Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (21): 3417-3423.doi: 10.3969/j.issn.2095-4344.0906
Previous Articles Next Articles
Type I collagen induction of bone marrow mesenchymal stem cells and the mechanism of osteogenesis
Xie Yu, Zhou Nuo
- Department of Oral and Maxillofacial Surgery, Stomatology Hospital of Guangxi Medical University, Nanning 530021, Guangxi Zhuang Autonomous Region, China
-
Revised:2018-05-21Online:2018-07-28Published:2018-07-28 -
Contact:Zhou Nuo, M.D., Professor, Department of Oral and Maxillofacial Surgery, Stomatology Hospital of Guangxi Medical University, Nanning 530021, Guangxi Zhuang Autonomous Region, China -
About author:Xie Yu, Master candidate, Department of Oral and Maxillofacial Surgery, Stomatology Hospital of Guangxi Medical University, Nanning 530021, Guangxi Zhuang Autonomous Region, China -
Supported by:the National Natural Science Foundation of China, No. 8167040353
CLC Number:
Cite this article
Xie Yu, Zhou Nuo. Type I collagen induction of bone marrow mesenchymal stem cells and the mechanism of osteogenesis[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(21): 3417-3423.
share this article
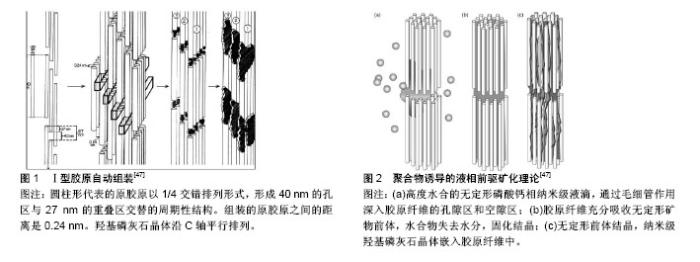
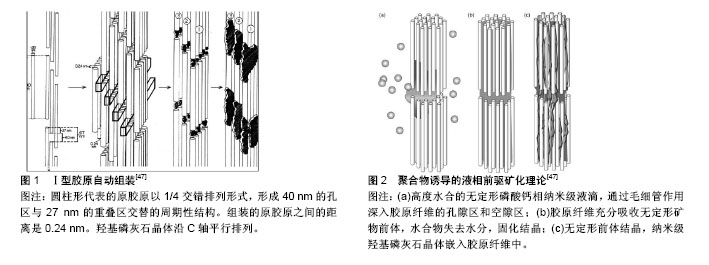
2.1 Ⅰ型胶原基因、结构及生物合成 2.1.1 Ⅰ型胶原基因、结构 Ⅰ型胶原由2条α1链及1条α2链交联构成,分别由COL1A1(17q21.3-q22)及COL1A2(7q21.3-q22)基因编码[3]。转化生长因子β1是众所周知的调节人体Ⅰ型胶原α1、α2的基因启动子,主要是通过细胞Smad信号转导通路介导[4-5]。Ⅰ型前胶原为Ⅰ型胶原的前体,为异源或同源三聚体结构,由2个proα1链和1个proα2链相互缠绕形成,它的结构是α1(Ⅰ)2α2,分别由COL1A1和COL1A2基因编码翻译而来。在结构上Ⅰ型前胶原比Ⅰ型胶原多C-前肽与N-前肽2个末端前肽。Ⅰ型胶原多肽链的一级氨基酸序列由(-Gly-X-Y-)n重复序列组成,X、Y通常为脯氨酸及羟脯氨酸。2条α1链及1条α2链通过氢键缠绕形成右手超螺旋结构的胶原前体。 2.1.2 Ⅰ型胶原的生物合成 α-多肽链在细胞核内经转录转运至粗面内质网(rER)中由核蛋白小体合成,在粗面内质网的囊腔中,经脯氨酰4-羟化酶(P4Hs)、脯氨酰3-羟化酶(P3Hs)和赖氨酰4位羟化酶(LHs)这3种酶羟基化,这3种酶需抗坏血酸(维生素C)作其辅助因子,缺乏抗坏血酸导致的坏血病其组织学及临床主要表现为骨骼及结缔组织发育不良[6]。HSP47是胶原特异性分子伴侣,对于原胶原三聚体正确构象及保持其结构的稳定性至关重要[7]。原胶原多肽链在内质网(ER)内至少受到9种分子伴侣和酶协助其折叠及三聚。构象正确的三股螺旋的原胶原分子与结合在其上的HSP47一并转移至高尔基体,之后HSP47从分子上解离下来,再次转运至内质网中[8]。在肽链的氨基末端和羧基末端各有一个非螺旋构象的结构域,这些结构称为非胶原域(NC),并从C端编号(NC1,NC2)。原胶原以前胶原特异性COPⅡ囊泡的形式排至胞外基质中,在N-蛋白酶及C-蛋白酶作用下切除C-前肽与N-前肽成为Ⅰ型胶原[9]。前肽的切割暴露了短肽序列,其是多肽链的非三重螺旋延伸端,端肽含有原纤维生成的结合位点。Ⅰ型原胶原在胞外基质中经过内切酶、羟化酶加工及交联等过程,自动组装成规律的致密胶原纤维,为后续钙盐等矿物质沉积和矿化提供场所[10]。 2.1.3 Ⅰ型胶原与骨相关疾病 Ⅰ型胶原蛋白基因的突变及代谢异常会导致相关的疾病。编码Ⅰ型胶原α1链的COL1A1或α2链的COL1A2基因突变[11-12],包括点突变、基因片段缺失及插入、剪切错误[13],引起肽链无法形成正确的构象或右旋三聚体结构异常,并进一步引起肽链修饰和转移异常,最终导致Ⅰ型胶原在结构或数量上发生异常[14]。与Ⅰ型胶原生物功能异常的疾病包括成骨不全症、软骨发育不良、Ehlers-Danlos综合征亚型、Alport综合征、Bethlem肌病、Knobloch综合征以及一些骨质疏松症、动脉瘤、骨关节病和椎间盘疾病等。 2.2 Ⅰ型胶原诱导骨髓间充质干细胞向成骨细胞分化 2.2.1 骨髓间充质干细胞 骨髓间充质干细胞主要存在于骨髓,四肢骨中的骨髓间充质干细胞来源中胚层间充质,颌骨中的骨髓间充质干细胞来源于颅神经脊源外胚层。骨髓间充质干细胞生物学特性表现为高度的增殖活性及多向谱系分化潜力,在体外特定的诱导环境下,骨髓间充质干细胞可向成骨细胞、软骨细胞、脂肪细胞、神经细胞、肝样细胞、髓核样细胞等方向分化[15-16],现已作为种子细胞被广泛应用于组织工程。 2.2.2 Ⅰ型胶原诱导骨髓间充质干细胞向成骨细胞分化 Ⅰ型胶原是健康骨组织细胞外基质的主要成分,对骨髓间充质干细胞的成骨分化、增殖以及骨骼的发育起至关重要的作用[17]。相关研究表明,Ⅰ型胶原蛋白基质培养的骨髓间充质干细胞显示出高的碱性磷酸酶活性和胶原合成能力,并且检测到骨钙素、骨涎蛋白及骨桥蛋白的表达,其为成骨细胞特异性蛋白[18]。碱性磷酸酶是干细胞向成骨细胞分化的重要标记酶,成骨细胞分化程度与碱性磷酸酶活性的高低呈正相关,常作为成骨细胞分化级联的相对早期标志物[19]。Hesse等[20]研究了Ⅰ型胶原水凝胶中大鼠骨髓间充质干细胞的迁移、增殖及成骨分化,大鼠骨髓间充质干细胞由凝胶表面向凝胶内部迁移,出现增殖活跃现象,碱性磷酸酶在第14天后开始增加,至第21天达到显著升高水平,碱性磷酸酶活性的增加出现适度的延迟。Mizuno等[21]以Ⅰ型胶原作为骨髓间充质干细胞培养基质,研究发现骨髓间充质干细胞可定向分化为成骨细胞,检测到新生矿化骨基质。Linsley等[22]将骨髓间充质干细胞接种于Ⅰ型胶原预涂层的24孔培养板底部,胶原与骨髓间充质干细胞具有良好的细胞相容性,且对细胞的生长、增殖和成骨分化都有积极的作用。 2.2.3 Ⅰ型胶原诱导骨髓间充质干细胞向成骨细胞分化的机制 Ⅰ型胶原是骨基质的主要成分,胶原-整合素相互作用是骨髓间充质干细胞向成骨细胞分化的重要信号。整合素(Integrins)为一异源二聚体跨膜糖蛋白,由α亚基和β亚基以非共价键构成。α亚基与基质黏附有关,β亚基与细胞信号传递有关,调控特定靶基因表达,同时也是黏着斑形成所必需的[23]。整合素α2β1是Ⅰ型胶原的主要受体。它可以特异性识别Ⅰ型胶原α1链的天冬氨酸-甘氨酸-谷氨酸-丙氨酸(DGEA)序列[24]。DGEA肽(Asp-Gly-Glu-Ala)具有与Ⅰ型胶原的细胞结合结构域相似的结构,并显示阻断胶原-整合素相互作用,从而抑制了骨髓间充质干细胞的成骨细胞表型的表达[16]。黏着斑处有多种重要的信号转导分子及蛋白,如黏着斑激酶、桩蛋白(paxillin)等,它们参与Integrin信号通路并激活下游相关因子[25],共同参与细胞黏附、增殖、分化及调节细胞骨架结构等生物学行为。整合素与肌动蛋白细胞骨架相互作用,通过蛋白talin和vincullin介导细胞外信息。Talin和vincullin与黏着斑激酶相互作用并激活黏着斑激酶。黏着斑激酶在此信号传导中起中心作用。黏着斑激酶为非受体蛋白酪氨酸激酶家族中的一员,其N端与整合素β亚基胞内段结合,C端可与桩蛋白等骨架蛋白结合[26-27]。黏着斑激酶活化及骨架蛋白磷酸化进一步引起含SH2结构域的胞内蛋白质发生磷酸化,进而启动Ras通路,通过Ras-Raf-MEK-ERKs这一序贯性激活途径[28],成骨特异性转录因子表达增加,提高Cbfa-1磷酸化水平,促进碱性磷酸酶的表达及矿物质沉积,增强骨髓间充质干细胞成骨分化[29-30]。 2.3 Ⅰ型胶原促成骨细胞成骨分化 发育过程中,骨的形成有两种形式:一种是膜内成骨,另一种是软骨内成骨。颅颌面骨的成骨形式主要是膜内成骨,而躯干长骨的成骨形式主要是软骨内成骨。成骨细胞是骨形成的主要功能细胞,它负责骨基质的生物合成及分泌多种生长因子、特殊蛋白,并且这些生长因子作用于成骨细胞的特异性受体发挥生物效应,共同参与骨发生、形成与再修复等生物行为。诸多蛋白及因子调控着成骨细胞的分化,如骨形态发生蛋白、转化生长因子β1、胰岛素样生长因子等。体内环境中,胶原由成骨细胞合成并分泌到细胞外基质中,与成骨细胞、骨细胞等共同作用使之形成骨雏形,并在其上沉积羟基磷灰石,逐渐完成骨的改建与成熟。成骨细胞分泌Ⅰ型胶原,Stein等[31]认为成骨细胞分泌胶原的顶峰时期为第8-10天,2周后出现明显下降。在成骨细胞增殖期,成骨细胞分泌碱性磷酸酶、骨钙素、整合素等,增加细胞间的链接及相互之间的信号转导,促进骨成熟。Ⅰ型胶原通过整合素对成骨细胞发挥促成骨分化和增强细胞间黏附的作用,并维持细胞的正常代谢活动,同时整合素可调节成骨细胞外胶原的合成[32]。杨志明等[33]通过用抗体阻断Ⅰ型胶原-整合素α2β1系统,破坏了成骨细胞与Ⅰ型胶原的黏附,发现成骨细胞的增殖能力减弱,同时检测出Ⅰ型胶原mRNA、骨钙素mRNA及α2β1mRNA 表达减弱,去除抗体作用后,成骨细胞的功能得到部分恢复。Ⅰ型胶原通过整合素α2β1系统启动黏着斑激酶介导的信号传导通路,增加成骨细胞的黏附能力,促进成骨细胞的分化,从而促进成骨。成骨细胞在分化阶段分泌大量的Ⅰ型胶原,在细胞外基质中起到了稳定的作用,利于细胞外基质的形成[34]。 2.4 Ⅰ型胶原在牵张成骨过程中的表达 2.4.1 牵张成骨 牵张成骨现如今已广泛应用于颅颌面部因感染、创伤、恶性肿瘤、唇腭裂术后所造成的继发骨缺损、颌骨畸形,并取得了良好的临床效果[35-37]。传统的颌骨缺损治疗方法有自体骨移植、异体骨移植、人工骨材料移植,但这些骨移植材料有术后感染并发症、排斥反应及成活率低等缺点。牵张成骨来源于自体骨新生,解决了自体和异体重建的局限性。牵张成骨是指通过外科手术截出新鲜骨断端,并且保持骨断端的稳定血供、骨膜及周围软组织,安置稳定牵张装置,经过一段间歇期后,以一定的速率及频率于骨断端施加牵引力进行牵张,以促进牵张间隙新骨形成[38]。牵张成骨的机制尚未明确,仍是国内外的前沿研究热点。在下颌骨牵引成骨区新骨的形成主要是膜内成骨,有局部的软骨内成骨,原因可能是局部血液循环较差引起的低氧环境,而致间充质细胞向软骨细胞分化[39]。 2.4.2 Ⅰ型胶原在牵张成骨过程中的表达 牵张成骨从骨切开至新骨成熟期间,参与的细胞不同,不同时期形成的胶原也不同。韦敏等[40]研究颅面骨牵张成骨过程中胶原生成及演变的机制和规律,结果表明牵张间隙早期出现成纤维细胞富集,其分泌Ⅲ型胶原,中后期大量成骨细胞生成,特异性分泌Ⅰ型胶原,最后Ⅰ型胶原完全替代Ⅲ型胶原成为骨组织的主要细胞外基质。丁宇翔等[41]观察牙槽突裂牵张成骨整复过程中不同时期胶原合成的种类及演变过程,研究牵张新骨生成的方式及组织学特点。结果显示牵张固定早期以分泌Ⅲ型胶原为主,随后Ⅰ型胶原增多并最终取代Ⅲ型胶原而成为骨基质的主要成分,Ⅱ型胶原在整个成骨过程中无明显阳性表现,牙槽突牵张成骨以膜内成骨方式为主,无明显的软骨内成骨现象。商洪涛等[42]研究上颌骨缝牵张成骨,牵张1周时骨缝区新生组织中Ⅲ、Ⅰ型胶原同时存在,牵张4周时Ⅰ型胶原比例升高,最终Ⅲ型胶原降解,Ⅰ型胶原完全成为骨基质的主要成分。Chen等[43]在牙周膜牵张成骨研究中发现牙周韧带Ⅰ型胶原含量在力加载开始时下降,随后增加,原因为高表达基质金属蛋白酶1介导的细胞外基质的部分降解及Ⅰ型胶原对牙槽间隔减阻所致的炎症反应。姜喜亮等[44]通过构建黏着斑激酶(focal adhesion kinase,FAK)特异性siRNA慢病毒表达载体,感染牵张前骨髓间充质干细胞,应用自行研制的多单元细胞拉伸与压缩装置,对感染后的骨髓间充质干细胞施加适当的机械张应力。结果表明黏着斑激酶基因沉默后,成骨转录因子Runx-2、碱性磷酸酶的mRNA和蛋白的表达水平明显降低,整合素信号通路抑制后,牵张成骨过程中骨髓间充质干细胞成骨分化减弱,提示整合素信号通路参与细胞力学信号向生物化学信号的转化。 2.5 Ⅰ型胶原蛋白在骨组织矿化中的作用 胶原蛋白的生物合成、胞外基质的自动组装及生物矿化是骨形成的重要步骤,目前胶原蛋白的合成及组装研究得较为透彻,矿化机制仍存有异议。目前有两大学说,分别为传统溶液结晶理论及聚合物诱导的液相前驱矿化理论。 2.5.1 Ⅰ型胶原自动组装 Ⅰ型胶原蛋白是骨细胞外基质的主要成分,胶原蛋白分子间通过交联的形式构建了有序排列和结构完整的胶原纤维,为钙、磷等矿物质的沉积提供了良好的内部微环境,Ⅰ型胶原在骨形成过程中起到生物矿化模板的作用,从而引导钙盐沉积、矿化,形成羟基磷灰石晶体[45]。Ⅰ型胶原纤维不仅为骨矿化提供基本结构场所,而且还保证了骨骼的韧性。Ⅰ型前胶原分泌到细胞外基质,各种蛋白酶加工最终形成成熟的胶原纤维,然后轴向平行聚集成4 nm的胶原微纤维,经过多级有序组装成直径在10-300 nm的胶原纤维,再与蛋白多糖等大分子交联形成纤维网状结构[46]。原胶原以1/4交错排列形式,形成40 nm的孔区(Hole)与27 nm的重叠区(Overlap)交替的周期性结构[47](图1)。 2.5.2 传统溶液结晶理论 孔区含有负电荷的氨基酸侧链。-COO-键合Ca2+,Ca2+被固定在带电荷的侧链,Ca2+再结合Pi离子(PO43-)形成网络,Ca-Pi之间通过静电力结合从而诱导成核[48]。负电荷的官能团加载于胶原纤维对成核和晶体生长有重要作用。带负电荷的羧基(-COOH)及羰基(C=O)是矿化过程中羟基磷灰石结晶的成核位点,同时羧基和Ca2+结合是形成成核位点的关键步骤[49]。涂姜磊等[50]证实Ⅰ型胶原确有体外诱导沉积类骨羟基磷灰石的矿化作用,胶原与其矿化产物类骨羟基磷灰石之间存在明显的配位相互作用,即胶原中的C=O与Ca2+的配位作用诱导羟基磷灰石在胶原中与羰基相对应的位点成核,进而促进类骨羟基磷灰石的形成。紫外光谱法和X射线衍射结果提示胶原矿化过程中存在非晶体相向晶体相的转变,并且晶体C轴与纤维长轴平行。成核位点的形成及非晶质的磷酸钙转变为有取向的羟基磷灰石晶体是传统溶液结晶机制的核心学说。矿化胶原纤维经过加工和组装,胶原纤维束的层状组织重排形成互相平行,最后再进行复杂的结构修饰形成骨组织。 2.5.3 聚合物诱导的液相前驱矿化理论 Olszta等[47]最初在骨质生物矿化中提出聚合物诱导的液相前驱矿化理论,认为无定形磷酸钙与非胶原蛋白形成聚合物,其形态为介于晶体与液体之间的无定形相,无定形前体相的流体特性使其能够通过毛细管而被吸入纳米级间隙和胶原纤维凹槽中,即液相矿物前体可以通过毛细作用引入胶原纤维的缝隙和沟槽中,随着水合物失去水分进入热力学稳定阶段,前体固化并结晶,留下嵌入胶原纤维纳米级的羟基磷灰石晶体(图2)。聚天冬氨酸(polyaspartic acid,pAsp)是一种非胶原蛋白的类似物,同时也是一种矿化导向剂,其与无定形磷酸钙形成带负电的复合物,二者相互作用从而介导无定形磷酸钙渗入胶原蛋白,最后在胶原内部转化为定向排列的磷灰石晶体[51-52]。Olszta等[47]将聚天冬氨酸加入到矿化液中,观察到取向磷灰石晶体的纤维内形成,且纤维内晶体与骨中的磷灰石晶体有相同的结构。为了探索胶原生物矿化的具体机制,国内外研究人员正致力于开展体外胶原矿化的深入研究[53-55]。 此外,也有研究显示Ⅰ型胶原蛋白能独立启动和定位磷灰石晶体的矿化,并影响磷灰石晶体的大小及在骨组织中的分布。Wang等[56]通过构建Ⅰ型胶原基质模型,排除了钙结合聚合物和其他非胶原蛋白的干预,结果表明不使用聚合物添加剂作为矿化导向剂下能够体外实现Ⅰ型胶原矿化,无机物的矿化形式类似于在动物机体内发现的骨矿化形式,并且可影响磷灰石晶体的尺寸和三维分布。在这项研究中,通过在高浓度Ⅰ型胶原中保持钙、磷酸盐和碳酸盐离子的离子强度恒定且接近于生理条件,氨蒸气中和控制pH值使胶原分子沉淀成纤维,观察到高致密、单分散的纤维均匀齐次矩阵,这与正常骨矿化早期相似。 2.6 Ⅰ型胶原在医疗领域的应用 目前基于对Ⅰ型胶原生物合成、组装及生物矿化机制的研究,研究人员正致力于研究与Ⅰ型胶原结构和组成类似的仿生材料。仿生胶原蛋白以多种形式的骨替代材料应用于医学、生物材料及组织工程领域。矿化胶原蛋白是胶原蛋白/羟基磷灰石(Col/HAp)复合材料,其具有良好的生物可降解性、骨传导、促骨再生的生物学性能,目前已作为商品应用于骨缺损修复治疗。矿化胶原替代脊髓融合手术中的传统植入材料钛合金,X射线下可见矿化胶原与脊柱有良好的骨性融合能力,最后出现降解,被新生骨组织所替代[57]。胶原海绵是一种新型生物材料,由动物结缔组织提取的海绵状多孔固体[58],经60 ℃消毒灭菌后,表现出良好的亲水性、低免疫原性,广泛应用于组织工程,其主要作用有止血、促细胞生长、促伤口愈合、抗感染、促骨修复等。Ⅰ型胶原蛋白水凝胶具有易诱导成软骨、良好可塑性的优点,最早应用于软骨缺损修复中。此外,根据组织损伤特点,将不同的生长因子特异性地结合到胶原材料上,可以起到特异性修复的作用。"
| [1] Gordon MK, Hahn RA. Collagens.Cell Tissue Res. 2010;339(1):247-257.[2] Roche P, Czubryt MP. Transcriptional control of collagen I gene expression. Cardiovasc Hematol Disord Drug Targets. 2014;14(2):107-120.[3] Vuorio E, de Crombrugghe B. The family of collagen genes. Annu Rev Biochem. 1990;59:837-872.[4] Jimenez SA, Varga J, Olsen A, et al. Functional analysis of human alpha 1(I) procollagen gene promoter. Differential activity in collagen-producing and -nonproducing cells and response to transforming growth factor beta 1. J Biol Chem. 1994;269(17):12684-12691.[5] Ghosh AK, Yuan W, Mori Y, et al. Antagonistic regulation of type I collagen gene expression by interferon-gamma and transforming growth factor-beta. Integration at the level of p300/CBP transcriptional coactivators. J Biol Chem. 2001; 276(14):11041-11048.[6] Canty EG, Kadler KE. Procollagen trafficking, processing and fibrillogenesis. J Cell Sci. 2005;118(Pt 7):1341-1353.[7] Tasab M, Batten MR, Bulleid NJ. Hsp47: a molecular chaperone that interacts with and stabilizes correctly-folded procollagen. EMBO J. 2000;19(10):2204-2211.[8] Sauk JJ, Norris K, Hebert C, et al. Hsp47 binds to the KDEL receptor and cell surface expression is modulated by cytoplasmic and endosomal pH. Connect Tissue Res. 1998; 37(1-2):105-119.[9] Ishikawa Y, Bächinger HP. A molecular ensemble in the rER for procollagen maturation. Biochim Biophys Acta. 2013;1833 (11):2479-2491.[10] Garnero P. Erratum to: The Role of Collagen Organization on the Properties of Bone. Calcif Tissue Int. 2015;97(3):241.[11] Cohen-Solal L, Zylberberg L, Sangalli A, et al. Substitution of an aspartic acid for glycine 700 in the alpha 2(I) chain of type I collagen in a recurrent lethal type II osteogenesis imperfecta dramatically affects the mineralization of bone. J Biol Chem. 1994;269(20):14751-14758.[12] Bardai G, Lemyre E, Moffatt P, et al. Osteogenesis Imperfecta Type I Caused by COL1A1 Deletions. Calcif Tissue Int. 2016; 98(1):76-84.[13] Prockop DJ. Mutations that alter the primary structure of type I collagen. The perils of a system for generating large structures by the principle of nucleated growth. J Biol Chem. 1990;265(26):15349-15352.[14] Gerhard DS, Wagner L, Feingold EA, et al. The status, quality, and expansion of the NIH full-length cDNA project: the Mammalian Gene Collection (MGC). Genome Res. 2004; 14(10B):2121-2127.[15] He W, Wu WK, Wu YL, et al. Ginsenoside-Rg1 mediates microenvironment-dependent endothelial differentiation of human mesenchymal stem cells in vitro. J Asian Nat Prod Res. 2011;13(1):1-11.[16] Mizuno M, Fujisawa R, Kuboki Y. Type I collagen-induced osteoblastic differentiation of bone-marrow cells mediated by collagen-alpha2beta1 integrin interaction. J Cell Physiol. 2000; 184(2):207-213.[17] Bortell R, Barone LM, Tassinari MS, et al. Gene expression during endochondral bone development: evidence for coordinate expression of transforming growth factor beta 1 and collagen type I. J Cell Biochem.1990;44(2):81-91.[18] Pittenger MF, Mackay AM, Beck SC, et al. Multilineage potential of adult human mesenchymal stem cells. Science. 1999;284(5411):143-147.[19] Aronow MA, Gerstenfeld LC, Owen TA, et al. Factors that promote progressive development of the osteoblast phenotype in cultured fetal rat calvaria cells. J Cell Physiol. 1990;143(2):213-221.[20] Hesse E, Hefferan TE, Tarara JE, et al. Collagen type I hydrogel allows migration, proliferation, and osteogenic differentiation of rat bone marrow stromal cells. J Biomed Mater Res A. 2010;94(2):442-449.[21] Mizuno M, Kuboki Y. Osteoblast-related gene expression of bone marrow cells during the osteoblastic differentiation induced by type I collagen. J Biochem. 2001;129(1):133-138.[22] Linsley C, Wu B, Tawil B. The effect of fibrinogen, collagen type I, and fibronectin on mesenchymal stem cell growth and differentiation into osteoblasts. Tissue Eng Part A. 2013; 19(11-12):1416-1423.[23] Salomon DS. Cell-cell and cell-extracellular matrix adhesion molecules communicate with growth factor receptors: an interactive signaling Web. Cancer Invest. 2000;18(6): 591-593.[24] Staatz WD, Fok KF, Zutter MM, et al. Identification of a tetrapeptide recognition sequence for the alpha 2 beta 1 integrin in collagen. J Biol Chem. 1991;266(12):7363-7367.[25] Puklin-Faucher E, Sheetz MP. The mechanical integrin cycle. J Cell Sci. 2009;122(Pt 2):179-186.[26] Schwartz MA. Integrins, oncogenes, and anchorage independence. J Cell Biol. 1997;139(3):575-578.[27] Wen LP, Fahrni JA, Troie S, et al. Cleavage of focal adhesion kinase by caspases during apoptosis. J Biol Chem. 1997; 272(41):26056-26061.[28] Kim EK, Choi EJ. Pathological roles of MAPK signaling pathways in human diseases. Biochim Biophys Acta. 2010; 1802(4):396-405.[29] Salasznyk RM, Klees RF, Williams WA, et al. Focal adhesion kinase signaling pathways regulate the osteogenic differentiation of human mesenchymal stem cells. Exp Cell Res. 2007;313(1):22-37[30] Jadlowiec J, Koch H, Zhang X, et al. Phosphophoryn regulates the gene expression and differentiation of NIH3T3, MC3T3-E1, and human mesenchymal stem cells via the integrin/MAPK signaling pathway. J Biol Chem. 2004;279(51): 53323-53330. [31] Stein GS, Lian JB, Stein JL, et al. Transcriptional control of osteoblast growth and differentiation. Physiol Rev. 1996;76(2): 593-629.[32] Ganta DR, McCarthy MB, Gronowicz GA. Ascorbic acid alters collagen integrins in bone culture. Endocrinology. 1997;138(9): 3606-3612.[33] 杨志明,屈艺.Ⅰ型胶原—整合素α2β1系统对成骨细胞生物学特性的调控[J].四川大学学报:医学版,2000,31(3):281-284.[34] Wenstrup RJ, Witte DP, Florer JB. Abnormal differentiation in MC3T3-E1 preosteoblasts expressing a dominant-negative type I collagen mutation. Connect Tissue Res.1996;35(1-4): 249-257.[35] Mofid MM, Manson PN, Robertson BC, et al. Craniofacial distraction osteogenesis: a review of 3278 cases. Plast Reconstr Surg. 2001;108(5):1103-1114.[36] Schreuder WH, Jansma J, Bierman MW, et al. Distraction osteogenesis versus bilateral sagittal split osteotomy for advancement of the retrognathic mandible: a review of the literature. Int J Oral Maxillofac Surg. 2007;36(2):103-110.[37] 王大章,陈刚,胡静.牵张成骨在矫治牙颌面畸形中的应用[J].华西口腔医学杂志,1998, 5(4):369-371.[38] Welch RD, Lewis DD. Distraction osteogenesis. Vet Clin North Am Small Anim Pract. 1999,29(5):1187-1205.[39] 柴本甫,汤雪明.实验性骨折愈合的细胞生物学[J].中华骨科杂志, 1991,11(3):203-206.[40] 韦敏,穆雄铮,张涤生,等.颅面牵拉成骨胶原合成的研究[J].口腔颌面外科杂志, 2001, 11(1):32-34.[41] 丁宇翔,刘彦普,敖建华.牙槽突裂牵张成骨胶原合成的研究[J].实用口腔医学杂志, 2005, 21(4):477-479.[42] 商洪涛,刘锐,孙建壮,等.上颌骨缝牵张的组织学变化[J].中国组织工程研究, 2007, 11(10):1833-1836.[43] Chen X, Li N, LeleYang, et al. Expression of collagen I, collagen III and MMP-1 on the tension side of distracted tooth using periodontal ligament distraction osteogenesis in beagle dogs. Arch Oral Biol. 2014;59(11):1217-1225.[44] 姜喜亮,朱晓文,胡静,等.整合素信号通路在大鼠骨髓间充质干细胞牵张中的作用和转导机制[J].中国口腔颌面外科杂志, 2015, 13(3):193-199.[45] Liebschner MA. Biomechanical considerations of animal models used in tissue engineering of bone. Biomaterials. 2004;25(9):1697-1714.[46] Cisneros DA, Hung C, Franz CM, et al. Observing growth steps of collagen self-assembly by time-lapse high-resolution atomic force microscopy. J Struct Biol. 2006;154(3):232-345.[47] Olszta MJ, Cheng X, Sang SJ, et al. Bone structure and formation: A new perspective. Materials Science & Engineering R. 2008; 58 (3) :77-116.[48] Xu Z, Yang Y, Zhao W, et al. Molecular mechanisms for intrafibrillar collagen mineralization in skeletal tissues. Biomaterials. 2015;39:59-66.[49] Wei Z, Huang ZL, Liao SS, et al. Nucleation Sites of Calcium Phosphate Crystals during Collagen Mineralization. Journal of the American Ceramic Society. 2003; 86(6):1052-1054.[50] 涂姜磊,郭富强,吕春春,等.胶原诱导沉积类骨HA及其机制研究[J].生物医学工程学杂志, 2011, 28(1):99-103.[51] Nudelman F, Pieterse K, George A, et al. The role of collagen in bone apatite formation in the presence of hydroxyapatite nucleation inhibitors. Nat Mater. 2010;9(12):1004-1009.[52] Cantaert B, Kim YY, Ludwig H, et al. Think Positive: Phase Separation Enables a Positively Charged Additive to Induce Dramatic Changes in Calcium Carbonate Morphology. Advanced Functional Materials. 2012; 22(5):907-915.[53] Zurick KM, Qin C, Bernards MT. Mineralization induction effects of osteopontin, bone sialoprotein, and dentin phosphoprotein on a biomimetic collagen substrate. J Biomed Mater Res A. 2013;101(6):1571-1581.[54] Qiu ZY, Cui Y, Tao CS, et al. Mineralized Collagen: Rationale, Current Status, and Clinical Applications. Materials (Basel). 2015;8(8):4733-4750.[55] Kerns JG, Buckley K, Churchwell J, et al. Is the Collagen Primed for Mineralization in Specific Regions of the Turkey Tendon? An Investigation of the Protein-Mineral Interface Using Raman Spectroscopy. Anal Chem. 2016;88(3): 1559-1563.[56] Wang Y, Azaïs T, Robin M, et al. The predominant role of collagen in the nucleation, growth, structure and orientation of bone apatite. Nat Mater. 2012;11(8):724-733.[57] Hvistendahl M. China's push in tissue engineering. Science. 2012;338(6109):900-902.[58] Kumar MS, Kirubanandan S, Sripriya R, et al. Triphala incorporated collagen sponge--a smart biomaterial for infected dermal wound healing. J Surg Res. 2010;158(1): 162-170. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [3] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [4] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [5] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [6] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [7] | Fan Quanbao, Luo Huina, Wang Bingyun, Chen Shengfeng, Cui Lianxu, Jiang Wenkang, Zhao Mingming, Wang Jingjing, Luo Dongzhang, Chen Zhisheng, Bai Yinshan, Liu Canying, Zhang Hui. Biological characteristics of canine adipose-derived mesenchymal stem cells cultured in hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1002-1007. |
| [8] | Geng Yao, Yin Zhiliang, Li Xingping, Xiao Dongqin, Hou Weiguang. Role of hsa-miRNA-223-3p in regulating osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1008-1013. |
| [9] | Lun Zhigang, Jin Jing, Wang Tianyan, Li Aimin. Effect of peroxiredoxin 6 on proliferation and differentiation of bone marrow mesenchymal stem cells into neural lineage in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1014-1018. |
| [10] | Zhu Xuefen, Huang Cheng, Ding Jian, Dai Yongping, Liu Yuanbing, Le Lixiang, Wang Liangliang, Yang Jiandong. Mechanism of bone marrow mesenchymal stem cells differentiation into functional neurons induced by glial cell line derived neurotrophic factor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1019-1025. |
| [11] | Duan Liyun, Cao Xiaocang. Human placenta mesenchymal stem cells-derived extracellular vesicles regulate collagen deposition in intestinal mucosa of mice with colitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1026-1031. |
| [12] | Pei Lili, Sun Guicai, Wang Di. Salvianolic acid B inhibits oxidative damage of bone marrow mesenchymal stem cells and promotes differentiation into cardiomyocytes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1032-1036. |
| [13] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [14] | Wang Shiqi, Zhang Jinsheng. Effects of Chinese medicine on proliferation, differentiation and aging of bone marrow mesenchymal stem cells regulating ischemia-hypoxia microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1129-1134. |
| [15] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||