Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (21): 3410-3416.doi: 10.3969/j.issn.2095-4344.0905
Previous Articles Next Articles
Research progress and tendency of mesenchymal stem cell transplantation in the treatment of spinal cord injury
Liu Ke-xun1, Huo Hong-jun2, Zhao Yan3, Zuo Yuan3
- 1Graduate School of Inner Mongolia Medical University, Hohhot 010000, Inner Mongolia Autonomous Region, China; 2Baotou Medical College, Inner Mongolia University of Science & Technology, Baotou 014000, Inner Mongolia Autonomous Region, China; 3Second Affiliated Hospital of Inner Mongolia University, Hohhot 010000, Inner Mongolia Autonomous Region, China
-
Revised:2018-05-30Online:2018-07-28Published:2018-07-28 -
Contact:Huo Hong-jun, Professor, Baotou Medical College, Inner Mongolia University of Science & Technology, Baotou 014000, Inner Mongolia Autonomous Region, China -
About author:Liu Ke-xun, Master candidate, Graduate School of Inner Mongolia Medical University, Hohhot 010000, Inner Mongolia Autonomous Region, China -
Supported by:the National Natural Science Foundation of Inner Mongolia Autonomous Region, No. 2016MS08132
CLC Number:
Cite this article
Liu Ke-xun, Huo Hong-jun, Zhao Yan, Zuo Yuan. Research progress and tendency of mesenchymal stem cell transplantation in the treatment of spinal cord injury[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(21): 3410-3416.
share this article

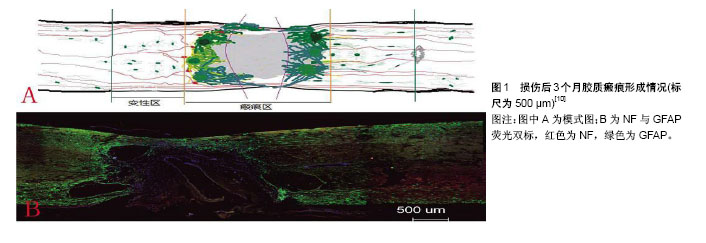
2.1 脊髓损伤概述 随着经济的迅猛发展,人们生活水平日新月异,交通业、建筑业的蓬勃发展。随之而来的脊髓损伤患者日趋增多,而目前对于这一疾病的治疗尚无突破,给患者本身带来痛苦的同时,也给其家庭与社会带来巨大的经济负担。在一些发达国家,如美国每年有近13 000起新的脊髓损伤发生,每年的直接医疗费用近200亿美元[1]。日本脊髓损伤患者占所有创伤患者的百分比从2004年的15.4%上升到2013年的17.6%,其中大多数由跌倒或车祸引起。这种损伤带来的疼痛、残疾、社会依赖和医疗费用相当高[2]。中国作为最大的发展中国家,同样面临这样的困扰。中国台湾有着亚洲创伤性脊髓损伤最高的发病率,高达为61.6/100万[3]。研究表明,随着年龄的增长脊髓损伤的发病率逐渐增加,在46-60岁年龄段达到顶峰[4]。中国正处于经济发展井喷时期,同时人口老龄化愈发显著,因此脊髓损伤的治疗与康复对今后发展的影响不言而喻。 脊髓损伤引起脊髓功能失调从而损害脊髓主要功能,即感觉、运动、自主和反射,完全或部分由于创伤、疾病或退化(非外伤)所致,其中90%是由创伤导致的,但近年来非创伤性因素所占比例不断增加。从剖析结构来看,颈椎损伤占急性脊髓损伤的55%左右,其余的胸椎、胸腰段和腰骶部损伤大约各占15%[5-6]。脊髓损伤分为原发性与继发性。原发性脊髓损伤是由压迫、挫伤或撕裂等各种外力引起,最常见的是脊柱骨折脱位。继发性损伤是由原发性脊髓损伤引起的一系列连锁反应,脊髓损伤后几分钟到数周均可发生。初级的连锁反应包括水肿、血流量减少、血管痉挛、氧自由基的产生、炎症、缺血、兴奋性毒性、脂质过氧化作用和引起神经细胞凋亡等[7]。星形胶质细胞也会帮助胶质瘢痕的形成,这些胶质瘢痕不仅防止渗透也会干扰轴突再生,产生大量炎症细胞,而且因组织缺损所形成的流体填充腔与瘢痕组织会抑制炎症分子上调,这导致防止神经再生的物理屏障形成(图1)[8-10]。 脊髓损伤的治疗方法大多停留在手术减压治疗、药物治疗及术后康复治疗。手术减压的目的在于解除脊髓压迫,维持脊柱稳定,尽量减少继发性脊髓损伤的发生,为后续脊髓功能恢复创造有利条件。由于神经的不可再生性,药物治疗仅仅是起到减轻神经细胞坏死与保护神经元功能的作用,如减轻脊髓神经水肿、增强脊髓神经血流量、减少炎症细胞浸润等等,但是治疗效果差强人意,同时一些药物的使用剂量及给药时间充满争议,其中最具代表性的是类固醇激素甲强龙和可以有效改善微循环功能的钙通道阻止剂尼莫地平等[11-12]。康复治疗通过后期康复锻炼,使脊髓损伤患者最大程度获得自理能力,尽可能减轻社会家庭负担,但是并没有从根本上恢复神经功能。为了解决神经修复的问题,细胞移植为脊髓损伤治疗提供了新的思路。间充质干细胞作为种子细胞进行细胞移植是最具代表性的治疗方向。"

2.2 间充质干细胞的不同来源及其生物学特征 间充质干细胞是多能成体干细胞,它具有分化成中胚层甚至非中胚层谱系的潜力,被视为组织修复和再生的营养因子,在1987年就已被Friedenstein等[13]在骨髓中发现并成功的分离培养。间充质干细胞可以分化为不同类型的细胞,如脂肪细胞、骨细胞、软骨细胞、肌肉细胞、肝细胞、心肌细胞、神经细胞、内皮细胞等[14]。根据国际细胞治疗协会的规定,间充质干细胞表达CD105(SH2)、CD73(SH3)、CD44和CD90,而不表达CD45、CD34、CD11b、CD19和HLA-DR,同时可以贴壁培养并能分化为以上不同类型的细胞[15]。间充质干细胞被发现存在于骨髓组织中,其主要来源还包括脂肪组织和围生组织(如脐血、羊膜、胎盘)等。大量的研究表明间充质干细胞在调控免疫反应、支持造血、抑制过度的炎症反应、促血管生成以及组织修复中起重要的作用[16-20]。这些独特的特点,使间充质干细胞成为干细胞治疗和组织工程中的理想种子细胞。 不同来源的间充质干细胞分化能力也略有不同,胎盘间充质干细胞和脐带间充质干细胞的增殖能力相似,且高于骨髓间充质干细胞和脂肪间充质干细胞,骨髓来源的间充质干细胞增殖能力最差;但在促进血管内皮增殖和血管分化的能力上,胎盘间充质干细胞和骨髓间充质干细胞的作用明显高于脂肪间充质干细胞和脐带间充质干细胞。在免疫调节方面,胎盘间充质干细胞表达前列腺素E2的水平最高,且抑制外周血单个核细胞合成γ-干扰素的作用最强[21]。 2.3 目前间充质干细胞治疗脊髓损伤的研究进展 间充质干细胞易于分离获得,且具有抗瘢痕组织增殖、抗神经细胞凋亡和抗炎作用,但目前干细胞功能性治疗的方法尚不明朗。越来越多的研究表明间充质干细胞拥有良好的免疫抑制性,可以减小病变腔,减少小胶质细胞/巨噬细胞反应等[22-23]。间充质干细胞还可以分泌神经营养因子如白细胞介素6、白细胞介素7和血管内皮生长因子等[24],这些因素都在修复神经损伤方面发挥着重要作用。据此间充质干细胞治疗脊髓损伤这一思路已逐步出现于治疗思维中。间充质干细胞的给药途径多种多样,如静脉注射间充质干细胞后,脊髓损伤大鼠的后肢截瘫症状出现改善,抗氧化、抗炎作用增强[25-26];腰穿经蛛网膜下腔注射间充质干细胞,其可在脊髓损伤处募集,有研究表明促红细胞生成素可以加速促进这一迁移,并修复脊髓损伤[27]。 对于这种治疗方案的实施,仅仅有充足的理论支持是远远不够的,还要有系统的、权威的安全性评估。人们在X射线引导下经皮椎管内注射同种异体脂肪间充质干细胞治疗犬慢性脊髓损伤,经术后16周随访观察其神经性病理疼痛与神经功能的恶化情况,并没有发现不良反应和并发症,从而也证实了该治疗方式的安全性[28]。随着间充质干细胞移植治疗脊髓损伤受到人们的重视,人们也意识到骨髓、脐带、脐血源性间充质干细胞虽有良好的治疗潜力,但是难免涉及伦理学与取材困难的问题。自体间充质干细胞移植自然而然的解决了这些问题,但是大多数研究依旧停留在基础实验和动物实验水平。 2.3.1 经基因修饰的间充质干细胞治疗脊髓损伤 将间充质干细胞进行基因修饰的目的是提高间充质干细胞向神经细胞的定向分化能力、抵抗脊髓损伤时神经样细胞凋亡、修复组织损伤以及降低损伤处的炎症反应等。信号转导通路参与生物体的各个生命活动过程,但是尚无将间充质干细胞进行基因修饰治疗脊髓损伤按信号转导通路归纳的相关综述。因此,作者试着将近年来最新研究结果按照信号转导通路进行总结。 MAPK信号转导通路是生物体内重要的转导通路之一,它参与了细胞增殖与分化的调控,介导炎症、凋亡、应激等反应。在治疗脊髓损伤的过程中,多数神经营养因子可激活该信号通路,如脑源性神经营养因子与胶质源性神经营养因子作用于酪氨酸激酶B,使酪氨酸激酶B自身磷酸化作用增强,从而激活Ras-MAPK通路,在cAMP反应元件结合蛋白(CPEB)的丝氨酸位点上激活CREB,促进脑源性神经营养因子、胶质源性神经营养因子基因与抗凋亡基因Bcl-2的表达,从而实现了对于神经样细胞的保护与抗神经样细胞凋亡。该通路不仅揭示了脑源性神经营养因子、胶质源性神经营养因子治疗脊髓损伤的作用机制,而且为基因修饰间充质干细胞治疗脊髓损伤提供了研究思路。血管内皮生长因子通过激活血管内皮生长因子受体,并使受体磷酸化从而激活MAPK实现内皮细胞的增殖,虽在促进神经细胞增殖过程中作用有限,但是却可明显缩小脊髓损伤时造成的空洞并抑制瘢痕组织形成[29]。MAPK通路上cAMP的激活也可由碱性成纤维生长因子来完成,碱性成纤维生长因子激活cAMP后进一步导致蛋白激酶C激活和Ca2+内流,碱性成纤维生长因子作为一种神经营养因子,可加速核蛋白体基因的转录,刺激细胞的DNA合成增强,促进细胞的分裂与增殖。MAPK通路不仅在细胞增殖、分化与凋亡中起到重要作用,更是调节着细胞的迁移与定植。基质细胞衍生因子1是通过激活MAPK促进间充质干细胞的定植来显著降低周围空泡病变范围[30-31]。学者们的研究并没有停留在MAPK信号转导通路上,人们试着从“源头”寻找可以增强相关因子表达的基因。近年来随着对基因研究的不断深入,人们发现原神经基因指导祖细胞分化为神经元过程中不可缺少的基因——神经元素2,它的表达上调亦可促进碱性成纤维生长因子和血管内皮生长因子的生成。mircoRNA-383基因可以增强胶质源性神经营养因子的表达[32]。将间充质干细胞经上述因子的基因修饰或者经可促进相关因子表达的基因修饰均可提高间充质干细胞对脊髓损伤的治疗效果[33-35]。 提高间充质干细胞向神经样细胞的定向分化能力也是增强间充质干细胞治疗脊髓损伤效果的方向。在体外实验中,间充质干细胞向神经样细胞定向分化的实验中已有许多成熟的方案,如生长因子诱导分化法、化学诱导方法、损伤脑组织匀浆诱导法等,但是这些方法并不能够全部运用到体内实验中。在体内实验中,除了上述生长因子外,多聚嘧啶序列结合蛋白2(polypyrimidine tract-binding protein 2,PTBP2)是对RNA起到辅助加工作用的hnRNP家族一员,它可以提高间充质干细胞向神经样细胞的分化效率。最近的研究表明,mircoRNA-9、mircoRNA-124表达水平可直接调控PTBP1的表达,而PTBP1是PTBP2的抑制因子,提高miR-124表达水平可降低PTBP1的表达,从而促进定向分化效率[36-38]。 在定向分化的过程中,TGF-β1/p-SMAD2通路同样起到至关重要的作用。转化生长因子β1(TGF-β1)配体与Ⅱ型受体结合,Ⅱ型受体招募并磷酸化Ⅰ型受体,进而诱导SMAD磷酸化并进入核内,从而增加GAP-43的表达。GAP-43通过对膜细胞的肌动蛋白、胞内蛋白的影响来促进突触前末梢的生长,活化的GAP-43进一步交换细胞骨架成分,调节神经末梢活性,从而调节细胞形态。血小板反应蛋白1(thrombospondin-1,TPS-1)就是从激活TGF-β1出发,经TGF-β1/p-SMAD2通路促进间充质干细胞定向分化时的轴突生长、神经元存活来治疗脊髓损伤[39-40]。精氨酸脱羧酶(arginine decarboxylase,ADC)是存在于哺乳动物中枢神经系统中合成胍丁胺的限速酶,通过移植过度表达ADC基因的间充质干细胞可通过激活Ras-MAPK通路与TGF-β1/p-SMAD2通路,增加神经营养因子产生的同时促进细胞存活、分化、轴突再生,从而促进运动功能恢复[41]。碱性成纤维生长因子激活cAMP导致的Ca2+内流,使细胞释放出钙调蛋白。GAP-43在第二信使的作用下发生磷酸化同时释放出钙调素,激活钙调磷酸酶可引起GAP-43的进一步磷酸化和与钙调素的重新组合。由此可见,碱性成纤维生长因子在促进细胞增殖的同时可以促进定向分化。各个信号通路的交叉现象普遍存在于治疗机制内,这些交叉点正是治疗脊髓损伤的关键,值得人们进一步系统研究。 2.3.2 间充质干细胞与其他联合治疗脊髓损伤 间充质干细胞联合其他细胞来治疗脊髓损伤,已从起初联合神经干细胞、许旺细胞这些常见的神经细胞发展到联合嗅鞘细胞、脱落乳牙牙髓干细胞以及联合不同来源的间充质干细胞等。研究目的从单纯的增加神经细胞数量、增加神经营养物质的分泌发展到促进受损轴突生长、血管生成、促凋亡因子和抗凋亡因子的平衡及减少损伤组织体积[42-46]。 研究发现脊髓损伤时过氧化物酶、丙二醛、肿瘤坏死因子α、核因子κB、硫酸软骨素蛋白聚糖等炎症因子高水平表达。近年来专家学者们发现将间充质干细胞与甲基强的松龙、白花丹醌、硫酸软骨素ABC、米诺环素、促红细胞生成素、粒细胞集落刺激因子等药物联合应用,不仅可逆转脊髓损伤所产生的微环境,还可增加一些神经营养因子的表达[47-65],见表1。 上述联合治疗方案虽然可以将间充质干细胞的优势发挥的淋漓尽致,并没有解决移植细胞生存率低及移植细胞定植困难的问题。组织工程支架的适时出现解决了该类问题,它可以通过给移植细胞构建一个适合的微环境,使间充质干细胞的诸多优点得以表现[66]。近来关于组织工程支架的研究不胜枚举,对于组织工程支架的利用也是趋于灵活多样,它们兼顾解决细胞移植中定植困难的同时,还增加了可促进间充质干细胞发挥优势的营养物质,如神经胶原调节支架、肽链水凝胶支架、脱细胞脊髓支架、神经营养支架等[67-70]。这些组织工程支架具有良好的神经诱导性,而且支架外层是强度大、降解速率慢的可降解材料,这为神经修复提供了长期稳定的微环境,同时避免了聚丙烯腈、聚氯乙烯支架在修复过程中造成的神经卡压。"

| [1] Ge L, Arul K, Ikpeze T, et al. Traumatic and Nontraumatic Spinal Cord Injuries. World Neurosurg. 2018;111:e142-e148.[2] Tafida MA, Wagatsuma Y, Ma E, et al. Descriptive epidemiology of traumatic spinal injury in Japan. J Orthop Sci. 2018;23(2): 273-276.[3] Ning GZ, Wu Q, Li YL, et al. Epidemiology of traumatic spinal cord injury in Asia: a systematic review. J Spinal Cord Med. 2012;35(4): 229-239.[4] Chen R, Liu X, Han S, et al. Current epidemiological profile and features of traumatic spinal cord injury in Heilongjiang province, Northeast China: implications for monitoring and control. Spinal Cord. 2017;55(4):399-404.[5] Sekhon LH, Fehlings MG. Epidemiology, demographics, and pathophysiology of acute spinal cord injury. Spine (Phila Pa 1976). 2001;26(24 Suppl):S2-12.[6] National Spinal Cord Injury Statistical Center. Spinal cord injury facts and figures at a glance. J Spinal Cord Med. 2013;36(1):1-2.[7] Mothe AJ, Tator CH. Advances in stem cell therapy for spinal cord injury. J Clin Invest. 2012;122(11):3824-3834.[8] Leal-Filho MB. Spinal cord injury: From inflammation to glial scar. Surg Neurol Int. 2011;2:112.[9] Fehlings MG, Tighe A. Spinal cord injury: the promise of translational research. Neurosurg Focus. 2008;25(5):E1.[10] 张海燕,杨朝阳,李晓光. 大鼠脊髓损伤后胶质瘢痕形成的病理学规律[J]. 中国康复理论与实践, 2011, 17(3):215-218.[11] Bracken MB. Steroids for acute spinal cord injury. Cochrane Database Syst Rev. 2012;1:CD001046.[12] Jia YF, Gao HL, Ma LJ, et al. Effect of nimodipine on rat spinal cord injury. Genet Mol Res. 2015;14(1):1269-1276.[13] Friedenstein AJ, Chailakhyan RK, Gerasimov UV. Bone marrow osteogenic stem cells: in vitro cultivation and transplantation in diffusion chambers. Cell Tissue Kinet. 1987;20(3):263-272.[14] Jiang Y, Jahagirdar BN, Reinhardt RL, et al. Pluripotency of mesenchymal stem cells derived from adult marrow. Nature. 2002; 418(6893):41-49.[15] Chamberlain G, Fox J, Ashton B, et al. Concise review: mesenchymal stem cells: their phenotype, differentiation capacity, immunological features, and potential for homing. Stem Cells. 2007;25(11):2739-2749.[16] Dominici M, Le Blanc K, Mueller I, et al. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy. 2006;8(4):315-317.[17] Lou G, Chen Z, Zheng M, et al. Mesenchymal stem cell-derived exosomes as a new therapeutic strategy for liver diseases. Exp Mol Med. 2017;49(6):e346.[18] Börger V, Bremer M, Ferrer-Tur R, et al. Mesenchymal Stem/Stromal Cell-Derived Extracellular Vesicles and Their Potential as Novel Immunomodulatory Therapeutic Agents. Int J Mol Sci. 2017;18(7): E1450.[19] Aqmasheh S, Shamsasanjan K, Akbarzadehlaleh P, et al. Effects of Mesenchymal Stem Cell Derivatives on Hematopoiesis and Hematopoietic Stem Cells. Adv Pharm Bull. 2017;7(2):165-177.[20] Liang L, Li Z, Ma T, et al. Transplantation of Human Placenta-Derived Mesenchymal Stem Cells Alleviates Critical Limb Ischemia in Diabetic Nude Rats. Cell Transplant. 2017; 26(1):45-61.[21] Han ZC, Du WJ, Han ZB, et al. New insights into the heterogeneity and functional diversity of human mesenchymal stem cells. Biomed Mater Eng. 2017;28(s1):S29-S45.[22] Shende P, Subedi M. Pathophysiology, mechanisms and applications of mesenchymal stem cells for the treatment of spinal cord injury. Biomed Pharmacother. 2017;91:693-706.[23] Gu C, Li H, Wang C, et al. Bone marrow mesenchymal stem cells decrease CHOP expression and neuronal apoptosis after spinal cord injury. Neurosci Lett. 2017;636:282-289.[24] Zhukareva V, Obrocka M, Houle JD, et al. Secretion profile of human bone marrow stromal cells: donor variability and response to inflammatory stimuli. Cytokine. 2010;50(3):317-321.[25] Morita T, Sasaki M, Kataoka-Sasaki Y, et al. Intravenous infusion of mesenchymal stem cells promotes functional recovery in a model of chronic spinal cord injury. Neuroscience. 2016;335: 221-231.[26] Kim Y, Jo SH, Kim WH, et al. Antioxidant and anti-inflammatory effects of intravenously injected adipose derived mesenchymal stem cells in dogs with acute spinal cord injury. Stem Cell Res Ther. 2015;6:229.[27] Li J, Guo W, Xiong M, et al. Erythropoietin facilitates the recruitment of bone marrow mesenchymal stem cells to sites of spinal cord injury. Exp Ther Med. 2017;13(5):1806-1812.[28] Escalhão CCM, Ramos IP, Hochman-Mendez C, et al. Safety of Allogeneic Canine Adipose Tissue-Derived Mesenchymal Stem Cell Intraspinal Transplantation in Dogs with Chronic Spinal Cord Injury. Stem Cells Int. 2017;2017:3053759.[29] Tang L, Lu X, Zhu R, et al. Adipose-Derived Stem Cells Expressing the Neurogenin-2 Promote Functional Recovery After Spinal Cord Injury in Rat. Cell Mol Neurobiol. 2016;36(5):657-667.[30] Wang GD, Liu YX, Wang X, et al. The SDF-1/CXCR4 axis promotes recovery after spinal cord injury by mediating bone marrow-derived from mesenchymal stem cells. Oncotarget. 2017; 8(7):11629-11640.[31] 谢桂琴,徐晓静,张焕相. 基质细胞衍生因子-1α对间充质干细胞迁移的影响[J]. 江苏大学学报:医学版, 2014, 24(4):277-282.[32] Wei GJ, An G, Shi ZW, et al. Suppression of MicroRNA-383 Enhances Therapeutic Potential of Human Bone-Marrow-Derived Mesenchymal Stem Cells in Treating Spinal Cord Injury via GDNF. Cell Physiol Biochem. 2017;41(4):1435-1444.[33] 王振宇,刘文革,黄朱宋. bFGF基因修饰骨髓间充质干细胞移植对急性脊髓损伤大鼠神经元的影响[J]. 福建医药杂志, 2010, 32(6):8-10.[34] Lee SH, Kim Y, Rhew D, et al. Impact of local injection of brain-derived neurotrophic factor-expressing mesenchymal stromal cells (MSCs) combined with intravenous MSC delivery in a canine model of chronic spinal cord injury. Cytotherapy. 2016 Oct 28. [Epub ahead of print][35] Lu Y, Gao H, Zhang M, et al. Glial Cell Line-Derived Neurotrophic Factor-Transfected Placenta-Derived Versus Bone Marrow-Derived Mesenchymal Cells for Treating Spinal Cord Injury. Med Sci Monit. 2017;23:1800-1811.[36] 代楠. miR-9、miR-124通过靶向PTBP1促进人脐带间充质干细胞成神经转分化的机制研究[D]. 苏州:苏州大学, 2017.[37] 孙孟坊,王丰,金孟浩, 等. miR-124修饰的骨髓间充质干细胞对大鼠脊髓损伤的作用研究[J]. 全科医学临床与教育, 2017, 15(4):372-375.[38] Zhao Y, Jiang H, Liu XW, et al. MiR-124 promotes bone marrow mesenchymal stem cells differentiation into neurogenic cells for accelerating recovery in the spinal cord injury. Tissue Cell. 2015; 47(2):140-146.[39] 朱奇. 慢病毒介导GAP-43基因修饰骨髓间充质干细胞移植治疗大鼠视神经损伤的实验研究[D]. 长春:吉林大学, 2014.[40] Pu Y, Meng K, Gu C, et al. Thrombospondin-1 modified bone marrow mesenchymal stem cells (BMSCs) promote neurite outgrowth and functional recovery in rats with spinal cord injury. Oncotarget. 2017;8(56):96276-96289.[41] Park YM, Han SH, Seo SK, et al. Restorative benefits of transplanting human mesenchymal stromal cells overexpressing arginine decarboxylase genes after spinal cord injury. Cytotherapy. 2015;17(1):25-37.[42] Stewart AN, Kendziorski G, Deak ZM, et al. Co-transplantation of mesenchymal and neural stem cells and overexpressing stromal-derived factor-1 for treating spinal cord injury. Brain Res. 2017;1672:91-105.[43] Yousefifard M, Nasirinezhad F, Shardi Manaheji H, et al. Human bone marrow-derived and umbilical cord-derived mesenchymal stem cells for alleviating neuropathic pain in a spinal cord injury model. Stem Cell Res Ther. 2016;7:36.[44] Oraee-Yazdani S, Hafizi M, Atashi A, et al. Co-transplantation of autologous bone marrow mesenchymal stem cells and Schwann cells through cerebral spinal fluid for the treatment of patients with chronic spinal cord injury: safety and possible outcome. Spinal Cord. 2016;54(2):102-109.[45] Wu S, Cui G, Shao H, et al. The Cotransplantation of Olfactory Ensheathing Cells with Bone Marrow Mesenchymal Stem Cells Exerts Antiapoptotic Effects in Adult Rats after Spinal Cord Injury. Stem Cells Int. 2015;2015:516215.[46] Lindsay SL, Toft A, Griffin J, et al. Human olfactory mesenchymal stromal cell transplants promote remyelination and earlier improvement in gait co-ordination after spinal cord injury. Glia. 2017;65(4):639-656.[47] Nicola FDC, Marques MR, Odorcyk F, et al. Neuroprotector effect of stem cells from human exfoliated deciduous teeth transplanted after traumatic spinal cord injury involves inhibition of early neuronal apoptosis. Brain Res. 2017;1663:95-105.[48] Yang W, Yang Y, Yang JY, et al. Treatment with bone marrow mesenchymal stem cells combined with plumbagin alleviates spinal cord injury by affecting oxidative stress, inflammation, apoptotis and the activation of the Nrf2 pathway. Int J Mol Med. 2016;37(4):1075-1082.[49] Lee SH, Kim Y, Rhew D, et al. Effect of the combination of mesenchymal stromal cells and chondroitinase ABC on chronic spinal cord injury. Cytotherapy. 2015;17(10):1374-1383.[50] Chen D, Zeng W, Fu Y, et al. Bone marrow mesenchymal stem cells combined with minocycline improve spinal cord injury in a rat model. Int J Clin Exp Pathol. 2015;8(10):11957-11969.[51] Rahimi A, Amiri I, Roushandeh AM, et al. Sublethal concentration of H2O2 enhances the protective effect of mesenchymal stem cells in rat model of spinal cord injury. Biotechnol Lett. 2018; 40(3):609-615.[52] Tan JW, Wang KY, Liao GJ, et al. Neuroprotective effect of methylprednisolone combined with placenta-derived mesenchymal stem cell in rabbit model of spinal cord injury. Int J Clin Exp Pathol. 2015;8(8):8976-8982.[53] Yang XM, Cheng YY, Zhang ZL, et al. Role of Methylprednisolone in Treatment of Spinal Cord Injured with Bone Marrow Mesenchymal Stem Cells Transplantation in Rats and Its Effect on the Expressions of Tumor Necrosis Factor-α and Interleukin-1β. Zhongguo Yi Xue Ke Xue Yuan Xue Bao. 2017;39(5):615-622.[54] Libro R, Bramanti P, Mazzon E. The combined strategy of mesenchymal stem cells and tissue-engineered scaffolds for spinal cord injury regeneration. Exp Ther Med. 2017;14(4): 3355-3368.[55] 徐志华,余勤,白月双,等. 补阳还五汤联合鞘内注射间充质干细胞治疗脊髓损伤的研究[J]. 中华中医药学刊,2012,30(12):2726-2729.[56] 吕华兵,牛伟,卜志勇,等. 红花黄素对骨髓间充质干细胞移植修复大鼠脊髓损伤的影响[J]. 湖北医药学院学报,2011,30(3):243-247.[57] 吴晓明,高文山,王静,等. 盐酸川芎嗪联合骨髓间充质干细胞移植对脊髓损伤模型大鼠的神经保护[J]. 中国组织工程研究, 2016, 20(1): 95-101.[58] 何珏,王天科. 丙泊酚联合骨髓间充质干细胞移植对脊髓损伤大鼠后肢功能及电生理的影响[J]. 中国组织工程研究, 2015, 19(41): 6659-6664.[59] 哈小琴,邓芝云,肖娜娜,等. 红景天联合KGF/HIF-1修饰的骨髓间充质干细胞对低氧条件下大鼠脊髓损伤修复的影响[J]. 解放军医药杂志,2015,27(10):15-19.[60] 杨迎桂,郭文家,买志福,等. 角质生长因子、缺氧诱导因子修饰的骨髓间充质干细胞对大鼠脊髓损伤修复的影响[J]. 解放军医药杂志, 2015, 27(10):20-24.[61] 何珏,王天科. 丙泊酚联合骨髓间充质干细胞移植对脊髓损伤大鼠后肢功能及电生理的影响[J]. 中国组织工程研究, 2015, 19(41): 6659-6664.[62] 张建军,王东,刘泽朋,等. 法舒地尔与骨髓间充质干细胞移植治疗大鼠脊髓损伤:有协同效应吗[J]. 中国组织工程研究, 2009, 13(45): 8901-8905.[63] 张振梁,杨新明,孟宪勇,等. 丙戊酸联合骨髓间充质干细胞移植对大鼠脊髓损伤的影响[J]. 中国临床药理学杂志, 2017, 33(8):714-717.[64] 张建军,梁辉,盛学东,等. 他克莫司对骨髓间充质干细胞移植治疗大鼠脊髓损伤的影响[J]. 国际神经病学神经外科学杂志, 2008, 35(5): 398-402.[65] 张占修,李志远,申勇. 乌司他丁联合脐带间充质干细胞移植脊髓损伤大鼠后肢运动功能和运动诱发电位的变化[J].中国组织工程研究, 2012,16(27):5076-5080.[66] Derakhshanrad N, Saberi H, Tayebi Meybodi K, et al. Case Report: Combination Therapy with Mesenchymal Stem Cells and Granulocyte-Colony Stimulating Factor in a Case of Spinal Cord Injury. Basic Clin Neurosci. 2015;6(4):299-305.[67] Peng Z, Gao W, Yue B, et al. Promotion of neurological recovery in rat spinal cord injury by mesenchymal stem cells loaded on nerve-guided collagen scaffold through increasing alternatively activated macrophage polarization. J Tissue Eng Regen Med. 2018;12(3):e1725-e1736.[68] Li LM, Han M, Jiang XC, et al. Peptide-Tethered Hydrogel Scaffold Promotes Recovery from Spinal Cord Transection via Synergism with Mesenchymal Stem Cells. ACS Appl Mater Interfaces. 2017;9(4):3330-3342.[69] Zhao Y, Tang F, Xiao Z, et al. Clinical Study of NeuroRegen Scaffold Combined With Human Mesenchymal Stem Cells for the Repair of Chronic Complete Spinal Cord Injury. Cell Transplant. 2017;26(5):891-900.[70] Wang N, Xiao Z, Zhao Y, et al. Collagen scaffold combined with human umbilical cord-derived mesenchymal stem cells promote functional recovery after scar resection in rats with chronic spinal cord injury. J Tissue Eng Regen Med. 2018;12(2):e1154-e1163.[71] Mortazavi Y, Sheikhsaran F, Khamisipour GK, et al. The Evaluation of Nerve Growth Factor Over Expression on Neural Lineage Specific Genes in Human Mesenchymal Stem Cells. Cell J. 2016;18(2):189-196. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [3] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [4] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [5] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [6] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [7] | Fan Quanbao, Luo Huina, Wang Bingyun, Chen Shengfeng, Cui Lianxu, Jiang Wenkang, Zhao Mingming, Wang Jingjing, Luo Dongzhang, Chen Zhisheng, Bai Yinshan, Liu Canying, Zhang Hui. Biological characteristics of canine adipose-derived mesenchymal stem cells cultured in hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1002-1007. |
| [8] | Geng Yao, Yin Zhiliang, Li Xingping, Xiao Dongqin, Hou Weiguang. Role of hsa-miRNA-223-3p in regulating osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1008-1013. |
| [9] | Lun Zhigang, Jin Jing, Wang Tianyan, Li Aimin. Effect of peroxiredoxin 6 on proliferation and differentiation of bone marrow mesenchymal stem cells into neural lineage in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1014-1018. |
| [10] | Zhu Xuefen, Huang Cheng, Ding Jian, Dai Yongping, Liu Yuanbing, Le Lixiang, Wang Liangliang, Yang Jiandong. Mechanism of bone marrow mesenchymal stem cells differentiation into functional neurons induced by glial cell line derived neurotrophic factor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1019-1025. |
| [11] | Duan Liyun, Cao Xiaocang. Human placenta mesenchymal stem cells-derived extracellular vesicles regulate collagen deposition in intestinal mucosa of mice with colitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1026-1031. |
| [12] | Pei Lili, Sun Guicai, Wang Di. Salvianolic acid B inhibits oxidative damage of bone marrow mesenchymal stem cells and promotes differentiation into cardiomyocytes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1032-1036. |
| [13] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [14] | Wang Shiqi, Zhang Jinsheng. Effects of Chinese medicine on proliferation, differentiation and aging of bone marrow mesenchymal stem cells regulating ischemia-hypoxia microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1129-1134. |
| [15] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||