Chinese Journal of Tissue Engineering Research ›› 2019, Vol. 23 ›› Issue (17): 2753-2761.doi: 10.3969/j.issn.2095-4344.1700
Previous Articles Next Articles
Three-dimensional culture of human pluripotent stem cells
Qin Liying1, Zhang Rui1, Ren Xiaolin1, Han Yu1, Chen Haofei2, Zhou Ping1
- 1School of Stomatology, Lanzhou University, Lanzhou 730000, Gansu Province, China; 2the First Clinical Medical College of Lanzhou University, Lanzhou 730000, Gansu Province, China
-
Revised:2019-01-11Online:2019-06-18Published:2019-06-18 -
Contact:Zhang Rui, Master, Lecturer, School of Stomatology, Lanzhou University, Lanzhou 730000, Gansu Province, China -
About author:Qin Liying, School of Stomatology, Lanzhou University, Lanzhou 730000, Gansu Province, China -
Supported by:the National Natural Science Foundation of China, No. 81801855 (to ZP); the Fundamental Research Funds for the Central Universities, No. lzujbky-2015-295 (to ZR); the National Innovation and Entrepreneurship Training Program for College Students in Lanzhou University, No. 20180730138 (to QLY)
CLC Number:
Cite this article
Qin Liying, Zhang Rui, Ren Xiaolin, Han Yu, Chen Haofei, Zhou Ping. Three-dimensional culture of human pluripotent stem cells[J]. Chinese Journal of Tissue Engineering Research, 2019, 23(17): 2753-2761.
share this article
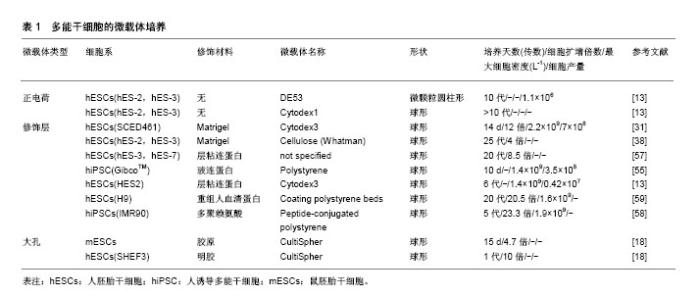
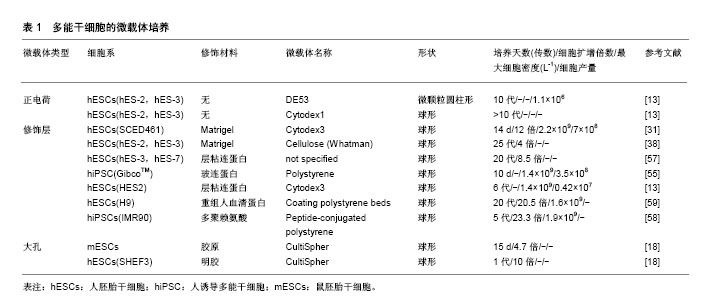
2.1 生物反应器系统 三维细胞培养包括静态和动态培养两种方法,Bardy等[21]发现,当使用Matrigel包被的DE-53微载体静态培养IMR90人诱导多能干细胞7 d,获得了7.7倍扩增和1.3×109 L-1的细胞产量,而在相同条件下改用旋转瓶培养,则能获得20倍扩增和6.1×109 L-1的细胞产量。三维培养更倾向于动态培养扩增细胞,除了简单的旋转瓶培养外,还有生物反应器悬浮动态培养。迄今为止,大部分生物反应器系统已被开发用于培养常规哺乳动物细胞系,包括波形生物反应器(也称为“细胞袋”),袋子主要分为两个区域:一个装满培养基,接种细胞,另一个装满空气,前者不断进行摇动的同时空气通过后者循环以提供必要的氧气交换[22]。填充/固定/纤维床生物反应器[23],通过在底部填充有固定支架/微载体/多孔纤维的空心柱或管将细胞接种在固定床上,同时使用泵连续循环新鲜培养基以获得足够的氧气转移和营养物供应。还有许多其他类型的生物反应器,但在人多能干细胞培养中以旋转壁式生物反应器和搅拌式生物反应器为主[24-25]。故而在此具体介绍这两种生物反应器。 旋转壁式生物反应器是将细胞及其培养基共同置于2个同轴的内外圆筒之间,沿水平轴连续旋转,在层流条件下产生微重力。通过培养基与细胞和内外圆筒的共同旋转,氧气和营养物质分布更均匀;通过加快旋转速度以减少细胞的沉降。此外,因为没有叶轮的搅拌,很大程度上降低了剪切应力对细胞活性的影响。Gerecht-Nir等[26]成功使用该系统促进拟胚体的形成。然而,旋转壁反应器的水平旋转限制了反应器体积的扩大,进而限制了其在大规模扩增细胞中的应用,该型生物反应器在人多能干细胞三维培养中应用逐渐减少。 搅拌式生物反应器由外部的玻璃容器和内部的叶轮组成,通过叶轮旋转以提供均匀的生长环境并且抵抗重力沉降,促进所有培养组分的均匀分布,将气体和营养物质转移到细胞聚集体中,并使人多能干细胞受控制的形成聚集,从而促进相对高密度下大规模扩增人多能干细胞[27-28]。研究发现,随着细胞聚集体直径的扩大,氧气和培养基中的营养物质难以到达细胞聚集体中央,细胞凋亡的数量逐渐增多[29]。因此,生物反应器系统还配备了用于监控和控制温度、pH值、营养物浓度(如葡萄糖)、代谢物(如乳酸盐、氨)和气体(如O2和CO2)等培养环境的技术。Silva等[30]使用搅拌式生物反应器以细胞聚集体的形式扩增SCED461人胚胎干细胞,100 mL体积内细胞数量达到3.01×108。Serra等[31]使用300 mL搅拌式生物反应器,在Matrigel包被的Cytodex3TM微载体上扩增SCED461人胚胎干细胞,可以获得6.78×108细胞。 然而叶轮的搅拌也会对细胞产生不利影响。例如,细胞聚集体培养中常见的细胞死亡,低细胞生长率和低产量[32]。因此,为了控制人多能干细胞聚集体的形成和促进其进一步生长,搅拌生物反应器中需要优化的参数很多。但是,由生物反应器搅动产生的流体动力学条件(例如流动方向、速度、剪切力和化学环境等)非常复杂,并且不断变化。同时,人多能干细胞对生物反应器叶轮的几何形状、尺寸和位置、容器几何形状和尺寸、搅拌速率等许多因素敏感,目前还没有研究明确这些因素如何影响人多能干细胞活性[33]。此外,尚不清楚细胞如何对这些复杂的水动力条件作出反应[34-35]。目前,仅在小鼠多能干细胞中描述了剪切应力对多能性和分化特性的影响[36-37]。Leung等[38]发现相同的剪切力对不同细胞系的影响是不同的,在搅拌的旋转瓶中以微载体形式培养人胚胎干细胞,发现与静态培养相比,HES-2细胞系即使在7次传代后也不会影响细胞活力、倍增时间和多能性标记物的表达。然而,HES-3细胞系在1次传代之后,多能标记OCT-4、MAB84和TRA-1-60表达下调。 在使用生物反应器培养人多能干细胞时,最常用的培养方法是重复批次培养,也可称为灌注培养,即每隔一定的时间部分换液(余留部分旧培养基)以提供人多能干细胞扩增所需的生长因子。然而这种培养方式并不能完全去除反应器中的代谢废物,即使缩短至每两小时间隔进行一次培养基更换[31]。近期开发出了一种新型灌注培养方式,通过将新鲜培养基连续加入生物反应器中,同时以相同的流速连续除去用过的培养基来不断为细胞提供新鲜营养素、生长因子,除去代谢废物,确保反应器中更均匀的条件,被称为循环灌注培养。Wan等[39]使用该种灌注方法获得了长期的(14-35 d)肿瘤外植体生长。然而这种灌注方式尚未被应用于人多能干细胞的扩增,并且循环灌注同样存在操作复杂、成本较高的缺点和污染的风险。 在大规模灌注过程的情况下培养基消耗较多,而培养基昂贵的生长因子添加将会产生过高的培养成本,Come等[40]曾将透析膜与灌注式培养相结合,将来有望将循环灌注系统与渗透性薄膜结合,通过膜的分子质量选择性滤出代谢废物,而将人多能干细胞释放的生长因子留在生物反应器中,使人多能干细胞通过其内源因子的分泌自我调节,减少昂贵的生长因子补充。此外,大规模扩增同样需要大体积的生物反应器,相应的叶轮体积也会增大,随之带来的剪切应力也是不能避免的。在未来,这些问题还需要更加深入的分析。 2.2 细胞聚集体 通常在涂布有疏水性和非黏附性材料的多孔板和半固体培养基(例如含有琼脂、琼脂糖或甲基纤维素等的培养基)中使用悬浮法或悬滴法进行细胞聚集体的形成。虽然其不需要附着表面和黏附分子,降低培养成本,但是处理半固体培养基和收获人多能干细胞聚集体并不容易。例如使用悬滴法,一个10 cm的培养皿仅能形成30-40个聚集体[41]。因此,这种方法下细胞不能够大规模扩增。Carpenedo等[42]发现使用旋转轨道悬浮培养系统,可以产生较悬滴法尺寸更为均匀的细胞聚集体,并且在7 d后实现了总细胞产量增加4倍。Watanabe等[43]发现在ROCK抑制剂(Y-27632)存在的情况下,可将单个分散的人多能干细胞直接接种到悬浮生物反应器,从那之后就有报道显示人多能干细胞在搅拌悬浮系统中以聚集体形式扩增。然而,人多能干细胞具有强烈的细胞间相互作用,聚集体常在培养过程中相互融合形成大的细胞团块(即团聚)。团聚导致细胞聚集体大小不均匀,对细胞培养不利甚至影响细胞表型和分化潜能。因此,这种培养方法最大的挑战之一是需要在初始和长期培养过程中控制细胞总量和聚集体直径。 在初始培养阶段调节细胞聚集体大小和形态的方法包括强制聚集和通过使用生物反应器的搅动进行调节。Choi等[44]发现圆柱形孔或矩形孔有利于产生圆盘形或圆柱形的人胚胎干细胞聚集体,而模拟悬滴液相边缘的凹形培养孔促进均匀球形聚集体的形成。Bauwens等[45]通过微孔强制聚集的方法限制了细胞聚集体的大小,并通过微孔的大小和数量调节细胞聚集体数目。Abbasalizadeh等[46]通过使用不同搅拌速率和剪切保护剂浓度的组合,优化生物反应器流体动力学条件,成功获得了特定尺寸的聚集体。Sargen等[47]发现在25 r/min下,细胞聚集体直径较大且分散,在培养7 d后聚集体大小增加。相比之下,更高的转速(55 r/min)促进形成更小和单分散的聚集体,显示有限的尺寸增加。Lock等[48]认为45 r/min至60 r/min的转速是最适宜形成尺寸均匀的聚集体。 长期培养过程中控制细胞量的方法主要是进行周期性解离。Xu等[49]证实使用Accutase添加Rho相关激酶(ROCK)抑制剂Y-27632可以极大地减少与细胞解离方法有关的细胞凋亡。同时发现ROCK抑制剂促进了静电悬浮培养中H9人胚胎干细胞聚集体的形成,使聚集体维持一定直径。Lee等[50]研究发现使用EDTA或ReLeSR的解离缓冲液将H9人胚胎干细胞聚集体解离成小团块,并在补充有Y-27632的mTESR-1培养基上培养,人胚胎干细胞的存活率比Accutase传代获得的人多能干细胞高六七倍。这些结果表明,简单的团块传代方法在不使用酶或其他补充条件下可显著提高三维悬浮培养过程中人多能干细胞聚集体的扩增效率。然而,由于人多能干细胞对物理应激和单细胞解离的敏感性,将人多能干细胞集落解离成单细胞或小团块进行传代培养会导致相当大的细胞损失。酶解离程序已被报道成为聚集培养物中细胞回收率低的主要原因[24],而机械解离技术似乎是一种有效的替代方法。Otsuji等[51]将细胞聚集体通过50 μm的网状过滤器,可以将较大的聚集体分成较小的团块,显著降低了253G1人多能干细胞的丧失,并在42 d之后达到1.65×107的细胞扩增。最近,Nath等[52]发现使用肉毒杆菌血凝素(HA)特异性结合E-钙黏蛋白可以破坏Tic人诱导多能干细胞聚集体间的细胞连接,并产生较传统解离方法更高的细胞数量、细胞密度和扩增倍数,最高细胞密度可达到(4.5±0.2)×106。此外,Chen等[53]通过将重组玻连蛋白修饰的温敏聚合物与Nkx2.5人多能干细胞混合,自发形成胚状体。当胚状体达到细胞因子和营养利用上限时,这些温敏聚合物通过从37 ℃到25 ℃的温度变化促进胚状体分解,使得细胞的循环解离和传代收集成为可能。实验重复该过程3次传代(超过18 d)提供了> 30倍的细胞扩增,为人多能干细胞的聚集体无损循环解离提供了新的研究思路。 但是如同之前所述,将细胞聚集体直接暴露于生物反应器中,其复杂的流体动力学对细胞状态影响的研究尚不清楚,可能是细胞扩增效率不高的主要原因。 2.3 微载体 微载体培养是指将人多能干细胞从培养板表面解离(常使用Accutase或EDTA)之后[48,54],以单细胞的形式接种在预先处理好的微载体上进行生长的培养方式。微载体技术具有独特的优势,因为它为细胞提供了均一的培养条件,细胞黏附和生长的表面积很大。 目前有两种微载体用于细胞培养:①实心球形或圆盘形颗粒,其中细胞仅附着于微载体表面;②由纤维素或明胶制备的大孔微载体,其允许细胞渗入。市售的大孔微载体(CultiSpherTM)可使细胞密度提高,并保护细胞免受来自生物反应器的剪切力的影响,虽然成功用于病毒疫苗和重组蛋白生产,但由于难以从大孔微载体内安全收集细胞,目前仅用于鼠胚胎干细胞的扩增[37]。 市售微载体依据其是否需要细胞外基质涂层分为两大类:一类需要涂覆Matrigel或其他细胞外基质组分,另一类不需要涂覆,通过自身电荷吸引而进行细胞培养。对于未涂覆的微载体可以观察到高的细胞附着效率,然而在连续传代期间细胞逐渐丧失多能性[18,55]。对于涂覆材料的研究多基于二维培养表面[13]。用Matrigel涂覆微载体,可以至少3次传代并保持多能性[31]。但是Matrigel成分复杂且不明确,随后有学者研究发现层粘连蛋白、胶原蛋白、重组玻连蛋白等涂覆于微载体表面而进行细胞培养[18,54-56],与Matrigel相比成分明确,不含异源成分。此外,Fan等[57]发现使用层粘连蛋白或玻连蛋白衍生的肽涂层可以显著增加H9人胚胎干细胞的附着,与Matrigel涂层相比,成分明确的涂层也引起更快的人胚胎干细胞倍增。同时他们发现,使用重组人血清白蛋白联合紫外照射包被的微载体,显著增强细胞的接种效率和存活率,H9人胚胎干细胞在多次连续传代中每次传代增长(20.5±2.0)倍[58]。然而,这些蛋白质可能会有批次间变异,除此之外,Lam等[59]发现常需要预先涂布阳离子聚-1-赖氨酸(PLL)之后涂覆细胞外基质蛋白(特别是层粘连蛋白111)才能确保细胞生长,在多次传代中实现类似Matrigel的细胞倍增。但是后来他们发现,由于人层粘连蛋白52的高亲和力,它可以以单层形式作为聚苯乙烯的涂层,实现高效的细胞黏附、扩增,而无需额外的正电荷[60]。随着研究进展,已经开发了合成聚合物和多肽修饰人工合成表面,避免了与重组底物生产相关的批次问题和成本过高的缺点,支持二维平面条件下人多能干细胞的扩增[9-10,61]。Badenes等[30, 62]开发的合成表面SynthemaxⅡ微载体(Corning)已经成功转化为新型商用微载体,而不需要表面涂层。他们使用合成的肽-丙烯酸酯表面微载体在无血清培养基中长期支持人诱导多能干细胞自我更新,并且在15 d内累计达到241倍扩增,获得3.3×108个细胞,而不需要额外的表面涂层[63]。现将常用微载体总结在表1中。"
| [1] Davidson KC, Mason EA, Pera MF. The pluripotent state in mouse and human. Development. 2015;142(18):3090-3099.[2] Takahashi K, Tanabe K, Ohnuki M,et al. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell. 2007;131(5):861-872.[3] Xu C, Inokuma MS, Denham J, et al. Feeder-free growth of undifferentiated human embryonic stem cells. Nat Biotechnol. 2001;19(10):971-874.[4] Prowse AB, Doran MR, Cooper-White JJ, et al. Long term culture of human embryonic stem cells on recombinant vitronectin in ascorbate free media. Biomaterials. 2010;31(32): 8281-8288.[5] Li L, Wang S, Jezierski A, et al. A unique interplay between Rap1 and E-cadherin in the endocytic pathway regulates self-renewal of human embryonic stem cells. Stem Cells. 2010;28(2):247-257.[6] Miyazaki T, Futaki S, Suemori H, et al. Laminin E8 fragments support efficient adhesion and expansion of dissociated human pluripotent stem cells. Nat Commun. 2012;3:1236.[7] Rodin S, Antonsson L, Niaudet C, et al. Clonal culturing of human embryonic stem cells on laminin-521/E-cadherin matrix in defined and xeno-free environment. Nat Commun. 2014;5:3195.[8] Shibata S, Hayashi R, Okubo T, et al. Selective Laminin-Directed Differentiation of Human Induced Pluripotent Stem Cells into Distinct Ocular Lineages. Cell Rep. 2018;25(6):1668-1679.[9] Klim JR, Li L, Wrighton PJ, et al. A defined glycosaminoglycan- binding substratum for human pluripotent stem cells. Nat Methods. 2010;7(12):989-994.[10] Melkoumian Z, Weber JL, Weber DM, et al. Synthetic peptide-acrylate surfaces for long-term self-renewal and cardiomyocyte differentiation of human embryonic stem cells. Nat Biotechnol. 2010;28(6):606-610.[11] Brafman DA, Chang CW, Fernandez A, et al. Long-term human pluripotent stem cell self-renewal on synthetic polymer surfaces. Biomaterials. 2010;31(34):9135-9144.[12] Lock LT, Tzanakakis ES. Stem/Progenitor cell sources of insulin-producing cells for the treatment of diabetes. Tissue Eng. 2007;13(7):1399-1412.[13] Chen AK, Chen X, Choo AB, et al. Critical microcarrier properties affecting the expansion of undifferentiated human embryonic stem cells. Stem Cell Res. 2011;7(2):97-111.[14] Sart S, Schneider YJ, Li Y, et al. Stem cell bioprocess engineering towards cGMP production and clinical applications. Cytotechnology. 2014;66(5):709-722.[15] Gattazzo F, Urciuolo A, Bonaldo P. Extracellular matrix: a dynamic microenvironment for stem cell niche. Biochim Biophys Acta. 2014;1840(8):2506-2519.[16] Chang CW, Hwang Y, Brafman D, et al. Engineering cell-material interfaces for long-term expansion of human pluripotent stem cells. Biomaterials. 2013;34(4):912-921.[17] Oh SK, Chen AK, Mok Y, et al. Long-term microcarrier suspension cultures of human embryonic stem cells. Stem Cell Res. 2009;2(3):219-230.[18] Storm MP, Orchard CB, Bone HK, et al. Three-dimensional culture systems for the expansion of pluripotent embryonic stem cells. Biotechnol Bioeng. 2010;107(4):683-695.[19] Rodrigues CA, Fernandes TG, Diogo MM, et al. Stem cell cultivation in bioreactors.Biotechnol Adv. 2011;29(6):815-829.[20] Jenkins MJ, Farid SS. Human pluripotent stem cell-derived products: advances towards robust, scalable and cost-effective manufacturing strategies. Biotechnol J. 2015;10(1):83-95.[21] Bardy J, Chen AK, Lim YM, et al. Microcarrier suspension cultures for high-density expansion and differentiation of human pluripotent stem cells to neural progenitor cells. Tissue Eng Part C Methods. 2013;19(2):166-180.[22] Hundt B, Best C, Schlawin N, et al. Establishment of a mink enteritis vaccine production process in stirred-tank reactor and Wave Bioreactor microcarrier culture in 1-10 L scale. Vaccine. 2007;25(20):3987-3995.[23] Liu N, Li Y, Yang ST. Expansion of embryonic stem cells in suspension and fibrous bed bioreactors. J Biotechnol. 2014; 178:54-64.[24] Zweigerdt R, Olmer R, Singh H, et al. Scalable expansion of human pluripotent stem cells in suspension culture. Nat Protoc. 2011;6(5):689-700.[25] Chen KG, Mallon BS, McKay RD, et al. Human pluripotent stem cell culture: considerations for maintenance, expansion, and therapeutics. Cell Stem Cell. 2014;14(1):13-26.[26] Gerecht-Nir S, Cohen S, Itskovitz-Eldor J. Bioreactor cultivation enhances the efficiency of human embryoid body (hEB) formation and differentiation. Biotechnol Bioeng. 2004; 86(5):493-502.[27] Amit M, Chebath J, Margulets V, et al. Suspension culture of undifferentiated human embryonic and induced pluripotent stem cells. Stem Cell Rev. 2010;6(2):248-259.[28] Singh H, Mok P, Balakrishnan T, et al. Up-scaling single cell-inoculated suspension culture of human embryonic stem cells. Stem Cell Res. 2010;4(3):165-179.[29] Kempf H, Olmer R, Kropp C, et al. Controlling expansion and cardiomyogenic differentiation of human pluripotent stem cells in scalable suspension culture. Stem Cell Reports. 2014;3(6): 1132-1146.[30] Silva MM, Rodrigues AF, Correia C, et al. Robust Expansion of Human Pluripotent Stem Cells: Integration of Bioprocess Design With Transcriptomic and Metabolomic Characterization. Stem Cells Transl Med. 2015;4(7):731-742.[31] Serra M, Brito C, Sousa MF, et al. Improving expansion of pluripotent human embryonic stem cells in perfused bioreactors through oxygen control. J Biotechnol. 2010;148(4):208-215.[32] Kinney MA, Sargent CY, McDevitt TC. The multiparametric effects of hydrodynamic environments on stem cell culture. Tissue Eng Part B Rev. 2011;17(4):249-262.[33] Liu N, Zang R, Yang ST, et al. Stem cell engineering in bioreactors for large-scale bioprocessing. Engineering in Life Sciences. 2014; 14(1): 4-15.[34] Olmer R, Lange A, Selzer S, et al. Suspension culture of human pluripotent stem cells in controlled, stirred bioreactors. Tissue Eng Part C Methods. 2012;18(10):772-784.[35] Teo A, Mantalaris A, Lim M, et al. Influence of culture pH on proliferation and cardiac differentiation of murine embryonic stem cells. Biochemical Engineering Journal. 2014; 90(15): 8-15.[36] Shafa M, Krawetz R, Zhang Y, et al. Impact of stirred suspension bioreactor culture on the differentiation of murine embryonic stem cells into cardiomyocytes. BMC Cell Biol. 2011;12:53.[37] Gareau T, Lara GG, Shepherd RD, et al. Shear stress influences the pluripotency of murine embryonic stem cells in stirred suspension bioreactors. J Tissue Eng Regen Med. 2014;8(4):268-278.[38] Leung HW, Chen A, Choo AB, et al. Agitation can induce differentiation of human pluripotent stem cells in microcarrier cultures. Tissue Eng Part C Methods. 2011; 17(2):165-172.[39] Wan X, Ball S, Willenbrock F, et al. Perfused Three-dimensional Organotypic Culture of Human Cancer Cells for Therapeutic Evaluation. Sci Rep. 2017;7(1):9408.[40] Côme J, Nissan X, Aubry L, et al. Improvement of culture conditions of human embryoid bodies using a controlled perfused and dialyzed bioreactor system. Tissue Eng Part C Methods. 2008;14(4):289-298.[41] Kurosawa H. Methods for inducing embryoid body formation: in vitro differentiation system of embryonic stem cells. J Biosci Bioeng. 2007;103(5):389-398.[42] Carpenedo RL, Sargent CY, McDevitt TC. Rotary suspension culture enhances the efficiency, yield, and homogeneity of embryoid body differentiation. Stem Cells. 2007;25(9): 2224-2234.[43] Watanabe K, Ueno M, Kamiya D, et al. A ROCK inhibitor permits survival of dissociated human embryonic stem cells. Nat Biotechnol. 2007;25(6):681-686.[44] Choi YY, Chung BG, Lee DH, et al. Controlled-size embryoid body formation in concave microwell arrays. Biomaterials. 2010;31(15):4296-4303.[45] Bauwens CL, Toms D, Ungrin M. Aggregate Size Optimization in Microwells for Suspension-based Cardiac Differentiation of Human Pluripotent Stem Cells. J Vis Exp. 2016;(115):e54308.[46] Abbasalizadeh S, Larijani MR, Samadian A, et al. Bioprocess development for mass production of size-controlled human pluripotent stem cell aggregates in stirred suspension bioreactor. Tissue Eng Part C Methods. 2012;18(11):831-851.[47] Sargent CY, Berguig GY, Kinney MA, et al. Hydrodynamic modulation of embryonic stem cell differentiation by rotary orbital suspension culture. Biotechnol Bioeng. 2010;105(3): 611-626.[48] Lock LT, Tzanakakis ES. Expansion and differentiation of human embryonic stem cells to endoderm progeny in a microcarrier stirred-suspension culture. Tissue Eng Part A. 2009;15(8):2051-2063.[49] Xu RH, Peck RM, Li DS, et al. Basic FGF and suppression of BMP signaling sustain undifferentiated proliferation of human ES cells. Nat Methods. 2005;2(3):185-190.[50] Lee MO, Jeon H, Son MY, et al. Clump-passaging-based efficient 3D culture of human pluripotent stem cells under chemically defined conditions. Biochem Biophys Res Commun. 2017;493(1):723-730.[51] Otsuji TG, Bin J, Yoshimura A, et al. A 3D sphere culture system containing functional polymers for large-scale human pluripotent stem cell production. Stem Cell Reports. 2014;2(5): 734-745.[52] Nath SC, Tokura T, Kim MH, et al. Botulinum hemagglutinin-mediated in situ break-up of human induced pluripotent stem cell aggregates for high-density suspension culture. Biotechnol Bioeng. 2018;115(4):910-920.[53] Chen X, Prowse AB, Jia Z, et al. Thermoresponsive worms for expansion and release of human embryonic stem cells. Biomacromolecules. 2014;15(3):844-855.[54] Badenes SM, Fernandes TG, Cordeiro CS, et al. Defined Essential 8™ Medium and Vitronectin Efficiently Support Scalable Xeno-Free Expansion of Human Induced Pluripotent Stem Cells in Stirred Microcarrier Culture Systems. PLoS One. 2016;11(3):e0151264.[55] Fernandes AM, Marinho PA, Sartore RC, et al. Successful scale-up of human embryonic stem cell production in a stirred microcarrier culture system. Braz J Med Biol Res. 2009;42(6): 515-522.[56] Heng BC, Li J, Chen AK, et al. Translating human embryonic stem cells from 2-dimensional to 3-dimensional cultures in a defined medium on laminin- and vitronectin-coated surfaces. Stem Cells Dev. 2012;21(10):1701-1715.[57] Fan Y, Hsiung M, Cheng C, et al. Facile engineering of xeno-free microcarriers for the scalable cultivation of human pluripotent stem cells in stirred suspension. Tissue Eng Part A. 2014;20(3-4):588-599.[58] Fan Y, Zhang F, Tzanakakis ES, et al. Engineering Xeno-Free Microcarriers with Recombinant Vitronectin, Albumin and UV Irradiation for Human Pluripotent Stem Cell Bioprocessing. ACS Biomater Sci Eng. 2017;3(8):1510-1518.[59] Lam AT, Li J, Chen AK, et al. Cationic surface charge combined with either vitronectin or laminin dictates the evolution of human embryonic stem cells/microcarrier aggregates and cell growth in agitated cultures. Stem Cells Dev. 2014;23(14):1688-1703.[60] Lam AT, Li J, Chen AK, et al. Improved Human Pluripotent Stem Cell Attachment and Spreading on Xeno-Free Laminin-521-Coated Microcarriers Results in Efficient Growth in Agitated Cultures. Biores Open Access. 2015;4(1):242-257.[61] Villa-Diaz LG, Nandivada H, Ding J, et al. Synthetic polymer coatings for long-term growth of human embryonic stem cells. Nat Biotechnol. 2010;28(6):581-583.[62] Badenes SM, Fernandes TG, Rodrigues CA, et al. Scalable expansion of human-induced pluripotent stem cells in xeno-free microcarriers. Methods Mol Biol. 2015;1283:23-29.[63] Badenes SM, Fernandes TG, Miranda CC, et al. Long-term expansion of human induced pluripotent stem cells in amicrocarrier-based dynamic system. Journal of Chemical Technology and Biotechnology. 2017; 92(3): 482-493.[64] Chen AK, Reuveny S, Oh SK. Application of human mesenchymal and pluripotent stem cell microcarrier cultures in cellular therapy: achievements and future direction. Biotechnol Adv. 2013;31(7):1032-1046.[65] Pasparakis G, Bouropoulos N. Swelling studies and in vitro release of verapamil from calcium alginate and calcium alginate-chitosan beads. Int J Pharm. 2006;323(1-2):34-42.[66] Serra M, Correia C, Malpique R, et al. Microencapsulation technology: a powerful tool for integrating expansion and cryopreservation of human embryonic stem cells. PLoS One. 2011;6(8):e23212.[67] Siti-Ismail N, Bishop AE, Polak JM, et al. The benefit of human embryonic stem cell encapsulation for prolonged feeder-free maintenance. Biomaterials. 2008;29(29): 3946-3952.[68] Wilson JL, McDevitt TC. Stem cell microencapsulation for phenotypic control, bioprocessing, and transplantation. Biotechnol Bioeng. 2013;110(3):667-682.[69] Wilson JL, Najia MA, Saeed R, et al. Alginate encapsulation parameters influence the differentiation of microencapsulated embryonic stem cell aggregates. Biotechnol Bioeng. 2014; 111(3):618-631.[70] Jing D, Parikh A, Tzanakakis ES. Cardiac cell generation from encapsulated embryonic stem cells in static and scalable culture systems. Cell Transplant. 2010;19(11):1397-1412.[71] Ikeda K, Nagata S, Okitsu T, et al. Cell fiber-based three-dimensional culture system for highly efficient expansion of human induced pluripotent stem cells. Sci Rep. 2017;7(1):2850.[72] Dang SM, Gerecht-Nir S, Chen J, et al. Controlled, scalable embryonic stem cell differentiation culture. Stem Cells. 2004; 22(3):275-282.[73] Gerecht S, Burdick JA, Ferreira LS, et al. Hyaluronic acid hydrogel for controlled self-renewal and differentiation of human embryonic stem cells. Proc Natl Acad Sci U S A. 2007;104(27):11298-11303.[74] Li Q, Wang Q, Wang O, et al. A simple and scalable hydrogel-based system for culturing protein-producing cells. PLoS One. 2018;13(1):e0190364.[75] Lei Y, Jeong D, Xiao J, et al. Developing Defined and Scalable 3D Culture Systems for Culturing Human Pluripotent Stem Cells at High Densities. Cell Mol Bioeng. 2014;7(2): 172-183.[76] Lin H, Li Q, Lei Y. Three-dimensional tissues using human pluripotent stem cell spheroids as biofabrication building blocks. Biofabrication. 2017;9(2):025007.[77] Lei Y, Schaffer DV. A fully defined and scalable 3D culture system for human pluripotent stem cell expansion and differentiation. Proc Natl Acad Sci U S A. 2013;110(52): E5039-5048. |
| [1] | Li Shao, Liang Yongkang, Gao Yi, Peng Qing. Establishment and functional in vitro characteristics of three-dimensional collagen HepaRG microsphere [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(16): 2541-2547. |
| [2] | Han Ningning, Zuo Jinfu, Sun Miao, Tang Shengjian, Liu Fangjun. Application and progress of umbilical cord mesenchymal stem cells in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(13): 2079-2086. |
| [3] | Zhu Mingqi, Zhou Jingxu, Lin Lizhu, Chen Zeren, Liao Zhixiao . In vitro co-culture of decellularized matrix of porcine colon with colon cancer HCT116 cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(10): 1533-1538. |
| [4] | Zhao Chuntao, Qing Mingsong, Yu Langbo, Peng Jiachen . Meta-analysis of total knee arthroplasty guided by kinematic alignment and mechanical alignment [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(9): 1435-1442. |
| [5] |
Zhang Cong, Zhao Yan, Du Xiaoyu, Du Xinrui, Pang Tingjuan, Fu Yining, Zhang Hao, Zhang Buzhou, Li Xiaohe, Wang Lidong.
Biomechanical analysis of the lumbar spine and pelvis in adolescent
idiopathic scoliosis with lumbar major curve |
| [6] |
Cen Yanhui, Xia Meng, Jia Wei, Luo Weisheng, Lin Jiang, Chen Songlin, Chen Wei, Liu Peng, Li Mingxing, Li Jingyun, Li Manli, Ai Dingding, Jiang Yunxia.
Baicalein inhibits the biological behavior of hepatocellular
carcinoma stem cells by downregulation of Decoy receptor 3 expression |
| [7] | He Yujie, Wang Haiyan, Li Zhijun, Li Xiaohe, Cai Yongqiang, Dai Lina, Xu Yangyang, Wang Yidan, Xu Xuebin. Digital measurements of the anatomical parameters of pedicle-rib unit screw fixation in thoracic vertebrae of preschoolers [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(6): 869-876. |
| [8] | Yan Shu, Lu Yan, Ouyang Zhaolian. Analysis of programs on tissue engineering funded by the National Natural Science Foundation of China between 2013 and 2018 [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(5): 731-735. |
| [9] | Yu Xingge, Lin Kaili. Application of nanocomposite hydrogels in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(34): 5441-5446. |
| [10] | Gu Yubo, Zhang Dongliang, Yuan Wei, , Song Lujie. Exosomes from adipose-derived stem cells can promote cavernous nerve regeneration in rats [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(31): 4979-4985. |
| [11] | Sun Jian, Fang Chao, Gao Fei, Wei Laifu, Qian Jun. Clinical efficacy and complications of short versus long segments of internal fixation for the treatment of degenerative scoliosis: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(3): 438-445. |
| [12] | Gao Yangyang, Che Xianda, Han Pengfei, Liang Bin, Li Pengcui. Accuracy of robot-assisted and fluoroscopy-guided pedicle screw placement: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(3): 446-452. |
| [13] | Zhang Jian, Wang Xiaojian, Qin Dean, Zhao Zhongtao, Liang Qingyuan, An Qijun, Song Jiefu. Risk factors for proximal junctional kyphosis after spinal deformity surgery: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(3): 460-468. |
| [14] |
Yang Jiayi, Zhang Guanghao, Zhang Cheng, Li Ke, Huo Xiaolin, Wu Changzhe.
Stem cell expansion and hepatic differentiation: measurement of related indicators and detection methods [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(25): 4039-4045. |
| [15] | Wang Qi, Yang Xiaoshuang, Wang Dali. Effects of hydrogel three-dimensional culture on the characteristics of human amniotic mesenchymal stem cells and paracrine effect [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(22): 3460-3466. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||