Chinese Journal of Tissue Engineering Research ›› 2013, Vol. 17 ›› Issue (20): 3738-3745.doi: 10.3969/j.issn.2095-4344.2013.20.018
Previous Articles Next Articles
Application of hyaluronic acid in bone tissue engineering
Mao Qiu-hua, Xu Pu
- Department of Oral Implantology, Hainan Provincial Stomatology Center, Affiliated Haikou Hospital, Xiangya School of Medicine, Central South University, Haikou 570208, Hainan Province, China
-
Received:2012-12-01Revised:2013-03-03Online:2013-05-14Published:2013-05-14 -
Contact:Xu Pu, M.D., Chief physician, Master’s supervisor, Department of Oral Implantology, Hainan Provincial Stomatology Center, Affiliated Haikou Hospital, Xiangya School of Medicine, Central South University, Haikou 570208, Hainan Province, China hnxupu@163.com -
About author:Mao Qiu-hua★, Studying for master’s degree, Department of Oral Implantology, Hainan Provincial Stomatology Center, Affiliated Haikou Hospital, Xiangya School of Medicine, Central South University, Haikou 570208, Hainan Province, China
CLC Number:
Cite this article
Mao Qiu-hua, Xu Pu. Application of hyaluronic acid in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2013, 17(20): 3738-3745.
share this article

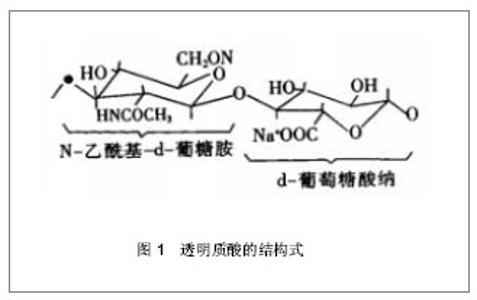
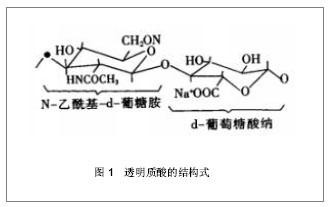
透明质酸是结缔组织中的细胞外基质的重要的组成成分,广泛分布于动物和人体结缔组织、上皮组织、神经组织中,在眼玻璃体、脐带、皮肤、软骨和滑液中含量较高。Meyer等[5]于1934年首次从牛眼玻璃体中提取得到透明质酸。随后Kendall等[6]于1937年成功地从细菌如链球菌中分离出了透明质酸,从细菌发酵生产透明质酸的方法为其广泛应用于骨组织工程奠定了基础。 2.2 透明质酸在骨组织工程中的应用 透明质酸是一种可降解生物材料,具有良好的生物相容性,其亲水性对细胞吸附、生长和分化具有重要的作用,可以作为一种临时的骨架支持和刺激新骨组织的生长,在完成一定时间的机械支撑作用后逐步降解被新生的骨组织替代[7-8]。有研究表明,透明质酸可以通过增强细胞迁移、黏附、增殖和分化来诱导新骨形成[9],也可以诱导间充质细胞分化从而加速骨缺损形成新 骨[10]。Mendes等[11]在拔牙后的牙槽窝内灌注1% 透明质酸钠0.1 mL,组织形态学及免疫组织化学分析显示,早期骨小梁沉积明显增加,骨形成蛋白2和骨桥蛋白的表达明显提高,说明透明质酸可以加速拔牙后牙槽窝愈合,诱导成骨蛋白的表达;Baldini等[12]将透明质酸混合自体骨和血液形成一凝胶状复合物充填拔牙后的牙槽窝,研究结果表明,透明质酸可以加速骨沉积和骨改建。但Maus等[13]的研究结果显示,注射用透明质酸及其复合骨形态发生蛋白-2都不能促进骨缺损修复。 2.2.1 透明质酸结合生长因子 将生长因子以合适的基质输送,诱导宿主组织中未分化的多潜能干细胞形成新骨,可促进局部骨缺损修复。在生理条件下, 透明质酸分子中糖醛酸基上的羟基呈游离状态, 当与钠离子结合后, 该多聚糖便以钠盐形式存在,即透明质酸钠(sodium hyaluronate,SH),杨峰等[14]观察了透明质酸钠凝胶为载体的碱性成纤维细胞生长因子对骨折愈合的影响,发现碱性成纤维细胞生长因子/透明质酸钠复合物比碱性成纤维细胞生长因子能更快地促进骨折愈合;透明质酸钠可作为碱性成纤维细胞生长因子的有效载体,使碱性成纤维细胞生长因子从透明质酸钠载体上缓慢释放。张力等[15]将转基因成肌细胞与骨形成蛋白2结合应用于修复兔挠骨缺损,发现透明质酸钠凝胶可以作为构建组织工程骨的良好载体。李守宏等[16-17]发现重组人类转化生长因子β1加入透明质酸钠凝胶可促进微血管的发生和血循环的再建,诱导新骨形成,能充分发挥转化生长因子的成骨效应和透明质酸钠凝胶的缓释功能,促进骨缺损修复。此外,路正刚等[18]在放疗后3个月的狗下颔无牙区植入钛种植体,局部使用骨形成蛋白和透明质酸钠凝胶复合物,结果显示种植体骨结合、骨小梁体积百分率均较对照侧高。 2.2.2 化学改性透明质酸 透明质酸的降解时间与其分子量的大小紧密相关,因此,要想延长透明质酸分子在机体中的降解时间,势必就需要通过化学修饰制备出一种相对分子质量远高于天然透明质酸钠分子的衍生物, 即交联的透明质酸钠衍生物,交联透明质酸钠的制备原理是使用一种或多种组合的化学交联剂, 利用交联剂自身存在的相关官能团使透明质酸分子发生化学反应, 从而使透明质酸分子之间交联在一起。通过交联反应使透明质酸分子延长、增大或降低其溶解性能, 从而提高机械强度和抵御机体的降解作用[19]。因此,学者们对透明质酸进行了多种化学改性,并应用于骨组织工程的研究。 Martinez-Sanz等[20]以氨基丙三醇为交联剂,通过酰胺化反应形成腙-HA基质,体外实验证明这种透明质酸衍生物复合骨形成蛋白2没有细胞毒性,组织相容性较好,将复合物注射在鼠颅骨表面,8周后组织学检查显示颅骨表面有新骨形成,而且有较高骨钙素、骨髓血管生成的表达,即腙-透明质酸可以作为骨形成蛋白2的运输载体,并能促进骨扩增。其后续研究也证实了这一发现[21]。Bae等[2]观察了光化透明质酸复合辛伐他汀对骨再生的影响,光固化透明质酸是2-氨基乙基甲基丙烯酸酯与透明质酸羧酸共轭形成,结果显示2-氨基乙基甲基丙烯酸酯-透明质酸基质的粘弹性较透明质酸具有显著的提高,并能调节辛伐他汀稳定缓慢释放,而辛伐他汀可以促进 MC3T3-E1细胞增殖和分化,从而诱导新骨形成,即光化透明质酸复合辛伐他汀可以作为构建组织工程骨的良好支架。Lisignoli等[22]将透明质酸通过酯化衍生而来的透明质酸苄基酯与骨髓基质细胞结合研究鼠模型中大面积骨缺损中的骨生成,细胞是在碱性成纤维生长因子补充或没有补充的矿化介质中培养,分别在40,60,80,200 d后进行缺损愈合评价,体内研究证明透明质酸苄基酯是骨缺损修复的合适载体,并且当和骨髓基质细胞以及碱性成纤维生长因子结合时能显著加速骨的矿化。此外,Kim等[23]将透明质酸丙烯酸酯、骨形成蛋白2和人骨髓间充质干细胞结合研究鼠颅骨缺损中的骨生成,细胞在透明质酸丙烯酸酯与骨形成蛋白2复合物中培养的存活率较高,4周后进行组织学及免疫组织化学评价,结果发现人骨髓间充质干细胞/透明质酸/骨形成蛋白2基质具有最高的骨钙素表达、成熟骨形成及血管内皮标记物(如CD31和血管内皮生长因子),即透明质酸丙烯酸酯可以作为细胞和生长因子的输送载体。 2.2.3 透明质酸-胶原纤维复合物 胶原是细胞外最重要的水不溶性纤维蛋白,是构成细胞外基质的骨架,含有特定的细胞结合氨基酸基团,可和细胞表面的整合黏附受体相互作用,支持细胞黏附、迁移和生长;胶原和透明质酸结合可提供更接近天然组织的复合材料,促进细胞增殖和分化。Liu等[24]用高碘酸钠改性透明质酸得到具有活性甲酰基的透明质酸,然后和胶原纤维交联得到一种新型的胶原蛋白-透明质酸基质,这种基质具有高度的多孔结构,较大的表面积和可控的降解速度,从而可以允许修复细胞的生长、增殖和分化,植入小鼠颅骨缺损模型的实验证明,胶原-透明质酸可促进骨完全愈合,并且显示有极好的生物相容性和骨传导活性,能完全整合到新修复的骨中,表明胶原基质中透明质酸的存在能支持新骨的形成,因此,胶原蛋白-透明质酸基质可作为生长因子的输送载体或可植入的细胞基质。Fischer等[25]也发现,胶原/透明质酸复合材料能促进细胞附着和生长,可以作为组织工程支架材料。de Brito等[26]观察了1%透明质酸凝胶复合可吸收胶原蛋白海绵对威斯塔鼠特定大小(5 mm直径)的颅骨缺损的愈合效果,60 d后组织学观察显示,1% 透明质酸复合可吸收胶原蛋白海绵能明显促进新骨形成。Kang等[27]用纤维蛋白/透明质酸复合物表面涂层支架作为骨形成蛋白2和脂肪基质细胞的输送载体,发现纤维蛋白-透明质酸复合物表面涂层支架明显提高了初始细胞黏附,保留骨形成蛋白2缓慢释放,并能持续刺激脂肪基质细胞的碱性磷酸酶活性,从而促进骨形成和骨矿化,即纤维蛋白-透明质酸复合物可作为生长因子和种子细胞的输送载体,促进脂肪基质细胞分化,诱导新骨再生。也有研究发现,磷酸三钙人工骨/透明质酸/Ⅰ型胶原/骨髓基质细胞复合物与自体骨具有相同的骨损修复能力, 可作为自体骨移植的替代物[28-29]。 2.2.4 透明质酸复合材料结合生长因子 透明质酸是骨修复中生长因子的良好运输载体,但其作为支架的主要缺陷是它的低细胞黏附性能,而整合素是细胞表面受体的主要家族,对细胞和细胞外基质的黏附起介导作用。Kisie等[30]将透明质酸与整合素上特定配体以共价键结合形成透明质酸-整合素基质,研究鼠模型中大面积骨缺损中的骨生成,与对照组相比,透明质酸-整合素水凝胶的细胞黏附性能和运载骨生长因子能力显著提高了,进一步提高了重组人骨形成蛋白2的成骨潜能,因此,透明质酸-整合素,基质可作为生长因子的输送载体,并具有潜在的临床应用价值。 肝素是一种酸性黏多糖,与生长因子具有高亲和性,可以增强生长因子的生物活性,控制生长因子缓慢释放。Liu等[31]将结构稳定的聚合物透明质酸与硫酸化的糖胺聚糖肝素结合合成新型的碱性成纤维细胞生长因子2输送体系, 肝素具有与碱性成纤维细胞生长因子2固有的特定结合部位,借助于稳定的胺或不稳定的亚胺将胺改性的透明质酸与氧化的肝素偶合形成二元共聚物,碱性成纤维细胞生长因子2与透明质酸-肝素轭合物的肝素部分快速键合,稳定的胺键合的凝胶能增加生长因子的稳定性和活性,控制有生物活性的骨形成蛋白在骨修复过程中持续缓慢释放。Xu等[32]通过反相乳化聚合技术,采用乙烯基砜为交联剂合成肝素-透明质酸水凝胶微粒,通过调协集成微粒中肝素的量来改变组合物的液相,使所得的微观粒子呈球形,形成纳米孔结构而适合于对生长因子封装;共价键固化肝素能稳定骨形态发生蛋白2,并通过调整微粒组合物来调整它的释放动力,相比纯透明质酸颗粒,混合动力透明质酸/肝素水凝胶微粒显示较高的骨形成蛋白2承载能力;透明质酸/肝素水凝胶微粒能控制骨形成蛋白2释放,诱导小鼠骨髓间充质干细胞分化形成成骨细胞,促进特异性的胞外基质分泌。基于透明质酸/肝素水凝胶粒子系统能调节有生物活性的骨形成蛋白2的持续缓慢释放,并能刺激多潜能干细胞分化的能力, 是骨修复和骨再生中合适的骨形成蛋白2输送载体。Bhakta等[33]的体外研究也显示透明质酸复合肝素可以控制骨形成蛋白2持续缓慢释放,延长生长因子骨诱导活性时间,而透明质酸载体初期释放骨形成蛋白2较多,释放持续时间相对偏短;但体内实验发现,与透明质酸/肝素基质相比,透明质酸为载体的骨形成效果较好,即异体骨移植主要受最初骨生长因子释放量影响。 2.3 透明质酸在骨组织工程中研究应用文献分析 2.3.1 透明质酸在骨组织工程中的应用文献的出版时间、资助基金、机构分析 CNKI数据库2002至2011年共收录了关于透明质酸在骨组织工程中研究应用的文献799篇。在这10年间,每年出版的文献数量变化不大,见图2。国家自然科学基金资助了16篇相关文献,其余基金资助的文献少于5篇,包括辽宁省科学技术基金、国家重点基础研究发展计划(973计划)、上海科技发展基金、上海市重点学科建设基金、国家高技术研究发展计划(863计划)等,见表1。"
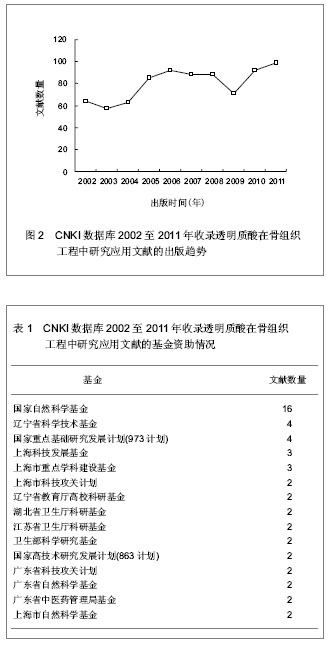

在发表相关文献的机构中,湖北中医学院、解放军总医院、广州中医药大学、华南理工大学发表文献较多,超过10篇。其他发表较多的有西安市红十字会医院、辽宁中医药大学、辽宁医学院、华中科技大学同济医学院附属协和医院、南方医科大学、中南大学湘雅医学院第二附属医院,见表2。 2.3.2 透明质酸在骨组织工程中研究应用的典型文献的研究方法和目的分析 CNKI数据库收录的关于透明质酸在骨组织工程中的应用的典型文献的研究方法和目的分析见表3,4。这些文献主要研究在3方面:①采用含透明质酸钠及其复合物的培养基在体外培养细胞,研究透明质酸钠对细胞体外成骨诱导分化的影响。②将透明质酸及其复合物注入骨缺损动物损伤处,探讨透明质酸植入体内后诱导成骨的能力,对骨缺损的修复效果。③种植体周围植入透明质酸钠及其复合物,观察局部应用透明质酸钠及其复合物对植体骨愈合作用的影响。这些文献表明近年透明质酸在骨组织工程上的应用研究引起了越来越多的关注,但其临床研究较少。"
| [1] O'Brien FJ. Biomaterials & scaffolds for tissue engineering. Materials Today. 2011;14(3):88-95.[2] Bae MS, Yang DH, Lee JB, et al. Photo-cured hyaluronic acid-based hydrogels containing simvastatin as a bone tissue regeneration scaffold. Biomaterials. 2011;32(32):8161-8171.[3] Kogan G, Soltes L, Stern R, et al. Hyaluronic acid: a natural biopolymer with a broad range of biomedical and industrial applications. Biotechnol Lett. 2007;29(1):17-25.[4] Laurent TC, Laurent UB, Fraser JR. Functions of hyaluronan. Ann Rheum Dis. 1995;54(5): 429-432.[5] Meyer K, Palmer JW. The polysaccharide of the vitreous humor. J Biol Chem. 1934;107:629-634.[6] Schiller S. Synthesis of hyaluronic acid by a soluble enzyme system from mammalian tissue. Biochem Biophys Res Commun. 1964;15(3):250-255.[7] Collins MN, Birkinshaw C. Hyaluronic Acid based Scaffolds for Tissue Engineering–A Review. Carbohydrate Polymers. 2013;92(2):1262-1279.[8] Depan D, Girase B, Shah JS , et al. Structure-process-property relationship of the polar graphene oxide-mediated cellular response and stimulated growth of osteoblasts on hybrid chitosan network structure nanocomposite scaffolds. Acta Biomater. 2011;7(9):3432-3445.[9] Aslan M, Simsek G, Dayi E. The effect of hyaluronic acid-supplemented bone graft in bone healing: experimental study in rabbits. J Biomater Appl. 2006;20(3):209-220.[10] Sasaki T, Watanabe C. Stimulation of osteoinduction in bone wound healing by high-molecular hyaluronic acid. Bone. 1995;16(1):9-15.[11] Mendes RM, Silva GA, LimaMF, et al. Sodium hyaluronate accelerates the healing process in tooth sockets of rats. Arch Oral Biol. 2008;53(12):1155-1162.[12] Baldini A, Zaffe D, Nicolini G. Bone-defects healing by high-molecular hyaluronic acid: preliminary results. Ann Stomatol (Roma). 2010;1(1):2-7.[13] Maus U, Andereya S, Gravius S, et al. Lack of effect on bone healing of injectable BMP-2 augmented hyaluronic acid. Arch Orthop Trauma Surg. 2008;128(12):1461-1466.[14] 杨峰,谢富强,谢秋玲,等.bFGF复合透明质酸钠凝胶对兔骨折愈合的X线评估[J].中国兽医杂志,2008,44(11):44-46.[15] 张力,王伟,崔福斋,等.新型组织工程化骨修复骨缺损的实验研究[C].第七届全国康复医学工程与康复工程学术研讨会,2010:168-175.[16] 李守宏,庞光明,陈建钢,等.TGF-β1复合物诱导颌骨成骨过程中微血管的形成与表达[J]. 临床口腔医学杂志,2007,23(3): 137-139.[17] 李守宏,庞光明,陈建钢,等. 透明质酸钠凝胶复合TGF-β1修复兔下颌骨缺损的实验研究[J].临床口腔医学杂志,2008,24(3): 157-159.[18] 路正刚,刘洪臣,王所亭,等.BMP与透明质酸钠复合物对放疗后种植体骨愈合影响的实验研究[J].口腔颌面外科杂志,2002, 12(4):297-299,308.[19] Ossipov DA, Piskounova S, Varghese OP, et al. Functionalization of hyaluronic acid with chemoselective groups via a disulfide-based protection strategy for in situ formation of mechanically stable hydrogels. Biomacromolecules. 2010;11(9):2247-2254.[20] Martinez-Sanz E, Ossipov DA, Hilborn J, et al. Bone reservoir: Injectable hyaluronic acid hydrogel for minimal invasive bone augmentation. J Control Release. 2011;152(2):232-240.[21] Martinez-Sanz E, Varghese OP, Kisiel M, et al. Improving the osteogenic potential of BMP-2 with hyaluronic acid hydrogel modified with integrin-specific fibronectin fragment. Biomaterials. 2013;34(3):704-712.[22] Lisignoli G, Fini M, Giavaresi G, et al. Osteogenesis of large segmental radius defects enhanced by basic fibroblast growth factor activated bone marrow stromal cells grown on non-woven hyaluronic acid-based polymer scaffold. Biomaterials. 2002;23(4):1043-1051.[23] Kim J, Kim IS, Cho TH, et al. One regeneration using hyaluronic acid-based hydrogel with bone morphogenic protein-2 and human mesenchymal stem cells. Biomaterials. 2007;28(10):1830-1837.[24] Liu LS, Thompson AY, Heidaran MA, et al. An osteoconductive collagen/hyaluronate matrix for bone regeneration. Biomaterials. 1999;20(12):1097-1108.[25] Fischer RL, McCoy MG, Grant SA. Electrospinning collagen and hyaluronic acid nanofiber meshes. J Mater Sci Mater Med. 2012;7(23):1645-1654.[26] de Brito BB, Mendes BMA, de Campos ML, et al. Association of hyaluronic acid with a collagen scaffold may improve bone healing in critical-size bone defects. Clin Oral Implants Res. 2012;23(8):938-942.[27] Kang SW, Kim JS, Park KS, et al. Surface modification with fibrin/hyaluronic acid hydrogel on solid-free form-based scaffolds followed by BMP-2 loading to enhance bone regeneration. Bone. 2011;48(2):298-306.[28] 卫爱林,叶小丰,刘世清,等.磷酸三钙/透明质酸/Ⅰ型胶原/骨髓基质胞复合物修复骨缺损与自体骨的比较(英文) [J].中国组织工程研究与临床康复,2007,(9):1779-1782.[29] 刘璐,李瑞欣,张莉,等.透明质酸改性壳聚糖-胶原-羟基磷灰石复合支架力学特性及成骨细胞的增殖(英文) [J].中国组织工程研究与临床康复,2011,15(38):7127-7131. [30] Kisiel M, Martino MM, Ventura M, et al. Improving the osteogenic potential of BMP-2 with hyaluronic acid hydrogel modified with integrin-specific fibronectin fragment. Biomaterials. 2013;34(3):704-712.[31] Liu LS, Ng CK, Thompson AY, et al. Hyaluronate-heparin conjugate gels for the delivery of basic fibroblast growth factor (FGF-2). J Biomed Mater Res. 2002;62(1):128-135.[32] Xu X, Jha AK, Duncan RL, et al. Heparin-decorated, hyaluronic acid-based hydrogel particles for the controlled release of bone morphogenetic protein 2. Acta Biomater. 2011;7(8):3050-3059.[33] Bhakta G, Rai B, Lim ZX, et al. Hyaluronic acid-based hydrogels functionalized with heparin that support controlled release of bioactive BMP-2. Biomaterials. 2012;33(26): 6113-6122. [34] 张玉强,李游,王岩峰,等.透明质酸钠对大鼠成肌细胞体外成骨分化的影响[J].中国医科大学学报,2011,(5):408-411.[35] 周薇娜,杜一飞,武和明,等.透明质酸钠复合rhBMP-2促进骨质疏松症种植体骨结合的实验研究[J].南京医科大学学报:自然科学版,2010,(7):957-961.[36] 梅晰凡,王岩松,吕刚,等.透明质酸钠可作为成骨诱导后骨髓间充质干细胞的载体[J].解剖学杂志,2009,32(02):152-154,179. [37] 张梅霞,姚瑶,闵少雄,等.生物活性玻璃/胶原蛋白/透明质酸/磷酸丝氨酸仿生复合支架的成骨及矿化效应[J].中国组织工程研究与临床康复,2008,12(14):2611-2614. [38] 章军辉,陈永强,汤亭亭,等.透明质酸复合BMP-2转染骨髓间充质干细胞修复兔桡骨干骨缺损[J].中华骨科杂志,2005,25(10): 608-612.[39] 卫爱林,刘世清,彭昊,等.磷酸三钙-透明质酸-Ⅰ型胶原-骨髓基质细胞复合修复骨缺损的实验研究[J].中国修复重建外科杂志, 2005,19(06):468-472.[40] 张双越,陈宁,宋晓玲, 等.重组人骨形成蛋白-2复合1%透明质酸钠骨膜内诱导成骨的实验研究[J].口腔医学,2004,24(02):71-73.[41] 蔡春水,肖平,李家元,等.透明质酸复合自体骨髓移植修复兔桡骨缺损[J]中国矫形外科杂志,2002,9(6):597-599.[42] Purcell BP, Elser JA, Mu A, et al. Synergistic effects of SDF-1α chemokine and hyaluronic acid release from degradable hydrogels on directing bone marrow derived cell homing to the myocardium. Biomaterials. 2012;33(31): 7849-7857.[43] Hempel U, Hintze V, Möller S, et al. Artificial extracellular matrices composed of collagen I and sulfated hyaluronan with adsorbed transforming growth factor β1 promote collagen synthesis of human mesenchymal stromal cells. Acta Biomater. 2012;8(2):659-666.[44] Yoon JS, Lee HJ, Choi SH, et al. Quercetin inhibits IL-1β-induced inflammation, hyaluronan production and adipogenesis in orbital fibroblasts from Graves' orbitopathy. PLoS One. 2011;6(10):e26261.[45] Ruffell B, Poon GF, Lee SS, et al. Differential use of chondroitin sulfate to regulate hyaluronan binding by receptor CD44 in Inflammatory and Interleukin 4-activated Macrophages. J Biol Chem. 2011;286(22):19179-19190.[46] Guo N, Woeller CF, Feldon SE, et al. Peroxisome proliferator-activated receptor gamma ligands inhibit transforming growth factor-beta-induced, hyaluronan-dependent, T cell adhesion to orbital fibroblasts. J Biol Chem. 2011;286(21):18856-18867.[47] Liu XW, Hu J, Man C, et al. Insulin-like growth factor-1 suspended in hyaluronan improves cartilage and subchondral cancellous bone repair in osteoarthritis of temporomandibular joint. Int J Oral Maxillofac Surg. 2011;40(2):184-190.[48] Kang SW, Kim JS, Park KS, et al. Surface modification with fibrin/hyaluronic acid hydrogel on solid-free form-based scaffolds followed by BMP-2 loading to enhance bone regeneration. Bone. 2011;48(2):298-306.[49] Docherty-Skogh AC, Bergman K, Waern MJ, et al. Bone morphogenetic protein-2 delivered by hyaluronan-based hydrogel induces massive bone formation and healing of cranial defects in minipigs. Plast Reconstr Surg. 2010; 125(5):1383-1392.[50] Hintze V, Moeller S, Schnabelrauch M, et al. Modifications of hyaluronan influence the interaction with human bone morphogenetic protein-4 (hBMP-4). Biomacromolecules. 2009;10(12):3290-3297.[51] Hannan FM, Athanasou NA, Teh J, et al. Oncogenic hypophosphataemic osteomalacia: biomarker roles of fibroblast growth factor 23, 1,25-dihydroxyvitamin D3 and lymphatic vessel endothelial hyaluronan receptor 1. Eur J Endocrinol. 2008;158(2):265-271. |
| [1] | Wu Xun, Meng Juanhong, Zhang Jianyun, Wang Liang. Concentrated growth factors in the repair of a full-thickness condylar cartilage defect in a rabbit [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1166-1171. |
| [2] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [3] | Li Cai, Zhao Ting, Tan Ge, Zheng Yulin, Zhang Ruonan, Wu Yan, Tang Junming. Platelet-derived growth factor-BB promotes proliferation, differentiation and migration of skeletal muscle myoblast [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1050-1055. |
| [4] | Luo Xuanxiang, Jing Li, Pan Bin, Feng Hu. Effect of mecobalamine combined with mouse nerve growth factor on nerve function recovery after cervical spondylotic myelopathy surgery [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 719-722. |
| [5] | Nie Huijuan, Huang Zhichun. The role of Hedgehog signaling pathway in transforming growth factor beta1-induced myofibroblast transdifferentiation [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 754-760. |
| [6] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [7] | Liu Liu, Zhou Qingzhu, Gong Zhuo, Liu Boyan, Yang Bin, Zhao Xian. Characteristics and manufacturing techniques of collagen/inorganic materials for constructing tissue-engineered bone [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 607-613. |
| [8] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [9] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [10] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [11] | Jiang Tao, Ma Lei, Li Zhiqiang, Shou Xi, Duan Mingjun, Wu Shuo, Ma Chuang, Wei Qin. Platelet-derived growth factor BB induces bone marrow mesenchymal stem cells to differentiate into vascular endothelial cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 3937-3942. |
| [12] | Liu Jinwei, Chen Yunzhen, Wan Chunyou. Changes of osteogenic growth factors in the broken end of bone nonunion under stress [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3619-3624. |
| [13] | Zhou Wu, Wang Binping, Wang Yawen, Cheng Yanan, Huang Xieshan. Transforming growth factor beta combined with bone morphogenetic protein-2 induces the proliferation and differentiation of mouse MC3T3-E1 cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3630-3635. |
| [14] | Xie Yang, Lü Zhiyu, Zhang Shujiang, Long Ting, Li Zuoxiao. Effects of recombinant adeno-associated virus mediated nerve growth factor gene transfection on oligodendrocyte apoptosis and myelination in experimental autoimmune encephalomyelitis mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3678-3683. |
| [15] | Zhou Anqi, Tang Yufei, Wu Bingfeng, Xiang Lin. Designing of periosteum tissue engineering: combination of generality and individuality [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3551-3557. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||