Chinese Journal of Tissue Engineering Research ›› 2019, Vol. 23 ›› Issue (21): 3294-3301.doi: 10.3969/j.issn.2095-4344.1760
Previous Articles Next Articles
Bone marrow mesenchymal stem cell-derived exosomes reduce the activation of type A1 astrocytes after spinal cord injury
Zhou Yan1, Wang Lin2, Pei Shuang2, Li Yanfei2, Chen Xuemei3, Jia Yanjie2
- 1Department of Radiology, 2Department of Neurology, the First Affiliated Hospital of Zhengzhou University, Zhengzhou 450052, Henan Province, China; 3Department of Human Anatomy, School of Basic Medical Sciences, Zhengzhou University, Zhengzhou 450001, Henan Province, China
-
Revised:2019-02-01Online:2019-07-28Published:2019-07-28 -
Contact:Jia Yanjie, MD, Doctoral supervisor, Professor, Chief physician, Department of Neurology, the First Affiliated Hospital of Zhengzhou University, Zhengzhou 450052, Henan Province, China -
About author:Zhou Yan, Master, Lecturer, Department of Radiology, the First Affiliated Hospital of Zhengzhou University, Zhengzhou 450052, Henan Province, China -
Supported by:the National Natural Science Foundation of China, No. U1604170 (to JYJ)
CLC Number:
Cite this article
Zhou Yan, Wang Lin, Pei Shuang, Li Yanfei, Chen Xuemei, Jia Yanjie. Bone marrow mesenchymal stem cell-derived exosomes reduce the activation of type A1 astrocytes after spinal cord injury[J]. Chinese Journal of Tissue Engineering Research, 2019, 23(21): 3294-3301.
share this article
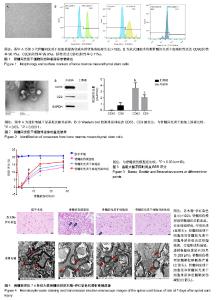
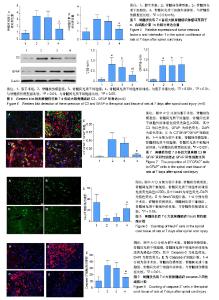
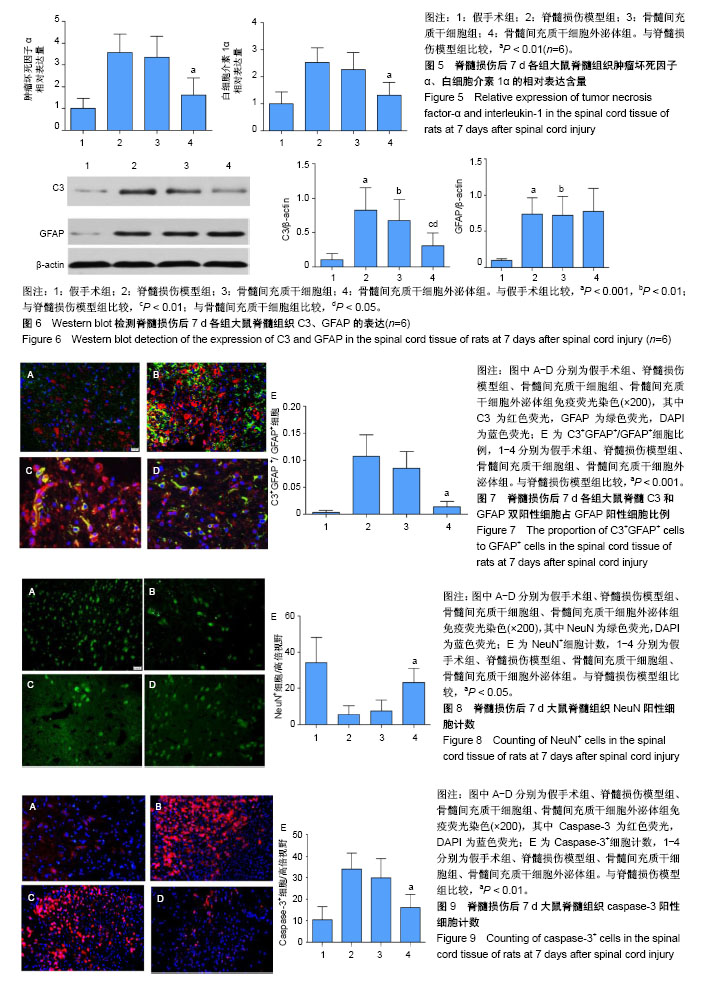
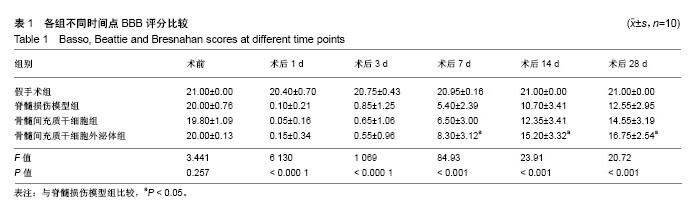
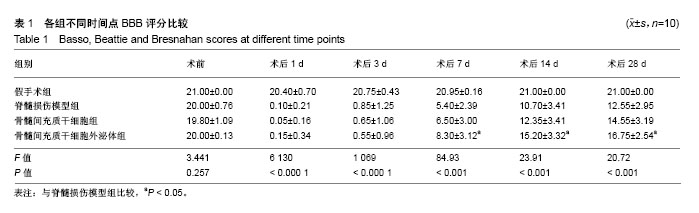
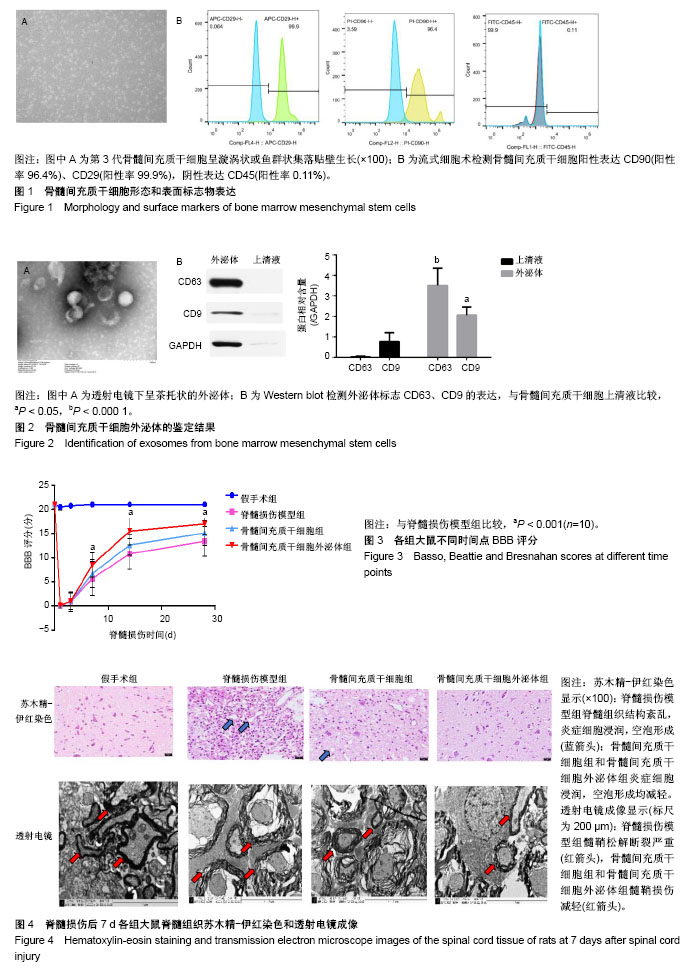
2.1 骨髓间充质干细胞的鉴定结果 细胞培养至第3代,于显微镜下观察呈漩涡状或鱼群状集落贴壁生长,见图1A。利用流式细胞术检测骨髓间充质干细胞表面标志物,每管1×105个细胞,检测3次,结果显示细胞阳性表达CD90(阳性率96.4%)、CD29(阳性率99.9%),阴性表达CD45(阳性率0.11%),见图1B。 2.2 骨髓间充质干细胞外泌体的鉴定结果 透射电镜下,可观察到多个直径为40-100 nm的茶托状囊泡结构,见图2A。Western blot检测结果显示,骨髓间充质干细胞外泌体CD63的相对表达量(3.504±0.848)明显高于骨髓间充质干细胞上清液(0.026±0.042),差异有显著性意义(t=8.295,P < 0.000 1);骨髓间充质干细胞外泌体CD9的相对表达量(2.065±0.391)明显高于骨髓间充质干细胞上清液(0.779±0.426),差异有显著性意义(t=3.067,P < 0.05)。因此,骨髓间充质干细胞外泌体表面标志蛋白CD63、CD9表达为阳性,见图2B。 2.3 实验动物一般情况分析 脊髓损伤模型制作成功80只,在各组注射PBS、骨髓间充质干细胞和外泌体过程及后续研究中,未发现大鼠出现发热、过敏和死亡等不良结局。 2.4 各组大鼠BBB评分 脊髓损伤术前、术后1,3,7,14,28 d对各组大鼠进行BBB评分,每组10只大鼠,利用GraphPad Prism软件处理BBB评分数据,见图3。各组BBB评分比较采用双因素方差分析,并计算F值和P值,见表1。结果显示:术前各组大鼠均可正常行走,评分>20分;术后1 d,除假手术组以外,其余3组均表现为双下肢瘫痪,评分<2分,且3组评分差异无显著性意义(P > 0.05);术后3 d,此3组BBB评分均有增高,但3组评分差异无显著性意义(P > 0.05);术后7,14,28 d,骨髓间充质干细胞外泌体组BBB评分显著高于脊髓损伤组和骨髓间充质干细胞组(P < 0.001);在各时间点,脊髓损伤组与骨髓间充质干细胞组BBB评分差异无显著性意义(P > 0.05)。 2.5 组织学检测结果 苏木精-伊红染色结果显示:与假手术组相比,脊髓损伤模型组大鼠脊髓组织结构紊乱、大量炎性细胞浸润、空泡形成;与脊髓损伤模型组相比,骨髓间充质干细胞组浸润的炎症细胞减少,组织结构有所改善,但仍有较多空泡存在;与脊髓损伤模型组相比,骨髓间充质干细胞外泌体组浸润炎症细胞明显减少,组织空泡、结构紊乱都有明显减轻。透射电镜成像显示:假手术组大鼠脊髓组织的髓鞘、神经元、轴突均表现为完整正常结构;脊髓损伤模型组和骨髓间充质干细胞组髓鞘呈“洋葱皮”样松解、断裂、脱失,轴突萎缩退化,神经元胞浆空泡化;骨髓间充质干细胞外泌体组与上述两组相比,髓鞘脱失、轴突退变均有较为明显的改善,见图4。 2.6 肿瘤坏死因子α、白细胞介素1α蛋白水平 ELISA检测结果显示:与假手术组相比,脊髓损伤模型组脊髓组织中肿瘤坏死因子α、白细胞介素1α相对含量显著增高(P < 0.01);与脊髓损伤模型组相比,骨髓间充质干细胞组肿瘤坏死因子α、白细胞介素1α相对含量差异均无显著性意义(P > 0.05);与脊髓损伤模型组相比,骨髓间充质干细胞外泌体组肿瘤坏死因子α、白细胞介素1α相对含量显著降低(P < 0.01),见图5。 2.7 Western blot检测脊髓组织C3、GFAP的表达 与假手术组相比,脊髓损伤模型组大鼠脊髓组织A1型星形胶质细胞标志物C3、星形胶质细胞标志物GFAP蛋白表达显著增高(P < 0.001);骨髓间充质干细胞组与脊髓损伤模型组相比,C3、GFAP表达水平差异无显著性意义(P > 0.05);骨髓间充质干细胞外泌体组与脊髓损伤模型组相比,C3水平明显降低(P < 0.01);除假手术组外,其余3组的GFAP表达水平差异无显著性意义(P > 0.05),见图6。 2.8 免疫荧光染色结果 免疫荧光双标C3和GFAP结果显示:与假手术组相比,脊髓损伤模型组C3+GFAP+/GFAP+的比例显著增大(P < 0.001);骨髓间充质干细胞组与脊髓损伤模型组相比,C3+GFAP+/GFAP+的比例差异无显著性意义(P > 0.05);骨髓间充质干细胞外泌体组与脊髓损伤模型组相比,C3+GFAP+/GFAP+的比例显著降低,差异有显著性意义(P < 0.001),见图7。 NeuN+细胞计数显示:与假手术组相比,脊髓损伤模型组NeuN+细胞数量显著下降(P< 0.001);骨髓间充质干细胞组与脊髓损伤模型组相比,NeuN+细胞数量差异无显著性意义(P > 0.05);骨髓间充质干细胞外泌体组与脊髓损伤模型组相比,NeuN+细胞数量增加,差异有显著性意义(P < 0.05),见图8。 Caspase-3+细胞计数显示:与假手术组相比,脊髓损伤模型组Caspase-3+细胞数量显著增加(P < 0.001);骨髓间充质干细胞组与脊髓损伤模型组相比,Caspase-3+细胞数量差异无显著性意义(P > 0.05);骨髓间充质干细胞外泌体组与脊髓损伤模型组相比,Caspase-3+细胞数量显著降低,差异有显著性意义(P < 0.01),见图9。"
| [1] 贾延劼,李燕飞,陈雪梅.脊髓损伤后神经病理性疼痛的研究现状[J].内科理论与实践,2017,12(2):92-96.[2] Anderson MA, Burda JE, Ren Y, et al. Astrocyte scar formation aids central nervous system axon regeneration. Nature. 2016;532(7598): 195-200.[3] Sofroniew MV. Astrogliosis. Cold Spring Harb Perspect Biol. 2014;7(2): a020420.[4] Liddelow SA, Guttenplan KA, Clarke LE, et al. Neurotoxic reactive astrocytes are induced by activated microglia. Nature. 2017;541(7638): 481-487.[5] Caplan AI, Correa D. The MSC: an injury drugstore. Cell Stem Cell. 2011;9(1):11-15.[6] Torres-Espín A, Hernández J, Navarro X. Gene expression changes in the injured spinal cord following transplantation of mesenchymal stem cells or olfactory ensheathing cells. PLoS One. 2013;8(10):e76141. [7] Riazifar M, Pone EJ, Lötvall J, et al. Stem Cell Extracellular Vesicles: Extended Messages of Regeneration. Annu Rev Pharmacol Toxicol. 2017;57:125-154.[8] Wang L, Pei S, Han L, et al. Mesenchymal Stem Cell-Derived Exosomes Reduce A1 Astrocytes via Downregulation of Phosphorylated NFκB P65 Subunit in Spinal Cord Injury. Cell Physiol Biochem. 2018;50(4):1535-1559.[9] 裴双,王琳,陈雪梅,等.骨髓间充质干细胞来源的外泌体静脉移植对脊髓损伤的修复作用[J]. 中国脊柱脊髓杂志, 2017,27(12): 1119-1127. [10] Ekström EJ, Bergenfelz C, von Bülow V, et al. WNT5A induces release of exosomes containing pro-angiogenic and immunosuppressive factors from malignant melanoma cells. Mol Cancer. 2014;13:88. [11] Franzen CA, Blackwell RH, Todorovic V, et al. Urothelial cells undergo epithelial-to-mesenchymal transition after exposure to muscle invasive bladder cancer exosomes. Oncogenesis. 2015;4:e163.[12] Hu Y, Rao SS, Wang ZX, et al. Exosomes from human umbilical cord blood accelerate cutaneous wound healing through miR-21-3p-mediated promotion of angiogenesis and fibroblast function. Theranostics. 2018; 8(1):169-184.[13] Anderson AJ, Najbauer J, Huang W, et al. Upregulation of complement inhibitors in association with vulnerable cells following contusion-induced spinal cord injury. J Neurotrauma. 2005;22(3):382-397. [14] Liddelow SA, Barres BA. Reactive Astrocytes: Production, Function, and Therapeutic Potential. Immunity. 2017; 46(6):957-967.[15] Phinney DG, Prockop DJ. Concise review: mesenchymal stem/multipotent stromal cells: the state of transdifferentiation and modes of tissue repair--current views. Stem Cells. 2007;25(11):2896-2902.[16] Lavoie JR, Rosu-Myles M. Uncovering the secretes of mesenchymal stem cells. Biochimie. 2013;95(12):2212-2221.[17] Riazifar M, Pone EJ, Lötvall J, et al. Stem Cell Extracellular Vesicles: Extended Messages of Regeneration. Annu Rev Pharmacol Toxicol. 2017;57:125-154.[18] Zhang B, Yin Y, Lai RC, et al. Mesenchymal stem cells secrete immunologically active exosomes. Stem Cells Dev. 2014;23(11): 1233-1244.[19] EL Andaloussi S, Mäger I, Breakefield XO, et al. Extracellular vesicles: biology and emerging therapeutic opportunities. Nat Rev Drug Discov. 2013;12(5):347-357.[20] van den Boorn JG, Schlee M, Coch C, et al. SiRNA delivery with exosome nanoparticles. Nat Biotechnol. 2011;29(4): 325-326.[21] Otero-Ortega L, Gómez de Frutos MC, Laso-García F, et al. Exosomes promote restoration after an experimental animal model of intracerebral hemorrhage. J Cereb Blood Flow Metab. 2018;38(5):767-779.[22] Xin H, Katakowski M, Wang F, et al. MicroRNA cluster miR-17-92 Cluster in Exosomes Enhance Neuroplasticity and Functional Recovery After Stroke in Rats. Stroke. 2017;48(3):747-753.[23] Xiong Y, Mahmood A, Chopp M. Emerging potential of exosomes for treatment of traumatic brain injury. Neural Regen Res. 2017;12(1):19-22. [24] Huang JH, Yin XM, Xu Y, et al. Systemic Administration of Exosomes Released from Mesenchymal Stromal Cells Attenuates Apoptosis, Inflammation, and Promotes Angiogenesis after Spinal Cord Injury in Rats. J Neurotrauma. 2017;34(24):3388-3396.[25] Badner A, Vawda R, Laliberte A, et al. Early Intravenous Delivery of Human Brain Stromal Cells Modulates Systemic Inflammation and Leads to Vasoprotection in Traumatic Spinal Cord Injury. Stem Cells Transl Med. 2016;5(8):991-1003.[26] Menge T, Zhao Y, Zhao J, et al. Mesenchymal stem cells regulate blood-brain barrier integrity through TIMP3 release after traumatic brain injury. Sci Transl Med. 2012;4(161): 161ra150.[27] Zhang R, Liu Y, Yan K, et al. Anti-inflammatory and immunomodulatory mechanisms of mesenchymal stem cell transplantation in experimental traumatic brain injury. J Neuroinflammation. 2013;10:106.[28] Watanabe J, Shetty AK, Hattiangady B, et al. Administration of TSG-6 improves memory after traumatic brain injury in mice. Neurobiol Dis. 2013;59:86-99.[29] Urdzíková LM, R??i?ka J, LaBagnara M, et al. Human mesenchymal stem cells modulate inflammatory cytokines after spinal cord injury in rat. Int J Mol Sci. 2014;15(7):11275-11293.[30] Rosado IR, Carvalho PH, Alves EG, et al. Immunomodulatory and neuroprotective effect of cryopreserved allogeneic mesenchymal stem cells on spinal cord injury in rats. Genet Mol Res. 2017;16(1):e12751.[31] Ylöstalo JH, Bartosh TJ, Coble K, et al. Human mesenchymal stem/stromal cells cultured as spheroids are self-activated to produce prostaglandin E2 that directs stimulated macrophages into an anti-inflammatory phenotype. Stem Cells. 2012;30(10):2283-2296.[32] Ruppert KA, Nguyen TT, Prabhakara KS, et al. Human Mesenchymal Stromal Cell-Derived Extracellular Vesicles Modify Microglial Response and Improve Clinical Outcomes in Experimental Spinal Cord Injury. Sci Rep. 2018;8(1):480.[33] Drommelschmidt K, Serdar M, Bendix I, et al. Mesenchymal stem cell-derived extracellular vesicles ameliorate inflammation-induced preterm brain injury. Brain Behav Immun. 2017;60:220-232.[34] Huang W, Lv B, Zeng H, et al. Paracrine Factors Secreted by MSCs Promote Astrocyte Survival Associated With GFAP Downregulation After Ischemic Stroke via p38 MAPK and JNK. J Cell Physiol. 2015;230(10): 2461-2475.[35] Park HJ, Shin JY, Kim HN, et al. Mesenchymal stem cells stabilize the blood-brain barrier through regulation of astrocytes. Stem Cell Res Ther. 2015;6:187.[36] He H, Zhou Y, Zhou Y, et al. Dexmedetomidine Mitigates Microglia-Mediated Neuroinflammation through Upregulation of Programmed Cell Death Protein 1 in a Rat Spinal Cord Injury Model. J Neurotrauma. 2018;35(21):2591-2603.[37] Xue H, Zhang XY, Liu JM, et al. NDGA reduces secondary damage after spinal cord injury in rats via anti-inflammatory effects. Brain Res. 2013; 1516:83-92. |
| [1] | Min Youjiang, Yao Haihua, Sun Jie, Zhou Xuan, Yu Hang, Sun Qianpu, Hong Ensi. Effect of “three-tong acupuncture” on brain function of patients with spinal cord injury based on magnetic resonance technology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-8. |
| [2] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [3] | Jiang Hongying, Zhu Liang, Yu Xi, Huang Jing, Xiang Xiaona, Lan Zhengyan, He Hongchen. Effect of platelet-rich plasma on pressure ulcers after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1149-1153. |
| [4] | Wan Ran, Shi Xu, Liu Jingsong, Wang Yansong. Research progress in the treatment of spinal cord injury with mesenchymal stem cell secretome [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1088-1095. |
| [5] | Zhao Min, Feng Liuxiang, Chen Yao, Gu Xia, Wang Pingyi, Li Yimei, Li Wenhua. Exosomes as a disease marker under hypoxic conditions [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1104-1108. |
| [6] | Wang Shiqi, Zhang Jinsheng. Effects of Chinese medicine on proliferation, differentiation and aging of bone marrow mesenchymal stem cells regulating ischemia-hypoxia microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1129-1134. |
| [7] | Kong Desheng, He Jingjing, Feng Baofeng, Guo Ruiyun, Asiamah Ernest Amponsah, Lü Fei, Zhang Shuhan, Zhang Xiaolin, Ma Jun, Cui Huixian. Efficacy of mesenchymal stem cells in the spinal cord injury of large animal models: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1142-1148. |
| [8] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [9] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [10] | Geng Yao, Yin Zhiliang, Li Xingping, Xiao Dongqin, Hou Weiguang. Role of hsa-miRNA-223-3p in regulating osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1008-1013. |
| [11] | Lun Zhigang, Jin Jing, Wang Tianyan, Li Aimin. Effect of peroxiredoxin 6 on proliferation and differentiation of bone marrow mesenchymal stem cells into neural lineage in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1014-1018. |
| [12] | Zhu Xuefen, Huang Cheng, Ding Jian, Dai Yongping, Liu Yuanbing, Le Lixiang, Wang Liangliang, Yang Jiandong. Mechanism of bone marrow mesenchymal stem cells differentiation into functional neurons induced by glial cell line derived neurotrophic factor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1019-1025. |
| [13] | Pei Lili, Sun Guicai, Wang Di. Salvianolic acid B inhibits oxidative damage of bone marrow mesenchymal stem cells and promotes differentiation into cardiomyocytes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1032-1036. |
| [14] | Ma Binxiang, He Wanqing, Zhou Guangchao, Guan Yonglin. Triptolide improves motor dysfunction in rats following spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 701-706. |
| [15] | Chen Junyi, Wang Ning, Peng Chengfei, Zhu Lunjing, Duan Jiangtao, Wang Ye, Bei Chaoyong. Decalcified bone matrix and lentivirus-mediated silencing of P75 neurotrophin receptor transfected bone marrow mesenchymal stem cells to construct tissue-engineered bone [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 510-515. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||