Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (28): 7418-7427.doi: 10.12307/2026.812
Previous Articles Next Articles
Bibliometric analysis of application of artificial intelligence in orthopedic imaging diagnosis
Yue Yuhang1, Xie Liangyu2, Shi Liupeng1, Yin Zuozhen3, Cao Shengnan3, Shi Bin3, Sun Guodong4, 5
- 1School of Medical Information Engineering, Shandong University of Traditional Chinese Medicine, Jinan 250355, Shandong Province, China; 2College of Medical Information and Artificial Intelligence, Shandong First Medical University, Jinan 250117, Shandong Province, China; 3Neck-Shoulder and Lumbocrural Pain Hospital of Shandong First Medical University, Jinan 250062, Shandong Province, China; 4Department of Rehabilitation, Third Affiliated Hospital (Affiliated Hospital of Shandong Academy of Medical Sciences) of Shandong First Medical University, Jinan 250031, Shandong Province, China; 5School of Medical Engineering and Translational Medicine, Tianjin University, Tianjin 300072, China
-
Received:2025-10-15Revised:2025-12-04Online:2026-10-08Published:2026-02-25 -
Contact:Sun Guodong, MD, Chief physician, Professor, Department of Rehabilitation, Third Affiliated Hospital (Affiliated Hospital of Shandong Academy of Medical Sciences) of Shandong First Medical University, Jinan 250031, Shandong Province, China; School of Medical Engineering and Translational Medicine, Tianjin University, Tianjin 300072, China -
About author:Yue Yuhang, MS candidate, School of Medical Information Engineering, Shandong University of Traditional Chinese Medicine, Jinan 250355, Shandong Province, China -
Supported by:Natural Science Foundation of Shandong Province, No. ZR2023QH042 (to CSN); Shandong Provincial Higher Education Youth Innovation and Technology Support Program, No. 2024KJN012 (to CSN); Shandong Provincial Key Research and Development Program, No. 2022CXG020510 (to SB); Jinan Municipal Clinical Medical Science and Technology Innovation Program, No. 202328051 (to CSN); Jinan Municipal Clinical Medical Science and Technology Innovation Program, No. 202430007 (to SGD)
CLC Number:
Cite this article
Yue Yuhang, Xie Liangyu, Shi Liupeng, Yin Zuozhen, Cao Shengnan, Shi Bin, Sun Guodong. Bibliometric analysis of application of artificial intelligence in orthopedic imaging diagnosis[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(28): 7418-7427.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
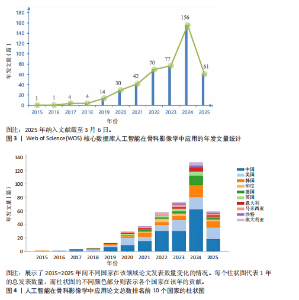
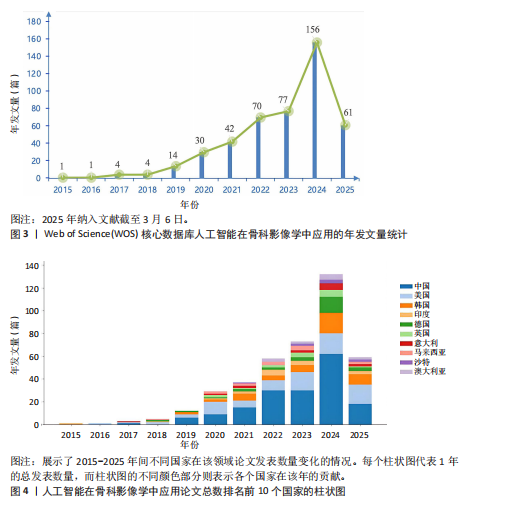
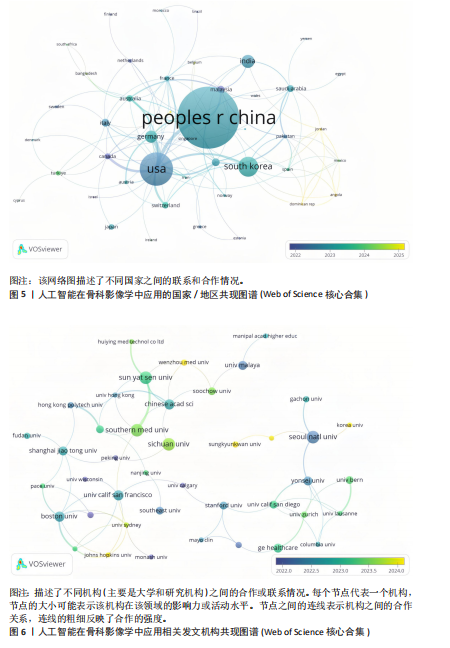
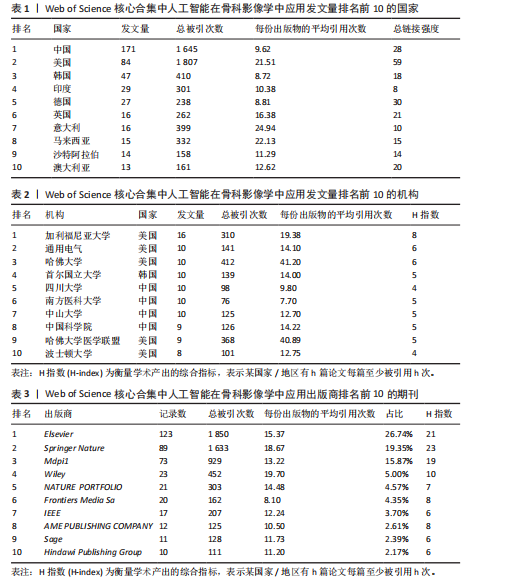
2.1 发文量分析 通过对人工智能在骨科影像学中的应用相关研究的统计分析,可以客观评估该研究领域的发展脉络和受关注程度,预测未来研究趋势。如图3所示,检索时间截至2025年3月,初步检索出1 026篇文献,经筛选清洗后,最终纳入文献共460篇。近年来该领域的累计发文量呈现稳步增长的趋势。具体而言,发表数量从2015年的1篇增加到峰值2024年的156篇。根据图表数据显示,2015-2024年发文量呈现持续增长态势。鉴于2025年仅统计前3个月的发文量已达61篇,可以谨慎预测2025年全年发文量将继续增长,考虑到不确定性因素,可以采用更为保守的估计方法。例如,假设2025年剩余9个月的发文量与2024年同期持平,即每月约18篇,则全年发文量约为175篇。初步数据显示2025年全年发文量有潜力超越2024年。这些结果表明,该领域的研究关注度和国际认知度持续提升。图4进一步显示了 2014-2024年间,全球前10个国家的年发文量均呈现上升趋势,其中中国的年发文量增幅尤为显著。这些结果表明,该领域的研究关注度和国际认知度持续提升。其中印度开展的研究最早,2015年就开始了研究。国家竞争呈现三级梯队格局:中美德三国构成第一梯队,主导技术发展;沙特阿拉伯凭借能源资本转化异军突起,与英国位列第二梯队;澳大利亚、韩国稳定跟进,意大利和马来西亚则代表新兴区域力量。 2.2 国家/地区发文量统计 2015-2025年全球范围内总计52个国家或地区参与了人工智能在骨科领域的研究,亚洲地区的中国和韩国位列其中。颜色越深代表着该国家发文量越多。由图5可知,美国的发文量虽然排在第2位,然而,在被引频次和链接强度上处于领先水平,并且作为最早开展该领域研究的国家在该领域的影响力首屈一指。意大利尽管发文量较少,但其平均引文量高达24.94,为各国最高,这一数据表示虽然意大利在该领域发文量有限,但成果质量颇高,获得了全球专家学者的广泛接受与认可。 在更细化的比较中,中国贡献了460篇文献中的171篇,占37.2%,而美国为84篇。然而,当以被引次数衡量影响力时,中国的总被引次数为1 645,占前10国总被引总和5 713"
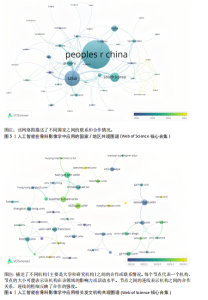
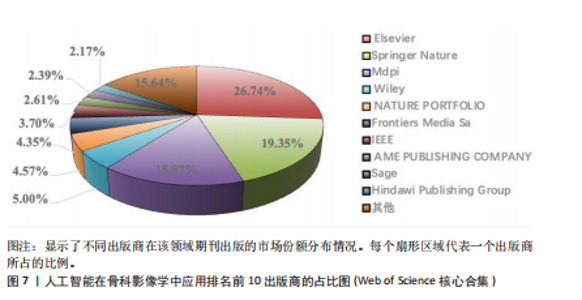
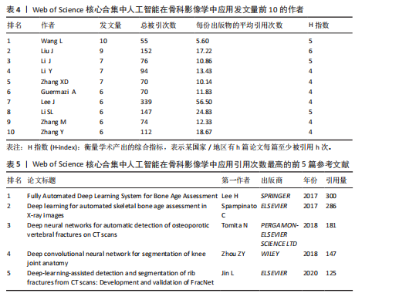
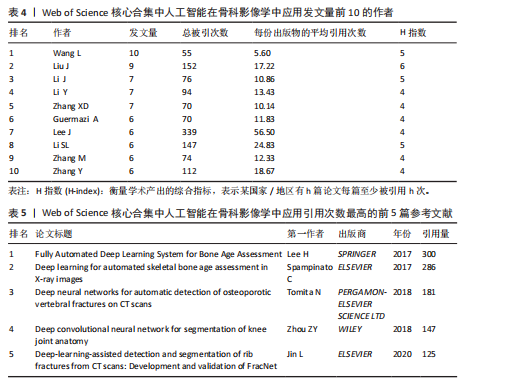
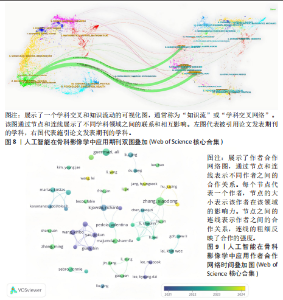
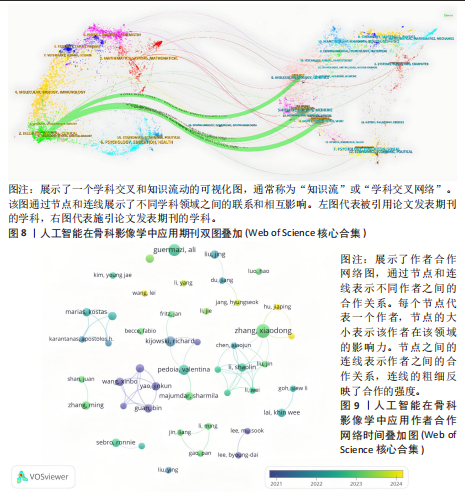
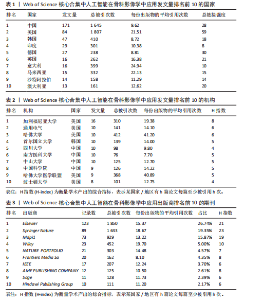
次的28.8%,而美国为1 807次占31.6%。换言之,中国在产量上占优,但在按被引数衡量的影响力上并未同比例领先。同时,中国的平均每篇被引为9.62明显低于美国的21.51,表明影响力方面仍有提升空间。为评估国际合作密度与被引影响力的关系,以国家间的总链接强度来表示国家之间的合作强度。如美国既具较高总链接强度又具较高每份出版物的平均引用次数,说明强国际合作往往能提升可见度与引用,但这种关系并非完全线性的或在所有国家通用,见表1。 2.3 机构发文量分析 如图6所示,各节点之间的连线较少且较细,说明各个机构之间的合作发文较少。通过分析得知人工智能在骨科影像应用领域的研究主要集中在各个国家的高等院校。如表2所示,发文量排名前10的机构中加利福尼亚大学发文量和H指数均位列第一。哈佛大学的平均引用次数高达41.20,表明其成果斐然得到了广泛的认可。南方医科大学的发文量有10篇,但其总引用次数和平均引用次数都仅排名第10,表明其成果并未得到国际社会的广泛认可,需要提高自身的学术水平,加深与其他机构的合作来扩大影响力。由图6可知,图中密集的点代表这些机构合作比较紧密。波士顿大学虽然发表的文章不是最多的,但从图中可知其开展的时间是是最早的一批。加利福尼亚大学、哈佛大学、中山大学等这些机构多集中于2023年。虽然哈佛大学不是发文量最多的,但其被引频次和平均被引批次都是最高的,说明其获得了国际社会的广泛认可,具有深远的影响力。 2.4 发文来源分析 对WOS文献以出版商为节点进行分析,数据显示共有191种期刊发表了相关文献。表3列出了在人工智能在骨科应用领域主要发文来源的相关信息,并按发表数量进行排序。《Elsevier》以163篇(26.74%)的发文量位居该领域的首位,其次是《Springer Nature》(89篇,19.35%)和《Mdpi1》(73篇,15.87%),这两家出版商在H指数方面表现突出,尤其是《Springer Nature》这本期刊在H指数位列首位,体现了该出版商在该领域的较高学术质量和影响力。图7直观地显示出排名前10出版商的占比。利用CiteSpace对460篇文献进行期刊双图叠加分析,如图8所示,左图的标签代表被引用论文发表期刊的学科,其中主要来自数学、医学、生态学、分子生物学、物理学等学科相关的期刊;右图的标签代表施引论文发表期刊的学科,大部分发表于计算机、毒理学、医学、数学、分子生物学相关学科的期刊上。每条线的颜色和粗细都反映了特定的数据集或类别以及流动的数量或强度。这种流线图不仅展示了复杂的数据流动和关联关系,还揭示了跨学科研究的重要性和发展趋势。 2.5 作者发文量分析 通过图9可以直观了解到该领域研究的作者具有较为稳定的合作团队,但各个团队之间的合作较少各节点之间的连线较少且较细,说明各个作者之间的合作发文较少。表4展示了发文量最多的10位作者,其中Wang L和 Liu J分别以10篇和9篇论文位居前两位,显示出他们在该领域的高产出,平"
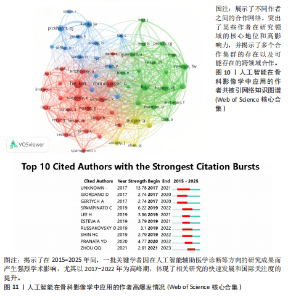
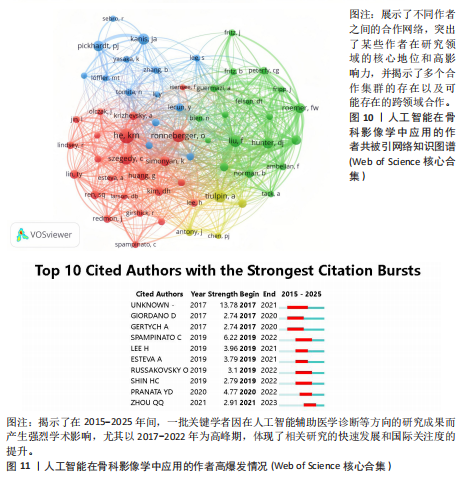
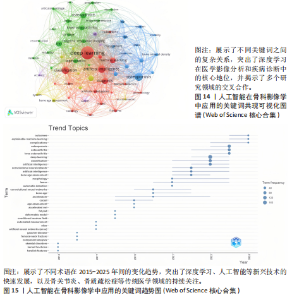
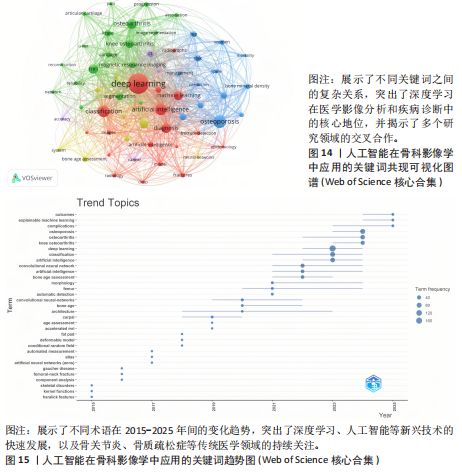
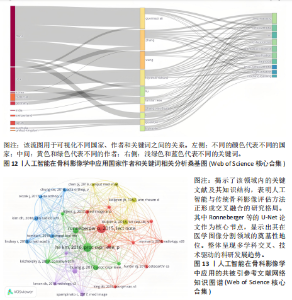
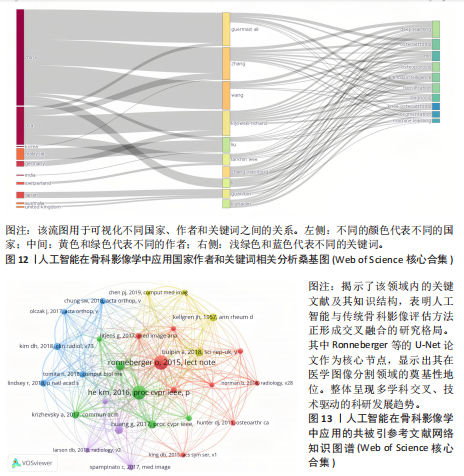
均每篇文献被引用分别为5.60次和17.22。虽然Lee J只有6篇文章,但其被引次数为339次,平均被引频次为56.50位列榜首,表明Lee J在该领域的卓越贡献。图10呈现了被引频次≥50的作者共被引网络。节点大小对应作者被引频次,连线反映共被引关系,颜色区分聚类。结果显示,Ronneberger Olaf作为高被引核心作者,他关于卷积神经网络与医学影像分割的研究形成紧密聚类,凸显该方向在连接骨科临床与人工智能算法中的枢纽作用,推动了影像诊断向智能识别、精准分析和手术规划方向发展(图11)。图12通过桑基图展示了国家、作者与关键词间的复杂关联。中美两国为主要研究力量,深度学习、骨关节炎和分类等是当前热点。该可视化有助于理解领域内研究主体贡献与合作模式,为未来研究方向提供参考。 作者共被引与合作网络分析显示,方法学核心作者与临床骨科派学者之间存在明显的学术桥接结构:Ronneberger 被识别为分割方法学的高被引核心,而Lee J 等临床作者则以较高的平均被引凸显临床应用的重要贡献。 2.6 共被引参考文献分析 文献共被引分析是一种衡量文献间关联程度的方法,当2篇或更多论文同时被一篇或多篇其他论文引用时,它们之间就形成了共被引关系。图13展示了28篇被引用次数不少于20次的参考文献的共被引网络。其中,2017年Lee H等在《SPRINGER》上发表的题目为“Fully Automated Deep Learning System for Bone Age Assessment”的论文引用次数最高,达300次。表5为排名前5位的共被引参考文献,通过分析可以发现深度学习是人工智能在骨科影像研究的核心领域。 2.7 关键词共现分析 关键词是对文章的主题和研究内容的高度概括,是作者对某一具体研究的学术观点的反映,通过对该领域中关键词出现频率的统计,能够判定该领域的研究热点、研究内容的更新速度和学科研究的活力[9]。图14的关键词共现网络显示,deep learning作为中心节点,在骨科影像人工智能研究中占据核心地位。与之紧密相连的convolutional neural networks,segmentation,MRI,CT关键词构成图像分割与多模态成像子网,反映研究者正通过融合二维与三维数据提升结构与软组织识别精度。右侧子网中,osteoporosis、bone mineral density、fracture高频共现,表明基于人工智能的骨质疏松筛查与骨折风险预测已成为新热点。从时间演变看,早期研究偏重feature extraction和network算法探索,近年则转向radiomics和risk精准筛查与预测分析方向。各子网间的高密度连线体现出算法创新与临床应用的深度融合,为智能骨科诊疗系统构建提供了多维度技术基础。表6列出了前20个高频关键词。图15显示,2015-2025年间机器学习、深度学习和人工智能等技术方向关注度持续上升,而骨龄评估、股骨等骨科相关话题则保持相对稳定。 2.8 关键词聚类分析 通过对关键词各属性进行聚类分析,得到了聚类模块值Q=0.473 9、聚类平均轮廓值S=0.745的权重,Q > 0.3则聚类结构显著,S > 0.7则聚类结构合理[10],在关键"
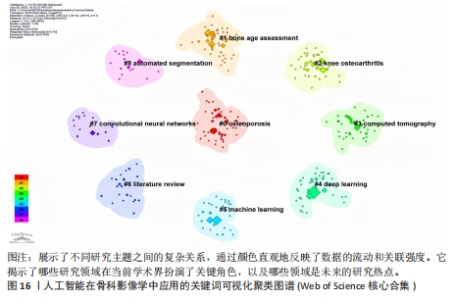
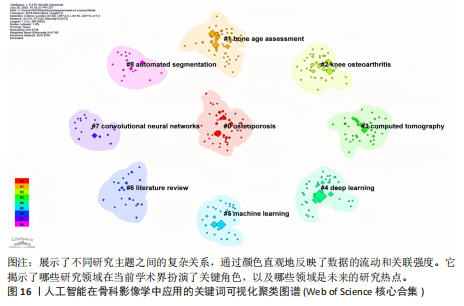
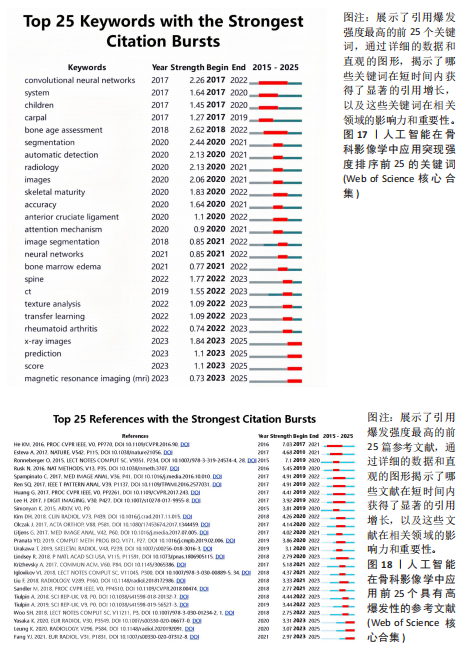
词聚类图谱中每个色块代表1个聚类。聚类的数目越小,则说明聚类越大,所含的关键词数据越多。通过关键词聚类分析,发现人工智能在骨科影像中的应用关键词共有9个聚类[11]。 在图16的关键词聚类图谱中,共识别出9个主题模块。每个色块代表一个高密度研究子领域:浅绿色模块(图16 #0)聚焦于模型结构与卷积神经网络;蓝色模块(图16 #1)对应图像分割任务;橙色模块(图16 #2)涵盖骨龄评估与疾病分级;红色模块(图16 #3)展示多模态融合策略;紫色模块(图16 #4)集中于注意力机制与生成对抗网络等算法创新;浅蓝色模块(图16 #5)关联软体执行器与应变传感器的设计与控制;黄色模块(图16 #6)反映电子皮肤等新型材料与制造工艺;灰色模块(图16 #7)映射外骨骼及可穿戴助力设备的研发;深绿色模块(图16 #8)则强调手部康复、脑卒中辅助等具体康复应用场景[12-13]。结果表明,人工智能在骨科影像学领域的研究正呈现出一种清晰而富有层次的发展轨迹——从最初的算法构建,逐步迈向具体任务的实现,再到多模态数据的融合,最终走向临床实际应用。这一多维扩展路径不仅体现了技术发展的逻辑演进,也反映出研究者在推动技术落地过程中对实用性与有效性的深度思考。可以说人工智能在骨科影像学领域的发展,正在为打造更加高效、可解释、并且真正贴合临床需求的智能诊疗系统打下了坚实的基础。人工智能在骨科影像学中应用关键词聚类代表性的原始文献见表7。 2.9 关键词突现分析 关键词的突现能够体现在短时间内的研究频次激增的状况,揭示了一定时期内研究者们所关心的领域。通过对突现词的分析,能够直观地观察到研究领域中的热点是随着时间推移而发生的变化,而突现词的变化能够对这个领域的发展趋势进行判断[14]。在图17的关键词突现分析中,设置最短爆发持续时间为3年,检测到20个爆发强度最高的关键词。2015–2017年,“convolutional neural networks,bone age assessment,fracture detection”率先突现反映了早期对核心网络架构与平面识别任务的探索;2018–2020年,deep learning,automated segmentation,osteoporosis成为新爆发词,表明研究重心向深度学习驱动的自动分割与骨质疏松筛查转移;进入2021–2025年,“multimodal,transfer learning”强劲突现显示出对多模态融合、模型泛化与可解释性评估的高度关注。图18中2023–2025年又出现一轮技术演进:foundation models、自监督学习以缓解标注瓶颈、联邦学习以支持跨中心隐私协同训练,以及面向临床部署的边缘计算与轻量化网络,正成为推动算法从方法学验证向多中心临床落地的重要路 径[15]。总体来看,领域已由早期的架构与任务优化,平滑过渡到面向临床决策支持的多源信息集成与工程化应用阶段。"
| [1] ASSIS LO, DE PAULA JJ, ASSIS MG, et al. Psychometric properties of the Brazilian version of Pfeffer’s Functional Activities Questionnaire. Front Aging Neurosci. 2014;6:255. [2] 吴惠一,袁琴,张洋,等.1990―2019年我国肌肉骨骼疾病负担分析[J].中华疾病控制杂志, 2023,27(6):655-661. [3] SING CW, LIN TC, BARTHOLOMEW S, et al. Global epidemiology of hip fractures: secular trends in incidence rate, post‐fracture treatment, and all‐cause mortality. J Bone Miner Res. 2023;38(8): 1064-1075. [4] LITJENS G, KOOI T, BEJNORDI BE, et al. A survey on deep learning in medical image analysis. Med Image Anal. 2017;42:60-88. [5] ZHANG J, LIU F, XU J, et al. Automated detection and classification of acute vertebral body fractures using a convolutional neural network on computed tomography. Front Aging Neurosci. 2023;14:1132725. [6] FENG E, JAYASURIYA NM, NATHANI KR, et al. Artificial intelligence image analysis for Hounsfield units in preoperative thoracolumbar CT scans: an automated screening for osteoporosis in patients undergoing spine surgery. J Neurosurg Spine. 2025;43(1):1-8. [7] WANG J, ZHOU S, CHEN S, et al. Prediction of osteoporosis using radiomics analysis derived from single source dual energy CT. BMC Musculoskelet Disord. 2023;24(1):100. [8] GYFTOPOULOS S, LIN D, KNOLL F, et al. Artificial Intelligence in Musculoskeletal Imaging: Current Status and Future Directions. AJR Am J Roentgenol. 2019;213(3):506-513. [9] JIANG P, RUAN X, FENG Z, et al. Research on online collaborative problem-solving in the last 10 years: Current status, hotspots, and outlook—A knowledge graph analysis based on CiteSpace. Mathematics. 2023;11(10):2353. [10] 陈悦,陈超美,刘则渊,等.CiteSpace知识图谱的方法论功能[J].科学学研究,2015,33(2):242-253. [11] HUANG X, HAN F, CHEN YF, et al. Bibliometric analysis of the application of artificial intelligence in orthopedic imaging. Quant Imaging Med Surg. 2025;15(5): 3993. [12] 唐欣尧,刘晓宜,王旭鹏,等.可穿戴腰椎外骨骼研究现状及关键技术分析[J].信息与控制, 2025,54(2):161-183. [13] 李辽远,韩建海,李向攀,等.上肢康复机器人关键技术研究进展[J].机械设计与研究,2021, 37(6):28-34. [14] 左安琪,庄贺,孔畅,等.人工智能应用于帕金森病研究的文献计量学分析[J/OL].神经损伤与功能重建,2025,1-6. [15] RYU JS, KANG H, CHU Y, et al. Vision-language foundation models for medical imaging: a review of current practices and innovations. Biomed Eng Lett. 2025;15(5):809-830. [16] 郭马超, 谢章家, 李宝林,等.人工智能技术在骨科应用的现状[J].中国矫形外科杂志,2024, 32(22):2067-2071. [17] GITTO S, SERPI F, ALBANO D, et al. AI applications in musculoskeletal imaging: a narrative review. Eur Radiol Exp. 2024; 8(1): 22. [18] 陶婷, 邹琦, 韩国栋,等.基于Web of Science数据库心肌纤维化研究现状及研究热点的文献计量学分析[J].中国循证心血管医学杂志, 2025,17(2):134-140. [19] LEE H, TAJMIR S, LEE J, et al. Fully automated deep learning system for bone age assessment. Radiology. 2017;285(3):1146-1154. [20] LARSON DB, CHEN MC, LUNGREN MP, et al. Performance of a deeplearning neural network model in assessing skeletal maturity on pediatric hand radiographs. Radiol Artif Intell. 2019;1(4):e180053. [21] SPAMPINATO C, PALAZZO S, GIORDANO D, et al. Deep learning for automated skeletal bone age assessment in Xray images. Pattern Recognition. 2019;94:228-234. [22] HALABI SS, PREVEDELLO LM, KALPATHYCRAMER J, et al. The RSNA Pediatric Bone Age Machine Learning Challenge. Radiology. 2019;291(1):164-172. [23] ISENSEE F, JAEGER PF, KOHL SAA, et al. nnUNet: a selfconfiguring method for deep learningbased biomedical image segmentation. Nat. Methods. 2021;18(2):203-211. [24] LI J, HU W, WU H, et al. Detection of hidden pediatric elbow fractures in X-ray images based on deep learning. J Radiat Res Appl Sci. 2024;17(2): 100893. [25] PI SW, LEE BD, LEE MS, et al. Ensemble deep-learning networks for automated osteoarthritis grading in knee X-ray images. Sci Rep. 2023;13(1): 22887. [26] PENG T, ZENG X, LI Y, et al. A study on whether deep learning models based on CT images for bone density classification and prediction can be used for opportunistic osteoporosis screening. OSINEP. 2024;35(1):117-128. [27] CHEN HY, HSU BWY, YIN YK, et al. Application of deep learning algorithm to detect and visualize vertebral fractures on plain frontal radiographs. Plos One. 2021;16(1):e0245992. [28] AYANA G, DESE K, ABAGARO AM, et al. Multistage transfer learning for medical images. Artif Intell Rev. 2024;57(9):232. [29] SALEHI AW, KHAN S, GUPTA G, et al. A study of CNN and transfer learning in medical imaging: Advantages, challenges, future scope. Sustainability. 2023;15(7):5930. [30] GULUWADI S. Enhancing brain tumor detection in MRI images through explainable AI using Grad-CAM with Resnet 50. BMC Med Imaging. 2024;24(1):1-19. [31] PRZĄDKA M, PAJĄK W, KLEINROK J, et al. Advances in 3D Printing Applications for Personalized Orthopedic Surgery: From Anatomical Modeling to Patient-Specific Implants. J Clin Med. 2025; 14(11):3989. [32] SUN J, YAO K, ZHU H, et al. Deep learning for polymer scaffold bioimage analysis: Opportunities and challenges. Int J Bioprint. 2024;11(2):16-33. [33] PETTIT RW, MARLATT BB, CORR SJ, et al. nnU-Net deep learning method for segmenting parenchyma and determining liver volume from computed tomography images. Ann Surg Open. 2022;3(2):e155. [34] 尹艺晓,马金刚,张文凯,等.从U-Net到Transformer:混合模型在医学图像分割中的应用进展[J].激光与光电子学进展,2025;62(2): 11-33. [35] WOO S, PARK J, LEE JY, et al. Cbam: Convolutional block attention module. ECCV. 2018:3-19. [36] HOWARD A, SANDLER M, CHU G, et al. Searching for mobilenetv3. ICCV. 2019:1314-1324. [37] HAN K, WANG Y, TIAN Q, et al. Ghostnet: More features from cheap operations. ICCV. 2020: 1580-1589. [38] MARTÍN PÉREZ IM, BOURHIM S, MARTÍN PÉREZ SE. Artificial Intelligence-Based Models for Automated Bone Age Assessment from Posteroanterior Wrist X-Rays: A Systematic Review. Appl Sci. 2025;15(11):5978. [39] ALAIMO D, TERRANOVA MC, PALIZZOLO E, et al. Performance of two different artificial intelligence (AI) methods for assessing carpal bone age compared to the standard Greulich and Pyle method. Radiol Med. 2024;129(10):1507-1512. [40] YE H. Artificial Intelligence Combined With Chest Radiography: New Hope for the Opportunistic Screening for Osteoporosis. KJR. 2025;26(4):390. [41] PADERNO A, ATAIDE GOMES EJ, GILBERG L, et al. Artificial intelligence-enhanced opportunistic screening of osteoporosis in CT scan: a scoping review. OSINEP. 2024;35(10):1681-1692. [42] YOON JS, YON CJ, LEE D, et al. Assessment of a novel deep learning-based software developed for automatic feature extraction and grading of radiographic knee osteoarthritis. BMC Musculoskelet Disord. 2023;24(1):869. [43] PI SW, LEE BD, LEE MS, et al. Ensemble deep-learning networks for automated osteoarthritis grading in knee X-ray images. Sci Rep. 2023;13(1): 22887. [44] HUO Y, XU Z, BAO S, et al. Adversarial synthesis learning enables segmentation without target modality ground truth. ISBI2018. 2018:1217-1220. [45] WANG Y, CHEN X, LI J, et al. Convolutional block attention module–multimodal feature-fusion action recognition: enabling miner unsafe action recognition. Sensors. 2024;24(14):4557. [46] NIE D, TRULLO R, LIAN J, et al. Medical image synthesis with context-aware generative adversarial networks. Med Image Comput Comput Assist Interv. 2017;10435:417-425. [47] MOHAMMED AS, HASANAATH AA, LATIF G, et al. Knee osteoarthritis detection and severity classification using residual neural networks on preprocessed x-ray images. Diagnostics. 2023; 13(8):1380. [48] ZHAO F, ZHANG C, GENG B. Deep multimodal data fusion. Acm Comput Surv. 2024;56(9):1-36. [49] CHADDAD A, HU Y, WU Y, et al. Generalizable and explainable deep learning for medical image computing: An overview. Cobe. 2025;33:100567. [50] ENNAB M, MCHEICK H. Advancing AI interpretability in medical imaging: a comparative analysis of pixel-level interpretability and Grad-CAM models. MAKE. 2025;7(1):12. [51] ZHANG K, KHOSRAVI B, VAHDATI S, et al. FDA review of radiologic AI algorithms: process and challenges. Radiology. 2024;310(1):e230242. [52] SANNI B. Edge-AI Systems for Medical Imaging: Real-Time Diagnostics in Low-Bandwidth and Resource-Constrained Environments. Diagnostic Radiology. 2023. |
| [1] | Xu Canli, He Wenxing, Wang Yuping, Ba Yinying, Chi Li, Wang Wenjuan, Wang Jiajia. Research context and trend of TBK1 in autoimmunity, signaling pathways, gene expression, tumor prevention and treatment [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(在线): 1-11. |
| [2] | Huang Jie, Zeng Hao, Wang Wenchi, Lyu Zhucheng, Cui Wei. Visualization analysis of literature on the effect of lipid metabolism on osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1558-1568. |
| [3] | Zhang Haiwen, Zhang Xian, Xu Taichuan, Li Chao. Bibliometric and visual analysis of the research status and trends of senescence in osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1580-1591. |
| [4] | Yang Zeyu, Zhi Liang, Wang Jia, Zhang Jingyi, Zhang Qingfang, Wang Yulong, Long Jianjun. A visualized analysis of research hotspots in high-frequency repetitive transcranial magnetic stimulation from the macroscopic perspective [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1320-1330. |
| [5] | Guan Yujie, Zhao Bin. Application and prospect of artificial intelligence in screening and diagnosis of scoliosis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(3): 721-730. |
| [6] | Peng Hao, Chen Qigang, Shen Zhen. A visual analysis of research hotspots of H-type vessels in various bone diseases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(3): 760-769. |
| [7] | Zhang Anqi, Hua Haotian, Cai Tianyuan, Wang Zicheng, Meng Zhuo, Zhan Xiaoqian, Chen Guoqian . Pain after total knee arthroplasty: current status and trend analysis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(3): 795-804. |
| [8] | Wang Xueting, Yang Wei, Wang Pengqin. Post-stroke rehabilitation robotics: current research status and hot topics in and outside China [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(28): 7404-7409. |
| [9] | Zhang Jingyi, Zhi Liang, Yang Zeyu, Li Yaning, Hu Jia, Wang Jia, Wang Yulong, Long Jianjun. Extracorporeal shock wave therapy: current research status, hotspots, and trends [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(28): 7410-7417. |
| [10] | Guo Jun, Lu Zheng, Yu Jinling, Hao Yuanyuan, Liu Kaishun, Liu Xuexia, Huang Yourong. Oxidative stress and osteoporosis: a bibliometric analysis of literature from SCI core database [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(28): 7428-7436. |
| [11] | Xu Canli, He Wenxing, Wang Yuping, Ba Yinying, Chi Li, Wang Wenjuan, Wang Jiajia. Research context and trend of TANK binding kinase 1 in autoimmunity and tumor prevention and treatment [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(28): 7456-7464. |
| [12] | Wang Feifei, Wang Zhennan. Scientometric deconstruction of developmental dynamics in upper-limb rehabilitation robotics: evidence network analysis via CiteSpace [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6400-6409. |
| [13] | Wang Fei, Wang Hangping, Dai Rongjuan, Gong Lingxuan. Visual analysis of the research literature on plantar fasciitis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6410-6420. |
| [14] | Yang Jun, Li Bin, Xing Guogang, Cai Jie, Liu Lu, Chen Peng, Zhang Tao, Fu Yuanbo, Liu Huilin. Application of patch-clamp technique in traditional Chinese medicine: a visual analysis of relevant literature [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(23): 6010-6020. |
| [15] | Li Qian, Li Zhenxing, Qiao Pengyan, Wang Pingzhi. Visual analysis of shear wave elastography in skeletal muscle research [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(23): 6021-6029. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||