Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (29): 7603-7611.doi: 10.12307/2026.215
Previous Articles Next Articles
Molecular mechanisms by which Fusobacterium nucleatum regulates colonic polyp formation in mice
Tian Yu1, Guo Ying1, Yiershatijiang Aniwaer1, Aihematijiang Refuhaiti1, Maierdana Maimaitireyimu2
- 1Department of Gastroenterology, the Sixth Affiliated Hospital of Xinjiang Medical University, Urumqi 830002, Xinjiang Uygur Autonomous Region, China; 2First Clinical Medical College, Shaanxi University of Chinese Medicine, Xi’an 712046, Shaanxi Province, China
-
Received:2025-07-06Revised:2025-11-27Online:2026-10-18Published:2026-03-04 -
Contact:Tian Yu, Department of Gastroenterology, the Sixth Affiliated Hospital of Xinjiang Medical University, Urumqi 830002, Xinjiang Uygur Autonomous Region, China -
About author:Tian Yu, MS, Associate chief physician, Master’s supervisor, Department of Gastroenterology, the Sixth Affiliated Hospital of Xinjiang Medical University, Urumqi 830002, Xinjiang Uygur Autonomous Region, China -
Supported by:Natural Science Foundation of Xinjiang Uygur Autonomous Region (General Program), No. 2022D01C332 (to TY)
CLC Number:
Cite this article
Tian Yu, Guo Ying, Yiershatijiang Aniwaer, Aihematijiang Refuhaiti, Maierdana Maimaitireyimu. Molecular mechanisms by which Fusobacterium nucleatum regulates colonic polyp formation in mice[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(29): 7603-7611.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
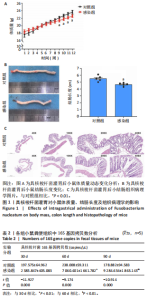
2.1 具核梭杆菌感染对小鼠生理及病理表型的影响 通过12周具核梭杆菌灌胃干预,发现具核梭杆菌长期感染可导致小鼠体质量增长抑制、结肠长度缩短及黏膜病理损伤。如图1A所示,对照组与感染组小鼠体质量随时间均呈递增趋势,前8周两组体质量差异无显著性意义(均P > 0.05);第9周起,感染组体质量显著低于对照组[第9周:(21.42±0.33) g vs. (22.16±0.31) g,t=3.277,P=0.011;第10周:(21.78±0.33) g vs. (22.72±0.32) g,t=4.130,P=0.003],且差异持续至第12周(P均 < 0.01)。干预12周后,感染组结肠长度较对照组显著缩短[(4.66±0.23) cm vs. (5.52±0.42) cm,t=4.054,P=0.004],见图1B。结肠缩短通常与慢性炎症导致的纤维化或组织重构相关,说明具核梭杆菌可能通过破坏结肠黏膜屏障引发结构性损伤。苏木精-伊红染色显示(图1C),对照组结肠黏膜结构完整,腺体排列紧密,杯状细胞丰富,仅见少量慢性炎症细胞浸润;感染组则出现腺体萎缩、排列松散,杯状细胞显著减少,固有层及黏膜下层中性粒细胞、嗜酸性粒细胞浸润增多,说明具核梭杆菌感染可通过诱导局部炎症微环境促进结肠黏膜异常增生。 2.2 具核梭杆菌肠道定植与增殖动态 通过荧光定量PCR技术定量检测小鼠粪便中具核梭杆菌的16S rRNA基因拷贝数,验证感染模型有效性及评估细菌负荷动态变化。在实验的不同时间点(30,60,90 d),分别测量了对照组和感染组的16S基因拷贝数。如表2所示,在30 d时,对照组的16S基因拷贝数为(197.575±64.962) copies/μL,而感染组则显著增高至(2 385.847±405.083) copies/μL(t=-11.927,P=0.000)。在60 d时,感染组的16S基因拷贝数继续大幅上升,达到(7 060.401±1 661.782) copies/μL,与对照组(238.008±59.311) copies/μL相比,差异极为显著(t= "

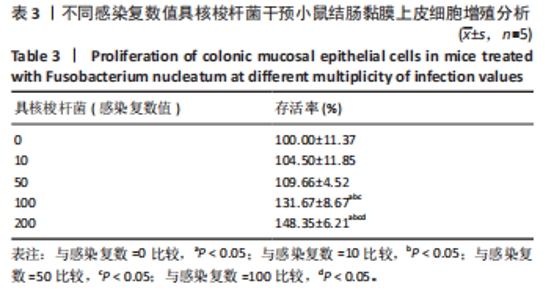
-9.174,P=0.000)。在90 d时,感染组的16S基因拷贝数进一步增加至(9 284.634±1 863.143) copies/μL,与对照组(178.882± 94.583) copies/μL相比,差异仍然极为显著(t=-10.914,P=0.000)。综上所述,此研究发现感染组16S rRNA基因拷贝数显著高于对照组(P < 0.01),且随时间递增,表明具核梭杆菌在肠道内持续增殖,导致小鼠肠道内细菌负荷量显著增加。 2.3 具核梭杆菌最佳感染复数值的筛选 此研究旨在筛选具核梭杆菌感染小鼠结肠黏膜上皮细胞的最佳感染复数值,以明确细菌负荷与细胞增殖的关系。实验设置感染复数值梯度为0,10,50,100,200,如表3所示,随着感染复数值增加,细胞存活率呈剂量依赖性显著升高(均P < 0.05)。感染复数=0(对照组)时存活率为(100.00±11.37)%;感染复数=10,50时存"
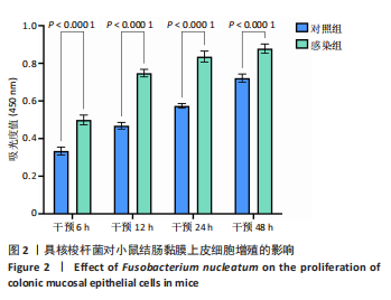
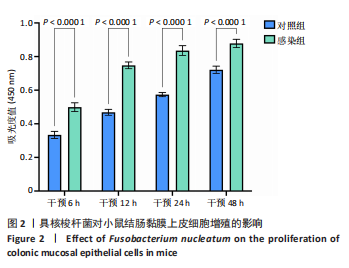
活率分别为(104.50±11.85)%和(109.66±4.52)%,差异无显著性意义(P > 0.05),表明低剂量感染对细胞增殖影响有限。感染复数=100时存活率升至(131.67±8.67)%(P < 0.05 vs. 感染复数=0);感染复数=200时达峰值(148.35±6.21)%(P < 0.05 vs. 所有组),表明高剂量具核梭杆菌显著促进细胞增殖。 2.4 具核梭杆菌对细胞活力时效性的影响 为评估具核梭杆菌对小鼠结肠黏膜上皮细胞活力的时效性影响,通过CCK-8法检测了不同干预时间点(6,12,24,48 h)的吸光度值。如图2所示,感染组(感染复数=200)的细胞活力显著高于对照组,且随时间延长呈现持续增强趋势。干预6 h时,对照组吸光度值为0.334±0.047,而感染组吸光度值显著升高至0.499±0.058(P < 0.000 1),表明具核梭杆菌在感染早期即可诱导细胞活力增强。随着干预时间延长,两组吸光度值均逐渐上升,但感染组上升更快。干预12 h,感染组吸光度值达0.748±0.045,较对照组(0.468±0.040)提升60.0%(P < 0.000 1)。直至干预48 h,感染组吸光度值达峰值0.878±0.055,较对照组(0.722±0.049)仍保持显著优势(P < 0.000 1)。上述结果表明,具核梭杆菌以时间依赖性方式显著促进结肠上皮细胞增殖。"
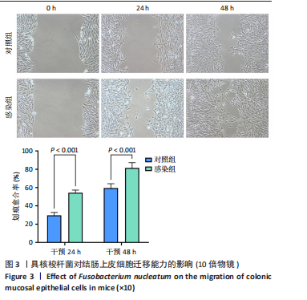
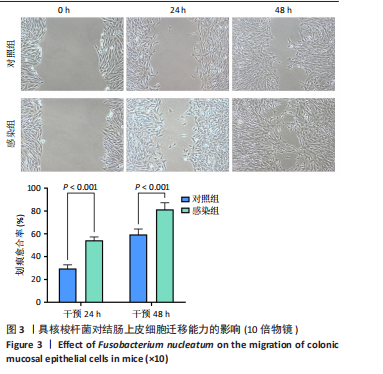
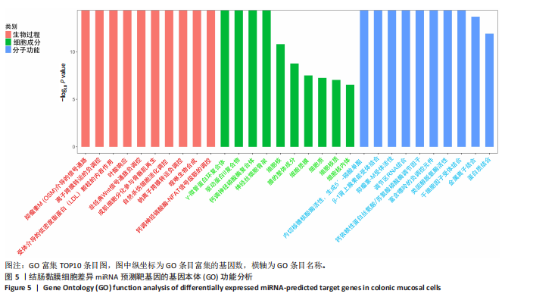
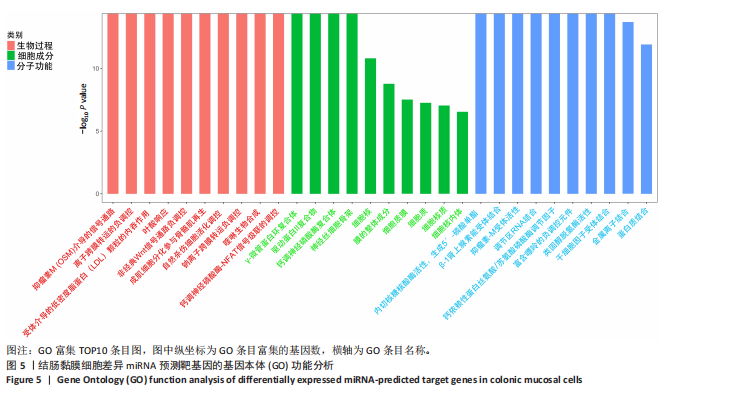
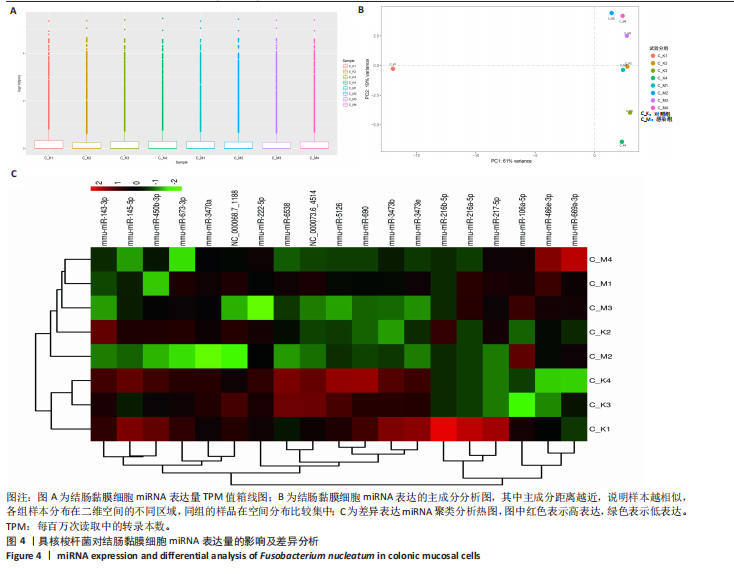
(29.68±3.15)%提升83.1%(P < 0.001);直至48 h,感染组划痕愈合率进一步升至(81.38±5.81)%,较对照组(59.44±4.78)%提升36.9%(P < 0.001),表明具核梭杆菌可显著增强结肠上皮细胞迁移能力,可能通过破坏细胞间连接或激活促迁移信号通路实现。 2.6 具核梭杆菌对结肠黏膜细胞miRNA表达量的影响及差异分析 每百万次读取中的转录本数(transcripts per million,TPM)箱线图显示样本间表达量分布相似(图4A),表明实验重复性良好。主成分分析结果显示,对照组与菌液感染组在二维空间中的分布呈现显著分离趋势(图4B),表明具核梭杆菌处理后结肠黏膜细胞的miRNA整体表达谱发生显著改变。第一主成分(PC1)解释了61%的变异,第二主成分(PC2)贡献了19%的变异,说明实验干预对miRNA表达的影响具有高度特异性。组内样本分布集中,表明实验重复性良好。这一结果提示,具核梭杆菌可能通过调控特定miRNA的表达,驱动结肠黏膜细胞的分子表型变化,为后续差异miRNA的筛选提供了数据支持。 基于差异表达的19个miRNA(3个上调,16个下调)进行非监督层次聚类分析(图4C),结果显示对照组与实验组样本分别聚为独立簇,进一步验证了主成分分析的结论。热图中红色区域(高表达)富集于对照组样本,绿色区域(低表达)富集于感染组样本,表明具核梭杆菌感染可能显著抑制多数miRNA的表达。值得注意的是,miR-143-3p、miR-145-5p等已知与结直肠肿瘤发生相关的miRNA在实验组显著下调,表明它们可能通过靶向促癌基因参与结肠息肉形成的调控。 2.7 差异miRNA预测靶基因功能分析 对差异miRNA的预测靶基因进行功能富集分析,GO分析(图5)显示,预测靶基因显著富集于“细胞增殖调控”(GO:0042127)和“信号转导”(GO:0007165)等生物学过程,表明miRNA可能通过调控生长相关通路发挥作用,促进细胞异常增殖并抑制凋亡,与结肠息肉的发生密切相关。 KEGG分析(图6A)表明,预测靶基因主要参与“丝裂原活"
| [1] 王佳佳,刘杰,王敏.构建具核梭杆菌诱导的实验性根尖周炎小鼠模型[J].中国组织工程研究,2022,26(2):176-181. [2] MONDAL T, CHATTOPADHYAY D, SAHA MONDAL P, et al. Fusobacterium nucleatum modulates the Wnt/β-catenin pathway in colorectal cancer development. Int J Biol Macromol. 2025;299:140196. [3] 郑文香,丛立春,韩斌.沉默信息调节因子1在具核梭杆菌诱导肠上皮细胞炎症和凋亡中的作用[J].中国病原生物学杂志,2019,14(6):643-649+655. [4] HUH JW, ROH TY. Opportunistic detection of Fusobacterium nucleatum as a marker for the early gut microbial dysbiosis. BMC Microbiol. 20203;20(1):208. [5] 王子奇,谭诗云.溃疡性结肠炎患者结直肠息肉复发的相关危险因素分析[J].胃肠病学和肝病学杂志,2025,34(3):373-376. [6] SHI L, LI H, LI S, et al. Pseudoinvasion and squamous metaplasia/morules in colorectal adenomatous polyp: a case report and literature review. Diagn Pathol. 2024;19(1):126. [7] LIU Y, LIN XX, HU SS, et al. The microbiota comparative analysis of the characteristics between colorectal adenomatous polyps and normal mucosal intestinal. Eur J Gastroenterol Hepatol. 2024;36(11):1305-1313. [8] BHATTACHARJEE R, PRABHAKAR N, KUMAR L, et al. Crosstalk between long noncoding RNA and microRNA in Cancer. Cell Oncol (Dordr). 2023;46(4):885-908. [9] 张林,高林波.miR-143和miR-145与肿瘤的研究进展[J].西安交通大学学报(医学版),2013,34(1):1-6. [10] 刘久莲,刘童,高善语.microRNA在结直肠癌耐药及治疗中作用的研究进展[J].中国肿瘤生物治疗杂志,2023,30(5):438-444. [11] 叶伟标,徐咏强,李妤玲,等.MicroRNA-200c在结直肠癌中的表达及对侵袭和转移的影响[J].中山大学学报(医学版),2018,39(3):335-340. [12] GHOFRANI-SHAHPAR M, PAKRAVAN K, RAZMARA E, et al. Cancer-associated fibroblasts drive colorectal cancer cell progression through exosomal miR-20a-5p-mediated targeting of PTEN and stimulating interleukin-6 production. BMC Cancer. 2024;24(1):400. [13] GUO S, CHEN J, CHEN F, et al. Exosomes derived from Fusobacterium nucleatum-infected colorectal cancer cells facilitate tumour metastasis by selectively carrying miR-1246/92b-3p/27a-3p and CXCL16. Gut. 2020. doi: 10.1136/gutjnl-2020-321187. [14] AKTAN Ç, TEKIN F, ORUÇ N, et al. CHRM3-Associated miRNAs May Play a Role in Bile Acid-Induced Proliferation of H508 Colon Cancer Cells. Turk J Gastroenterol. 2023;34(3):298-307. [15] 周锋,沙德胜,卢瑗瑗,等.基于Wnt/β-catenin通路探究迷迭香酸对结直肠癌上皮-间质转化的抑制作用[J].西安交通大学学报(医学版),2024,45(5): 726-733. [16] WANG N, FANG JY. Fusobacterium nucleatum, a key pathogenic factor and microbial biomarker for colorectal cancer. Trends Microbiol. 2023;31(2):159-172. [17] 邵雨琪,洪洁.肠道菌群和结直肠癌发生发展及治疗的研究进展[J].中国肿瘤临床,2024,51(17):881-887. [18] ZEPEDA-RIVERA M, MINOT SS, BOUZEK H, et al. A distinct Fusobacterium nucleatum clade dominates the colorectal cancer niche. Nature. 2024;628(8007): 424-432. [19] ELLAKWA DE, MUSHTAQ N, KHAN S, et al. Molecular functions of microRNAs in colorectal cancer: recent roles in proliferation, angiogenesis, apoptosis, and chemoresistance. Naunyn Schmiedebergs Arch Pharmacol. 2024;397(8):5617-5630. [20] 张晓云,王立峰.miR-143在结肠癌组织中的差异表达及其对结肠癌细胞凋亡和迁移的影响[J].上海交通大学学报(医学版),2017,37(3):325-329. [21] BAUER KM, HUMMON AB. Effects of the miR-143/-145 microRNA cluster on the colon cancer proteome and transcriptome. J Proteome Res. 2012;11(9):4744-4754. [22] XU C, FAN L, LIN Y, et al. Fusobacterium nucleatum promotes colorectal cancer metastasis through miR-1322/CCL20 axis and M2 polarization. Gut Microbes. 2021;13(1):1980347. [23] ZHANG L, LENG XX, QI J, et al. The adhesin RadD enhances Fusobacterium nucleatum tumour colonization and colorectal carcinogenesis. Nat Microbiol. 2024;9(9):2292-2307. [24] XIANG Z, LI X, WANG X, et al. Fusobacterium nucleatum exacerbates colitis via STAT3 activation induced by Acetyl-CoA accumulation. Gut Microbes. 2025; 17(1):2489070. [25] SAMENI F, ELKHICHI PA, DADASHI A, et al. Global prevalence of Fusobacterium nucleatum and Bacteroides fragilis in patients with colorectal cancer: an overview of case reports/case series and meta-analysis of prevalence studies. BMC Gastroenterol. 2025;25(1):71. [26] SUN L, ZHANG Y, CAI J, et al. Bile salt hydrolase in non-enterotoxigenic Bacteroides potentiates colorectal cancer. Nat Commun. 2023;14(1):755. [27] PROENÇA MA, BISELLI JM, SUCCI M, et al. Relationship between Fusobacterium nucleatum, inflammatory mediators and microRNAs in colorectal carcinogenesis. World J Gastroenterol. 2018;24(47):5351-5365. [28] 冯志平,陈富坤,杨传周,等.miR-143-3p靶向KRAS阻滞PI3K/Akt信号通路抑制分化型甲状腺癌细胞增殖、侵袭和迁移[J].现代肿瘤医学,2021,29(14): 2387-2393. [29] ZHANG L, SHAO W. MicroRNA-143-3p Inhibits Wilms’ Tumor Cell Growth By Targeting the Ras/Raf/MEK/ERK Pathway. Altern Ther Health Med. 2023;29(2): 140-147. [30] ABD ELHAFEEZ AS, GHANEM HM, SWELLAM M, et al. Involvement of FAM170B-AS1, hsa-miR-1202, and hsa-miR-146a-5p in breast cancer. Cancer Biomark. 2024;39(4):313-333. [31] 李震,姚弘虞,王世豪,等.WNT信号通路在KRAS基因突变型结直肠癌中的作用[J].郑州大学学报(医学版),2021,56(3):417-422. [32] CHENG X, SHEN T, LIU P, et al. mir-145-5p is a suppressor of colorectal cancer at early stage, while promotes colorectal cancer metastasis at late stage through regulating AKT signaling evoked EMT-mediated anoikis. BMC Cancer. 2022;22(1):1151. [33] YIN Y, YAN ZP, LU NN, et al. Downregulation of miR-145 associated with cancer progression and VEGF transcriptional activation by targeting N-RAS and IRS1. Biochim Biophys Acta. 2013;1829(2):239-247. [34] YOON JH, BAE E, NAGAFUCHI Y, et al. Repression of SMAD3 by STAT3 and c-Ski induces conventional dendritic cell differentiation. Life Sci Alliance. 2024;7(9): e201900581. [35] SHAN H, TIAN G, ZHANG Y, et al. Exploring the molecular mechanisms and therapeutic potential of SMAD4 in colorectal cancer. Cancer Biol Ther. 2024; 25(1):2392341. [36] LIU L, WANG Y, YU S, et al. Transforming Growth Factor Beta Promotes Inflammation and Tumorigenesis in Smad4-Deficient Intestinal Epithelium in a YAP-Dependent Manner. Adv Sci(Weinh). 2023;10(23):e2300708. [37] ROSHANI ASL E, RASMI Y, BARADARAN B. MicroRNA-124-3p suppresses PD-L1 expression and inhibits tumorigenesis of colorectal cancer cells via modulating STAT3 signaling. J Cell Physiol. 2021;236(10):7071-7087. [38] NIKOLAIEVA N, SEVCIKOVA A, OMELKA R, et al. Gut Microbiota-MicroRNA Interactions in Intestinal Homeostasis and Cancer Development. Microorganisms. 2022;11(1):107. [39] SHI H, LIANG GF, LI Y, et al. Preparation and Evaluation of Upconversion Nanoparticles Based miRNA Delivery Carrier in Colon Cancer Mice Model. J Biomed Nanotechnol. 2019;15(11):2240-2250. |
| [1] | Wang Baiyan, Yang Shu, Wang Yiming, Wu Mengqing, Xiao Yu, Guo Zixuan, Zhang Boyi, Feng Shuying. Exosome-delivered CRISPR/Cas system enables gene editing in target cells [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1839-1849. |
| [2] | Wang Zhengye, Liu Wanlin, Zhao Zhenqun. Advance in the mechanisms underlying miRNAs in steroid-induced osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1207-1214. |
| [3] | Wang Zhengye, Liu Wanlin, Zhao Zhenqun. Mechanisms of miRNAs involved in cartilage development: new strategies and targets [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6289-6296. |
| [4] | Wu Lingjie, Zheng Kaiyuan, Wang Guangrong, Yin Chong . Strategies for the application of miRNA-targeted therapy in the treatment of osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(22): 5792-5803. |
| [5] | Li Tianbo, Yu Zeyang, Qin Xinyuan, Wang Jiangning, Gao Lei. Exosomes derived from human umbilical cord mesenchymal stem cells in treatment of diabetic foot ulcers [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(19): 4897-4901. |
| [6] | Liu Yingzhao, Ma Yanxia, Lin Yaofa, Zhang Guoqiao, Miao Weiliang, Jia Yanli, Chi Chenshen, Song Wangsheng, Li Di, Liu Chenglong, Zhang Haonan. miR-9 regulates the differentiation of neural stem cells in mouse cerebral cortex [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(19): 4942-4948. |
| [7] | Liu Yuxuan, Guan Dongsheng, Wang Jing, Ren Yihan. Exosomal miRNA as an early diagnostic biomarker and potential therapeutic target for cerebral small vessel disease [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(19): 5000-5006. |
| [8] | Jian Xichao, Shao Jingjie, Tang Shihan, Qi Fang, Deng Chengliang. Exosomes promote diabetic wound healing: a visual analysis of research hotspots and evolutionary trends [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(19): 5072-5081. |
| [9] | Wang Xinyue, Li Hongli, Guo Chunhui, Chen Jibing, Yu Hua. Changes in the expression of six microRNAs in ovarian tissue from animal models of premature ovarian failure and in peripheral blood of patients with premature ovarian failure [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(18): 4675-4684. |
| [10] | Li Wenhui, Fan Weijing, Liu Guobin. Impact of Zi-Zhu ointment on the miRNA expression profile in mouse models of diabetic ulcers: a high-throughput sequencing analysis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(17): 4337-4346. |
| [11] | Lu Biqiong, Wei Zhongjian. Skeletal muscle-derived exosome-mediated regulation of bone formation and role of exercise intervention [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(13): 3379-3391. |
| [12] | Huang Jiayao, Gu Yu. Application of exosomes in the diagnosis and monitoring of oral diseases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(13): 3392-3401. |
| [13] | Wu Xianyuan, Zhang Nini, Huang Guilin. Gene transfection technology and tissue fibrosis repair [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(13): 3424-3434. |
| [14] | Wumiti·Taxi, Wang Lining, Li Muzhe, Sun Jie, Chen Shuangliu, Zhu Yihua, Zhou Shijie, Ma Yong, Guo Yang. Wenshen Tongluo Zhitong decoction regulates the bone fat differentiation balance of bone marrow mesenchymal stem cells through exosomal miR-342-3p [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(13): 3258-3269. |
| [15] | Jiang Yidi, Zhao Jianwei, Zhou Jue, Lyu Jinpeng, Wang Datao, Li Xunsheng, Yue Zhigang, Cui Bo, Sun Hongmei. Regulation of antler stem cell exosomes miRNA-145 on inflammatory chondrocytes [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(13): 3288-3297. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||