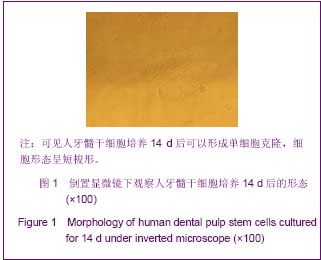
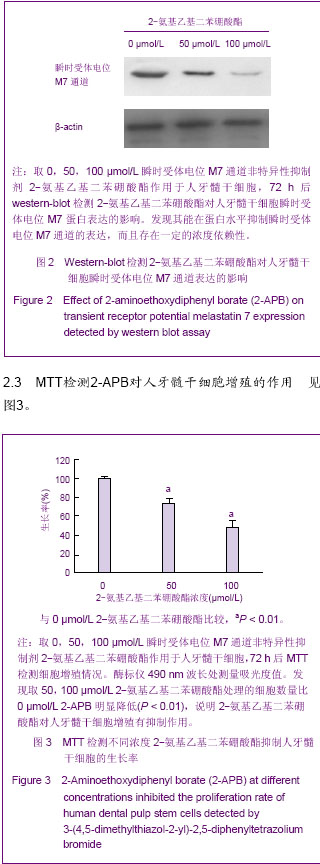
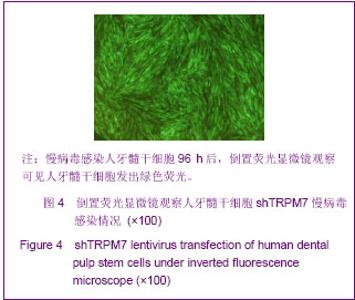
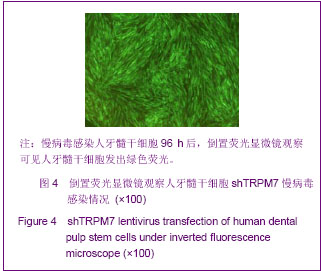
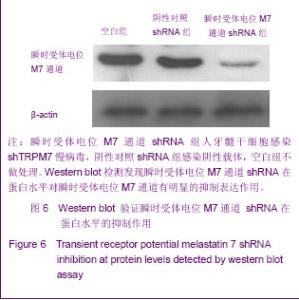
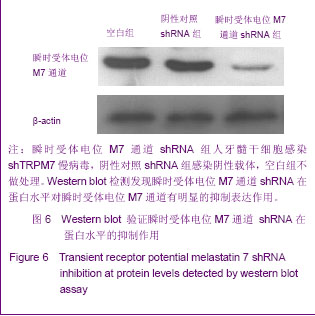
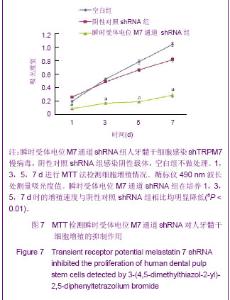
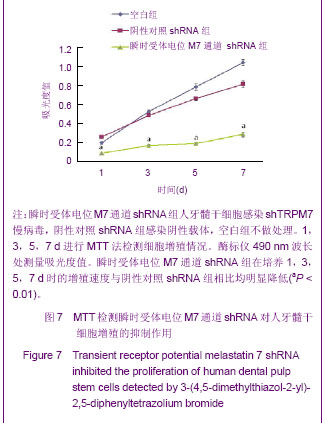
Chinese Journal of Tissue Engineering Research ›› 2013, Vol. 17 ›› Issue (19): 3495-3502.doi: 10.3969/j.issn.2095-4344.2013.19.012
Previous Articles Next Articles
Role of transient receptor potential melastatin 7 in the proliferation of human dental pulp stem cells
Cui Li1, 2, Xu Shuai-mei1, 2, Wu Bu-ling1, 2
- 1 Nanfang Hospital, Southern Medical University, Guangzhou 510515, Guangdong Province, China
2 College of Stomatology, Southern Medical University, Guangzhou 510515, Guangdong Province, China
-
Received:2012-12-10Revised:2013-02-21Online:2013-05-07Published:2013-05-07 -
Contact:r, Chief physician, Doctoral supervisor, Nanfang Hospital, Southern Medical University, Guangzhou 510515, Guangdong Province, China; College of Stomatology, Southern Medical University, Guangzhou 510515, Guangdong Province, China wubuling@yahoo.com -
About author:Cui Li★, Studying for master’s degree, Nanfang Hospital, Southern Medical University, Guangzhou 510515, Guangdong Province, China; College of Stomatology, Southern Medical University, Guangzhou 510515, Guangdong Province, China -
Supported by:Program of Introducing Talents of Discipline to Universities in Guangdong Province, No. C1010047*
CLC Number:
Cite this article
Cui Li, Xu Shuai-mei, Wu Bu-ling. Role of transient receptor potential melastatin 7 in the proliferation of human dental pulp stem cells[J]. Chinese Journal of Tissue Engineering Research, 2013, 17(19): 3495-3502.
share this article
| [1] Tecles O, Laurent P, Zygouritsas S, et al. Activation of human dental pulp progenitor/stem cells in response to odontoblast injury. Arch Oral Biol. 2005;50(2):103-108.[2] Fonfria E, Murdock PR, Cusdin FS, et al. Tissue distribution profiles of human TRPM cation channel family. J recept signal transduct re. 2006;26(3):159-178.[3] Ryazanova LV, Rondon LJ, Zierler S, et al. TRPM7 is essential for Mg2+ homeostasis in mammals. Nat Commun. 2010;1:109.[4] Penner R, Fleig A. The Mg2+ and Mg(2+)-nucleotide- regulated Channel-Kinase TRPM7. Handb Exp Pharmacol. 2007;(179):313-328.[5] Cheng H, Feng JM, Beck A, et al. Transient receptor potential melastatin type 7 is critical for the survival of bone marrow derived mesenchymal stem cell .Stem cell devel. 2011;19(9): 1393-1403.[6] Abed E, Moreau R. Importance of melastatin-like transient potential 7 and cations(magnesium, calcium)in human osteoblast-like cell proliferation. Cell Prolif. 2007;40(6): 849-865. [7] Lam DH, Grant CE, Hill CE. Differential expression of TRPM7 in rat hepatoma and embryonic and adult hepatocytes. Can J Physiol Pharmacol. 2012 ;90(4):435-444.[8] Du J, Xie J, Zhang Z, et al. TRPM7-mediated Ca2+ signals confer fibrogenesis in human atrial fibrillation. Circ Res. 2010; 106(5):992-1003.[9] Nilius B, Owsianik G. The transient receptor potential family of ion channels. Genome Biol. 2011;12(3):218. [10] Zholos A, Johnson C, Burdyga T, et al. TRPM channels in the vasculature.Adv Exp Med Biol. 2011;704:707-729. [11] Paravicini TM, Chubanov V, Gudermann T. TRPM7: A unique channel involved in magnesium homeostasis Int J Biochem Cell Biol. 2012;44(8):1381-1384. [12] Yogi A, Callera GE, Antunes TT, et al. Transient receptor potential melastatin 7 (TRPM7) cation channels, magnesium and the vascular system in hypertension. Circ J. 2011;75(2): 237-245 .[13] Runnels LW. TRPM6 and TRPM7: A Mul-TRP-PLIK-cation of channel functions. Curr Pharm Biotechnol. 2011;12(1):42-53.[14] TSchlingmann KP, Waldegger S, Konrad M, et al. TRPM6 and TRPM7--Gatekeepers of human magnesium metabolism.Biochim Biophys Acta. 2007;1772(8):813-821. [15] Scharenberg AM. TRPM2 and TRPM7: channel/enzyme fusions to generate novel intracellular sensors. Pflugers Arch. 2005;451(1):220-227.[16] Chubanov V, Mederos y, Schnitzler M, et al. Emerging roles of TRPM6/TRPM7 channel kinase signal transduction complexes. Naunyn Schmiedebergs Arch Pharmacol. 2005;371(4):334-341.[17] Runnels LW, Yue L, Clapham DE. The TRPM7 channel is inactivated by PIP(2) hydrolysis. Nat Cell Biol. 2002;4(5):329- 336.[18] Gwanyanya A, Sipido KR, Vereecke J, Mubagwa K. ATP and PIP2 dependence of the magnesium-inhibited, TRPM7-like cation channel in cardiac myocytes.Am J Physiol Cell Physiol. 2006 ;291(4):C627-635.[19] Macianskiene R, Gwanyanya A, Vereecke J , et al. inhibition of the magnesium-sensitive TRPM7-like channel in cardiac myocytes by nonhydrolysable GTP analogs: involvement of phosphoinositide metabolism. Cell Physiol Biochem. 2008; 22(1-4):109-118. [20] Park KS, Pang B, Park SJ, et al. Identification and functional characterization of ion channels in CD34(+) hematopoietic stem cells from human peripheral blood. Mol Cells. 2011;32 (2): 181-188.[21] Chubanov V, Gudermann T, Schlingmann KP. Essential role for TRPM6 in epithelial magnesium transport and body magnesium homeostasis. Pflugers Arch. 2005;451(1): 228-234.[22] Gronthos S, Mankani M, Brahim J, et al. Postnatal human dental pulp stem cells (DPSCs) in vitro and in vivo. Proc Natl Acad Sci U S A. 2000;97(25):13625-13630.[23] Gronthos S, Brahim J, Li W, et al. Stem cell properties of human dental pulp stem cells. J Dent Res. 2002;81(8): 531-535.[24] Zhang W, Walboomers XF, Wolke JG, et al. Differentiation ability of rat postnatal dental pulp cells in vitro. Tissue Eng. 2005;11(3-4):357-368. [25] Yang X, Zhang W, van den Dolder J, et al. Multilineage potential of STRO-1+ rat dental pulp cells in vitro. J Tissue Eng Regen Med. 2007;1(2):128-135.[26] Arthur A, Rychkov G, Shi S, et al. Adult human dental pulp stem cells differentiate toward functionally active neurons under appropriate environmental cues. Stem Cells. 2008; 26(7):1787-1795.[27] Paino F, Ricci G, De Rosa A, et al. Ecto-mesenchymal stem cells from dental pulp are committed to differentiate into active melanocytes. Eur Cell Mater. 2010;7(20):295-305 .[28] Gomes JA, Geraldes Monteiro B, Melo GB, et al. Corneal reconstruction with tissue-engineered cell sheets composed of human immature dental pulp stem cells. Invest Ophthalmol Vis Sci. 2010;51(3):1408-1414. [29] Cordeiro MM, Dong Z, Kaneko T, et al. Dental pulp tissue engineering with stem cells from exfoliated deciduous teeth. J Endod. 2008;34(8):962-969.[30] Sakai VT, Zhang Z, Dong Z, et al. SHED differentiate into functional odontoblasts and endothelium. J Dent Res. 2010; 89(8):791-796.[31] Bates-Withers C, Sah R, Clapham DE.TRPM7, the Mg2+ Inhibited Channel and Kinase. Adv Exp Med Biol. 2011;704: 173-183.[32] Chokshi R, Fruasaha P, Kozak JA. 2-aminoethyl diphenyl borinate (2-APB) inhibits TRPM7 channels through an intracellular acidification mechanism. Channels. 2012;6(5): 362-369.[33] Dreyer JL. Lentiviral vector-mediated gene transfer and RNA silencing technology in neuronal dysfunctions. Mol Biotechnol. 2011;47(2):169-187.[34] Manjunath N, Wu H, Subramanya S, et al. Lentiviral delivery of short hairpin RNAs. Adv Drug Deliv Rev. 2009;61(9):732- 745.[35] Nishitsuji H, Ikeda T, Miyoshi H, et al. Expression of small hairpin RNA by lentivirus-based vector confers efficient and stable gene-suppression of HIV-1 on human cells including primary non-dividing cells. Microbes Infect. 2004;6(1):76-85.[36] Jin J, Wu LJ, Clapham DE, et al.The channel kinase, TRPM7, is required for early embryonic development. PNAS. 2012; 109 (5):E225-233.[37] Schmitz C, Perraud AL, Johnson CO, et al. Regulation of vertebrate cellular Mg2+ homeostasis by TRPM7. Cell. 2003; 114(2):191-200.[38] Jiang J, Li MH, Inoue K, et al. Transient receptor potential melastatin 7-like current in human head and neck carcinoma cells:role in cell proliferation. Cancer Res. 2007;67(22): 10929-10938.[39] Hanano T, Hara Y, Shi J, et al. Involvement of TRPM7 in cell growth as a spontaneously activated Ca2+ entry pathway in human retinoblastoma cell. J Pharmacol Sci. 2004;95(4): 403-419.[40] Guilbert, A, Sevestre H, Ouadid-Ahidouch H, et al. Evidence that TRPM7 is required for breast cancer cell proliferation. Am J Physiol Cell Physiol. 2009;297(3):493-502.[41] Kim BJ, Park EJ, Lee JH, et al. Suppression of transient receptor potential melastatin 7 channel induces cell death in gastric cancer. Cancer Sci. 2008;99(12):2502-2509.[42] Yee NS, Zhou W, Liang IC. Transient receptor potential ion channel TRPM7 regulates exocrine pancreatic epithelial proliferation by Mg2+-sensitive Socs3a signaling in development and cancer. Dis Model Mech. 2011;4(2):240-254. |
| [1] | Li Cai, Zhao Ting, Tan Ge, Zheng Yulin, Zhang Ruonan, Wu Yan, Tang Junming. Platelet-derived growth factor-BB promotes proliferation, differentiation and migration of skeletal muscle myoblast [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1050-1055. |
| [2] | Liu Cong, Liu Su. Molecular mechanism of miR-17-5p regulation of hypoxia inducible factor-1α mediated adipocyte differentiation and angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1069-1074. |
| [3] | Wang Shiqi, Zhang Jinsheng. Effects of Chinese medicine on proliferation, differentiation and aging of bone marrow mesenchymal stem cells regulating ischemia-hypoxia microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1129-1134. |
| [4] | Ma Zetao, Zeng Hui, Wang Deli, Weng Jian, Feng Song. MicroRNA-138-5p regulates chondrocyte proliferation and autophagy [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 674-678. |
| [5] | Wang Yujiao, Liu Dan, Sun Song, Sun Yong. Biphasic calcium phosphate loaded with advanced platelet rich fibrin can promote the activity of rabbit bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 504-509. |
| [6] | Chen Junyi, Wang Ning, Peng Chengfei, Zhu Lunjing, Duan Jiangtao, Wang Ye, Bei Chaoyong. Decalcified bone matrix and lentivirus-mediated silencing of P75 neurotrophin receptor transfected bone marrow mesenchymal stem cells to construct tissue-engineered bone [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 510-515. |
| [7] | Zhou Jihui, Yao Meng, Wang Yansong, Li Xinzhi, Zhou You, Huang Wei, Chen Wenyao. Influence of novel nanoscaffolds on biological behaviors of neural stem cells and the related gene expression [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 532-536. |
| [8] | Zhou Wu, Wang Binping, Wang Yawen, Cheng Yanan, Huang Xieshan. Transforming growth factor beta combined with bone morphogenetic protein-2 induces the proliferation and differentiation of mouse MC3T3-E1 cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3630-3635. |
| [9] | Liu Keke, Duan Xin, Ma Xiangrui, Zhang Yuntao. Effect of cinnamaldehyde on osteoblasts in high glucose environment with the electrospinning membrane as a carrier [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3500-3504. |
| [10] | Song Shilei, Chen Yueping, Zhang Xiaoyun, Li Shibin, Lai Yu, Zhou Yi. Potential molecular mechanism of Wuling powder in treating osteoarthritis based on network pharmacology and molecular docking [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(20): 3185-3193. |
| [11] | Yu Chengshuai, Du Gang, Pang Shenning, Lao Shan. Chemerin, a pro-inflammatory adipokine, regulates chondrocyte proliferation and metabolism by increasing production of nitric oxide [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(2): 258-263. |
| [12] | Gao Yuanhui, Xiang Yang, Cao Hui, Wang Shunlan, Zheng Linlin, He Haowei, Zhang Yingai, Zhang Shufang, Huang Denggao. Comparison of biological characteristics of adipose derived mesenchymal stem cells in Wuzhishan inbreed miniature pigs aged two different months [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(19): 2988-2993. |
| [13] | Dai Min, Wang Shuai, Zhang Nini, Huang Guilin, Yu Limei, Hu Xiaohua, Yi Jie, Yao Li, Zhang Ligang. Biological characteristics of hypoxic preconditioned human amniotic mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(19): 3004-3008. |
| [14] | Dai Yaling, Chen Lewen, He Xiaojun, Lin Huawei, Jia Weiwei, Chen Lidian, Tao Jing, Liu Weilin. Construction of miR-146b overexpression lentiviral vector and the effect on the proliferation of hippocampal neural stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(19): 3024-3030. |
| [15] | Ailimaierdan·Ainiwaer, Wang Ling, Gu Li, Dilidaer•Taxifulati, Wang Shan, Yin Hongbin. Effect of transforming growth factor-beta3 on the proliferation and osteogenic capability of osteoblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(17): 2664-2669. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||