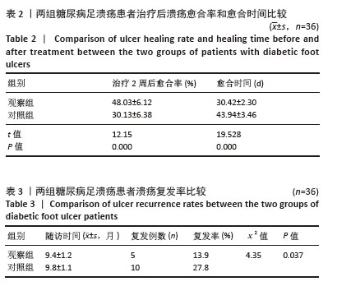
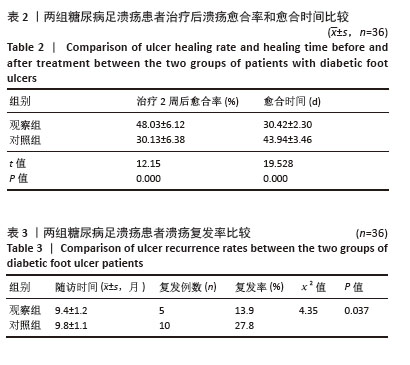
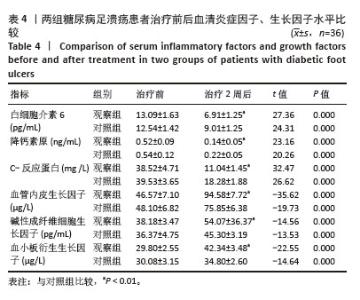
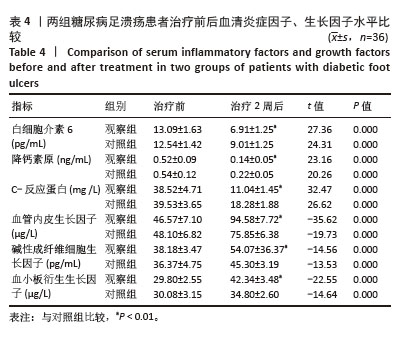
Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (19): 4897-4901.doi: 10.12307/2026.797
Previous Articles Next Articles
Exosomes derived from human umbilical cord mesenchymal stem cells in treatment of diabetic foot ulcers
Li Tianbo1, 2, Yu Zeyang1, 2, Qin Xinyuan1, 2, Wang Jiangning1, Gao Lei1, 2
- 1Department of Orthopedic Surgery, 2Department of Plastic Surgery, Beijing Shijitan Hospital, Capital Medical University, Beijing 100038, China
-
Received:2025-09-23Accepted:2025-12-03Online:2026-07-08Published:2026-02-14 -
Contact:Gao Lei, MD, Associate chief physician, Master’s supervisor, Department of Orthopedic Surgery, and Department of Plastic Surgery, Beijing Shijitan Hospital, Capital Medical University, Beijing 100038, China -
About author:Li Tianbo, Attending physician, Department of Orthopedic Surgery, and Department of Plastic Surgery, Beijing Shijitan Hospital, Capital Medical University, Beijing 100038, China -
Supported by:Beijing Yanhua Vascular and Wound Treatment Technology Development Foundation, No. 2022-B01 (to WJN)
CLC Number:
Cite this article
Li Tianbo, Yu Zeyang, Qin Xinyuan, Wang Jiangning, Gao Lei. Exosomes derived from human umbilical cord mesenchymal stem cells in treatment of diabetic foot ulcers[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(19): 4897-4901.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
| [1] ARMSTRONG DG, TAN TW, BOULTON AJM, et al. Diabetic Foot Ulcers: A Review. JAMA. 2023;330(1):62-75. [2] YIN K, QIAO T, ZHANG Y, et al. Unraveling shared risk factors for diabetic foot ulcer: a comprehensive Mendelian randomization analysis. BMJ Open Diabetes Res Care. 2023;11(6):e003523. [3] REHMAN ZU, KHAN J, NOORDIN S. Diabetic Foot Ulcers: Contemporary Assessment And Management. J Pak Med Assoc. 2023;73(7):1480-1487. [4] 张璟琳,冷敏,朱博恒,等.干细胞源外泌体促进糖尿病创面愈合的机制及应用[J].中国组织工程研究,2022,26(7):1113-1118. [5] LOTFY A, ABOQUELLA NM, WANG H. Mesenchymal stromal/stem cell (MSC)-derived exosomes in clinical trials. Stem Cell Res Ther. 2023;14(1):66. [6] ZHANG K, CHENG K. Stem cell-derived exosome versus stem cell therapy. Nat Rev Bioeng. 2023. doi: 10.1038/s44222-023-00064-2. [7] SALEEM M, SHAHZAD KA, MARRYUM M, et al. Exosome-based therapies for inflammatory disorders: a review of recent advances. Stem Cell Res Ther. 2024; 15(1):477. [8] LEE JH, WON YJ, KIM H, et al. Adipose Tissue-Derived Mesenchymal Stem Cell-Derived Exosomes Promote Wound Healing and Tissue Regeneration. Int J Mol Sci. 2023;24(13):10434. [9] WANG H, WU S, BAI X, et al. Mesenchymal Stem Cell-Derived Exosomes Hold Promise in the Treatment of Diabetic Foot Ulcers. Int J Nanomedicine. 2025;20: 5837-5857. [10] MCCOMB WD JR. Wagner Grading of Diabetic Foot Ulcers and National Pressure Injury Advisory Panel Staging of Pressure Injuries: A Comparison for Clinical Use. Adv Skin Wound Care. 2023;36(5):278-279. [11] CHESHIRE BL, MESSEDER SJ, PEPPER CJ, et al. Association of cognitive impairment and peripheral artery disease (PAD): A systematic review. Vasc Med. 2025:1358863X251336736. doi: 10.1177/1358863X251336736. [12] SENNEVILLE É, ALBALAWI Z, VAN ASTEN SA, et al. IWGDF/IDSA guidelines on the diagnosis and treatment of diabetes-related foot infections (IWGDF/IDSA 2023). Diabetes Metab Res Rev. 2024;40(3):e3687. [13] JOHNSON J, LAW SQK, SHOJAEE M, et al. First-in-human clinical trial of allogeneic, platelet-derived extracellular vesicles as a potential therapeutic for delayed wound healing. J Extracell Vesicles. 2023;12(7):e12332. [14] WU Y, SHEN G, HAO C. Negative pressure wound therapy (NPWT) is superior to conventional moist dressings in wound bed preparation for diabetic foot ulcers: A randomized controlled trial. Saudi Med J. 2023;44(10):1020-1029. [15] HE J, WANG X, WANG Z, et al. Interpretable deep learning method to predict wound healing progress based on collagen fibers in wound tissue. Comput Biol Med. 2025;191:110110. [16] ZHU S, ZENG C, ZOU Y, et al. The Clinical Diagnostic Values of SAA, PCT, CRP, and IL-6 in Children with Bacterial, Viral, or Co-Infections. Int J Gen Med. 2021;14: 7107-7113. [17] SKOULIDIS F, LI BT, HOCHMAIR M, et al. Pooled safety analysis and management of sotorasib-related adverse events in KRAS G12C-mutated advanced non-small cell lung cancer. Oncologist. 2025;30(1):oyae356. [18] HUANG C, LUO W, WANG Q, et al. Human mesenchymal stem cells promote ischemic repairment and angiogenesis of diabetic foot through exosome miRNA-21-5p. Stem Cell Res. 2021;52:102235. [19] SUN J, LIAO Z, LI Z, et al. Down-regulation miR-146a-5p in Schwann cell-derived exosomes induced macrophage M1 polarization by impairing the inhibition on TRAF6/NF-κB pathway after peripheral nerve injury. Exp Neurol. 2023;362:114295. [20] YANG H, XU H, WANG Z, et al. Analysis of miR-203a-3p/SOCS3-mediated induction of M2 macrophage polarization to promote diabetic wound healing based on epidermal stem cell-derived exosomes. Diabetes Res Clin Pract. 2023; 197:110573. [21] LI T, GAO L, WANG J. Identification of Exosome-Associated Biomarkers in Diabetic Foot Ulcers: A Bioinformatics Analysis and Experimental Validation. Biomedicines. 2025;13(7):1687. [22] SANADA A, YAMADA T, HASEGAWA S, et al. Enhanced Type I Collagen Synthesis in Fibroblasts by Dermal Stem/Progenitor Cell-Derived Exosomes. Biol Pharm Bull. 2022;45(7):872-880. [23] WANG Y, ZHANG Y, LI T, et al. Adipose Mesenchymal Stem Cell Derived Exosomes Promote Keratinocytes and Fibroblasts Embedded in Collagen/Platelet-Rich Plasma Scaffold and Accelerate Wound Healing. Adv Mater. 2023;35(40):e2303642. [24] YU X, LIU P, LI Z, et al. Function and mechanism of mesenchymal stem cells in the healing of diabetic foot wounds. Front Endocrinol (Lausanne). 2023;14:1099310. [25] SEGURA-EGEA JJ, CABANILLAS-BALSERA D, MARTÍN-GONZÁLEZ J, et al. Impact of systemic health on treatment outcomes in endodontics. Int Endod J. 2023;56 Suppl 2:219-235. [26] YARAHMADI A, SAEED MODAGHEGH MH, MOSTAFAVI-POUR Z, et al. The effect of platelet-rich plasma-fibrin glue dressing in combination with oral vitamin E and C for treatment of non-healing diabetic foot ulcers: a randomized, double-blind, parallel-group, clinical trial. Expert Opin Biol Ther. 2021;21(5):687-696. [27] CHEN L, ZHENG B, XU Y, et al. Nano hydrogel-based oxygen-releasing stem cell transplantation system for treating diabetic foot. J Nanobiotechnology. 2023; 21(1):202. [28] 王国燕,李勤雨,贾玉萍,等.外周血干细胞联合外泌体治疗糖尿病足溃疡创面疗效观察[J].中国美容医学,2024,33(8):33-37. [29] 梁荟玲,马立人,王晓歌,等.生肌玉红膏联合脐带间充质干细胞外泌体治疗糖尿病足创面的临床研究[J].血管与腔内血管外科杂志,2024,10(9): 1114-1117. [30] WANG X, ZHANG W, MAO L, et al. Inflammation-related gene ITGA5 affects the healing of diabetic foot ulcers through PI3K-AKT signaling pathway. Eur J Pharmacol. 2025;1002:177865. [31] MOHSIN F, JAVAID S, TARIQ M, et al. Molecular immunological mechanisms of impaired wound healing in diabetic foot ulcers (DFU), current therapeutic strategies and future directions. Int Immunopharmacol. 2024;139:112713. [32] WORSLEY AL, LUI DH, NTOW-BOAHENE W, et al. The importance of inflammation control for the treatment of chronic diabetic wounds. Int Wound J. 2023;20(6):2346-2359. [33] ZHA W, WANG J, GUO Z, et al. Efficient delivery of VEGF-A mRNA for promoting diabetic wound healing via ionizable lipid nanoparticles. Int J Pharm. 2023;632: 122565. [34] LIN J, LIN Z, HUANG A, et al. Effects of a PDGF-stem cell-hydrogel compound on skin wound healing in mice. Cytotherapy. 2025;27(5):609-618. [35] ZHANG S, LU X, CHEN J, et al. Promotion of angiogenesis and suppression of inflammatory response in skin wound healing using exosome-loaded collagen sponge. Front Immunol. 2024;15:1511526. [36] XIAO Y, LIU J, DAI F, et al. Dual Exosome Coating for Modulating Endothelial Function and Inflammation. ACS Appl Mater Interfaces. 2025;17(21):30492-30504. [37] 王宸宸,李天博,郭瑞敏,等.可吸收支架负载外泌体方法研究[J].中国修复重建外科杂志,2024,38(2):206-210. [38] YANG J, CHEN Z, PAN D, et al. Umbilical Cord-Derived Mesenchymal Stem Cell-Derived Exosomes Combined Pluronic F127 Hydrogel Promote Chronic Diabetic Wound Healing and Complete Skin Regeneration. Int J Nanomedicine. 2020;15:5911-5926. [39] PSARROU M, MITRAKI A, VAMVAKAKI M, et al. Stimuli-Responsive Polysaccharide Hydrogels and Their Composites for Wound Healing Applications. Polymers (Basel). 2023;15(4):986. [40] HU N, CAI Z, JIANG X, et al. Hypoxia-pretreated ADSC-derived exosome-embedded hydrogels promote angiogenesis and accelerate diabetic wound healing. Acta Biomater. 2023;157:175-186. |
| [1] | Jiang Xinghai, Song Yulin, Li Dejin, Shao Jianmin, Xu Junzhi, Liu Huakai, Wu Yingguo, Shen Yuehui, Feng Sicheng. Vascular endothelial growth factor 165 genes transfected into bone marrow mesenchymal stem cells to construct a vascularized amphiphilic peptide gel module [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 1903-1911. |
| [2] | Wang Baiyan, Yang Shu, Wang Yiming, Wu Mengqing, Xiao Yu, Guo Zixuan, Zhang Boyi, Feng Shuying. Exosome-delivered CRISPR/Cas system enables gene editing in target cells [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1839-1849. |
| [3] | Guo Jiachen, Gao Jun, Dai Wenhao, Liao Huayuan, Jiang You, Zhang Xi . Effect of compressive stress microenvironment on cytokines during fracture healing [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 908-916. |
| [4] | Wang Zhengye, Liu Wanlin, Zhao Zhenqun. Mechanism by which vascular endothelial growth factor A targets regulation of angiogenesis in the treatment of steroid-induced osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(3): 671-679. |
| [5] | Zhou Shibo, Yu Xing, Chen Hailong, Xiong Yang. Nanocrystalline collagen-based bone combined with Bushen Zhuangjin Decoction repairs bone defects in osteoporotic rats [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(2): 354-361. |
| [6] | Zhang Shujuan, Xu Qianqian, Wang Chao, Li Yunhui, Zhu Yanping. Transplantation of human umbilical cord mesenchymal stem cells to repair myelination disorders in neonatal rats with white matter injury [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(19): 4890-4896. |
| [7] | Jian Xichao, Shao Jingjie, Tang Shihan, Qi Fang, Deng Chengliang. Exosomes promote diabetic wound healing: a visual analysis of research hotspots and evolutionary trends [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(19): 5072-5081. |
| [8] | Wang Xinyue, Li Hongli, Guo Chunhui, Chen Jibing, Yu Hua. Changes in the expression of six microRNAs in ovarian tissue from animal models of premature ovarian failure and in peripheral blood of patients with premature ovarian failure [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(18): 4675-4684. |
| [9] | Wang Zhengye, Liu Wanlin, Zhao Zhenqun. Multidimensional target regulation of vascular endothelial growth factor A in articular cartilage development [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(16): 4193-4203. |
| [10] | Duan Cheng, Cheng Jie. Effect of miR-223-3p on biological behavior of tendon stem cells under hypoxic conditions [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(13): 3298-3307. |
| [11] | Lu Biqiong, Wei Zhongjian. Skeletal muscle-derived exosome-mediated regulation of bone formation and role of exercise intervention [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(13): 3379-3391. |
| [12] | Huang Jiayao, Gu Yu. Application of exosomes in the diagnosis and monitoring of oral diseases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(13): 3392-3401. |
| [13] | Wu Xianyuan, Zhang Nini, Huang Guilin. Gene transfection technology and tissue fibrosis repair [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(13): 3424-3434. |
| [14] | Sun Huiwen, Guo Qiangqiang, Wang Wei, Wu Jie, Xi Kun, Gu Yong. Engineered stem cell bionic periosteum coordinates immune inflammation and vascularization to promote bone regeneration [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(1): 21-33. |
| [15] | Jin Kai, Tang Ting, Li Meile, Xie Yuan. Effects of conditioned medium and exosomes of human umbilical cord mesenchymal stem cells on proliferation, migration, invasion, and apoptosis of hepatocellular carcinoma cells [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(7): 1350-1355. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||