]. In 2008, the first generation of bioresorbable everolimus-eluting coronary stent (BVS 1.0) was implanted in the patients with single de-novo native coronary artery lesions. In 2009, researchers revealed 2-year outcomes and results from a clinical trial (ABSORB Cohort A)[27]. The outcomes of this research demonstrated somewhat higher acute and late scaffold shrinkage compared to metallic drug-eluting stents, which was the principal concern of ABSORB BVS 1.0. The design of the second generation ABSORB BVS was changed in modified manufacturing process of the polymer and geometric changes in the polymeric platform. Consequently, the new revision possessed a more uniform strut distribution and provided increased radial strength[28-31]. In 2010 and 2011, the term published the clinical and imaging outcomes of the 6- and 12-month follow-up of the second generation of bioresorbable vascular scaffold (ABSORB BVS 1.1) for the treatment of de novo coronary artery stenosis[32-33]. Of note, the scaffold area of the second revision was unchanged at 6 months estimated by OCT in both preclinical and clinical trials[29, 32, 34]. In addition, other three clinical trials are underway: the ABSORB Physiology, the ABSORB II, and the ABSORB EXTEND. The ABSORB Physiology setting focuses on estimating the short- and long-term effects of an Absorb BVS and a Xience V® (Abbott Vascular, Santa Clara, USA) stent. The ABSORB II study is the first randomized trial designed to compare the safety and efficacy of the Absorb BVS and Xience prime (Abbott Laboratories) in 500 patients with de novo coronary artery disease. The ABSORB EXTEND trial is a large-scale, single-arm trial that enrolls 1 000 patients with complex coronary artery disease which aims to evaluate the occurrence of side branch blockage after the implantation of bioresorbable vascular scaffold. The results show that bioresorbable vascular scaffolds can provide durable support force, allow persistent drug release and have no late in-stent restenosis. However, the research results also show that the bioresorbable vascular scaffold posses a higher rate of small side branch blockage compared with XIENCE V® which calls for further improvement of stent design strategy[35]. Data from this trial may be used to support approval in various markets around the world[36] (Figures 2, 3). REVA scaffolds (Reva Medical INC, San Diego, CA, USA): The first generation of REVA scaffold has a unique “slide and lock” design, which means its expansion is based on sliding, locking parts (Figure 4A). REVA’s bioresorbable polymer delivers exceptional stent performance, and also doubles as a drug-delivery matrix with complete drug (sirolimus) release from the polymer. Moreover, the REVA polymer is also visible under x-ray because it is impregnated with iodine. The stent possesses high radial strength and negligible recoil with standard balloon deployment. Other features include its non-toxin breakdown products and its ability to vary resorption rate. Complete degradation time of REVA stent is about 18 to 24 months after implantation. However, the rate can be modified for meeting different needs, such as diseased coronaries, vulnerable plaque, diabetic lesions, and drug eluting mechanic[37]. Yet, the 4- and 6-month follow-up of the first revision REVA also show high target lesion revascularization in clinical trial RESORB (the REVA endovascular study of a bioresorbable coronary stent), which was contribute to the invention of the second revision REVA scaffold, ReZolve. The ReZolve stent has a spiral slide-and-lock mechanism and carries the anti-proliferative drug sirolimus. Currently, this new scaffold is undergoing evaluation in the RESTORE (ReZolve sirolimus-eluting bioresorbable coronary scaffold) clinical trial, which aims to pilot investigate its safety and efficacy in 50 patients. Clinical data from the RESTORE trial will become available throughout 2012 as patients pass the 1- and 6-month clinical evaluation stages. Furthermore, the RESTORE II clinical trial which aims to assess the safety and performance of the ReZolve2 Bioresorbable Coronary Scaffold in native coronary arteries started in April 2013. Totally 125 patients are estimated to enrolled in this trial. The preliminary outcomes are supposed to publish in 2014. REVA Medical INC tries to assuming acceptable results of these studies to provide sufficient evidence for CE mark approval (Figure 4B). DESolve™ BRS (Elixir Medical Corporation, Sunnyvale, CA, USA): The DESolve BRS has a backbone of PLLA polymer coated with a polylactide-based resorbable polymer-drug matrix which contains two anti-proliferative drugs (Novolimus and Myolimus). Novolimus, a metabolite of sirolimus, was developed internally by Elixir, while myolimus is a Novartis Pharma AG compound. Both these two drugs belong to the powerful macrocyclic lactone class of drugs, the family of which is widely used for DES applications[38-39]. The initial researches had confirmed the safety and efficacy of novolimus and myolimus[40-41], as well as PLLA[22]. The degradation of DESolve scaffold occurs in about 1 year. Its fully resorption process takes approximately 2-3 years, leaving behind a thin neointimal lining and well-maintained lumen. Additionally, potential benefits include a functional endothelium enabling vasomotion and uniform flow dynamics in the vessel. The FIM research of DESolve which enrolled 16 patients with a single de novo coronary artery lesion also demonstrates the radial strength of the device comparable with the Elixir’s BMS. The encouraging results from the previous researches motivate a multi-center, prospective study enrolling 120 patients at up to 15 centers in Germany, Belgium, Poland, Brazil and New Zealand designed to evaluate the safety and efficacy of the DESolve scaffold. A 6-month follow-up is the principal endpoint. This study focuses on the long-term assessment of the scaffold and surrounding vessel assessed by QCA, intravascular ultrasound (IVUS), optical coherence tomography (OCT) and MSCT[42]. Meanwhile, the DeSolve NX II trial has been designed to evaluate the effectiveness of the device in a larger number of patients in order to apply for CE mark. Recently, the Elixir Medical INC announced that the DESolve is CE mark approved (Figure 5). XINSORB (Weite Biotechnology Co., Ltd., China): XINSORB has a bioresorbable PLLA backbone coated with the mixture of poly-D-Llactic acid (PDLLA) and PLLA. The coating carried and controlled the release of anti-proliferative drug, sirolimus. The molecular weight of PLLA used for XINSORB stent was 300 kDa. Recently, the acute recoil between XINSORB stent and metallic stent was compared in a porcine model. Efficacy of XINSORB is confirmed while raising questions with regard to the durability of this novel medical device[43]. Moreover, to investigate acute recoil of bioabsorbable PLLA stent, PLLA XINSORB stents (Huaan Biotechnology Co., Ltd., China) and metallic stents EXCEL (Jiwei Medical Co, Shandong, China) were implanted into the coronary arteries of 16 mini porcines. The results showed that the acute stent recoil of XINSORB scaffold is similar to that of stainless-steel- based EXCEL stent. No acute stent mal-apposition or collapse appeared in both kinds of stent[44]. The 2-years follow-up results of the animal trial are supposed to be public this year. The FIM trial (with no compare group) has been started on September 5, 2013, which was the first case of independent bioresorbable stent (Xinsorb) implanted in China (Figure 6). The 6 month follow-up results conclude from the clinical trial which includes 30 patients with single de novo lesion using coronary angiograpgy, IVUS and OCT show the effectiveness and safety of the Xinsorb stent (Figure 7). Although the current findings are encouraging, long-term follow-up researches in larger studies are extremely necessary before widely applied in the clinic. Other Bioresorbable Polymer Scaffolds: IDEAL (Xenogenics, Canton, MA, USA)[46-47] is also made up of polymer materials which are undergoing clinical research. Besides these, there are other biodegradable polymer stents that are undergoing pre-clinical studies, such as ART BRS[48],Amaranth[49], Acute BRS[50]. The metallic alloys mainly utilized for BRSs are iron and magnesium alloyed with some rare metals[51]. Metal bioabsorbable stents are intuitively attractive since they have the potential to perform similarly to stainless steel metal stents[52]."
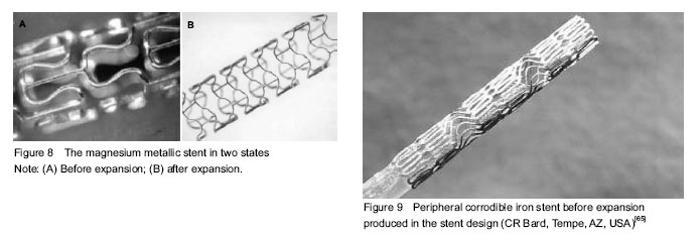
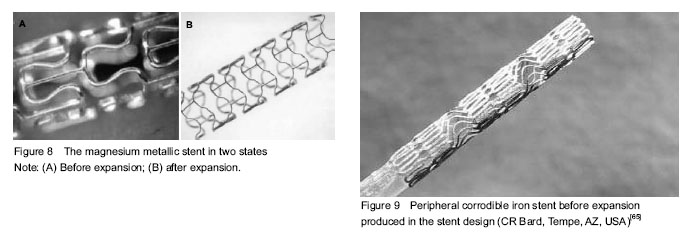
Magnesium metallic stents (AMS): Mg based alloy has been used for more than 14 years since the first implantation in humans. Magnesium metallic stents are the dominating bioresorbable metallic stents at present. As an essential element in the biological system, magnesium is a splendid candidate for the bioresorbable metallic stents. Biocorrosion of magnesium based alloys is a prospect in new technology for improving cardiovascular implants as an effective temporary system with inherent or hybrid local drug delivery functions[53-56]. The first animal trial utilized a magnesium alloy which contained 2% aluminum and 1% rare earths (Ce, Pr, Nd). Nevertheless, the concept yielded some of the expected results involving thrombogenicity, biocompatibility, and local tissue response during different periods of time[57]. In 2005, the first successful implantation of a biodegradable metal stent, rescued a child, who received the implantation of a biodegradable magnesium stent, from an extremely severe clinical problem who received the implantation of a magnesium stent[58]. In 2006, the first biodegradable stent based on a magnesium alloy that allows controlled corrosion with release to the vessel wall and the blood stream of a natural body component (such as magnesium with beneficial antithrombotic, anti-arrhythmic, and anti-proliferative properties) was implanted in 20 patients below the knee[59]. In 2007, prospective multicentre clinical trials of coronary implantations of absorbable magnesium stents were reported by Erbel et al [57, 60]. The efficacy of the first revision AMS (AMS 1.0) was evaluated in the PROGRESS-AMS (Clinical Performance and Angiographic Results of Coronary Stenting) study, which declared that complete biocorrosion occurred within 2 months after the implantation in humans and contributed to the early reduction in the radial strength of the devices, following with high late lumen loss and target lesion reconstruction[61]. To overcome previous limitations, a new generation AMS, DREAMS (drug-eluting absorbable metallic stents) 1.0 was innovated. Anti-proliferative drug paclitaxel was added to the device. The design of the scaffold and the composition of the magnesium alloy also changed, thus providing the device with increased radial strength, and a prolongation resorption process[61]. However, late lumen loss, although improved, remained high. DREAMS 2.0 incorporates sirolimus elution instead of paclitaxel. Preclinical evaluation of the device revealed reduced inflammation, and a higher endothelization rate compared with the DREAMS 1.0. In the latest revision, the degradation process demonstrated a prolonging to 6 months[62] (Figure 8). Iron alloy stents: Although having not progressed as far as magnesium stents, the first animal study of iron stents was completed earlier than AMS[63]. The preclinical study of iron stents implanted in porcine coronary arteries was limited to a 28-day follow-up, but claimed to have less neointima formation at this time point[64] (Figure 9). The potential advantages of bioabsorbable iron alloy[66]: (1) Iron is a essential element in human body, (2) biocorrodible metal, (3) possess appropriate degradation rate, (4) excellent comprehensive mechanical properties, (5) not radiolucent for the high density of iron base alloy compared with other metal, (6) good nuclear magnetic resonance compatibility, (7) associated with lower late stent thrombosis. Other BRSs: Other technologies using polymer other than poly-lactide, as well as using other resorbable metal alloys, are also being investigated. These BRSs are currently undergoing testing, such as Avatar BRS, Sahajanand BRS, MeRes BRS, Zorion BRS."

The main problems of current BRS Intra-coronary stent implantation reduced both restenosis rates and acute complications of coronary angioplasty but increased sub-acute thrombosis rates and hemorrhagic complications when used with antithrombotic drugs. Biocompatibility Acquiring biocompatibility is a complex chain process. The interaction among polymer composition, degradation products and implanted stents is the principal reason for inflammation. In addition, reactions to the biological material implantation, which are considered to be inherent defense mechanisms, may be produced by the body’s neuro-endocrine system. Biodegradable materials possess excellent blood compatibility. However, its histocompatibility is still controversial. Histocompatibility represents the ability of allowing adhesion and proliferation of the endothelial cells pseudo intima formation. Today, the concept of biocompatibility is moving from a “do no harm” mission (i.e., non-toxic, non-antigenic, non-mutagenic, etc.) to one of doing “good”, which means encouraging positive healing responses instead of merely delayed the healing[67]. The local toxicity is related to the local concentration of the elements over time. The tissue tolerance for physiologically occurring metals depends on the change of their tissue concentrations induced by corrosion. The reasons for using magnesium as the main alloy component are its surface characteristics and expected local tissue tolerance. Thus, elements with high tissue concentrations such as magnesium supposedly have a better biocompatibility during degradation[68]. Changes in local oxygen and pH can be neglected if the degradation is slow. However, formation of reactive oxygen species through Fenton-type chemical reactions merit requires more attention, as in the case of non-degradable metals. Degradation rate An appropriate degradation rate of scaffold backbone is essential for BRS. The BRSs are supposed to exist for a certain period of time and then completely disappear. Two dominant factors influencing the degradation rate are inherent properties of materials and implanted environment (Table 1)."
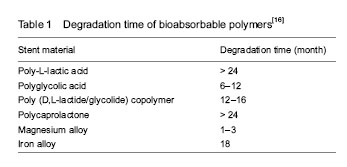

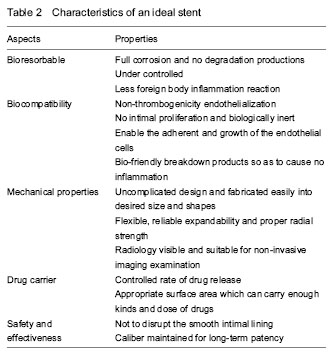
It was speculated that coronary stents have no clear function six months after implantation. As a result, the appropriate complete degradation time which is supposed to meet the requirements of clinical practice is about 3-6 months after implantation[10]. Previous studies confirmed that PLA is hard and brittle with slow degradation, while PGA is soft with fast degradation rate and insufficient support force. A current study implanted in porcine coronary arteries claimed that stents made of bio-corrodible iron are associated with less neo-intimal formation than cobalt-chromium stents[64]. The corrosion of iron appears to be too slow. The stents are still present at 18 months[69]. The research showed that the mechanical properties and degradation behavior of Fe-35%Mn alloy closely approach the clinical requirements[70]. In 2010, Schinhammer et al [71] put forward two design strategies to make iron based alloy more susceptible to corrosion: (1) the formation of a solid solution such that the Fe matrix, and (2) the formation of noble IMP particles that generate micro-galvanic corrosion and promote active dissolution of the matrix. Mn and Pd demonstrated to be suitable candidate for alloying additions for this approach. In 2011, Liu et al [72] found that the elements Co, W, C, and S are suitable on a comprehensive consideration of the improved mechanical properties, appropriate corrosion rates and good biocompatibility, however, the degradation rate of Fe-alloy show no significant clinical difference when it was compared with pure iron. Despite of the fast degradation rate of the early AMS, the degradation times of current magnesium systems proved to be closer to the requirements. Drug-eluting absorbable metallic stents (DREAMS) 1.0 and 2.0, which have changed the alloy of magnesium, were innovated and are undergoing preclinical researches. They are supposed to have an increasing radial strength, and prolongation resorption processes. As mentioned above, the fully degradation time of the latest magnesium alloy has prolonged to over 6 months with the evolution in the stent design[62]. Bio-corrosion of magnesium based alloys may be a promising new technology for improving cardiovascular implants as effective temporary systems with inherent or hybrid local drug delivery functions. In conclusion, the methods to control the degradation rate include changing the mixture proportion of alloy, as well as the design strategies of polymers or metal stents. Mechanical properties At present, scholars tend to use the finite element analysis to investigate and analyze the parameter of the bioresorbable materials, such as the elastic modulus, the yield strength, and the influence of the collapse behavior. Biodegradable polymers were widely used as delivery vehicles for drug coating and utilized to control the release of drugs[16, 51-52]. The superiority of providing a possibility for combining physical treatment with pharmaceutical therapy makes polymeric stent more attractive than metallic BRS in drug coating[73]. However, none of the degradable polymer materials currently used is beyond reproach. Common to all is the lack of long-term clinical evidence demonstrating a clear advantage of providing sufficient radial strength over other stents[64]. Compared with metallic scaffold, polymer BRS proved to be more vulnerable to early recoil after implantation, which was potentially due to the differences in material properties: (1) polymers are more flexible and potential to be more directly affected by the elastic properties of the arterial wall, (2) in addition, as the polymer BRS is designed to be gradually metabolized, the polymer backbone will lose its structural integrity during degradation, which contributes to the reduction of its radial strength[74]. Furthermore, as the PLLA used in both Igaki-Tamai and BVS 1.0, the main reason of stent recoil might be associated with differences in stent design. Because the Igaki-Tamai stent was self-expandable, whereas the BVS was balloon-expandable, the Igaki-Tamai stent tends to expand gradually in time until it reaches full unconstrained dimensions. In short, polymer bioresorbable scaffolds are confronted with challenges in achieving appropriate strength and resistance to recoil. In the previous preclinical and clinical studies, metallic stents have manifested their excellent radial strength. Both magnesium and iron frequently applied in the researches of metallic BRS proved inappropriate bioresorbable rate resulting in many complications. Because of the high degradation rate, the former magnesium alloy stents could not provide sufficient support before they were completely corroded. However, the latest AMS overcome this drawback and take possession of a prolonged bioresorbable time which is supposed to satisfy clinical practices. Modifications of stent mechanical characteristics are currently in development[75]. Imaging properties Biodegradable metal stents hamper non-invasive imaging of coronary arteries with multislice computed tomography and MRI before complete corrosion. IVUS and OCT appear to be much more suitable. Compared with metal stents, the density of biodegradable polymer material is extremely low. All of the biodegradable polymers mentioned above are radiolucent, which may impair accurate positioning. It is difficult to deploy the stent precisely without fluoroscopic visualization[16]. Currently, most investigators prefer to use angiography, IVUS or OCT after the implant of the polymer stents. Besides, some scholars add radio-paque metal markers at the two tips of the polymer stents in order to be visible under the X-rays. As a result, the invention of a new kind of polymer material which is radiology visible is badly needed. Drug carrier system The mechanisms of controlled drug release are varied, such as diffusion, dissolution or degradation, ion exchange, osmosis and prodrug[76]. A precise control of the drug-release kinetics is extremely important, since the release kinetics should be tailored to the pathophysiologic phases of restenosis depending on the specific mechanism of drug action. In addition, appropriate surface areas which can carry various kinds and doses of drugs are also of great importance. Polymer BRSs might be superior to polymer-coated metallic stents as local drug delivery stents in terms of biodegradation and the amount of loaded drug. Drug-mixed polymer stents can be loaded with a larger amount of drugs than drug-coated metallic stents because the polymer stent struts can contain the drug[52]. Compared with drug-eluting stents, biodegradable stents can carry various kinds of drugs, achieve a longer release time, which is one of the trends in the development of the endovascular stents. The drug of the BRS may not only inhibit the proliferation of smooth muscle cells, but also hamper the healing of the lesion vessels and the endothelialization of the vessel wall which results in the recovery delay. Some researchers considered that drug-coated stents only delay rather than completely eliminate the appearance of restenosis. In brief, the ideal biomaterial is expected to overcome all the drawbacks shown as follow (Table 2)."
| [1] Yazdani SK, Farb A, Nakano M, et al. Pathology of drug-eluting versus bare-metal stents in saphenous vein bypass graft lesions. JACC Cardiovasc Interv. 2012; 5(6):666-674. [2] Gruntzig A. Transluminal dilatation of coronary-artery stenosis. Lancet. 1978;1(8058):263. [3] Roubin GS, Cannon AD, Agrawal SK, et al. Intracoronary stenting for acute and threatened closure complicating percutaneous transluminal coronary angioplasty. Circulation. 1992;85(3):916-927. [4] Serruys PW, de Jaegere P, Kiemeneij F, et al. A comparison of balloon-expandable-stent implantation with balloon angioplasty in patients with coronary artery disease. Benestent Study Group. N Engl J Med. 1994;331(8): 489-495. [5] Fischman DL, Leon MB, Baim DS, et al. A randomized comparison of coronary-stent placement and balloon angioplasty in the treatment of coronary artery disease. Stent Restenosis Study Investigators. N Engl J Med. 1994; 331(8):496-501. [6] Serruys PW, Keane D. The bailout stent. Is a friend in need always a friend indeed? Circulation. 1993;88(5 Pt 1): 2455-2457. [7] Serruys PW, Strauss BH, Beatt KJ, et al. Angiographic follow-up after placement of a self-expanding coronary-artery stent. N Engl J Med. 1991;324(1):13-17. [8] Rensing BJ, Vos J, Smits PC, et al. Coronary restenosis elimination with a sirolimus eluting stent: first European human experience with 6-month angiographic and intravascular ultrasonic follow-up. Eur Heart J. 2001;22(22):2125-2130. [9] Serruys PW, Daemen J. Are drug-eluting stents associated with a higher rate of late thrombosis than bare metal stents? Late stent thrombosis: a nuisance in both bare metal and drug-eluting stents. Circulation. 2007;115(11):1433-1439. [10] Colombo A, Karvouni E. Biodegradable stents: "fulfilling the mission and stepping away". Circulation. 2000;102(4): 371-373. [11] Stack RS, Califf RM, Phillips HR, et al. Interventional cardiac catheterization at Duke Medical Center. Am J Cardiol. 1988;62(10 Pt 2):3F-24F. [12] Karrillon GJ, Morice MC, Benveniste E, et al. Intracoronary stent implantation without ultrasound guidance and with replacement of conventional anticoagulation by antiplatelet therapy. 30-day clinical outcome of the French Multicenter Registry. Circulation. 1996;94(7):1519-1527. [13] Hassell ME, van de Hoef TP, Damman P, et al. The bioresorbable coronary scaffold. Ned Tijdschr Geneeskd. 2012;156(36):A4994. [14] Heublein B, Rohde R, Kaese V, et al. Biocorrosion of magnesium alloys: a new principle in cardiovascular implant technology? Heart. 2003;89(6):651-656. [15] Bourantas CV, Zhang Y, Farooq V, et al. Bioresorbable scaffolds: current evidence and ongoing clinical trials. Curr Cardiol Rep. 2012;14(5):626-634. [16] Waksman R. Update on bioabsorbable stents: from bench to clinical. J Interv Cardiol. 2006;19(5):414-421. [17] Vert M. Bioabsorbable polymers in medicine: an overview. EuroIntervention. 2009;5 Suppl F:F9-F14. [18] Onuma Y, Serruys PW. Bioresorbable scaffold: the advent of a new era in percutaneous coronary and peripheral revascularization? Circulation. 2011;123(7):779-797. [19] Zidar JP, Lincoff A, Stack R. Biodegradable stents. In: Topol EJ, ed. Textbook of Interventional Cardiology. 2nd ed. Philadelphia: Saundars,1994. [20] van der Giessen WJ, Slager CJ, van Beusekom HM, et al. Development of a polymer endovascular prosthesis and its implantation in porcine arteries. J Interv Cardiol. 1992;5(3): 175-185. [21] van der Giessen WJ, Lincoff AM, Schwartz RS, et al. Marked inflammatory sequelae to implantation of biodegradable and nonbiodegradable polymers in porcine coronary arteries. Circulation. 1996;94(7):1690-1697. [22] Tamai H, Igaki K, Kyo E, et al. Initial and 6-month results of biodegradable poly-l-lactic acid coronary stents in humans. Circulation. 2000;102(4):399-404. [23] Nishio S, Kosuga K, Igaki K, et al. Long-Term (>10 Years) clinical outcomes of first-in-human biodegradable poly-l-lactic acid coronary stents: Igaki-Tamai stents. Circulation. 2012;125(19):234323-53. [24] Onuma Y, Garg S, Okamura T, et al. Ten-year follow-up of the IGAKI-TAMAI stent. A posthumous tribute to the scientific work of Dr. Hideo Tamai. EuroIntervention. 2009;5 Suppl F:F109-111. [25] Zhang Y, Bourantas CV, Farooq V, et al. Bioresorbable scaffolds in the treatment of coronary artery disease. Med Devices (Auckl). 2013;6:37-48. [26] Ormiston JA, Serruys PW, Regar E, et al. A bioabsorbable everolimus-eluting coronary stent system for patients with single de-novo coronary artery lesions (ABSORB): a prospective open-label trial. Lancet. 2008;371(9616): 899-907. [27] Ormiston JA, Serruys PW, Regar E, et al. A bioabsorbable everolimus-eluting coronary stent system for patients with single de-novo coronary artery lesions (ABSORB): a prospective open-label trial. Lancet. 2008;371(9616): 899-907. [28] Serruys PW, Ormiston JA, Onuma Y, et al. A bioabsorbable everolimus-eluting coronary stent system (ABSORB): 2-year outcomes and results from multiple imaging methods. Lancet. 2009;373(9667):897-910. [29] Onuma Y, Serruys PW, Gomez J, et al. Comparison of in vivo acute stent recoil between the bioresorbable everolimus-eluting coronary scaffolds (revision 1.0 and 1.1) and the metallic everolimus-eluting stent. Catheter Cardiovasc Interv. 2011;78(1):3-12. [30] Gomez-Lara J, Brugaletta S, Diletti R, et al. A comparative assessment by optical coherence tomography of the performance of the first and second generation of the everolimus-eluting bioresorbable vascular scaffolds. Eur Heart J. 2011;32(3):294-304. [31] Brugaletta S, Gomez-Lara J, Serruys PW, et al. Serial in vivo intravascular ultrasound-based echogenicity changes of everolimus-eluting bioresorbable vascular scaffold during the first 12 months after implantation insights from the ABSORB B trial. JACC Cardiovasc Interv. 2011;4(12):1281-1289. [32] Ormiston JA, Serruys PW, Onuma Y, et al. First serial assessment at 6 months and 2 years of the second generation of absorb everolimus-eluting bioresorbable vascular scaffold: a multi-imaging modality study. Circ Cardiovasc Interv. 2012;5(5):620-632. [33] Serruys PW, Onuma Y, Ormiston JA, et al. Evaluation of the second generation of a bioresorbable everolimus drug-eluting vascular scaffold for treatment of de novo coronary artery stenosis: six-month clinical and imaging outcomes. Circulation. 2010;122(22):2301-2312. [34] Serruys PW, Onuma Y, Dudek D, et al. Evaluation of the second generation of a bioresorbable everolimus-eluting vascular scaffold for the treatment of de novo coronary artery stenosis: 12-month clinical and imaging outcomes. J Am Coll Cardiol. 2011;58(15):1578-1588. [35] Tearney GJ, Bouma BE. Shedding light on bioabsorbable stent struts seen by optical coherence tomography in the ABSORB trial. Circulation. 2010;122(22):2234-2235. [36] Muramatsu T, Onuma Y, van Geuns RJ, et al. One-Year Clinical Outcomes of Diabetic Patients Treated With Everolimus-Eluting Bioresorbable Vascular Scaffolds: A Pooled Analysis of the ABSORB and the SPIRIT Trials. JACC Cardiovasc Interv. 2014. in press. [37] Farooq V, Gomez-Lara J, Brugaletta S, et al. Proximal and distal maximal luminal diameters as a guide to appropriate deployment of the ABSORB everolimus-eluting bioresorbable vascular scaffold: a sub-study of the ABSORB Cohort B and the on-going ABSORB EXTEND Single Arm Study. Catheter Cardiovasc Interv. 2012;79(6): 880-888. [38] Pollman MJ. Engineering a bioresorbable stent: REVA programme update. EuroIntervention. 2009;5 Suppl F: F54-57. [39] Stefan V. Desolve First in Man Study–Preliminary Results. Rotterdam: EuroPCR, 2012. [40] Yan J, Bhat VD. Elixir Medical's bioresorbable drug eluting stent (BDES) programme: an overview. EuroIntervention. 2009;5 Suppl F:F80-82. [41] Serruys PW, Garg S, Abizaid A, et al. A randomised comparison of novolimus-eluting and zotarolimus-eluting coronary stents: 9-month follow-up results of the EXCELLA II study. EuroIntervention. 2010;6(2):195-205. [42] Costa JR Jr, Abizaid A, Feres F, et al. EXCELLA First-in-Man (FIM) study: safety and efficacy of novolimus-eluting stent in de novo coronary lesions. EuroIntervention. 2008;4(1):53-58. [43] Shen L, Wang QB, Wu YZ, et al. Short-term effects of fully bioabsorbable PLLA coronary stents in a porcine model. Polymer Bulletin. 2012;68(4):1171-1181. [44] Wu Y, Shen L, Wang Q, et al. Comparison of acute recoil between bioabsorbable poly-L-lactic acid XINSORB stent and metallic stent in porcine model. J Biomed Biotechnol. 2012;2012:413956. [45] Ge JB. The first case of independent bioresorbable stent (Xinsorb) implanted in China. 2013-10-09 . http://www.menzhen.org/. [46] Jabara R, Chronos N, Robinson K. Novel bioabsorbable salicylate-based polymer as a drug-eluting stent coating. Catheter Cardiovasc Interv. 2008;72(2):186-194. [47] Jabara R, Pendyala L, Geva S, et al. Novel fully bioabsorbable salicylate-based sirolimus-eluting stent. EuroIntervention. 2009;5 Suppl F:F58-64. [48] Lafont A, Durand E. A.R.T.: concept of a bioresorbable stent without drug elution. EuroIntervention. 2009;5 Suppl F:F83-87. [49] cvPIPELINE. Santa Barbara: Market Monitors Inc. 2012. http://www.cvpipeline.com. [50] Cottone RJ, Thatcher GL, Parker SP, et al. OrbusNeich fully absorbable coronary stent platform incorporating dual partitioned coatings. EuroIntervention. 2009;5 Suppl F: F65-71. [51] Vogt F, Stein A, Rettemeier G, et al. Long-term assessment of a novel biodegradable paclitaxel-eluting coronary polylactide stent. Eur Heart J. 2004;25(15):1330-1340. [52] Tsuji T, Tamai H, Igaki K, et al. Biodegradable stents as a platform to drug loading. Int J Cardiovasc Intervent. 2003;5(1):13-16. [53] Wittchow E, Adden N, Riedmüller J, et al. Bioresorbable drug-eluting magnesium-alloy scaffold: design and feasibility in a porcine coronary model. EuroIntervention. 2013;8(12):1441-1450. [54] Serruys PW, Garcia-Garcia HM, Onuma Y. From metallic cages to transient bioresorbable scaffolds: change in paradigm of coronary revascularization in the upcoming decade? Eur Heart J. 2012;33(1):16-25b. [55] Waksman R. Current state of the absorbable metallic (magnesium) stent. EuroIntervention. 2009;5 Suppl F: F94-97. [56] Williams D. New interests in magnesium. Med Device Technol. 2006;17(3):9-10. [57] Heublein B, Rohde R, Kaese V, et al. Biocorrosion of magnesium alloys: a new principle in cardiovascular implant technology? Heart. 2003;89(6):651-656. [58] Zartner P, Cesnjevar R, Singer H, et al. First successful implantation of a biodegradable metal stent into the left pulmonary artery of a preterm baby. Catheter Cardiovasc Interv. 2005;66(4):590-594. [59] Di Mario C, Griffiths H, Goktekin O, et al. Drug-eluting bioabsorbable magnesium stent. J Interv Cardiol. 2004; 17(6):391-395. [60] Erbel R, Di Mario C, Bartunek J, et al. Temporary scaffolding of coronary arteries with bioabsorbable magnesium stents: a prospective, non-randomised multicentre trial. Lancet. 2007;369(9576):1869-1875. [61] Ghimire G, Spiro J, Kharbanda R, et al. Initial evidence for the return of coronary vasoreactivity following the absorption of bioabsorbable magnesium alloy coronary stents. EuroIntervention. 2009;4(4):481-484. [62] Haude M, Erbel R, Erne P, et al. Safety and performance of the drug-eluting absorbable metal scaffold (DREAMS) in patients with de-novo coronary lesions: 12 month results of the prospective, multicentre, first-in-man BIOSOLVE-I trial. Lancet. 2013;381(9869):836-844. [63] Peuster M, Wohlsein P, Brügmann M, et al. A novel approach to temporary stenting: degradable cardiovascular stents produced from corrodible metal-results 6-18 months after implantation into New Zealand white rabbits. Heart. 2001;86(5):563-569. [64] Waksman R, Pakala R, Baffour R, et al. Short-term effects of biocorrodible iron stents in porcine coronary arteries. J Interv Cardiol. 2008;21(1):15-20. [65] Peuster M, Hesse C, Schloo T, et al. Long-term biocompatibility of a corrodible peripheral iron stent in the porcine descending aorta. Biomaterials. 2006;27(28): 4955-4962. [66] Pierson D, Edick J, Tauscher A, et al. A simplified in vivo approach for evaluating the bioabsorbable behavior of candidate stent materials. J Biomed Mater Res B Appl Biomater. 2012;100(1):58-67. [67] Helmus MN, Gibbons DF, Cebon D. Biocompatibility: meeting a key functional requirement of next-generation medical devices. Toxicol Pathol. 2008;36(1):70-80. [68] Waksman R, Pakala R, Kuchulakanti PK, et al. Safety and efficacy of bioabsorbable magnesium alloy stents in porcine coronary arteries. Catheter Cardiovasc Interv. 2006;68(4): 607-619. [69] O'Brien B, Carroll W. The evolution of cardiovascular stent materials and surfaces in response to clinical drivers: a review. Acta Biomater. 2009;5(4):945-958. [70] Hermawan H, Dubé D, Mantovani D. Degradable metallic biomaterials: design and development of Fe-Mn alloys for stents. J Biomed Mater Res A. 2010;93(1):1-11. [71] Schinhammer M, Hänzi AC, Löffler JF, et al. Design strategy for biodegradable Fe-based alloys for medical applications. Acta Biomater. 2010;6(5):1705-1713. [72] Liu B, Zheng YF. Effects of alloying elements (Mn, Co, Al, W, Sn, B, C and S) on biodegradability and in vitro biocompatibility of pure iron. Acta Biomater. 2011;7(3): 1407-1420. [73] Peng T, Gibula P, Yao KD, et al. Role of polymers in improving the results of stenting in coronary arteries. Biomaterials. 1996;17(7):685-694. [74] Tanimoto S, Serruys PW, Thuesen L, et al. Comparison of in vivo acute stent recoil between the bioabsorbable everolimus-eluting coronary stent and the everolimus-eluting cobalt chromium coronary stent: insights from the ABSORB and SPIRIT trials. Catheter Cardiovasc Interv. 2007;70(4):515-523. [75] Zhu S1, Xu L, Huang N. Development of biodegradable magnesium-based biomaterial]. Sheng Wu Yi Xue Gong Cheng Xue Za Zhi. 2009;26(2):437-439, 451. [76] Acharya G, Park K. Mechanisms of controlled drug release from drug-eluting stents. Adv Drug Deliv Rev. 2006;58(3): 387-401. |
| [1] | Shi Yehong, Wang Cheng, Chen Shijiu. Early thrombosis and prevention of small-diameter blood vessel prosthesis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1110-1116. |
| [2] | Cheng Jianjun, Ding Ya, Dong Lei, Pan Tan, Li Xinglong, Yu Haiyang, Wang Hongliang. Efficacy and safety of combined use of tranexamic acid in total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(22): 3514-3520. |
| [3] | Wu Qian, Liu Lingfeng, Li Lisong, Lu Yingjie, Zhou Liyu, Xu Wu, Huang Lixin, Jiang Dinghua. Deep vein thrombosis distribution and risk factors after total knee arthroplasty during enhanced recovery after surgery [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(13): 2044-2050. |
| [4] | Jing Jinpeng, Zhang Yue, Liu Xiaomin, Liu Yi. Traditional Chinese medicine injection for promoting blood circulation in prevention of deep vein thrombosis after orthopedic surgery: network meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1467-1476. |
| [5] | Yang Yang, Li Naxi, Zhang Jian, Wang Mian, Gong Taifang, Gu Liuwei. Effect of tourniquet combined with exsanguination band use on short-term lower extremity venous thrombosis after knee arthroscopy [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(6): 898-903. |
| [6] | Yang Feng, Zhao Qian, Zhang Shixuan, Zhao Tienan, Feng Bo. Effectiveness and safety of rapamycin combined with CD133 antibody stent in preventing vascular restenosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 579-584. |
| [7] | Rong Yi, Yu Hao, Yang Junfeng, Wang Lan, Zhang Jiannan, Shao Yang. Risk factor analysis and prediction of deep venous thrombosis of lower extremity in elderly patients with hip fracture after operation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(33): 5357-5363. |
| [8] | Ma Jing, Zheng Liping, Sun Ji, Yang Xiaoqin, Pu Linyun, Yuan Tun, Liang Jie. Evaluation of thrombosis in thoracic aortic stent system with covered stent and bare stent [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(22): 3474-3479. |
| [9] | Guo Yangyan, Yu Zhengwen, Zhang Jian. Research hotspots of magnesium alloy biomaterials in an in vivo animal [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(22): 3556-3565. |
| [10] | Chen Jinping, Li Kui, Chen Qian, Guo Haoran, Zhang Yingbo, Wei Peng. Meta-analysis of the efficacy and safety of tranexamic acid in open spinal surgery [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1458-1464. |
| [11] | Xu Jianxia, Wang Zhaoxu, Wang Chunren. Blood compatibility of disposable blood perfusion device in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 588-592. |
| [12] | Li Fang, Wu Ketong, Zhao Jun, Li Gang. Advances of endovascular stent and its treatment for aneurysms [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(34): 5561-5569. |
| [13] | Fan Xinchao, Bao Wenjuan, Zhang Kai, Sun Xilong, Huang Teng, Gao Bo, Zhai Jinshuai, Zhou Yibin, Qiu Changmao, Li Wenyi, Li Xicheng. Diagnostic value of D-dimer, erythrocyte sedimentation rate and C-reactive protein in deep vein thrombosis of lower extremity after hip and knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(33): 5324-5328. |
| [14] | Jing Jinpeng, Zhang Yue, Liu Xiaomin, Liu Yi. Buyang Huanwu Decoction in prevention of deep venous thrombosis after orthopedic surgery: meta-analysis and trial sequential analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(20): 3267-3274. |
| [15] | Zhou Qi, Gao Yi, Wei Kang, Li Jun, Xu Jianda, Jiang Yang, Qu Yuxing. Total knee arthroplasty for rheumatoid arthritis: knee function and biochemical index changes [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(9): 1337-1341. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||