Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (32): 5117-5122.doi: 10.3969/j.issn.2095-4344.0387
Previous Articles Next Articles
Numerical analysis of fluid field changes in articular cartilage with micro-defects under compressive load
Li Xiao-ming1, 2, Men Yu-tao1, 2, Zhang Chun-qiu1, 2
- 1Tianjin Key Laboratory for Advanced Mechatronic System Design and Intelligent Control, 2National Demonstration Center for Experimental Mechanical and Electrical Engineering Education, Tianjin University of Technology, Tianjin 300384, China
-
Received:2018-01-11Online:2018-11-18Published:2018-11-18 -
Contact:Men Yu-tao, PhD, Lecturer, Tianjin Key Laboratory for Advanced Mechatronic System Design and Intelligent Control, National Demonstration Center for Experimental Mechanical and Electrical Engineering Education, Tianjin University of Technology, Tianjin 300384, China -
About author:Li Xiao-ming, Master candidate, Tianjin Key Laboratory for Advanced Mechatronic System Design and Intelligent Control, National Demonstration Center for Experimental Mechanical and Electrical Engineering Education, Tianjin University of Technology, Tianjin 300384, China -
Supported by:the National Natural Science Foundation of China, No. 11402171, 11672208, 11432016 and 81741141
CLC Number:
Cite this article
Li Xiao-ming1, 2, Men Yu-tao1, 2, Zhang Chun-qiu1, 2. Numerical analysis of fluid field changes in articular cartilage with micro-defects under compressive load [J]. Chinese Journal of Tissue Engineering Research, 2018, 22(32): 5117-5122.
share this article
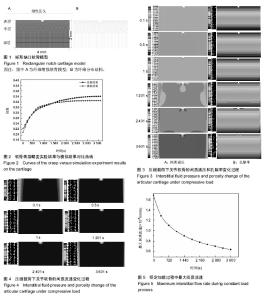
该文主要从间质液压力、间质液流速以及间质液流动轨迹等力学性能来研究微缺损关节软骨的流体场变化。 2.1 间质液压力 在软骨承受压缩载荷过程中,首先做出反应的并不是关节软骨的固体基质,而是软骨组织内液相的间质液,称之为间质液压效应[34-36]。这种效应与它的多孔特性是密切相关的。由于关节软骨内部的孔隙极小,导致它的渗透率只有10-15 m4/Ns,当关节软骨刚受到压力时,软骨组织中的间质液不能快速地从承载区流出,反而形成了液压来承受软骨表面的压力。又由于渗透率的变化缓慢,所以这种液压效应可以维持很长一段时间。这样不但很大程度上分担了软骨表面的载荷,阻碍了软骨发生大的变形,而且减少了软骨表面间固体部分的接触,从而保证了摩擦系数在关节在运动过程始终可以保持的很低[37-39]。因此,研究关节软骨损伤后间质液压的分布状况对于骨关节炎的治疗和预防具有重要意义。 图3为微缺损关节软骨不同压缩时间下的间质液压和孔隙率的分布云图,其中0-1 s为线性加载过程,1-3 601 s为恒定加载过程。从图3A可以看出整个加载过载中间质液压的最大值始终位于缺损底部的两侧底角处,这是由于此处存在挤压应力,使得缺损处的孔隙率变小(见图3B),更大程度上的限制了间质流动,进而在此处形成了极高的间质液压。孔隙率变小导致的间质流动受阻,一方面减少了软骨细胞代谢所需营养物质及其分泌的各种生物分子的流失;另一方面间质液压的增大,使得此处的承载能力提高,进而可以阻碍损伤向深处扩展,这其实可以看作是关节软骨在损伤后作出的自我保护。此外,从图3A可以看出缺损边缘处间质液压较低,这是由于缺损边缘处基质被拉伸,孔隙率变大,间质流动加快,从而间质液压变小。由上述分析可知,孔隙率变大,间质流动加快,间质压力变小,不利于软骨自身的修复,反而会进一步促使损伤的扩展。 从图3A中可以看出在线性加载过程中,关节软骨整体的间质压力随着时间的推移逐渐增大,这是由于随着载荷的增大,软骨内部压力梯度也逐渐增大,间质流动有加快的趋势,而软骨内部极小的孔隙率却限制了这种趋势,进而在软骨内形成的间质压力也逐渐增大;在恒定加载过程中,关节软骨整体的间质压力随着时间的推移逐渐减小,这是由于随着时间的推移软骨间质液逐渐流出,间质液压效果衰竭,载荷逐步转由固相部分承担。在这个过程中,胶原纤维和基质开始承受载荷,与软骨表层平行走向的胶原纤维就像鼓面的蒙皮一样,使软骨面上载荷的分布趋于均匀。 2.2 间质液流速 图4显示了微缺损关节软骨不同压缩时间下的间质流速变化云图,从图中可以看出在线性加载阶段,关节软骨整体的间质流速随着时间的推移逐渐增大,这是由于随着载荷的增大,软骨内部压力梯度也逐渐增大,进而间质液的流动速度也逐渐增大;在恒定加载过程中,关节软骨整体的间质流速随着时间的推移逐渐减小,这一"


方面是由于随着时间的推移软骨间质液逐渐流出,软骨内部的间质液不再饱和,另一方面随着时间的推移载荷开始转由固相的胶原纤维承担,软骨间质液受到的压力梯度也逐渐减小,进而导致间质流速逐渐减小。此外,在图中还可以看出在整个加载过程中,软骨缺损边缘的间质流速始终大于周边未缺损部位,这是由于此处存在应力集中,压力梯度大于未缺损部位,同时应力集中使得该出产生了大变形,孔隙率变大,这使得缺损边缘的间质流速增大,进而加速了软骨细胞代谢所需营养物质及其分泌的各种生物分子的流失。 图5为恒定加载过程中软骨间质液的最大流速变化情况。当线性加载结束时,关节软骨的间质流速达到最大值1.67×10-6 mm/s,而在整个恒定加载过程中,间质流速呈现出指数形式的衰减趋势,这种逐渐趋于平缓的衰减趋势反映出了间质液压效应和固相变形的互补过程。即在线性加载阶段,由于间质液的快速响应机制使得间质流速产生了一个峰值,但在恒定加载过程中,随着间质液的减少,关节软骨开始产生变形,而软骨的变形会压缩软骨内部的蛋白多糖类分子,从而软骨内部的孔隙率减小,孔隙的减小又会反过来阻碍间质液的流出。这种互补机制既能实现营养物质的传递又能延长液相承担载荷的时间。 2.3 间质液流动轨迹 图6是整个加载过程中各个时刻对应的间质流动的方向,从图6A中可以看出整个加载过程中关节软骨未缺损部位的间质流动方向始终朝向软骨两侧,并且随着时间的推移,间质流动的范围由中间向两侧逐渐缩小,最终集中在软骨的两侧区域,这是由于随着时间的推移软骨间质液逐渐向外流出导致的。从图6B(缺口局部放大5倍)中可以看出整个加载过程中缺损部位软骨间质液的流动轨迹表现出现很大的不同,具体表现为软骨间质液始终以缺损底部的两侧底角处的应力集中处为中心向四周流动,尤其向软骨表面和中间的间质流动更为明显,这是由于应力集中处的间质液压高于四周区域(见图6A),由于液压差的存在,软骨间质液自然从高压处流向低压处,而表面和中间的间质流动更为明显是因为缺损边缘处基质的孔隙率较大(见图3B)。这种现象加速了软骨细胞代谢所需营养物质及其分泌的各种生物分子的流失。"

| [1] 翟文杰,翟中勇. 软骨组织无约束压缩的有限元分析[J]. 医用生物力学, 2012,27(6): 630-635.[2] Le TK, Montejano LB, Cao Z, et al. Health care costs in US patients with and without a diagnosis of osteoarthritis. J Pain Res. 2012;5:23-30.[3] Mow VC, Huiskes R. 骨科生物力学暨力学生物学[M]. 3版.汤亭亭等,译.济南:山东科学出版社,2009.[4] 周海宇.关节软骨的生物摩擦学机理研究[D]. 上海:上海交通大学,2014.[5] Mow VC, Kuei SC, Lai WM, et al.Biphasic creep and stress relaxation of articular cartilage in compression? Theory and experiments.J Biomech Eng. 1980;102(1):73-84.[6] Frank EH, Grodzinsky AJ.Cartilage electromechanics--I. Electrokinetic transduction and the effects of electrolyte pH and ionic strength.J Biomech.1987;20(6):615-627. [7] Frank EH, Grodzinsky AJ. Cartilage electromechanics--II. A continuum model of cartilage electrokinetics and correlation with experiments. J Biomech.1987;20(6):629-639.[8] Korhonen RK, Laasanen MS, Töyräs J, et al. Fibril reinforced poroelastic model predicts specifically mechanical behavior of normal, proteoglycan depleted and collagen degraded articular cartilage. J Biomech. 2003;36(9):1373-1379.[9] Li LP, Buschmann MD, Shirazi-Adl A. A fibril reinforced nonhomogeneous poroelastic model for articular cartilage: inhomogeneous response in unconfined compression. J Biomech. 2000;33(12):1533-1541. [10] Moger CJ, Arkill KP, Ellis RE, et al. Cartilage collagen matrix reorientation and displacement in response to surface loading. J Biomech Eng. 2009;131(3):031008.[11] 周海宇,李元超,王成焘. 力学参数随深度变化的关节软骨数值模拟[J]. 医用生物力学, 2011, 26(2): 168-172.[12] Lai WM, Hou JS, Mow VC. A triphasic theory for the swelling and deformation behaviors of articular cartilage. J Biomech Eng. 1991; 113(3):245-258. [13] Abazari A, Elliott JA, Law GK,et al. A biomechanical triphasic approach to the transport of nondilute solutions in articular cartilage. Biophys J. 2009;97(12):3054-3064.[14] 蒋彦龙,门玉涛,陈玲,等. 矩形微缺损关节软骨在压缩载荷下损伤演化的数值分析[J]. 医用生物力学. 2017.32(2):62-68.[15] 田倩倩. 缺损软骨损伤演化过程的研究[D].天津:天津理工大学,2014.[16] 伏虎,陈玲,门玉涛,等. 缺损软骨在滚压载荷下的实验与有限元分析[J]. 山东大学学报(理学版), 2017,52(5):37-40.[17] Accardi MA, Mccullen SD, Callanan A, et al. Effects of fiber orientation on the frictional properties and damage of regenerative articular cartilage surfaces. Tissue Eng Part A. 2013;19(19-20):2300-2310.[18] 孟迪,张春秋,刘海英,等. 软骨缺损形状对组织工程修复区的力学影响[J]. 生物医学工程与临床, 2016,33(1):9-14.[19] Xiao PP, Gao LL, Liu ZD, et al. The Micro-Mechanical Behavior of Articular Cartilage under Continuous Sliding Load. App Mech Mater.2013;395-396:654-657.[20] Men YT, Jiang YL, Chen L, et al. On mechanical mechanism of damage evolution in articular cartilage. Mater Sci Eng C Mater Biol Appl. 2017;78:79-87. [21] Wilson W, van Burken C, van Donkelaar C, et al. Causes of mechanically induced collagen damage in articular cartilage. J Orthop Res. 2006;24(2):220-228.[22] Klika V, Gaffney EA, Chen YC, et al. An overview of multiphase cartilage mechanical modelling and its role in understanding function and pathology. J Mech Behav Biomed Mater. 2016;62:139-157.[23] Hay ED.Extracellular matrix.J Cell Biol. 1981;91(3 Pt 2):205s-223s.[24] Tian QQ, Ye JD, Li X, et al. Numerical simulation to the process of damage evolution of defect cartilage. App Mecha Mater. 2013;397-400 (6): 629-632.[25] 孟迪. 压缩载荷下软骨缺损修复后力学状态的研究[D]. 天津:天津理工大学, 2016.[26] Zhou Y, Liu HY, Men YT, et al. Numerical Study on Mechanical Behavior of Tissue-Engineering Repaired Cartilage in Sliding Load Condition. App Mecha Mater.2014;441:598-601.[27] Accardi MA, Dini D, Cann PM. Experimental and numerical investigation of the behaviour of articular cartilage under shear loading—Interstitial fluid pressurisation and lubrication mechanisms. Tribology International. 2011;44(5):565-578.[28] Sakai N, Hagihara Y, Furusawa T, et al. Analysis of biphasic lubrication of articular cartilage loaded by cylindrical indenter.Tribology International. 2012;46(1):225-236.[29] 周玉. 修复的缺损软骨在滑动条件下力学性质的研究[D]. 天津:天津理工大学, 2014.[30] van der Voet A. A comparison of finite element codes for the solution of biphasic poroelastic problems. Proc Inst Mech Eng H. 1997;211(2): 209-211.[31] 刘馨燕,严波,刘海京. 关节软骨两相多孔介质非线性模型的有限元方法[J]. 应用力学学报, 2004, 21(2):61-65.[32] 张玉,俞然刚.手工计算与仿真技术在土木工程教学中的应用[J].中国电力教育, 2013,29(31):79-81.[33] Gu WY, Yao H, Huang CY, et al. New insight into deformation- dependent hydraulic permeability of gels and cartilage, and dynamic behavior of agarose gels in confined compression. J Biomech. 2003; 36(4):593-598.[34] Suh JK, Li Z, Woo SL.Dynamic behavior of a biphasic cartilage model under cyclic compressive loading.J Biomech. 1995;28(4):357-364. [35] Gadomski A, Be?dowski P, RubÌ JM, et al. Some conceptual thoughts toward nanoscale oriented friction in a model of articular cartilage. Math Biosci. 2013;244(2):188-200.[36] McCann, Laura. Tribological Investigation of Articular Cartilage Substitution in the Medial Compartmental Knee. University of Leeds, 2009.[37] Ateshian GA.The role of interstitial fluid pressurization in articular cartilage lubrication.J Biomech. 2009;42(9):1163-1176. [38] Neu CP, Reddi AH, Komvopoulos K, et al. Increased friction coefficient and superficial zone protein expression in patients with advanced osteoarthritis. Arthritis Rheum.2010;62(9):2680-2687.[39] Grad S, Loparic M, Peter R , et al. Sliding motion modulates stiffness and friction coefficient at the surface of tissue engineered cartilage. Osteoarthritis Cartilage. 2012;20(4):288-295.[40] 段航天. 压缩载荷作用下关节软骨修复区细胞的力学环境研究[D]. 天津:天津理工大学, 2017.[41] Sakai N, Hashimoto C, Yarimitsu S, et al. A functional effect of the superficial mechanical properties of articular cartilage as a load bearing system in a sliding condition. Biosurface Biotribology. 2016; 2(1):26-39. |
| [1] | Hu Kai, Qiao Xiaohong, Zhang Yonghong, Wang Dong, Qin Sihe. Treatment of displaced intra-articular calcaneal fractures with cannulated screws and plates: a meta-analysis of 15 randomized controlled trials [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1465-1470. |
| [2] | Wu Xun, Meng Juanhong, Zhang Jianyun, Wang Liang. Concentrated growth factors in the repair of a full-thickness condylar cartilage defect in a rabbit [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1166-1171. |
| [3] | Li Jiacheng, Liang Xuezhen, Liu Jinbao, Xu Bo, Li Gang. Differential mRNA expression profile and competitive endogenous RNA regulatory network in osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1212-1217. |
| [4] | Geng Qiudong, Ge Haiya, Wang Heming, Li Nan. Role and mechanism of Guilu Erxianjiao in treatment of osteoarthritis based on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1229-1236. |
| [5] | He Xiangzhong, Chen Haiyun, Liu Jun, Lü Yang, Pan Jianke, Yang Wenbin, He Jingwen, Huang Junhan. Platelet-rich plasma combined with microfracture versus microfracture in the treatment of knee cartilage lesions: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 964-969. |
| [6] | Liu Xin, Yan Feihua, Hong Kunhao. Delaying cartilage degeneration by regulating the expression of aquaporins in rats with knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 668-673. |
| [7] | Xie Chongxin, Zhang Lei. Comparison of knee degeneration after anterior cruciate ligament reconstruction with or without remnant preservation [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 735-740. |
| [8] | Deng Zhenhan, Huang Yong, Xiao Lulu, Chen Yulin, Zhu Weimin, Lu Wei, Wang Daping. Role and application of bone morphogenetic proteins in articular cartilage regeneration [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 798-806. |
| [9] | Chen Lei, Zheng Rui, Jie Yongsheng, Qi Hui, Sun Lei, Shu Xiong. In vitro evaluation of adipose-derived stromal vascular fraction combined with osteochondral integrated scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3487-3492. |
| [10] | Tian Guangzhao, Yang Zhen, Zha Kangkang, Sun Zhiqiang, Li Xu, Sui Xiang, Huang Jingxiang, Guo Quanyi, Liu Shuyun. Regulatory effect of decellularized cartilage matrix on macrophage polarization [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3545-3550. |
| [11] | Ren Wenbo, Liao Yuanpeng. Visualization analysis of traumatic osteoarthritis research hotspots and content based on CiteSpace [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(21): 3374-3381. |
| [12] | Song Shilei, Chen Yueping, Zhang Xiaoyun, Li Shibin, Lai Yu, Zhou Yi. Potential molecular mechanism of Wuling powder in treating osteoarthritis based on network pharmacology and molecular docking [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(20): 3185-3193. |
| [13] | Wei Jinqiang, Huang Dengcheng, Cao Xuewei, Zhou Jianwei, Sun He, Li Zehui. Analysis of researches on TCM treatments for cartilage diseases in recent 20 years by mapping knowledge domains [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(20): 3202-3209. |
| [14] | Lu Mingfeng, Zhao Lilian, Xing Jisi, He Lilei, Xu Ting, Wang Changbing. Posttraumatic progression of cartilage degeneration following anterior cruciate ligament reconstruction: a second-look arthroscopic analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(2): 222-227. |
| [15] | Zhang Jianhui, Ma Heran, Tan Yi, Wang Zhihui. Knee injury repair using human adipose-derived mesenchymal stem cells-based scaffold-free three-dimensional gel-like construct in pigs [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(19): 2969-2975. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||