Chinese Journal of Tissue Engineering Research ›› 2019, Vol. 23 ›› Issue (20): 3229-3234.doi: 10.3969/j.issn.2095-4344.1238
Previous Articles Next Articles
Application of platelet-rich plasma in repair of Achilles tendon injury
Zhao Maosheng1, 2, Feng Guochao1, 2, Deng Yinshuan2, Zhen Ping2
- 1Gansu University of Chinese Medicine, Lanzhou 730000, Gansu Province, China; 2Lanzhou General Hospital of Lanzhou Military Region of Chinese PLA, Lanzhou 730050, Gansu Province, China
-
Online:2019-07-18Published:2019-07-18 -
Contact:Zhen Ping, Chief physician, Professor, Lanzhou General Hospital of Lanzhou Military Region of Chinese PLA, Lanzhou 730050, Gansu Province, China -
About author:Zhao Maosheng, Master candidate, Gansu University of Chinese Medicine, Lanzhou 730000, Gansu Province, China; Lanzhou General Hospital of Lanzhou Military Region of Chinese PLA, Lanzhou 730050, Gansu Province, China -
Supported by:the National Natural Science Foundation of China, No. 81371983 (to ZP)| the Military Science Research Program, No. CWH17J007 (to DYS)
CLC Number:
Cite this article
Zhao Maosheng, Feng Guochao, Deng Yinshuan, Zhen Ping. Application of platelet-rich plasma in repair of Achilles tendon injury[J]. Chinese Journal of Tissue Engineering Research, 2019, 23(20): 3229-3234.
share this article
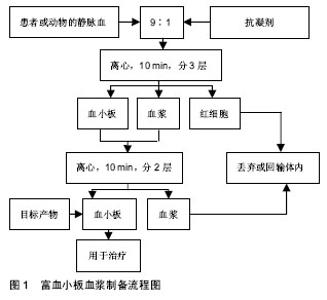
2.1 富血小板血浆的研究现状 富血小板血浆是一种含高体积分数血小板的血液制品,有液体和凝胶两种形式。有研究发现血小板能够促进血管生成、胶原合成,并慢慢开始在临床上使用,治疗各种原因导致的血小板减少和出血性疾病。1977年,Harke等[5]首次成功地将富血小板血浆应用于心脏外科手术,降低了血小板在体外循环过程中的受损率并且减少了术后失血量。大量的实验表明,血小板除了在止血和血栓形成过程中发挥作用外,血小板被激活后会释放大量的生长因子,参与损伤组织的再生修复,在组织再生、创面修复、感染治疗和功能重建等领域都有广泛的应用[6-7]。 富血小板血浆的制备:由于全血中不同的组分沉降系数各异,全血在离心机的作用下分层,从而制备出富血小板血浆。正常人全血中血小板的浓度为(1-3)× 108 L-1,经离心浓缩后的富血小板血浆中血小板浓度可为全血的3-6倍[8]。目前临床上使用的各种富血小板血浆产品的制备方法及组分存在差别,国际上尚无标准化的提取方法。根据其内含的组分的不同理论上可以将其分为4个亚型,分别是纯富血小板血浆、含白细胞的富血小板血浆、富血小板纤维基质和含白细胞的富血小板纤维基质[9]。多数研究者选择前2种,在临床上最常用的是纯富血小板血浆。制备的方法可分为手工制备法和设备制备法,由于离心次数的差异,还可以将手工制备分为一次、二次和三次离心法[10],其中二次离心法是最常用的制备富血小板血浆的方法,见图1。"
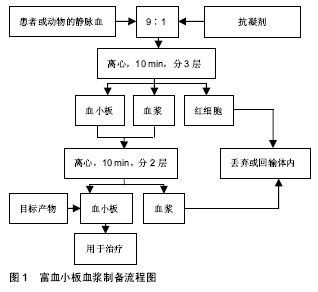
首先在无菌条件下采静脉血置于离心管中,加入抗凝剂枸橼酸钠[11],全血与抗凝剂的比例约为9∶1。然后开始准备离心,第一阶段在相对低速条件下,离心 10 min后离心管中分为3层,上层为贫血小板血浆,下层为红细胞,上、下两层交界的浅淡黄色层,即富血小板血浆层。然后抽取下层红细胞,混匀后开始第二阶段离心。第二阶段在相对高速条件下,离心10 min后离心管中分为两层,上层为贫血小板血浆,下层吸取混匀后即为富血小板血浆。但是否进行第二阶段的离心,目前还存在争议。 富血小板血浆的作用机制:富血小板血浆中除了含有大量的血小板之外,还含有白细胞、少量红细胞以及纤维蛋白[12-13]。血小板由骨髓中成熟的巨核细胞脱落而来,具有生物活性,内含有多种被膜颗粒。富血小板血浆中的血小板被凝血酶、胶原等激活剂激活后,血小板中的α-颗粒通过脱颗粒作用能够释放多种信号蛋白,这些信号蛋白与相应细胞的膜受体结合后,通过细胞内、外的信号转导通路来参与细胞增殖、基质形成、骨形成及胶原蛋白的合成等过程,进而促进组织的再生修 复[14-15]。这种调节机制在骨、软骨、韧带及肌腱等组织修复愈合和再生过程中起着重要的作用。 血小板在富血小板血浆中不仅含量最多,而且也是发挥主要功能的成分。富血小板血浆中的血小板被激活后释放大量的信号转导蛋白,包括血小板源性生长因子、转化生长因子β、血管内皮生长因子、表皮生长因子、类胰岛素生长因子及成纤维细胞生长因子等[16]。这些生长因子在组织损伤修复过程中发挥的作用不尽相同。 血小板源性生长因子是组织愈合初期发挥作用的关键生长因子,其的主要效应细胞是干细胞系,能够促使干细胞向肌腱细胞分化,有利于组织塑形[17];转化生长因子β在人体中存在3种亚型(转化生长因子β1,β2,β3),其中转化生长因子β1能够调节Ⅰ,Ⅲ型胶原的合成及分泌,在组织的再生修复中发挥重要作用[18];血管内皮生长因子是胱氨酸生长因子,能够直接调控血管内皮细胞的迁移、增殖、分化和存活,与成纤维细胞生长因子共同作用刺激血管等重要结构在修复早期形成[19];成纤维细胞生长因子还具有促进成纤维细胞的增殖、迁移以及分泌胶原蛋白的功能;类胰岛素生长因子表达于肌腱修复的各个阶段,特别是在炎症阶段能够促进腱样细胞的迁徙和增殖,促进细胞外基质的分泌[20]。研究发现,这几种生长因子的表达的峰值表现在组织修复的不同时期,有必要研究这些生长因子之间的相互作用,从而更好地明确富血小板血浆在组织愈合修复中所发挥的作用。 由于白细胞和血小板的沉降速度接近,因此富血小板血浆中会含有一定量的白细胞。白细胞在机体防御中发挥着重要的作用,白细胞可以帮助清除局部坏死组织,从而加快组织的损伤修复速度。含白细胞的富血小板血浆具有抗菌作用,很可能与含白细胞的富血小板血浆中含有大量的白细胞有关。但有研究表明,含有高浓度白细胞的富血小板血浆增加了促炎因子、分解代谢的介质释放,并影响胶原合成,不利于肌腱病和退行性关节病变的治疗,反而延长了修复的时间,甚至会增加患者的局部疼痛[21-23]。Dovi等[24]在中性粒细胞清除的小鼠研究中,发现中性粒细胞过多或在局部停留过久对无菌性伤口的愈合不利。兔的跟腱实验表明,局部注射含白细胞的富血小板血浆注射含白细胞的富血小板血浆会造成更严重的炎症反应[25]。因此,白细胞的存在一直是富血小板血浆疗法争议所在,众多研究学者较一致的观点是:组织损伤合并感染,建议使用含白细胞的富血小板血浆;针对肌腱病等退行性疾病未合并感染的,建议使用纯富血小板血浆。 富血小板血浆中含有少量的可溶性的纤维蛋白原。纤维蛋白原被裂解转变为不可溶的纤维蛋白单体后,会形成一种立体的网状结构。这种结构与人类天然纤维蛋白的结构十分相似,能够为细胞附着、增殖和分化提供理想的场所,有利于组织结构的重建[26-27]。另外,纤维蛋白结合血小板释放的生长因子,随着蛋白的降解缓慢释放,延长作用时间[28]。富血小板血浆中还包含有纤维连接蛋白、黏连蛋白及凝血酶致敏蛋白等[14],它们主要是发挥细胞连接作用,影响细胞的迁徙。 2.2 富血小板血浆在跟腱病治疗中的应用 肌腱本身缺乏血供,基础代谢率低,受伤后再生和修复能力极差。肌腱损伤后愈合需要经过炎症期、增殖期以及修复或重塑期3个时期[2],这3个时期人体内各种生长因子对肌腱的修复均起到调节作用,而被激活后的富血小板血浆能够在病变部位持续释放大量的生长因子,加快了损伤肌腱的愈合与修复[29],从而达到修复和重建损伤跟腱的目的,见表1。"
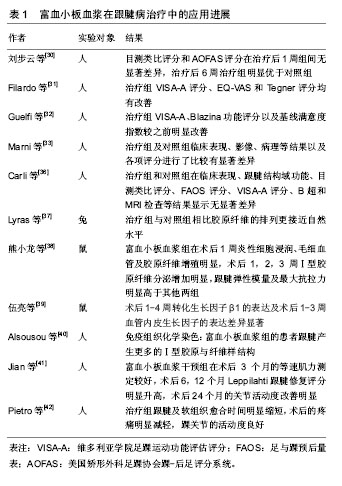

2.2.1 跟腱炎 跟腱炎属于非感染性肌腱炎性疾病,即无菌性炎症。现临床上常用的治疗方法为局部注射小剂量糖皮质激素封闭治疗,但有研究表明,局部使用激素短期内疗效较好,但从长远来看不利于跟腱的愈合,能够抑制胶原纤维的增生和细胞间质的合成,使之更容易断裂[30]。相比之下,富血小板血浆能构造良好的纤维骨架促进胶原纤维的增生和细胞增殖、分化,被众多研究者所推崇。 Filardo等[31]一项前瞻性的临床研究中,共纳入了27例患者(34个跟腱),患者接受3次超声引导下腱内注射富血小板血浆(每次间隔2周),对患者进行了最多平均54.1个月(最少30个月)的随访观察,发现患者的维多利亚学院足踝运动功能评估评分(Victorian Institute of Sport Assessment Score-Achillea,VISA-A)显著改善,并且EQ目测类比和Tegner评分也显示出了积极地趋势,因此认为富血小板血浆在治疗跟腱病上总体中期疗效较好。Guelfi等[32]在临床试验中,对73例患者(83个跟腱)进行了单次富血小板血浆注射治疗后进行了平均为50.1个月的随访观察,除了7例进行了第2次注射外,并没有跟腱断裂的发生VISA-A及Blazina功能评分以及基线满意度指数较之前明显改善,因此认为单次注射富血小板血浆对于慢性跟腱病的长期疗效较好,能够降低不良反应的发生率,是治疗慢性跟腱病的一个不错的替代方案。Marni等[33]一项随机对照研究中,收纳了178例慢性肌腱病患者(其中跟腱病占25%)分为了富血小板血浆组和对照组,对临床表现、影像及病理等结果以及各项评分进行了比较分析(其中65例患者因数据缺失而被排除结果),结果表明富血小板血浆组和对照组有显著差异。刘步云等[30]一项临床随机对照试验中,收治了36例患者,随机分为了富血小板血浆治疗组和激素对照组,分别对治疗前和治疗后1,6周进行了目测类比评分和美国矫形外科足踝协会踝-后足评分系统(American orthopedic foot and ankle society Ankle Hindfoot Scale,AOFAS)系统,治疗前两组各项评分无显著差异,治疗后1周较治疗前目测类比评分和AOFAS评分均具有显著性意义,但组间无显著差异,治疗后6周组间比较治疗组明显优于对照组,因此认为富血小板血浆联合运动疗法较激素有明显的远期疗效。但也有一些研究表明富血小板血浆在治疗肌腱病上治疗效果不明显[34-35],同时有人提出异议,认为其方法存在问题。 2.2.2 跟腱断裂 跟腱断裂是常见的肌腱断裂性损伤之一。跟腱属于人体腱纤维最厚、机械性能最强的肌腱,自起点至止点逐渐变窄变厚,距跟骨结节上2-6 cm处最窄,且此处的血供最差,因此最易发生断裂。可分为急性断裂和慢性断裂。急性断裂主要是由强烈的外力造成,如剧烈运动时,跟腱受到突然扭转的作用力而发生断裂;慢性的断裂主要是跟腱组织的变性,胶原纤维减少,使跟腱变脆,最终发生断裂。临床上跟腱断裂后常常是手术治疗,但随之伴随的是各种不良反应,如瘢痕愈合、术后恢复慢、术后难护理、极易发生感染和组织粘连等。Carli等[36]一项临床前瞻系统研究中,纳入了30例经微创手术修复断裂跟腱的患者,随机分为了富血小板血浆组和对照组,富血小板血浆组分别于术中和术后14 d腱内注射富血小板血浆,对两组患者分别进行了长达24个月的随访和体检,两组在临床表现、跟腱结构域功能、目测类比评分、FAOS:足与踝预后量表(The Foot and Ankle Outcome Score,FAOS)、VISA-A评分、B超和MRI检查等结果显示无差异,从而得到富血小板血浆在跟腱断裂手术治疗中未起到良好促进作用的结论。 Lyras等[37]在兔跟腱断裂的随机对照实验中,在肌腱愈合的炎症期和增殖期富血小板血浆组血管形成明显增多,与对照组相比胶原纤维的排列更接近自然水平,第四周富血小板血浆组血管形成明显减少,因此认为富血小板血浆能够缩短损伤跟腱的愈合时间,且胶原纤维的排列更接近原组织水平。有熊小龙以及伍亮等的大鼠跟腱断裂实验认为跟腱断裂的早期在跟腱周围注射富血小板血浆能够促进跟腱的愈合修复[38-39],但中、远期的疗效观察有待进一步的研究。Alsousou等[40]临床研究中,免疫组织化学结果显示,富血小板血浆组较对照组能够促使患者跟腱产生更多的Ⅰ型胶原与纤维样结构,能够促进人体跟腱早期的愈合,缩短成熟时间。Jian 等[41]在一项临床前瞻性随机对照实验中,对急性跟腱断裂改良Krackow缝合后,使用了富血小板血浆干预,分别在3,6,12,24个月对患者评估等速肌力、踝关节活动度、小腿腿围及Leppilahti跟腱修复评分,术后3个月富血小板血浆干预组的等速肌力测定较对照组好,术后6,12个月富血小板血浆干预组Leppilahti跟腱修复评分较对照组明显升高,术后24个月富血小板血浆干预组的关节活动度改善明显,表明富血小板血浆能够促进断裂跟腱早期的修复,并可改善患者踝关节短、中期的功能。Pietro等[42]一项富血小板血浆联合透明质酸治疗跟腱断裂修补术后的临床试验中,选择了10例跟腱术后发生不良反应的患者,在富血小板血浆联合透明质酸治疗后,跟腱及软组织愈合时间明显缩短,术后的疼痛明显减轻,踝关节的活动度达到了令人满意的程度,说明富血小板血浆联合透明质酸治疗有效。"

| [1] Wang J,周义钦.肌腱干细胞研究新进展[J].国际骨科学杂志,2015, 36(3):163-167.[2] Wu F, Nerlich M, Docheva D. Tendon injuries: basic science and new repair proposals. EFORT Open Rev. 2017;2(7):332-342.[3] Rodeo SA, Nguyen JT, Cavanaugh JT, et al. Clinical and Ultrasonographic Evaluations of the Shoulders of Elite Swimmers, Am J Sports Med. 2016;44(12):3214-3221.[4] Buda R, Castagnini F, Pagliazzi G, et al. Treatment algorithm for chronic achilles tendon lesions review of the literature and proposal of a new classification. J Am Podiatr Med Assoc. 2017; 107(2):144-149.[5] Harke H, Tanger D, Furst-Denzer S, et al. Effect of a preoperative separation of platelets on the postoperative blood loss subsequent to extracorporeal circulation in open heart surgery. Anaesthesist. 1977;26(2):64-71.[6] 潘红娟,王雅丽,吕爽,等.富血小板血浆制备方法稳定性的研究[J].中国实验诊断学,2016,20(6):1008-1010.[7] Dhillon M, Patel S, Shetty V, et al. Orthobiologics and platelet rich plasma, Indian J Orthop. 2014;48(1):1-9.[8] Ayatollahi A, Hosseini H, Shahdi M, et al. Platelet-rich plasma by single spin process in male pattern androgenetic alopecia: is it an effective treatment? Indian Dermatol Online J. 2017;8(6):460-464.[9] Ehrenfest DMD, Andia I, Zumstein MA, et al. Classification of platelet concentrates (Platelet-Rich Plasma-PRP, Platelet-Rich Fibrin-PRF) for topical and infiltrative use in orthopedic and sports medicine: current consensus, clinical implications and perspectives. Muscles Ligaments Tendons J. 2014;4(1):3-9.[10] 福嘉欣,王树森.自体富血小板血浆治疗骨关节与运动损伤疾病的机制与应用[J].中国组织工程研究,2017,21(30):4908-4914.[11] Moran J, Sun S, Khababa I, et al. A randomized trial comparing gentamicin/citrate and heparin locks for central venous catheters in maintenance hemodialysis patients. Am J Kidney Dis. 2012; 59(1):102-107.[12] Nurden AT, Nurden P, Sanchez M, et al. Platelets and wound healing. Front Biosci. 2008;13:3532-3548.[13] Drago L, Bortolin M, Vassena C, et al. Plasma components and platelet activation are essential for the antimicrobial properties of autologous platelet-rich plasma: an in vitro study. PLoS One. 2014; 9(9):e107813.[14] 吕汝举,刘美静.富血小板血浆(富血小板血浆)治疗技术及应用[J].中国医疗器械信息,2013,19(10):50-55.[15] Brass L. Understanding and evaluating platelet function. Hematology Am Soc Hematol Educ Program. 2010;2010(1): 387-396.[16] 潘红娟.富血小板血浆制备技术的优化及其组分的检测[D].长春:吉林大学,2017.[17] Younesi M, Knapik DM, Cumsky J, et al. Effects of PDGF-BB delivery from heparinized collagen sutures on the healing of lacerated chicken flexor tendon, in vivo. Acta Biomater. 2017; 63:200-209.[18] Lichtman MK, Otero-Vinas M, Falanga V. Transforming growth factor beta (TGF-β) isoforms in wound healing and fibrosis. Wound Repair Regen, 2016;4(2):215-222.[19] Ruhrberg C. Growing and shaping the vascular tree: multiple roles for VEGF. Bioessays. 2003;5(11):1052-1060.[20] Fujioka-Kobayashi M, Miron RJ, Hernandez M, et al. Optimized platelet rich fibrin with the low speed concept: growth factor release, biocompatibility and cellular response. J Periodontol. 2016;8(1):1-17.[21] Zhou Y, Zhang J, Wu H, et al. The differential effects of leukocyte-containing and pure platelet-rich plasma (PRP) on tendon stem/progenitor cells-implications of PRP application for the clinical treatment of tendon injuries. Stem Cell Res Ther. 2015;6(1):173.[22] Young A, Mcnaught CE. The physiology of wound healing. Surgery (Oxford). 2011;9(10):475-479.[23] Boswell SG, Schnabel LV, Mohammed HO, et al. Increasing platelet concentrations in leukocyte-reduced platelet-rich plasma decrease collagen gene synthesis in tendons. Am J Sports Med. 2014;42(1):42-49.[24] Dovi JV, He LK, DiPietro LA. Accelerated wound closure in neutrophil-depleted mice. J Leukoc Biol. 2003;73(4):448-455.[25] Dragoo JL, Braun HJ, Durham JL, et al. Comparison of the acute inflammatory response of two commercial platelet-rich plasma systems in healthy rabbit tendons. Am J Sports Med. 2012; 40(6):1274-1281.[26] Dohan DM, Choukroun J, Diss A, et al. Platelet-rich fibrin (PRF): asecond-generation platelet concentrate. Part I: Technological concepts and evolution. Am J Sports Med. 2006;101(3):e37-e44.[27] Choukroun J, Diss A, Simonpieri A, et al. Platelet-rich fibrin (PRF): a second-generation platelet concentrate. Part IV: clinical effects on tissue healing. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2006;101(3):e56-e60.[28] Dohan DM, Choukroun J, Diss A, et al. Platelet-rich fibrin (PRF): a second-generation platelet concentrate. Part II: platelet-related biologic features. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2006;101(3):e45-e50.[29] Mishra A, Woodall J, Vieira A. Treatment of tendon and muscle using platelet-rich plasma. Clin Sports Med. 2009;28(1):113-125.[30] 刘步云,咸杰,孙育良,等.局部注射自体富血小板血浆联合运动疗法和局部注射曲安奈德治疗跟腱病的临床疗效对比[J].四川医学, 2017,38(5):10-13.[31] Filardo G, Kon E, Matteo BD, et al. Platelet-rich plasma injections for the treatment of refractory achilles tendinopathy: results at 4 years. Blood Transfus. 2014;12(4):1-8.[32] Guelfi M, Pantalone A, Vanni D, et al. Long-term beneficial effects of platelet-rich plasma for non-insertional Achilles tendinopathy, Foot Ankle Surg. 2015;21(3):178-181.[33] Marni W, Terry D, Heather B, et al. A pilot study evaluating the effectiveness of platelet-rich plasma therapy for treating degenerative tendinopathies: a randomized control trial with synchronous observational cohort. PLoS One. 2016;11(2): e0147842.[34] Harmon KG, Rao AL. The use of platelet-rich plasma in the nonsurgical management of sports injuries: hype or hope? Hematol Am Soc Hematol Educ Program. 2013;2013(1):620-626.[35] Vos RJD, Windt J, Weir A. Strong evidence against platelet-rich plasma injections for chronic lateral epicondylar tendinopathy: a systematic review. Br J Sports Med. 2014;48(12):952-956.[36] Carli AD, Lanzetti R, Ciompi A, et al. Can platelet-rich plasma have a role in Achilles tendon surgical repair? Knee Surg Sports Traumatol Arthrosc. 2015;24(7):2231-2237.[37] Lyras DN, Kazakos K, Verettas D, et al. The influence of platelet-rich plasma on angiogenesis during the early phase of tendon healing. Foot Ankle Int. 2009;30(11):1101-1106.[38] 熊小龙,伍亮,相大勇,等.富血小板血浆对大鼠跟腱断裂早期愈合的影响[J].中国修复重建外科杂志,2012,26(4):466-471.[39] 伍亮,熊小龙,相大勇,等.TGF-β1和VEGF在富血小板血浆治疗跟腱断裂模型中的表达及意义[J].中华临床医师杂志(电子版), 2013, 7(4):1639-1645.[40] Alsousou J, Thompson M, Harrison P, et al. Effect of platelet-rich plasma on healing tissues in acute ruptured achilles tendon: a human immunohistochemistry study. Lancet. 2015;385:S19.[41] Jian Z, Xiaolian M, Zhongmin S, et al. A prospective study of platelet-rich plasma as biological augmentation for acute achilles tendon rupture repair. Biomed Res Int. 2016;2016:1-8.[42] Pietro G, Barbara DA, Annarita A, et al. Use of platelet rich plasma and hyaluronic acid in the treatment of complications of achilles tendon reconstruction. World J Plast Surg. 2016;5(2):124-132.[43] Lei H, Gui L, Xiao R. The effect of anticoagulants on the quality and biological efficacy of platelet-rich plasma. Clin Biochem, 2009;42(13-14):1452-1460.[44] Harrison S, Vavken P, Kevy S, et al. Platelet activation by collagen provides sustained release of anabolic cytokines. T Am J Sports Med. 2011;39(4):729-734.[45] 罗涛,李放,张宁.不同抗凝剂和激活剂联合应用对富血小板血浆凝胶释放生长因子影响的比较[J].中国组织工程研究, 2012,16(16): 2893-2897. |
| [1] | Jiang Hongying, Zhu Liang, Yu Xi, Huang Jing, Xiang Xiaona, Lan Zhengyan, He Hongchen. Effect of platelet-rich plasma on pressure ulcers after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1149-1153. |
| [2] | Wu Xun, Meng Juanhong, Zhang Jianyun, Wang Liang. Concentrated growth factors in the repair of a full-thickness condylar cartilage defect in a rabbit [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1166-1171. |
| [3] | Xie Wenjia, Xia Tianjiao, Zhou Qingyun, Liu Yujia, Gu Xiaoping. Role of microglia-mediated neuronal injury in neurodegenerative diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1109-1115. |
| [4] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [5] | Li Cai, Zhao Ting, Tan Ge, Zheng Yulin, Zhang Ruonan, Wu Yan, Tang Junming. Platelet-derived growth factor-BB promotes proliferation, differentiation and migration of skeletal muscle myoblast [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1050-1055. |
| [6] | He Xiangzhong, Chen Haiyun, Liu Jun, Lü Yang, Pan Jianke, Yang Wenbin, He Jingwen, Huang Junhan. Platelet-rich plasma combined with microfracture versus microfracture in the treatment of knee cartilage lesions: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 964-969. |
| [7] | Luo Xuanxiang, Jing Li, Pan Bin, Feng Hu. Effect of mecobalamine combined with mouse nerve growth factor on nerve function recovery after cervical spondylotic myelopathy surgery [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 719-722. |
| [8] | Nie Huijuan, Huang Zhichun. The role of Hedgehog signaling pathway in transforming growth factor beta1-induced myofibroblast transdifferentiation [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 754-760. |
| [9] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [10] | Jiang Tao, Ma Lei, Li Zhiqiang, Shou Xi, Duan Mingjun, Wu Shuo, Ma Chuang, Wei Qin. Platelet-derived growth factor BB induces bone marrow mesenchymal stem cells to differentiate into vascular endothelial cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 3937-3942. |
| [11] | Hao Xiaona, Zhang Yingjie, Li Yuyun, Xu Tao. Bone marrow mesenchymal stem cells overexpressing prolyl oligopeptidase on the repair of liver fibrosis in rat models [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 3988-3993. |
| [12] | Liu Jinwei, Chen Yunzhen, Wan Chunyou. Changes of osteogenic growth factors in the broken end of bone nonunion under stress [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3619-3624. |
| [13] | Zhou Wu, Wang Binping, Wang Yawen, Cheng Yanan, Huang Xieshan. Transforming growth factor beta combined with bone morphogenetic protein-2 induces the proliferation and differentiation of mouse MC3T3-E1 cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3630-3635. |
| [14] | Xie Yang, Lü Zhiyu, Zhang Shujiang, Long Ting, Li Zuoxiao. Effects of recombinant adeno-associated virus mediated nerve growth factor gene transfection on oligodendrocyte apoptosis and myelination in experimental autoimmune encephalomyelitis mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3678-3683. |
| [15] | Bi Qingwei, Liu Chengpu, Li Yan, Zhao Wenwen, Han Mei. Structure analysis of platelet-rich fibrin derived from two centrifugation procedures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3534-3539. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||