Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (19): 5057-5065.doi: 10.12307/2026.790
Previous Articles Next Articles
In vitro simulation of cellular exercise environments: advancements in methodology and signal simulation
Chen Bingao1, Chen Hongbao1, Xie Hao1, Ding Xinglei2, Yuan Yu1, Zhang Jiahao1, Ban Weikang1, Xu Shenghao1, Yuan Yang1
- 1School of Physical Education, 2School of Life Sciences, Qufu Normal University, Qufu 273165, Shandong Province, China
-
Received:2025-10-15Accepted:2025-12-09Online:2026-07-08Published:2026-02-24 -
Contact:Yuan Yang, PhD, Associate professor, School of Physical Education, Qufu Normal University, Qufu 273165, Shandong Province, China -
About author:Chen Bingao, MS candidate, School of Physical Education, Qufu Normal University, Qufu 273165, Shandong Province, China -
Supported by:Taishan Scholars Talent Project, No. tsqn202312181 (to YY); Shandong Provincial Youth Innovation Team Project, No. 2023RW102 (to YY)
CLC Number:
Cite this article
Chen Bingao, Chen Hongbao, Xie Hao, Ding Xinglei, Yuan Yu, Zhang Jiahao, Ban Weikang, Xu Shenghao, Yuan Yang. In vitro simulation of cellular exercise environments: advancements in methodology and signal simulation[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(19): 5057-5065.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

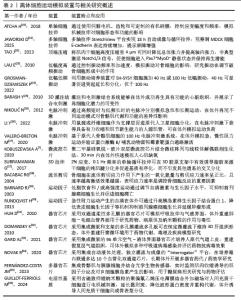
2.1 运动诱导的微环境变化与离体模拟需求 2.1.1 运动引发的细胞微环境动态特征 运动作为一种内源性的系统生理刺激,会在不同组织和器官内塑造出高度动态且时空异质的细胞微环境[9-11]。在骨骼肌和心肌中,肌纤维随肌肉收缩与舒张承受周期性机械拉伸,细胞骨架在应力传导下发生重排[12];血管内皮则在血流加速时感受显著增强的流体剪切力,诱导一氧化氮合成与血管扩张反应[13-14]。电信号在运动神经元活动中尤为重要,通过影响细胞微环境调节和基因表达模式来调节生长和分化过程[15]。同时,运动过程中局部血流分配的调整会造成组织氧分压的周期性波动,高强度或长时间运动更可导致瞬时缺氧与再氧合交替出现,促发氧化还原信号的动态变化[14]。伴随能量代谢速率提升,局部乳酸、ATP/ADP比值及离子浓度均呈现短时高幅波动,形成具有机械、化学和代谢信号交织特征的微环境[14]。这些变化不仅在分子水平上驱动代谢重编程、信号通路激活及基因表达调控,还在细胞群体水平上影响增殖、分化与凋亡等命运决定,从而构成运动诱发的细胞微环境的动态本质。 2.1.2 离体模拟技术的前沿发展与应用环境 随着生物工程和仿生技术的进步,离体细胞模拟运动环境的技术体系正向多维度、高还原性和智能化方向发展。首先,针对传统细胞培养缺乏动态机械力的局限,近年来新型柔性拉伸平台和微振动等装置能够从不同频率、幅度和方向上模拟肌肉与内皮细胞在运动中的力学刺激,实现细胞空间重排和功能表型的动态塑造[16]。研究发现,2%应变(0.1 Hz频率)的单轴机械加载,可诱导封装于3D胶原支架的脂肪干细胞腱向分化[17]。其次,电生理层面,微电极阵列与可编程脉冲发生器已广泛应用于离体心肌、神经与骨骼肌细胞,精准还原动作电位触发与信号同步传导过程,支持对兴奋-收缩耦合机制的系统性分析[18]。此外,生物化学因子模拟方面,越来越多的研究采用运动后血清、人工肌因子、外泌体以及多种代谢物进行培养环境的动态调控,从而更接近体内运动时体液环境对细胞行为的影响[19]。更进一步,随着多器官、芯片与微流控系统的发展,不同器官细胞可在同一平台实现物质和信号的实时交流,配合嵌入式传感器对微环境参数(如pH值、氧气、代谢物等)进行动态监测和反馈调节,有力推动了复杂运动生理状态下多器官相互作用的机制研究。整体来看,离体模拟技术正在突破单一应激、静态模型等传统瓶颈,朝着高通量、多维协同和生理相关性的方向持续创新,为精准解析运动介导的分子机制和转化医学应用提供了坚实支撑(表2和图2)[7,17-18,20-36]。 "


2.2 机械力刺激模拟技术与细胞反应 在运动过程中,人体会受到多种机械力刺激,包括拉伸、压缩、剪切力以及低强度振动等,这些力对身体各个部位和组织的功能有着重要影响[37],这些机械力通过物理信号传递,调控细胞的生长、分化、迁移等生物学过程,进而影响机体的适应性反应和功能调节[38]。例如,拉伸力可以通过细胞内信号通路调节细胞形态与功能,压缩力则可调控骨骼和软组织的力学响应,流体剪切力在血管和骨细胞中的作用尤为显著,而低强度振动刺激已成为骨组织工程的重要手段之一[20]。为了更好地模拟这些机械力刺激对细胞的影响,研究者们开发了多种实验装置,如拉伸装置、压缩平台、流体剪切力模型和振动平台等,并根据实验特点进行组合[39]。这些装置能够精确控制机械刺激的强度、频率和持续时间,为细胞在不同力学环境下的生物力学响应提供了有效的模拟工具,推动了细胞生物学、运动生理学及康复医学的研究进展。 2.2.1 拉伸和压缩 细胞通过机械转导感知并响应由器官功能、运动和生长引起的机械应变,影响细胞的生长、分化、迁移和侵袭等过程[38,40]。为了有效模拟细胞的微环境并深入研究细胞对机械刺激的反应,研究者开发了细胞拉伸和压缩装置[41]。拉伸应力通过周期性拉伸调节细胞形态和功能,而压应力则通过外部压缩力对细胞结构产生影响[42-43]。拉伸与压缩的联合应用为研究细胞在不同机械环境下的生物学反应提供了关键的实验工具,有助于更全面地理解机械应力在细胞行为中的作用[38,44]。细胞拉伸刺激通过机械信号激活细胞内信号通路,促进细胞形态重塑、基因表达调控及生物力学适应,广泛影响细胞功能和组织重塑。周期性拉伸通过整合素和机械敏感离子通道调节细胞骨架,黏着斑激酶信号通路在这一过程中发挥关键作用,促进细胞内重组和定向[20,45]。拉伸引发的机械刺激增强了炎症因子(如白细胞介素8、单核细胞趋化和激活因子/单核细胞趋化蛋白1)的分泌与基因表达,同时激活磷脂酶C、蛋白激酶C和酪氨酸激酶等关键分子,进一步调控细胞行为[46]。此外,周期性拉伸还通过改变细胞膜形态,增加细胞内钙离子浓度,这在调节细胞响应、促进细胞迁移、增殖和分化等生物学过程中起着重要作用。这些机制综合影响了细胞的适应性重塑和功能调控。单轴拉伸装置可在柔性膜上施加1 Hz、10%-20%的拉伸幅度,造成细胞顺应应力重新排列:低频(≤1 Hz)拉伸导致细胞平行排列,高频则因应变恢复时间不足而让细胞垂直于拉伸方向[20]。最近有文献报道了一款名为StretchView的多轴细胞拉伸平台,StretchView可以与自动化和时间分辨活细胞成像兼容,能够长时间(12 h)自动采集细胞图像,并展示稳定的应变场[21]。在18 h的循环拉伸实验中,发现拉伸增强了单层MDCK细胞中E-钙黏蛋白的定位,表明细胞屏障完整性增加,证明了StretchView在细胞机械生物学研究中的潜力。通过对肌肉干细胞的顶端表面施加物理压缩来模拟肌肉干细胞的体内机械环境,实验证明,无论基础弹性和化学成分如何,压缩都会使激活的肌肉干细胞恢复到静止干细胞状态[22]。针对不同组织需优化应力波形、幅度和持续时间,使它更接近运动中的肌肉拉伸和压缩模式,并考虑与电刺激及生物化学因子配合的整体策略[47-48]。 2.2.2 剪切力 剪切力在细胞机械转导中扮演着关键角色,尤其在骨细胞和血管内皮细胞等多种细胞类型的生理过程中[49],通过整合素等机制感知和响应外部施加的剪切力,进而影响细胞形态、基因表达、增殖和分化等生物学行为。在骨细胞中,剪切力能够调节骨骼的健康与重塑,促进骨基质的生成和矿化。剪切力会引发骨细胞突起膜上的钙离子释放,并通过信号传递作用影响细胞之间的交流[50],这一过程通过间隙连接调节骨细胞之间的电位和电流,从而调节整个骨细胞网络及骨母细胞的活动。剪切力影响未成熟以及成熟血管壁内皮细胞,并促进骨髓来源内皮祖细胞和胚胎干细胞分化为血管壁内皮细胞。为了方便对细胞机械传导的理解,研究者将目光投入到体外模型上,旨在受控细胞培养环境中重现流体剪切力[49]。体外模型大致分为平行板流动室、摇摆平台和3D灌注生物反应器,广泛应用于模拟流体剪切力对细胞的影响。平行板流动室用于单层细胞培养,采用聚碳酸酯室和载玻片封闭,通过静水压力或泵控制流体流动,产生脉动性、振荡性和单向流动等流动剖面,并产生0.001-3 Pa范围内的生理相关壁剪应力。摇摆平台用于在多孔板中培养细胞,通过摇摆或轨道摇动应用液体刺激,能够提供高通量样品培养[51]。该系统产生低幅度、不均匀图案的振荡流体剪切应力,范围为0.001-0.25 Pa。剪应力的大小受细胞在井中的位置、运动频率和液体量等因素的影响。摇摆平台已被证明能增加间充质干细胞的成骨活性、胶原蛋白分泌。3D灌注生物反应器通过将流体流动引入多孔支架,实现更自然的三维细胞培养环境,利用灌注方式在支架内部形成均匀微环境,模拟骨组织的孔隙率和刚度,可通过计算模型估算剪应力大小,并结合支架变形和压缩等因素影响细胞受力,是研究骨细胞力学响应的重要平台。通过这些模型,研究人员能够控制流动类型、剪切速率和持续时间,探讨流体剪切力对细胞的生物力学反应。这些模型的应用揭示了剪切力在细胞功能调控中的重要性,尤其是在骨细胞的力学感知和骨骼健康中。 2.2.3 振动 振动是骨细胞经历的最常见刺激,低强度高频率振动不会引起或较小引起膜应变和流体剪切。低强度高频振动是骨组织工程的新兴刺激模式,也是模拟步行或跳跃等运动产生微振的重要手段。研究表明,对骨细胞施加< 1×g加速度、30-90 Hz振动1 h,可显著提高环氧化酶2 mRNA表达并降低核因子κB受体活化因子配体水平,且振动条件下培养基能够抑制破骨细胞形成,这表明骨细胞可以感知振动并通过释放可溶性因子调节骨重建。为避免振动引起流体剪切,刺激装置采用密封培养孔和加速度反馈控制,剪切应力低至10-6 Pa。类似的振动刺激也可用于模拟跑步或力量训练中的微振动环境,如在肌肉片上施加低幅度振动可以诱导钙离子振荡和肌纤维增生。有研究认为振动促进间充质干细胞增殖和成骨分化的效果与流体剪切力大小无关,而与细胞骨架的预应力和重塑水平密切相关[52-54]。使用局部/直接振动可以显著改变肌肉细胞的结构特征,并显著提高肌肉力量,刺激效果与振幅和频率有着密切的关系[55]。有研究表明,适当强度的表面声波机械振动能显著促进细胞迁移,从而加快体外伤口愈合,而高强度则可能抑制生长[56]。适度振动能够触发细胞内钙离子浓度升高,激活钙依赖性蛋白酶,切割黏附与骨架相关蛋白,重塑肌动蛋白网络,导致丝状伪足收缩、细胞形态变圆,并可能启动迁移或应激相关的生物学过程[57]。有研究设计了一款利用低频低强度的生物反应平台,将神经细胞暴露于40 Hz和100 Hz振动环境中,发现神经细胞的分化和增殖能力显著增强[24]。未来的训练芯片应将振动与拉伸和电刺激组合,通过编程控制不同刺激在时间上的先后和强度,实现对运动模式的精确复刻。 2.3 电生理信号模拟技术与细胞反应 在运动模拟和细胞工程的研究中,电刺激一直是重要且有效的生理模拟手段,广泛应用于肌肉和心脏细胞的功能训练与分化[58]。近年来,研究者不仅仅关注单一电刺激属性对细胞的干预,而是发展出了多种联动机制。例如,器官芯片和电信号协同系统可以精准模拟器官和细胞层面的生理与病理生理特征,并再现运动干预中的一些关键属性[35]。为了更全面地模拟运动干预的多种物理属性,研究者开始结合机械刺激、电信号和流体力学等不同类型的信号,从多个角度完成对运动干预的精准再现。通过精细调控机械拉伸、电脉冲刺激和灌注以及联合应用的强度、频率和持续时间,不仅能够模拟运动对细胞的直接作用,还能实现对细胞行为的精准调控[25,39]。这些技术的不断优化与创新,尤其是引入多器官芯片和磁响应材料等新兴方法,为离体细胞运动环境的模拟设计提供了坚实基础,并在组织工程、药物开发及临床应用领域展现出广阔前景[59]。 2.3.1 电信号 电刺激可诱导肌纤维收缩、钙离子内流和代谢改变,是模拟神经-肌肉兴奋与收缩耦合不可或缺的手段。近年来,电脉冲刺激被广泛应用于人源肌管体外运动环境的模拟实验[60]。2002年,首次使用人肌肉管进行电脉冲刺激,研究显示电脉冲刺激对体外培养人肌管中的葡萄糖摄取具有与体内肌肉收缩相似的作用。具体而言,研究人员采用200 ms电脉冲,每隔5 s以100 Hz的频率施加,连续刺激5 min,结果肌管的葡萄糖摄取率提升了约30%,这种短时高频的刺激设计,成功模拟了机体急性高强度运动所产生的生理效应[61-62]。 此外,NIKOLI?等[18]研究进一步证实,急性高频电脉冲刺激除促进葡萄糖摄取外,还会导致ATP和磷酸肌酸等高能分子水平下降,同时细胞乳酸水平升高,表现出明显的能量代谢重塑特征。然而,现实生活和临床健康管理中,长期规律的运动训练对于代谢改善的效果更加持久[63]。基于这一认识,学者们开始尝试以慢性低频电脉冲刺激模拟长期运动适应。在短时间内,已有2种慢性低频电脉冲刺激模型:一种是LAMBERND等[64]采用1 Hz、2 ms、11.5 V的参数,刺激时长为2-24 h;另一种是NIKOLI?等[18]设置为1 Hz、2 ms、30 V的参数,刺激持续24-48 h。另一项研究提出用100 Hz、0.4 ms、15 V脉冲(间隔4 s)持续30 min模拟阻力训练,该方案激活蛋白激酶B/哺乳动物雷帕霉素靶蛋白/S6K1信号通路,促进肌蛋白合成[27]。 有研究建立了一种以小鼠成纤维细胞为饲养层的人源卫星细胞共培养体外运动模型,通过电脉冲刺激成功诱导了散发性包涵体肌炎患者来源肌管收缩,并揭示散发性包涵体肌炎肌管在收缩后表现出特异性的TDP-43细胞质聚集,为研究人类肌肉疾病的运动反应和病理机制提供了新平台[26]。这些结果表明,脉冲频率、波宽和持续时间决定了对细胞代谢与生长的不同调节效果,电刺激参数的选择应根据所模拟的运动类型和研究目标进行精细调控,并可与机械拉伸和生物化学刺激组合以实现更真实的模拟。 2.3.2 器官芯片 器官芯片作为一种前沿的微流体装置,内壁覆盖着在流体环境下培养的活细胞,能够极为精准地模拟出器官层面的生理及病理生理特征[31-32]。相对于静态微生理系统[65],器官芯片能更为准确地评估人体内的药物分布、疗效和毒性,并且现在器官芯片几乎可以模拟所有器官系统的各类疾病[66]。随着理论和技术的发展,科研人员开发了可以施加循环机械应变的芯片,如皮肤芯片[34,67]。运动涉及全身性的多器官系统响应,单一细胞类型无法再现复杂的激素和代谢调节,因此多器官芯片为研究运动中器官互作提供了理想平台。有团队发明了一种“健身房芯片(gym-on-a-chip)”将骨骼肌和胰岛细胞分别培养于2个微腔,通过微流道连接形成闭环循环。肌肉腔中的电极产生温和脉冲,使肌肉收缩并分泌肌因子(如白细胞介素6)等信号分子,下游胰岛腔感受到来自上游的代谢信号后通过细胞膜传感器触发胰岛素释放,研究者可借助集成的电化学传感器实时监测肌因子和胰岛素的释放动态,发现肌肉收缩后立即释放肌因子,而胰岛素释放延后数十分钟,证明了肌-胰岛间的串联反馈[68]。类似平台还能通过替换不同细胞或添加肝细胞、脂肪细胞扩展为肌肉-肝脏-胰腺-脂肪四器官环路,用于研究运动调节糖脂代谢、肌肉-肝脏协同和肌肉-大脑通讯等复杂生理过程,具有极高的前沿价值。此外,集成pH值、氧气和电化学传感器的多器官训练芯片能够实时监测肌因子和胰岛素的释放动态,为多器官协同研究提供精准的数据支撑[8]。在航天领域,器官芯片的研究也备受关注。在太空的微重力环境下,肌肉组织在芯片中进行为期10 d的实验表明,与地面对照相比,暴露于太空环境的老年受试者的激素肌肉代谢相关基因表达下调,年轻受试者炎症途径相关基因下调[69]。这一成果表明,微重力环境加速了与衰老相关的肌肉消退,强调了运动训练在微重力情境下的重要性。未来,结合电刺激、拉伸和内分泌因素的微重力肌肉芯片将有助于探索太空任务中如何通过地面运动或药物干预延缓肌肉萎缩。基于上述理论与技术,器官芯片中的电信号协同训练系统通过集成多器官组织、可控电刺激、动态流体和多参数传感等关键要素,能够同步还原运动过程中的机械收缩、电生理激活、信号分泌以及跨器官代谢反馈,高度仿真运动状态下的人体生理响应,为离体细胞层面的运动环境模拟提供了坚实的技术基础。 2.3.3 机械-电耦合 电刺激常与机械应力协同作用,例如体内的肌肉和心肌正是在机械伸缩和动作电位的耦合下完成收缩和能量代谢。有研究构建了一个由聚二甲基硅氧烷组织腔和气动驱动拉伸平台组成的实验装置。聚二甲基硅氧烷组织腔包含8个微孔(长8.05 mm、宽2.5 mm),每个微孔内设一对柱状物(高2.78 mm、直径0.8 mm),用于固定组织并测量收缩力;腔内还置入碳棒作为刺激电极(一对碳棒可刺激4个微孔)。实验将新生大鼠心肌细胞接种于胶原凝胶并填充至微孔内,凝胶压实3 d后,对微组织施加3种刺激(持续3 d):电刺激(3.0-4.0 V/cm、1 Hz)、机械刺激(5%静态应变)或机电联合刺激(电+机械刺激),研究结果显示,相较仅电/机械刺激或未刺激的对照组,接受电刺激(含机电联合刺激)的心脏微组织收缩幅度显著增大;经肌节α-辅肌动蛋白、肌动蛋白和肌钙蛋白T染色证实肌节结构改善,心房/脑利钠肽表达升高,验证了电刺激(尤其机电联合)对心脏微组织功能及结构的促进作用,为心脏组织工程等研究提供实验依据[70]。另一项研究中,使用去细胞化猪心外膜作为支架,接种间充质干细胞,并施加20%循环拉伸与5 V、1 Hz电脉冲刺激,发现细胞表现出典型的心肌细胞样表型,表达α-肌动蛋白、肌球蛋白重链、心肌肌钙蛋白T、连接素43及N-钙黏蛋白等标志物,说明电-机械协同刺激显著促进心肌向分化[71]。这些发现表明,同步的力学与电学信号相较于单一刺激,在驱动心肌细胞分化与功能成熟方面具有更强的诱导效应。除了传统电极直接刺激方式,近年来出现了一种新兴方法,即利用磁致伸缩/压电复合材料,将外部磁场转换为局部电信号,从而实现非接触式电刺激。例如,将钴铁氧体(CoFe?O?)磁致伸缩纳米颗粒与聚偏氟乙烯压电薄膜结合构建复合支架,在外加磁场作用下可产生稳定电信号[59]。研究显示,此类支架可促进间充质干细胞骨向分化,同时增强黏附斑形成,并激活细胞外信号调节激酶/丝裂原活化蛋白激酶信号通路,从而改善细胞功能和分化潜力。在这些研究中,明确指出了机械-电耦合刺激作为细胞培养和分化的有效手段,作用机制与运动干预中产生的机械-电刺激属性具有高度相似性。因此,未来研究中深入探讨和阐明运动干预所产生的机械-电刺激特性,将对运动模拟的设计与细胞工程领域的应用提供关键的理论支持和技术基础。 2.4 生物活性因子模拟技术 在离体细胞运动环境模拟中,很多研究者将目光放在了运动生化环境的构建中,并在这个过程中引入了很多新概念,例如运动因子和运动模拟物(表3)。运动因子被定义为响应急性或慢性运动而释放的信号部分,它们通过内分泌、旁分泌或自分泌途径发挥作用。运动模拟物作为一类拟议疗法,是药物开发中的一种前卫范式,有助于打破药物发现中的“反摩尔定律”[19,72]。运动模拟物通过模拟运动干预引起的肌因子分泌、信号转导网络及小分子靶标三大路径实现运动效应的体外复现[73]。基于此,在构建体外细胞运动模型时,可引入运动模拟物策略,精准模拟运动引发的内分泌因子谱系变化,并以此改造细胞培养体系,实现运动干预后细胞外微环境的动态模拟。随着大数据分析和人工智能(AI)等前沿技术的应用,可以对运动干预下多种细胞因子及功能变化进行高通量挖掘与精准建模,为运动模拟物的设计与优化提供坚实的数据基础。结合人工合成及规模化制造技术,这些数据驱动的运动模拟物能够更全面地还原体内运动微环境,应用于离体细胞实验,为基础研究和临床转化提供了更加真实、标准和高效的模拟平台,具有显著的理论与实际应用价值。 "

| [1] REISMAN EG, HAWLEY JA, HOFFMAN NJ. Exercise-Regulated Mitochondrial and Nuclear Signalling Networks in Skeletal Muscle. Sports Med. 2024; 54(5):1097-1119. [2] ZHANG W, HUANG G, XU F. Engineering Biomaterials and Approaches for Mechanical Stretching of Cells in Three Dimensions. Front Bioeng Biotechnol. 2020;8:589590. [3] SZABÓ MR, PIPICZ M, CSONT T, et al. Modulatory Effect of Myokines on Reactive Oxygen Species in Ischemia/Reperfusion. Int J Mol Sci. 2020;21(24):9382. [4] QIN H, LUO Z, SUN Y, et al. Low-intensity pulsed ultrasound promotes skeletal muscle regeneration via modulating the inflammatory immune microenvironment. Int J Biol Sci. 2023;19(4): 1123-1145. [5] GUNASEKARA N, CLAUSS D, BLOCH W. Effects of Exercise-Induced Changes in Myokine Expression on the Tumor Microenvironment. Sports Med Int Open. 2024;8:a22831663. [6] POTIER E, NOAILLY J, ITO K. Directing bone marrow-derived stromal cell function with mechanics. J Biomech. 2010;43(5):807-817. [7] KOBUSZEWSKA A, JASTRZĘBSKA E, ŻUKOWSKI K, et al. Simulation of hypoxia of myocardial cells in microfluidic systems. Sci Rep. 2020;10(1):15524. [8] CLARKE GA, HARTSE BX, NIARAKI ASLI AE, et al. Advancement of Sensor Integrated Organ-on-Chip Devices. Sensors (Basel). 2021;21(4):1367. [9] LAVIN KM, COEN PM, BAPTISTA LC, et al. State of Knowledge on Molecular Adaptations to Exercise in Humans: Historical Perspectives and Future Directions. Compr Physiol. 2022;12(2):3193-3279. [10] ROBERTS FL, MARKBY GR. New Insights into Molecular Mechanisms Mediating Adaptation to Exercise; A Review Focusing on Mitochondrial Biogenesis, Mitochondrial Function, Mitophagy and Autophagy. Cells. 2021;10(10):2639. [11] LU ZM, SUN ZY, ZHANG S, et al. Lipolysis is accompanied by immune microenvironment remodeling in adipose tissue of obesity with different exercise intensity. Eur Rev Med Pharmacol Sci. 2023;27(3):867-878. [12] FOWLER A, KNAUS KR, KHUU S, et al. Network model of skeletal muscle cell signalling predicts differential responses to endurance and resistance exercise training. Exp Physiol. 2024;109(6): 939-955. [13] HONG J, PARK Y. Microvascular Function and Exercise Training: Functional Implication of Nitric Oxide Signaling and Ion Channels. Pulse (Basel). 2024;12(1):27-33. [14] REDDY I, YADAV Y, DEY CS. Cellular and Molecular Regulation of Exercise-A Neuronal Perspective. Cell Mol Neurobiol. 2023;43(4):1551-1571. [15] SONG Y, SOTO J, CHEN B, et al. Cell engineering: Biophysical regulation of the nucleus. Biomaterials. 2020;234:119743. [16] BIGHI B, RAGAZZINI G, GALLERANI A, et al. Cell stretching devices integrated with live cell imaging: a powerful approach to study how cells react to mechanical cues. Prog Biomed Eng (Bristol). 2024;7(1). doi: 10.1088/2516-1091/ad9699. [17] SUBRAMANIAN G, STASUK A, ELSAADANY M, et al. Effect of Uniaxial Tensile Cyclic Loading Regimes on Matrix Organization and Tenogenic Differentiation of Adipose-Derived Stem Cells Encapsulated within 3D Collagen Scaffolds. Stem Cells Int. 2017;2017:6072406. [18] NIKOLIĆ N, BAKKE SS, KASE ET, et al. Electrical pulse stimulation of cultured human skeletal muscle cells as an in vitro model of exercise. PLoS One. 2012;7(3):e33203. [19] ZHU Y, SONG G. Molecular origin and biological effects of exercise mimetics. J Exerc Sci Fit. 2024; 22(1):73-85. [20] ATCHA H, DAVIS CT, SULLIVAN NR, et al. A Low-Cost Mechanical Stretching Device for Uniaxial Strain of Cells: A Platform for Pedagogy in Mechanobiology. J Biomech Eng. 2018;140(8):0810051-0810059. [21] JAWORSKI D, HUNDSDORFER L, BASTOUNIS E, et al. StretchView - A Multi-Axial Cell-Stretching Device for Long-Term Automated Videomicroscopy of Living Cells. Adv Sci (Weinh). 2025;12(9):e2408853. [22] TAO J, CHOUDHURY MI, MAITY D, et al. Mechanical compression creates a quiescent muscle stem cell niche. Commun Biol. 2023;6(1):43. [23] LAU E, AL-DUJAILI S, GUENTHER A, et al. Effect of low-magnitude, high-frequency vibration on osteocytes in the regulation of osteoclasts. Bone. 2010;46(6):1508-1515. [24] GROSMAN-DZIEWISZEK P, WIATRAK B, DZIEWISZEK W, et al. Influence of 40 Hz and 100 Hz Vibration on SH-SY5Y Cells Growth and Differentiation-A Preliminary Study. Molecules. 2022;27(10):3337. [25] BARASH Y, DVIR T, TANDEITNIK P, et al. Electric field stimulation integrated into perfusion bioreactor for cardiac tissue engineering. Tissue Eng Part C Methods. 2010;16(6):1417-1426. [26] LI Y, CHEN W, OGAWA K, et al. Feeder-supported in vitro exercise model using human satellite cells from patients with sporadic inclusion body myositis. Sci Rep. 2022;12(1):1082. [27] VALERO-BRETON M, WARNIER G, CASTRO-SEPULVEDA M, et al. Acute and Chronic Effects of High Frequency Electric Pulse Stimulation on the Akt/mTOR Pathway in Human Primary Myotubes. Front Bioeng Biotechnol. 2020;8:565679. [28] BACABAC RG, SMIT TH, MULLENDER MG, et al. Nitric oxide production by bone cells is fluid shear stress rate dependent. Biochem Biophys Res Commun. 2004;315(4):823-829. [29] BARNARD RJ, NGO TH, LEUNG PS, et al. A low-fat diet and/or strenuous exercise alters the IGF axis in vivo and reduces prostate tumor cell growth in vitro. Prostate. 2003;56(3):201-206. [30] RUNDQVIST H, AUGSTEN M, STRÖMBERG A, et al. Effect of acute exercise on prostate cancer cell growth. PLoS One. 2013;8(7):e67579. [31] HUH D, MATTHEWS BD, MAMMOTO A, et al. Reconstituting organ-level lung functions on a chip. Science. 2010;328(5986):1662-1668. [32] DOMANSKY K, INMAN W, SERDY J, et al. Perfused multiwell plate for 3D liver tissue engineering. Lab Chip. 2010;10(1):51-58. [33] GARD AL, LUU RJ, MILLER CR, et al. High-throughput human primary cell-based airway model for evaluating influenza, coronavirus, or other respiratory viruses in vitro. Sci Rep. 2021; 11(1):14961. [34] NOVAK R, INGRAM M, MARQUEZ S, et al. Robotic fluidic coupling and interrogation of multiple vascularized organ chips. Nat Biomed Eng. 2020; 4(4):407-420. [35] FERNÁNDEZ-COSTA JM, ORTEGA MA, RODRÍGUEZ-COMAS J, et al. Training-on-a-Chip: A Multi-Organ Device to Study the Effect of Muscle Exercise on Insulin Secretion in Vitro. Adv Mater Technol. 2023; 8:2200873. [36] GUILLOT-FERRIOLS M, COSTA CM, CORREIA DM, et al. Piezoelectric Stimulation Induces Osteogenesis in Mesenchymal Stem Cells Cultured on Electroactive Two-Dimensional Substrates. ACS Appl Polym Mater. 2024;6(22):13710-13722. [37] CARVAJAL-AGUDELO JD, MCNEIL A, FRANZ-ODENDAAL TA. Effects of simulated microgravity and vibration on osteoblast and osteoclast activity in cultured zebrafish scales. Life Sci Space Res (Amst). 2023;38:39-45. [38] KAMBLE H, BARTON MJ, JUN M, et al. Cell stretching devices as research tools: engineering and biological considerations. Lab Chip. 2016; 16(17):3193-3203. [39] STOPPEL WL, KAPLAN DL, BLACK LD 3RD. Electrical and mechanical stimulation of cardiac cells and tissue constructs. Adv Drug Deliv Rev. 2016;96:135-155. [40] WANG J, CHATTERJEE A, ZIGAN C, et al. Pneumatic Non-Equibiaxial Cell Stretching Device With Live-Cell Imaging. IEEE Trans Biomed Eng. 2024; 71(3):820-830. [41] HONG Y, PENG X, YU H, et al. Cell-matrix feedback controls stretch-induced cellular memory and fibroblast activation. Proc Natl Acad Sci U S A. 2025;122(12):e2322762122. [42] SONG M, JANG Y, KIM SJ, et al. Cyclic Stretching Induces Maturation of Human-Induced Pluripotent Stem Cell-Derived Cardiomyocytes through Nuclear-Mechanotransduction. Tissue Eng Regen Med. 2022;19(4):781-792. [43] LE ROUX AL, VENTURINI V, GÓMEZ-GONZÁLEZ M, et al. Equibiaxial Stretching Device for High Magnification Live-Cell Confocal Fluorescence Microscopy. J Vis Exp. 2025;(220). doi: 10.3791/67520. [44] ONAL S, ALKAISI MM, NOCK V. Microdevice-based mechanical compression on living cells. iScience. 2022;25(12):105518. [45] DHEIN S, SCHREIBER A, STEINBACH S, et al. Mechanical control of cell biology. Effects of cyclic mechanical stretch on cardiomyocyte cellular organization. Prog Biophys Mol Biol. 2014; 115(2-3):93-102. [46] OKADA M, MATSUMORI A, ONO K, et al. Cyclic stretch upregulates production of interleukin-8 and monocyte chemotactic and activating factor/monocyte chemoattractant protein-1 in human endothelial cells. Arterioscler Thromb Vasc Biol. 1998;18(6):894-901. [47] CHIU KH, KARPAT M, HAHN J, et al. Cyclic Stretching Triggers Cell Orientation and Extracellular Matrix Remodeling in a Periodontal Ligament 3D In Vitro Model. Adv Healthc Mater. 2023;12(30):e2301422. [48] LEE LW, LEE GH, SU IH, et al. Mechanobiological mechanism of cyclic stretch-induced cell columnarization. Cell Rep. 2025;44(5):115662. [49] WITTKOWSKE C, REILLY GC, LACROIX D, et al. In Vitro Bone Cell Models: Impact of Fluid Shear Stress on Bone Formation. Front Bioeng Biotechnol. 2016;4:87. [50] WEINBAUM S, COWIN SC, ZENG Y. A model for the excitation of osteocytes by mechanical loading-induced bone fluid shear stresses. J Biomech. 1994;27(3):339-360. [51] ZHOU X, LIU D, YOU L, et al. Quantifying fluid shear stress in a rocking culture dish. J Biomech. 2010;43(8):1598-1602. [52] LIN CY, SASSI A, WU Y, et al. Mechanotransduction pathways regulating YAP nuclear translocation under Yoda1 and vibration in osteocytes. Bone. 2025;190:117283. [53] WEN J, BAO M, TANG M, et al. Low magnitude vibration alleviates age-related bone loss by inhibiting cell senescence of osteogenic cells in naturally senescent rats. Aging (Albany NY). 2021;13(8):12031-12045. [54] KENNEDY IW, TSIMBOURI PM, CAMPSIE P, et al. Nanovibrational stimulation inhibits osteoclastogenesis and enhances osteogenesis in co-cultures. Sci Rep. 2021;11(1):22741. [55] MASUD AA, SHEN CL, LUK HY, et al. Impact of Local Vibration Training on Neuromuscular Activity, Muscle Cell, and Muscle Strength: A Review. Crit Rev Biomed Eng. 2022;50(1):1-17. [56] BRUGGER MS, BAUMGARTNER K, MAURITZ SCF, et al. Vibration enhanced cell growth induced by surface acoustic waves as in vitro wound-healing model. Proc Natl Acad Sci U S A. 2020;117(50):31603-31613. [57] LAGIER AJ, ELBE A, THAMKE A, et al. Vibration, a treatment for migraine, linked to calpain driven changes in actin cytoskeleton. PLoS One. 2022;17(4):e0262058. [58] ITO A, YAMAMOTO Y, SATO M, et al. Induction of functional tissue-engineered skeletal muscle constructs by defined electrical stimulation. Sci Rep. 2014;4:4781. [59] TANG B, SHEN X, YANG Y, et al. Enhanced cellular osteogenic differentiation on CoFe2O4/P(VDF-TrFE) nanocomposite coatings under static magnetic field. Colloids Surf B Biointerfaces. 2021;198:111473. [60] AAS V, BAKKE SS, FENG YZ, et al. Are cultured human myotubes far from home? Cell Tissue Res. 2013;354(3):671-682. [61] EGAN B, ZIERATH JR. Exercise metabolism and the molecular regulation of skeletal muscle adaptation. Cell Metab. 2013;17(2):162-184. [62] ZEBEDIN E, SANDTNER W, GALLER S, et al. Fiber type conversion alters inactivation of voltage-dependent sodium currents in murine C2C12 skeletal muscle cells. Am J Physiol Cell Physiol. 2004;287(2):C270-C280. [63] HAWLEY JA, HARGREAVES M, JOYNER MJ, et al. Integrative biology of exercise. Cell. 2014; 159(4):738-749. [64] LAMBERTUCCI RH, SILVEIRA LDOS R, HIRABARA SM, et al. Effects of moderate electrical stimulation on reactive species production by primary rat skeletal muscle cells: cross talk between superoxide and nitric oxide production. J Cell Physiol. 2012;227(6):2511-2518. [65] CLEVERS H. Modeling Development and Disease with Organoids. Cell. 2016;165(7):1586-1597. [66] ZARKOOB H, ALLUÉ-GUARDIA A, CHEN YC, et al. Modeling SARS-CoV-2 and influenza infections and antiviral treatments in human lung epithelial tissue equivalents. Commun Biol. 2022;5(1):810. [67] VARONE A, NGUYEN JK, LENG L, et al. A novel organ-chip system emulates three-dimensional architecture of the human epithelia and the mechanical forces acting on it. Biomaterials. 2021;275:120957. [68] SCHLÜNDER K, CIPRIANO M, ZBINDEN A, et al. Microphysiological pancreas-on-chip platform with integrated sensors to model endocrine function and metabolism. Lab Chip. 2024;24(7):2080-2093. [69] PARAFATI M, GIZA S, SHENOY TS, et al. Human skeletal muscle tissue chip autonomous payload reveals changes in fiber type and metabolic gene expression due to spaceflight. NPJ Microgravity. 2023;9(1):77. [70] MIKLAS JW, NUNES SS, SOFLA A, et al. Bioreactor for modulation of cardiac microtissue phenotype by combined static stretch and electrical stimulation. Biofabrication. 2014;6(2):024113. [71] WANG B, WANG G, TO F, et al. Myocardial scaffold-based cardiac tissue engineering: application of coordinated mechanical and electrical stimulations. Langmuir. 2013;29(35):11109-11117. [72] VAN NORMAN GA. Overcoming the Declining Trends in Innovation and Investment in Cardiovascular Therapeutics: Beyond EROOM’s Law. JACC Basic Transl Sci. 2017;2(5):613-625. [73] GUBERT C, HANNAN AJ. Exercise mimetics: harnessing the therapeutic effects of physical activity. Nat Rev Drug Discov. 2021;20(11): 862-879. [74] TORO-ROMÁN V, BARTOLOMÉ I, SIQUIER-COLL J, et al. Serum vanadium concentrations in different sports modalities. J Trace Elem Med Biol. 2021;68:126808. [75] BETTARIGA F, TAAFFE DR, GALVÃO DA, et al. Suppressive effects of exercise-conditioned serum on cancer cells: A narrative review of the influence of exercise mode, volume, and intensity. J Sport Health Sci. 2024;13(4):484-498. [76] DE MIGUEL Z, KHOURY N, BETLEY MJ, et al. Exercise plasma boosts memory and dampens brain inflammation via clusterin. Nature. 2021; 600(7889):494-499. [77] FINDEISEN M, ALLEN TL, HENSTRIDGE DC, et al. Treatment of type 2 diabetes with the designer cytokine IC7Fc. Nature. 2019;574(7776):63-68. [78] KURZ E, HIRSCH CA, DALTON T, et al. Exercise-induced engagement of the IL-15/IL-15Rα axis promotes anti-tumor immunity in pancreatic cancer. Cancer Cell. 2022;40(7):720-737.e5. [79] ZHANG B, LI F, SHI Y, et al. Single-cell RNA sequencing integrated with bulk RNA sequencing analysis reveals the protective effects of lactate-mediated lactylation of microglia-related proteins on spinal cord injury. CNS Neurosci Ther. 2024;30(9):e70028. [80] OSTROWSKI K, ROHDE T, ZACHO M, et al. Evidence that interleukin-6 is produced in human skeletal muscle during prolonged running. J Physiol. 1998;508 ( Pt 3)(Pt 3):949-953. [81] BOSTRÖM P, WU J, JEDRYCHOWSKI MP, et al. A PGC1-α-dependent myokine that drives brown-fat-like development of white fat and thermogenesis. Nature. 2012;481(7382):463-468. [82] RUAS JL, WHITE JP, RAO RR, et al. A PGC-1α isoform induced by resistance training regulates skeletal muscle hypertrophy. Cell. 2012;151(6): 1319-1331. [83] LUO Z, MEI J, WANG X, et al. Voluntary exercise sensitizes cancer immunotherapy via the collagen inhibition-orchestrated inflammatory tumor immune microenvironment. Cell Rep. 2024;43(9):114697. [84] OZAN M, BUZDAĞLI Y, BAYGUTALP NK, et al. Serum BDNF and Selenium Levels in Elite Athletes Exposed to Blows. Medicina (Kaunas). 2022;58(5):608. [85] CHIA E, MARINO FE. Serum brain-derived neurotrophic factor (BDNF) and self-paced time-trial performance in older untrained men. PLoS One. 2023;18(7):e0285628. [86] VAN DER STEDE T, VAN DE LOOCK A, TURIEL G, et al. Cellular deconstruction of the human skeletal muscle microenvironment identifies an exercise-induced histaminergic crosstalk. Cell Metab. 2025;37(4):842-856.e7. [87] LIU L, KIM S, BUCKLEY MT, et al. Exercise reprograms the inflammatory landscape of multiple stem cell compartments during mammalian aging. Cell Stem Cell. 2023;30(5): 689-705.e4. [88] SAFDAR A, TARNOPOLSKY MA. Exosomes as Mediators of the Systemic Adaptations to Endurance Exercise. Cold Spring Harb Perspect Med. 2018;8(3):a029827. [89] LÖTVALL J, HILL AF, HOCHBERG F, et al. Minimal experimental requirements for definition of extracellular vesicles and their functions: a position statement from the International Society for Extracellular Vesicles. J Extracell Vesicles. 2014;3:26913. [90] VITUCCI D, MARTONE D, ALFIERI A, et al. Muscle-derived exosomes and exercise in cancer prevention. Front Mol Med. 2023;3:1202190. [91] FU G, WANG Z, HU S. Exercise improves cardiac fibrosis by stimulating the release of endothelial progenitor cell-derived exosomes and upregulating miR-126 expression. Front Cardiovasc Med. 2024; 11:1323329. [92] LIANG X, FA W, WANG N, et al. Exosomal miR-532-5p induced by long-term exercise rescues blood-brain barrier function in 5XFAD mice via downregulation of EPHA4. Aging Cell. 2023; 22(1):e13748. [93] ZHAO K, CUI Y, DU Y, et al. Exercise Mimetic Exosomes Re-establish the Extracellular Matrix Metabolic Balance and Alleviate the Inflammatory Macrophage Infiltration in Intervertebral Disc Degeneration. Adv Healthc Mater. 2025;14(20): e2500219. [94] WANG J, FAN S, WANG J. Exercise and its beneficial impacts on mood disorders and neurodegeneration: a novel mechanistic perspective of the exosomes involvement. Mol Cell Biochem. 2025;480(11):5639-5658. [95] LAI Z, LIANG J, ZHANG J, et al. Exosomes as a delivery tool of exercise-induced beneficial factors for the prevention and treatment of cardiovascular disease: a systematic review and meta-analysis. Front Physiol. 2023;14:1190095. [96] ASWAD H, FORTERRE A, WIKLANDER OP, et al. Exosomes participate in the alteration of muscle homeostasis during lipid-induced insulin resistance in mice. Diabetologia. 2014;57(10):2155-2164. [97] OJIMA K, OE M, NAKAJIMA I, et al. Proteomic analysis of secreted proteins from skeletal muscle cells during differentiation. EuPA Open Proteomics. 2014;5:1-9. [98] FRÜHBEIS C, HELMIG S, TUG S, et al. Physical exercise induces rapid release of small extracellular vesicles into the circulation. J Extracell Vesicles. 2015;4:28239. [99] CHATURVEDI P, KALANI A, MEDINA I, et al. Cardiosome mediated regulation of MMP9 in diabetic heart: role of mir29b and mir455 in exercise. J Cell Mol Med. 2015;19(9):2153-2161. |
| [1] | Haonan Yang, Zhengwei Yuan, Junpeng Xu, Zhiqi Mao, Jianning Zhang. Preliminary study on the mechanisms and efficacy of deep brain stimulation in treating depression [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(在线): 1-9. |
| [2] | Sun Yaotian, Xu Kai, Wang Peiyun. Potential mechanisms by which exercise regulates iron metabolism in immune inflammatory diseases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1486-1498. |
| [3] | Wen Fan, Xiang Yang, Zhu Huan, Tuo Yanfang, Li Feng. Exercise improves microvascular function in patients with type 2 diabetes [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1225-1235. |
| [4] | Jiang Yang, Peng Hao, Song Yanping, Yao Na, Song Yueyu, Yin Xingxiao, Li Yanqi, Chen Qigang. Isometric exercise reduces resting blood pressure: a meta-analysis of moderating factors and dose effects [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 975-986. |
| [5] | Qin Di, Qin Xuelin, Li Zhu, Ye Jiachi, Chen Gan, Lin Yi, Peng Yong. Differential proteomic analysis of exercise-induced and pathological cardiac hypertrophy models in mice [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(28): 7287-7296. |
| [6] | Liang Zihan, Wang Rui, Sun Lei, Jin Ranran, Lyu Pengju, Li Yalong, Cheng Chaofei, Yue Han, Shen Sining. Overexpression of programmed death ligand 1 enhances immunosuppressive capacity of human umbilical cord mesenchymal stromal cells against T cells [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(19): 4873-4881. |
| [7] | Li Tianbo, Yu Zeyang, Qin Xinyuan, Wang Jiangning, Gao Lei. Exosomes derived from human umbilical cord mesenchymal stem cells in treatment of diabetic foot ulcers [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(19): 4897-4901. |
| [8] | Li Jiapeng, Zuleina·Abula, Jia Qianqian, Nigare·Yunusijiang, Sun Jiaqi, Zhao Jin, Wu Zeyu. Advantages and potential of cell-derived exosomes in oral tissue regeneration [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(19): 5015-5023. |
| [9] | Zhang Shuli, Hou Chaowen, Yuan Shanshan, Ma Yuhua . Mechanism by which exercise regulates autophagy in different physiological systems [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(18): 4737-4748. |
| [10] | Yang Jiangxi, Li Huangyan, Zhang Yeting, Yu Zuoyin . Research hotspots and thematic evolution in the field of exercise interventions for multiple sclerosis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(18): 4771-4781. |
| [11] | Wumiti·Taxi, Wang Lining, Li Muzhe, Sun Jie, Chen Shuangliu, Zhu Yihua, Zhou Shijie, Ma Yong, Guo Yang. Wenshen Tongluo Zhitong decoction regulates the bone fat differentiation balance of bone marrow mesenchymal stem cells through exosomal miR-342-3p [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(13): 3258-3269. |
| [12] | Duan Cheng, Cheng Jie. Effect of miR-223-3p on biological behavior of tendon stem cells under hypoxic conditions [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(13): 3298-3307. |
| [13] | Zhang Shilei, Qin Chuanhong, Wang Jianxu, Sun Shui. Osteogenic-adipogenic differentiation imbalance of bone marrow mesenchymal stem cells and osteonecrosis of the femoral head: from molecular mechanisms to therapeutic strategies [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(13): 3350-3358. |
| [14] | Mao Sujie, Gao Jie, Pan Zhuangli. Immune cells synergistically regulate inflammatory response, muscle regeneration and metabolic homeostasis in training-induced stress responses [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(10): 2671-2680. |
| [15] | Liu Ziwei, Nijati·Tursun, Yin Rui, Li Shuhui, Zhou Jing. Effect of cannabinoid type I receptors on neuronal differentiation of human apical papilla stem cells [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(1): 93-100. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||