Chinese Journal of Tissue Engineering Research ›› 2019, Vol. 23 ›› Issue (25): 4031-4036.doi: 10.3969/j.issn.2095-4344.1795
Previous Articles Next Articles
Research progress in stem cell-based tissue engineering technology assisting mandibular distraction osteogenesis
- Department of Oral and Maxillofacial Surgery, Stomatology Hospital of Guangxi Medical University, Nanning 530021, Guangxi Zhuang Autonomous Region, China
-
Revised:2019-03-19Online:2019-09-08Published:2019-09-08 -
Contact:Zhou Nuo, Professor, Doctoral supervisor, Department of Oral and Maxillofacial Surgery, Stomatology Hospital of Guangxi Medical University, Nanning 530021, Guangxi Zhuang Autonomous Region, China -
About author:Han Zhiqi, Master candidate, Department of Oral and Maxillofacial Surgery, Stomatology Hospital of Guangxi Medical University, Nanning 530021, Guangxi Zhuang Autonomous Region, China -
Supported by:the National Natural Science Foundation of China, No. 81670970 (to ZN)
CLC Number:
Cite this article
Han Zhiqi, Jiang Weidong, Zhou Nuo. Research progress in stem cell-based tissue engineering technology assisting mandibular distraction osteogenesis[J]. Chinese Journal of Tissue Engineering Research, 2019, 23(25): 4031-4036.
share this article

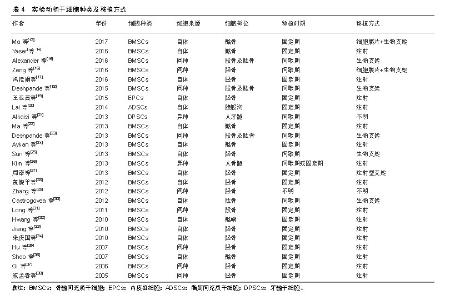
2.1 纳入文献基本情况 经过文献检索,干细胞移植辅助下颌牵张成骨动物实验共26篇文献[13-38],对全部文献的主要实验结论进行总结并制表,如表1所示,绝大多数干细胞移植都在下颌牵张成骨动物实验中起到了增强成骨或成血管的作用。 2.2 实验动物模型的选择 在全部26篇文献中,有20篇文献的实验动物是小型动物,如兔、大鼠等啮齿动物;仅有6篇文献的实验动物是大型动物,如犬、猪、绵羊、山羊等,见表2。这可能是由于小型实验动物易操作、易进行更深层次分子层面研究。但是,由于其与人下颌骨的大小、形态、功能上的巨大差异,使用小型动物进行下颌牵张成骨实验结果很难应用到临床中[39]。而就大型动物而言,其下颌骨的大小、形态等更接近人类,其实验结果更容易投入到临床应用中,进行进一步的临床试验。因此,未来使用大型哺乳动物进行下颌牵张成骨实验研究将会成为一种趋势,为多中心、大样本随机对照临床试验提供理论基础。 就实验动物性别而言,13篇文献为雄性动物,3篇文献为雌性动物,10篇文献并未提及动物性别。目前并未有学者对性别因素是否影响干细胞组织工程辅助下颌牵张成骨进行研究,且下颌牵张成骨手术标准并未提及性别差异。将来可能需要进一步研究性别因素是否影响干细胞辅助下颌牵张成骨的成骨成血管功能。"

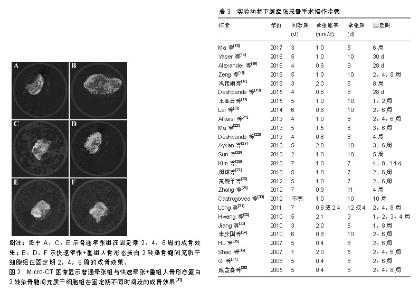
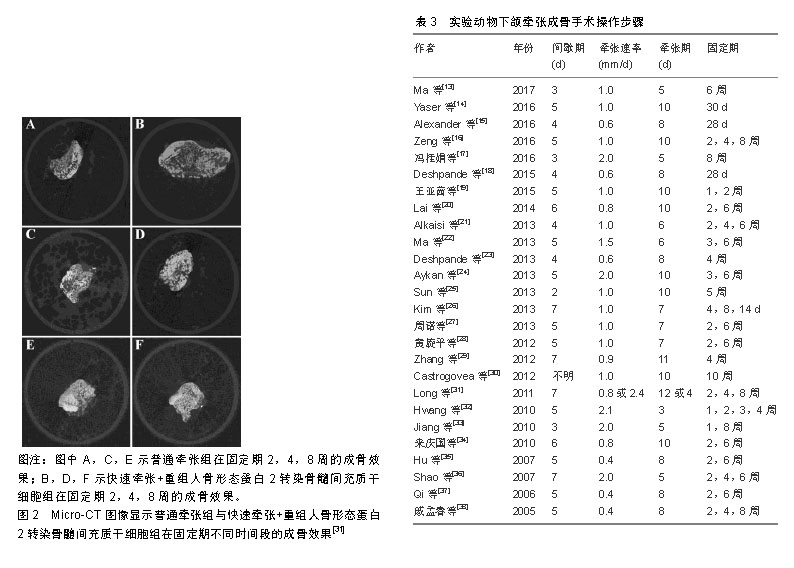
2.3 实验动物下颌牵张成骨手术步骤 在全部26篇文献中,就牵张器械而言,不同的学者使用了各不相同的手术器材,包括商品化牵张器与各种自制牵张器,目前学界没有一个明确的器械规范。下颌牵张成骨手术操作步骤一般分为3步,分别为间歇期、牵张期及固定期。如表3所示,间歇期的时间范围在2-7 d,牵张期的时间范围在3-12 d,牵张的速率范围在0.4-2.4 mm/d,固定期的时间范围在4 d-10周。 由于实验动物模型选择及构建的不同,实验动物下颌牵张成骨手术标准不完全相同。近10年学者们的研究表明,小型实验动物如大鼠、兔等,其最佳牵张速率可能为0.5 mm/d,而大型实验动物如犬、猪等,其最佳牵张速率可能应该与人类相似,为1 mm/d。为了探究干细胞组织工程辅助下颌牵张成骨在成骨速度上的效果,干细胞移植实验组需要进行更快的牵张速率与当前国际公认的最适牵张速率进行对比,而目前只有极少数实验进行了干细胞移植辅助快速牵张与普通牵张的对比,如Long等[31]在2011年日本兔下颌处进行的实验研究,其结论表明进行了重组人骨形成蛋白2转染骨髓间充质干细胞移植的快速牵张组与普通牵张组在成骨质量上大致一致,显著缩短了牵张期所需要的时间,这提示干细胞组织工程可以缩短下颌牵张成骨的牵张期时间,从而有效减少术区感染、纤维愈合等各种并发症的发生,提高疗效,见图2。"

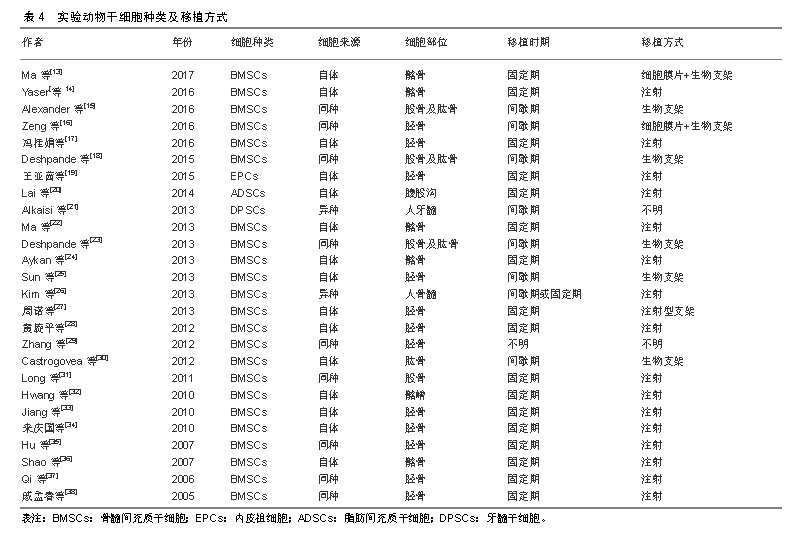
(1)骨髓间充质干细胞:自1970年Freidenstein等[40]发现在塑料培养皿上骨髓间充质干细胞可以分化成为骨及软骨后,骨髓间充质干细胞做为一种具有自我更新能力和多向分化潜能的细胞,逐渐被学界所熟悉,成为近年来干细胞辅助下颌牵张成骨的研究热点。大量研究表明局部应用骨髓间充质干细胞可以促进骨折愈合、缩短骨折愈合时间。由于骨髓间充质干细胞具有显著的成骨分化潜能,目前是组织工程中最常用的骨再生细胞[41]。在下颌牵张成骨的应用中,23篇文献全部证实骨髓间充质干细胞可以促进下颌牵张成骨的成骨速度,缩短固定期时间。陈燕等[42]将骨髓间充质干细胞注入实验组兔下颌牵张成骨处,生理盐水注入对照组,通过免疫组化法进行检测发现新生骨痂处骨保护素的表达显著高于对照组,这可能说明骨髓间充质干细胞促进下颌牵张成骨的作用可能是通过骨保护素的高表达来实现的。 (2)内皮祖细胞:内皮祖细胞是血管内皮细胞的前体细胞,在生理或病理因素刺激下,可从骨髓中动员到损伤处进行损伤修复。自1997年Asahara等[43]首次证明了循环外周血中存在能分化为血管内皮细胞的前体细胞,将其命名为内皮祖细胞以来,因其具有促血管新生的作用,越来越多的学者将其作为种子细胞用于骨组织工程学。Rozen等[44]将内皮祖细胞等移植于山羊胫骨骨缺损动物模型中,发现其修复缺损能力明显增强。王亚茜[19]首次使用内皮祖细胞移植辅助下颌牵张成骨,证实内皮祖细胞可以通过促进血管新生的方式增强下颌牵张成骨中的血管新生与血管生成,从而缩短治疗时间。然而有关内皮祖细胞辅助下颌牵张成骨的实验研究极少,体内外实验探究内皮祖细胞与下颌牵张成骨的关系将会成为一个研究热点。 (3)脂肪间充质干细胞:近年来,由于脂肪间充质干细胞数量丰富且易于提取的原因,逐渐受到学者们的大量关注。相比骨髓间充质干细胞的提取需要对患者髂骨、胫骨等骨髓进行穿刺,造成患者极大痛苦,脂肪间充质干细胞的提取则相对简单易行,且对患者创伤小。Halvorsen等[45]观测到脂肪间充质干细胞可以诱导分化为成骨细胞、成软骨细胞等多种细胞。骨髓间充质干细胞的成骨能力大于脂肪间充质干细胞,因为骨髓间充质干细胞具有固有的成骨特性,而脂肪间充质干细胞需要外部环境的刺激来诱导分化为成骨细胞。Mahmoudifar等[46]向培养脂肪间充质干细胞的培养基中添加地塞米松、骨形态发生蛋白等成功将脂肪间充质干细胞诱导为成骨细胞,但是这些体外诱导方式无法运用于干细胞体内移植。Lai等[20]将Osterix转染于脂肪间充质干细胞并移植于下颌牵张成骨处,证明Osterix转染的脂肪间充质干细胞可以增强下颌牵张成骨的成骨速度与质量,而未转染的脂肪间充质干细胞则与普通下颌牵张成骨无明显差别。 (4)牙髓干细胞:牙髓干细胞由Gronthos等[47]在2000年时首次发现并正式提出,并指出牙髓干细胞具有多向分化潜能,不仅可以分化为成牙本质细胞,而且可以在特殊刺激下诱导分化为成骨细胞。这可能是由于牙髓干细胞来源于颅面神经嵴,对颅面骨骼的发育具有促进作用。在提取方式上,相对于骨髓间充质干细胞、内皮祖细胞、脂肪间充质干细胞等需要对患者进行有创操作造成患者痛苦,牙髓干细胞可在脱落乳牙内提取,方便易行,且对患者无损伤。越来越多的国家已经建立了乳牙牙髓干细胞库[48],为日后治疗机体创伤及病变提供了一个很好的组织工程学种子细胞来源。在下颌牵张成骨研究方面,Alkaisi等[21]首次将人牙髓干细胞移植于兔下颌牵张成骨处,结果不仅证实牙髓干细胞显著增强兔下颌牵张成骨的质量与速度,而且发现在未使用免疫抑制剂的情况下向兔体内移植人牙髓干细胞并未造成免疫排斥反应,这为临床上使用同种或异种牙髓干细胞进行下颌牵张成骨的治疗打下了一定的基础。尽管上述研究证实脂肪间充质干细胞、牙髓干细胞等细胞在下颌牵张成骨应用中取得了积极的成果,但也有报道指出,这些细胞与骨髓间充质干细胞相比具有较低的成骨能力[49-50]。接下来需要将这些细胞与骨髓间充质干细胞辅助下颌牵张成骨进行对比,从成骨效率与方便易行两方面确定最适合进行干细胞治疗的细胞种类。 2.4.2 细胞移植方式 如表4所示,在26篇文献中,通过注射方式将干细胞悬液注射入下颌牵张成骨间隙的文献共有16篇,通过生物支架将干细胞放入下颌牵张成骨间隙的文献有8篇,通过细胞膜片将干细胞植入下颌牵张成骨间隙的文献有2篇(2篇同时使用了细胞膜片+生物支架),另有2篇未提及移植方式。Ma等[22]研究发现细胞膜片移植促进下颌牵张成骨的成骨作用要优于注射干细胞悬液,见图3,这可能得益于细胞膜片可以将干细胞更好地保留并固定在牵张成骨区,使得牵张成骨区的干细胞数量与浓度显著增加,一次性较大数量细胞移植得以实现。但相对于简单快捷的注射操作,使用细胞膜片或生物支架的方式不得不进行下颌牵张成骨区的二次开创手术,这会增加感染等并发症的风险。周诺等[27]使用一种注射型生物支架对氧化聚乙烯和聚丙烯共聚物(PluronicF-127)成功解决了使用生物支架需要二次植入的问题,并证实使用PluronicF-127要优于注射细胞悬液。细胞移植的最佳时机是截骨时植入细胞膜片或生物支架,牵张期、固定期注射干细胞悬液。这都说明了下颌牵张成骨区干细胞固定并保留的重要性,将来仍需进一步研究移植方法与移植时机的选择是否通过增加下颌牵张成骨区细胞数量影响成骨质量与速度。"

| [1]Primrose AC, Broadfoot E, Diner PA, et al. Patients' responses to distraction osteogenesis: a multi-centre study. Int J Oral Maxillofac Surg. 2005;34(3):238-242.[2]王良,李军,沈毅,等. 虚拟手术辅助的血管化腓骨肌(皮)瓣行下颌骨精确重建[J].中国耳鼻咽喉颅底外科杂志, 2016,22(3): 220-224.[3]张赛,张龙城.下颌骨缺损修复的研究进展[J].中国耳鼻咽喉颅底外科杂志, 2017,23(2):177-181.[4]邹霓.骨组织工程人工合成支架材料的研究现状[J].中国组织工程研究与临床康复, 2008,12(23):4481-4484.[5]Codivilla A. The classic: On the means of lengthening, in the lower limbs, the muscles and tissues which are shortened through deformity. 1905. Clin Orthop Relat Res. 2008;466(12):2903-2909.[6]Ilizarov GA. The principles of the Ilizarov method. 1988. Bull Hosp Jt Dis. 1997;56(1):49-53.[7]McCarthy JG, Schreiber J, Karp N, et al. Lengthening the human mandible by gradual distraction. Plast Reconstr Surg.1992; 89(1): 1-8; discussion 9-10.[8]Nørholt SE, Jensen J, Schou S, et al. Complications after mandibular distraction osteogenesis: a retrospective study of 131 patients. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2011; 111(4):420-427.[9]Ai-Aql ZS, Alagl AS, Graves DT, et al. Molecular mechanisms controlling bone formation during fracture healing and distraction osteogenesis. J Dent Res. 2008;87(2):107-118.[10]Khedr MS, Abdel Al SE, Osman MM, et al. The effect of electrical stimulation on mandibular distraction osteogenesis. Egyptian Journal of Oral & Maxillofacial Surgery. 2011; 2(2):41-49.[11]Xie LK, Wangrangsimakul K, Suttapreyasri S, et al. A preliminary study of the effect of low intensity pulsed ultrasound on new bone formation during mandibular distraction osteogenesis in rabbits. Int J Oral Maxillofac Surg. 2011;40(7):730-736.[12]Wang L, Zhou S, Liu B, et al. Locally applied nerve growth factor enhances bone consolidation in a rabbit model of mandibular distraction osteogenesis. J Orthop Res. 2006;24(12):2238-2245.[13]Ma G, Zhao JL, Mao M, et al. Scaffold-Based Delivery of Bone Marrow Mesenchymal Stem Cell Sheet Fragments Enhances New Bone Formation In Vivo. J Oral Maxillofac Surg. 2017;75(1): 92-104.[14]El Hadidi YN, El Kassaby M, El Fatah Ahmed SA, et al. Effect of Mesenchymal Stem Cell Application on the Distracted Bone Microstructure: An Experimental Study. J Oral Maxillofac Surg. 2016;74(7):1463.e1-1463.e11.[15]Zheutlin AR, Deshpande SS, Nelson NS, et al. Bone marrow stem cells assuage radiation-induced damage in a murine model of distraction osteogenesis: A histomorphometric evaluation. Cytotherapy. 2016;18(5):664-672.[16]Zeng JJ, Guo P, Zhou N, et al. Treatment of large bone defects with a novel biological transport disc in non-vascular transport distraction osteogenesis. Int J Oral Maxillofac Surg. 2016;45(5): 670-677.[17]冯桂娟,郑科,宋冬惠,等. Runt相关基因2修饰的骨髓间充质干细胞促进兔下颌骨牵张成骨的研究[J].华西口腔医学杂志, 2016,34(2): 125-129.[18]Deshpande SS, Gallagher KK, Donneys A, et al. Stem cells rejuvenate radiation-impaired vasculogenesis in murine distraction osteogenesis. Plast Reconstr Surg. 2015;135(3): 799-806. [19]王亚茜.EPCs在犬下颌骨非血管化输送盘牵张成骨血管新生中的作用的研究[D]. 南宁:广西医科大学, 2015.[20]Lai QG, Sun SL, Zhou XH, et al. Adipose-derived stem cells transfected with pEGFP-OSX enhance bone formation during distraction osteogenesis. J Zhejiang Univ Sci B. 2014;15(5): 482-490.[21]Alkaisi A, Ismail AR, Mutum SS, et al. Transplantation of human dental pulp stem cells: enhance bone consolidation in mandibular distraction osteogenesis. J Oral Maxillofac Surg. 2013;71(10): 1758.e1-13.[22]Ma D, Ren L, Yao H, et al. Locally injection of cell sheet fragments enhances new bone formation in mandibular distraction osteogenesis: a rabbit model. J Orthop Res. 2013;31(7): 1082-108.[23]Deshpande SS, Gallagher KK, Donneys A, et al. Stem cell therapy remediates reconstruction of the craniofacial skeleton after radiation therapy. Stem Cells Dev. 2013;22(11):1625-1632.[24]Aykan A, Ozturk S, Sahin I, et al. Biomechanical analysis of the effect of mesenchymal stem cells on mandibular distraction osteogenesis. J Craniofac Surg. 2013;24(2):e169-175.[25]Sun Z, Tee BC, Kennedy KS, et al. Scaffold-based delivery of autologous mesenchymal stem cells for mandibular distraction osteogenesis: preliminary studies in a porcine model. PLoS One. 2013;8(9):e74672.[26]Kim IS, Cho TH, Lee ZH, et al. Bone regeneration by transplantation of human mesenchymal stromal cells in a rabbit mandibular distraction osteogenesis model. Tissue Eng Part A. 2013;19(1-2):66-78.[27]周诺,黄旋平,江献芳,等.骨形态发生蛋白-2基因修饰骨髓间充质干细胞复合对氧化聚乙烯和聚丙烯共聚物移植促进兔下颌骨牵张成骨的实验研究[J]. 华西口腔医学杂志, 2013, 31(3): 247-252.[28]黄旋平,周诺,杨媛媛,等. hBMP-2基因修饰自体 BMSCs 移植促进兔下颌骨牵张成骨新骨形成的X线分析[J]. 实用口腔医学杂志, 2012,28(4):457-460.[29]Zhang WB, Zheng LW, Chua DT, et al. Treatment of irradiated mandibles with mesenchymal stem cells transfected with bone morphogenetic protein 2/7. J Oral Maxillofac Surg. 2012;70(7): 1711-1176.[30]Castro-Govea Y, Cervantes-Kardasch VH, Borrego-Soto G, et al. Human bone morphogenetic protein 2-transduced mesenchymal stem cells improve bone regeneration in a model of mandible distraction surgery. J Craniofac Surg. 2012;23(2):392-396.[31]Long J, Li P, Du HM, et al. Effects of bone morphogenetic protein 2 gene therapy on new bone formation during mandibular distraction osteogenesis at rapid rate in rabbits. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2011;112(1):50-57.[32]Hwang YJ, Choi JY. Addition of mesenchymal stem cells to the scaffold of platelet-rich plasma is beneficial for the reduction of the consolidation period in mandibular distraction osteogenesis. J Oral Maxillofac Surg. 2010;68(5):1112-1124.[33]Jiang X, Zou S, Ye B, et al. bFGF-Modified BMMSCs enhance bone regeneration following distraction osteogenesis in rabbits. Bone. 2010;46(4):1156-1161.[34]来庆国. OSX基因修饰的骨髓MSCs促进兔下颌骨牵张成骨的实验研究[D]. 济南:山东大学, 2010.[35]Hu J, Qi MC, Zou SJ, et al. Callus formation enhanced by BMP-7 ex vivo gene therapy during distraction osteogenesis in rats. J Orthop Res. 2007;25(2):241-251.[36]Shao Z, Liu B, Peng Q, et al. Transplantation of osteoblast-like cells to the distracted callus in the rabbit mandible. Plast Reconstr Surg. 2007;119(2):500-507.[37]Qi M, Hu J, Zou S, et al. Mandibular distraction osteogenesis enhanced by bone marrow mesenchymal stem cells in rats. J Craniomaxillofac Surg. 2006;34(5):283-289.[38]戚孟春,胡静,邹淑娟,等.骨髓间充质干细胞移植促进大鼠下颌骨牵张成骨的实验研究[J].中国口腔颌面外科杂志, 2005, 3(2): 151-154.[39]Pearce AI, Richards RG, Milz S, et al. Animal models for implant biomaterial research in bone: a review. Eur Cell Mater. 2007;13: 1-10.[40]Friedenstein AJ, Gorskaja JF, Kulagina NN. Fibroblast precursors in normal and irradiated mouse hematopoietic organs. Exp Hematol. 1976;4(5):267-274.[41]Isobe Y, Koyama N, Nakao K, et al. Comparison of human mesenchymal stem cells derived from bone marrow, synovial fluid, adult dental pulp, and exfoliated deciduous tooth pulp. Int J Oral Maxillofac Surg. 2016;45(1):124-131.[42]陈燕,李志刚. MSCs对兔下颌骨牵张成骨处OPG表达的影响[J]. 药物与人, 2014,27(7): 9-10.[43]Asahara T, Murohara T, Sullivan A, et al. Isolation of putative progenitor endothelial cells for angiogenesis. Science. 1997; 275(5302):964-967.[44]Rozen N, Bick T, Bajayo A, et al. Transplanted blood-derived endothelial progenitor cells (EPC) enhance bridging of sheep tibia critical size defects. Bone. 2009;45(5):918-924.[45]Halvorsen YD, Franklin D, Bond AL, et al. Extracellular matrix mineralization and osteoblast gene expression by human adipose tissue-derived stromal cells. Tissue Eng. 2001;7(6):729-741.[46]Mahmoudifar N, Doran PM. Osteogenic differentiation and osteochondral tissue engineering using human adipose-derived stem cells. Biotechnol Prog. 2013;29(1):176-185.[47]Gronthos S, Mankani M, Brahim J, et al. Postnatal human dental pulp stem cells (DPSCs) in vitro and in vivo. Proc Natl Acad Sci U S A. 2000;97(25):13625-13630.[48]霍永标. 乳牙牙髓干细胞库的构建及其研究进展[J]. 国际口腔医学杂志, 2011, 38(2): 188-191.[49]Wu W, Le AV, Mendez JJ, et al. Osteogenic performance of donor-matched human adipose and bone marrow mesenchymal cells under dynamic culture. Tissue Eng Part A. 2015;21(9-10): 1621-1632.[50]Ceccarelli G, Bloise N, Mantelli M, et al. A comparative analysis of the in vitro effects of pulsed electromagnetic field treatment on osteogenic differentiation of two different mesenchymal cell lineages. Biores Open Access. 2013;2(4):283-294.[51]Kitoh H, Kitakoji T, Tsuchiya H, et al. Transplantation of marrow-derived mesenchymal stem cells and platelet-rich plasma during distraction osteogenesis--a preliminary result of three cases. Bone. 2004;35(4):892-898. |
| [1] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [2] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [3] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [4] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [5] | Liu Cong, Liu Su. Molecular mechanism of miR-17-5p regulation of hypoxia inducible factor-1α mediated adipocyte differentiation and angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1069-1074. |
| [6] | Wang Zhengdong, Huang Na, Chen Jingxian, Zheng Zuobing, Hu Xinyu, Li Mei, Su Xiao, Su Xuesen, Yan Nan. Inhibitory effects of sodium butyrate on microglial activation and expression of inflammatory factors induced by fluorosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1075-1080. |
| [7] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [8] | Liao Chengcheng, An Jiaxing, Tan Zhangxue, Wang Qian, Liu Jianguo. Therapeutic target and application prospects of oral squamous cell carcinoma stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1096-1103. |
| [9] | Xie Wenjia, Xia Tianjiao, Zhou Qingyun, Liu Yujia, Gu Xiaoping. Role of microglia-mediated neuronal injury in neurodegenerative diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1109-1115. |
| [10] | Li Shanshan, Guo Xiaoxiao, You Ran, Yang Xiufen, Zhao Lu, Chen Xi, Wang Yanling. Photoreceptor cell replacement therapy for retinal degeneration diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1116-1121. |
| [11] | Jiao Hui, Zhang Yining, Song Yuqing, Lin Yu, Wang Xiuli. Advances in research and application of breast cancer organoids [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1122-1128. |
| [12] | Wang Shiqi, Zhang Jinsheng. Effects of Chinese medicine on proliferation, differentiation and aging of bone marrow mesenchymal stem cells regulating ischemia-hypoxia microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1129-1134. |
| [13] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [14] | Kong Desheng, He Jingjing, Feng Baofeng, Guo Ruiyun, Asiamah Ernest Amponsah, Lü Fei, Zhang Shuhan, Zhang Xiaolin, Ma Jun, Cui Huixian. Efficacy of mesenchymal stem cells in the spinal cord injury of large animal models: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1142-1148. |
| [15] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||