Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (19): 5000-5006.doi: 10.12307/2026.144
Previous Articles Next Articles
Exosomal miRNA as an early diagnostic biomarker and potential therapeutic target for cerebral small vessel disease
Liu Yuxuan1, Guan Dongsheng2, Wang Jing1, Ren Yihan1
- 1Second Clinical Medical College of Henan University of Chinese Medicine, Zhengzhou 450046, Henan Province, China; 2Henan Province Hospital of TCM, Zhengzhou 450002, Henan Province, China
-
Received:2025-06-16Accepted:2025-08-05Online:2026-07-08Published:2026-02-24 -
Contact:Guan Dongsheng, Professor, Chief physician, Doctoral supervisor, Henan Province Hospital of TCM, Zhengzhou 450002, Henan Province, China -
About author:Liu Yuxuan, MS, Second Clinical Medical College of Henan University of Chinese Medicine, Zhengzhou 450046, Henan Province, China -
Supported by:National Natural Science Foundation of China, No. 81673943 (to GDS); National Training Project for Innovative Key Talents in Traditional Chinese Medicine, No. (2019)128 (to GDS); Henan Province Key R&D and Promotion Project (Science and Technology Research), No. 242102310564 (to GDS); Henan Province Key R&D and Promotion Project (Science and Technology Research), No. 232102310444 (to GDS); Henan Province Key Scientific Research Project of Higher Education Institutions, No. 23A360021 (to GDS)
CLC Number:
Cite this article
Liu Yuxuan, Guan Dongsheng, Wang Jing, Ren Yihan. Exosomal miRNA as an early diagnostic biomarker and potential therapeutic target for cerebral small vessel disease[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(19): 5000-5006.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
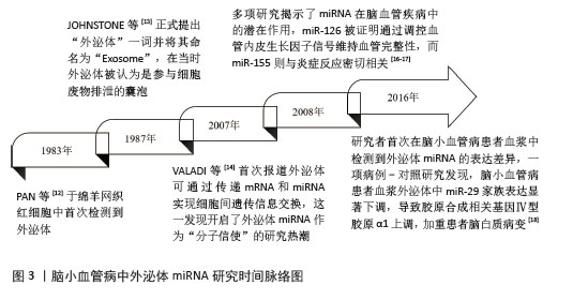
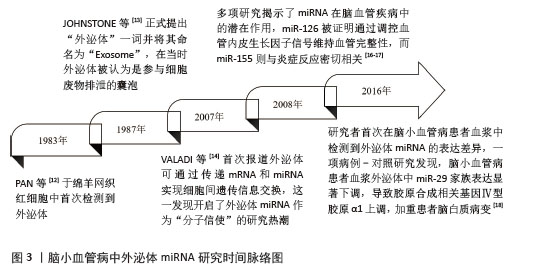
2.1 外泌体miRNA研究历史概要 外泌体的研究始于20世纪80年代,PAN等[12]在1983年的时候于绵羊网织红细胞中首次检测到外泌体。1987年,JOHNSTONE等[13]正式提出“外泌体”一词并将其命名为“Exosome”,在当时外泌体被认为是参与细胞废物排泄的囊泡。直至21世纪初,才明确外泌体作为细胞分泌的纳米级囊泡(直径30-150 nm)的特性,并发现其携带蛋白质、脂质和核酸(包括miRNA)的功能。2007年,VALADI等[14]首次报道外泌体可通过传递mRNA和miRNA实现细胞间遗传信息交换,这一发现开启了外泌体miRNA作为“分子信使”的研究热潮。与此同时,miRNA被证实通过结合靶基因mRNA的3’非翻译区调控基因表达,参与多种病理生理过程[15]。2008年,多项研究揭示了miRNA在脑血管疾病中的潜在作用,miR-126被证明通过调控血管内皮生长因子信号维持血管完整性,而miR-155则与炎症反应密切相关[16-17]。这些发现为后续探索外泌体miRNA在脑小血管病中的作用奠定了理论基础。2016年,研究者首次在脑小血管病患者血浆中检测到外泌体miRNA的表达差异,一项病例-对照研究发现,脑小血管病患者血浆外泌体中miR-29家族表达显著下调,导致胶原合成相关基因Ⅳ型胶原α1上调,加重了患者脑白质病变[18]。具体时间脉络见图3。 "
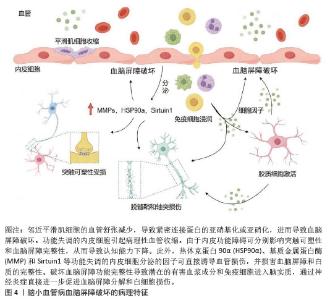
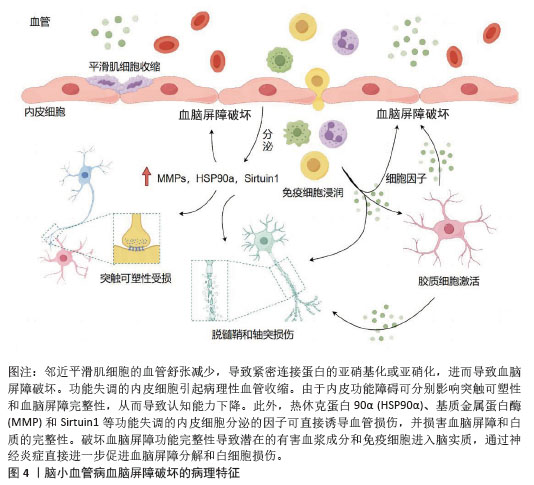
2.2 外泌体miRNA生物学功能及研究现状 外泌体miRNA是由来源于细胞核中的18-25个核苷酸组成的单链小非编码RNA分子,受体细胞中释放的成熟miRNA通过转录或切割靶向信使RNA特异性结合以调控多个靶基因表达,同时参与调节多种细胞和分子途径[9]。近几年随着转录组学研究和高通量测序技术迅猛发展,逐渐发现了外泌体miRNA的重要作用机制。与其他器官相比,脑组织中各种来源的外泌体miRNA类型更多、表达水平更高,主要调节脑内关键基因的表达,在细胞凋亡、神经分化、脑血管生成等过程中均发挥重要作用[19]。研究指出,外泌体miRNA可通过靶向调控多种信号通路参与疾病的病理生理过程从而影响表观症状,是预防和诊断阿尔茨海默病、帕金森病、血管性痴呆等多种脑血管疾病的重要生物标志物[20-21]。外泌体的脂质双分子层可保护内部miRNA免受核酸酶降解,使它在血液、脑脊液等体液中长期稳定存在,成为理想的液体活检标志物。进一步研究发现,在中枢神经系统内,外泌体miRNA参与调节血脑屏障功能稳态、内皮细胞退化、突触功能和炎症等诸多脑小血管病病理生理过程[22]。一个外泌体miRNA可以靶向多个mRNA,同时多个外泌体miRNA也可以协同调节同一个mRNA,表明外泌体miRNA具有的多通道和多靶点特征与脑小血管病多因素病理机制相吻合,提示外泌体miRNA可能通过参与脑小血管病的异质性发病机制进行治疗[23]。体内与体外研究发现,存在于唾液、尿液、血清、血液和脑脊液中的外泌体miRNA参与脑小血管病多种发病机制,是疾病发生发展过程中的重要相关成分,被认为是脑小血管病的潜在治疗靶点与生物学标志物[24]。 2.3 脑小血管病发病机制和诊疗方案的研究现状 脑小血管病是一种异质性疾病,其发病机制涉及多个方面,目前研究普遍认为可能与慢性脑灌注不足、血脑屏障受损、炎症反应、细胞凋亡等因素有关[25]。脑小血管病的病理学改变主要为脑组织和血管的病理改变,通常是由各种危险因素导致小动脉硬化、淀粉样变性、血管扩张能力下降、神经血管耦合衰减和血脑屏障渗漏等病理改变,使大脑深部白质和深灰质区域的穿支动脉出现血管密度降低、管壁增厚狭窄、自动调节能力下降、微血管病理性迂曲和毛细血管细线样改变等,脑组织呈现出慢性脑缺血状态,从而形成一系列脑组织神经影像学上可观察到的病理改变[26-27]。 目前,脑小血管病的临床诊断主要依靠神经影像学检查,如磁共振成像,普遍认可的典型特征包括皮质下梗死(即腔隙性梗死)、腔隙性白质高信号、脑微出血和脑萎缩[28]。脑小血管病的治疗手段主要是控制危险因素和对症治疗。通过对高血压、糖尿病、高脂血症和吸烟等危险因素的科学管理进行预防干预,应用降压药、抗血小板药、降脂药及改善认知障碍的药物如胆碱酯能抑制剂多奈哌齐等在一定程度上可以延缓症状进展[29]。当前针对脑小血管病治疗靶点的研究还不全面,仍需继续开展更多基于脑小血管病病理机制的特异性研究。非编码RNA中的miRNA作为一种新型生物标志物,可通过不同信号通路参与神经退行性疾病,miRNA是预测和诊断神经退行性疾病的主要生物标志物[20]。通过对外泌体miRNA的研究,有助于在基因水平了解脑小血管病的病理机制,为疾病的早期诊断及靶向治疗提供思路。 2.4 外泌体miRNA参与脑小血管病的病理机制 2.4.1 外泌体miRNA保护血脑屏障 血脑屏障主要由内皮细胞、基底膜、星形胶质细胞、周细胞紧密连接构成,密切调节离子、分子和细胞进出循环血液和脑脊液或脑实质的活动,维持中枢神经系统内稳态并保护大脑免受神经毒素、病原体和其他有害物质的侵害[30],见图4。血脑屏障功能障碍是脑小血管病发病机制中的关键环节[31]。研究表明,脑小血管病患者血浆外泌体中的miR-4685-3p负调控基质金属蛋白酶9的表达,增强细胞的活力和成管能力,改善内皮细胞损伤[32]。神经细胞来源外泌体miR-124-3p通过诱导长链非编码RNA-Malat1的表达激活了层粘连蛋白γ1/磷脂酰肌醇3激酶/蛋白激酶B信号通路,从而调节星形胶质细胞活性以改善血脑屏障的功能障碍[33]。血浆细胞外囊泡中miR-383-3p通过降低脑内皮细胞中紧密连接蛋白claudin 5的表达以可逆的方式显著增加血脑屏障通透性[34]。血浆外泌体中miR-148a-3p通过激活Smad2和抑制基质金属蛋白酶9信号通路改善血脑屏障损伤[35]。上调脑内皮细胞中外泌体miR-320a依赖性葡萄糖转运蛋白1的表达可以介导环磷酸腺苷/蛋白激酶A/环磷酸腺苷反应元件结合蛋白信号通路以改善血脑屏障功能障碍[36]。此外,周细胞外泌体miR-638通过抑制肿瘤坏死因子α诱导的周细胞迁移来保护血脑屏障的完整性[37]。上述研究表明,外泌体miRNA可以通过影响内皮细胞功能状态、调节紧密连接蛋白表达、上调星形胶质细胞活性和抑制周细胞迁移等方面实现对血脑屏障的保护,可能成为脑小血管病新的治疗靶点。 "

2.4.2 外泌体miRNA改善慢性脑灌注不足 在脑小血管病的发病过程中,慢性脑灌注不足被视为重要的发病机制,尤其是与年龄相关的脑小血管病。由多种因素引起的动脉硬化、管壁变厚和管腔狭窄闭塞这些脑小血管的结构损伤破坏了血管的自我调节能力,进而影响脑部的血液供应[38]。慢性脑灌注不足会导致神经元死亡,轴突、树突及突触损伤,从而导致认知和记忆障碍。研究表明,大脑皮质锥体细胞来源的外泌体miR-30a-5p可以通过靶向信息调节因子1和增加核因子红细胞相关因子2核转位来改善脑小血管病大鼠慢性脑灌注不足诱导的认知功能障碍[39]。海马神经元细胞外泌体miR-218a-5p通过靶向SH3和多个锚蛋白重复结构域2通路减轻低氧状态引起的突触损伤,改善脑小血管病大鼠慢性脑灌注不足诱导的认知功能缺陷和突触功能障碍[40]。内皮祖细胞来源外泌体miR-17-5p通过激活磷酸酶及张力蛋白同源物/磷脂酰肌醇3激酶/蛋白激酶B信号通路抑制脑内皮细胞损伤,显著增加脑微血管密度和脑血流量,从而减轻血管损伤,改善慢性脑灌注不足[41]。抑制海马神经细胞中外泌体miR-153通过上调双侧颈总动脉闭塞大鼠的突触蛋白1表达并延长CA3-CA1回路延迟反应增强期,提升了海马谷氨酸能囊泡运输功能,改善大鼠慢性脑灌注不足[42]。海马组织来源的外泌体miR-181c可能通过靶向上调神经丝光(NF-L)泛素化的三联基序2(TRIM2)的表达,增加了N-甲基-D-天冬氨酸受体1表达,导致海马神经元重塑并伴随着海马神经元树突分支和树突棘密度的增加,说明了外泌体miR-181c可能从慢性脑灌注不足的状态中拯救细胞活性,改善认知缺陷[43]。以上研究表明,外泌体miRNA可通过改善慢性脑灌注不足成为脑小血管病的潜在治疗靶点,见表1。 "


2.4.3 外泌体miRNA减轻炎症反应 随着对脑小血管病病理机制的深入研究,证实了炎症标志物和脑小血管病之间存在着紧密联系。基于此,可以将脑小血管病炎症反应分为全身性炎症与血管炎症,分别对应两种具有不同区域偏好的脑小血管病亚型[44-45]。炎症在诱导脑小血管病的过程中,会产生多种炎症递质、促炎因子以及募集免疫细胞,导致血脑屏障损伤和内皮细胞功能障碍,是脑小血管病的重要危险因素[45]。研究表明,脑小血管病患者外周血中外泌体miR-183的特异性表达可能通过调节炎症因子影响脑小血管病神经影像学病灶的改变[8]。海马神经元细胞来源的外泌体miR-138-5p靶向脂质运载蛋白2在海马中上调,抑制了海马中肿瘤坏死因子α、白细胞介素1β和白细胞介素6的分泌,减轻了炎症反应[46]。大鼠腹内侧前额叶皮质外泌体中miR-204-5p的上调抑制了Janus激酶2/信号转导和转录活化因子3信号通路,导致促炎细胞因子水平降低,从而减轻了神经炎症[47]。来源于间充质干细胞衍生外泌体的miR-330-5p通过靶向含SET域蛋白7以减少肿瘤坏死因子α、白细胞介素1β和白细胞介素6的表达,从而减轻炎症反应[48]。在动物实验中,通过激活Nespas/miR-383-3p/SHP2通路调节小胶质细胞来源外泌体miR-383-3p和核苷酸结合寡聚化结构域样受体蛋白1炎症小体表达,从而减轻神经炎症反应[49]。此外,miR-223、miR-146a、miR-124、miR-21等具有抗炎作用的巨噬细胞来源外泌体miRNA和miR-326、miR-155、miR-27b等具有促炎作用的T细胞来源外泌体miRNA,通过上调或下调免疫细胞和炎症因子蛋白的异常表达来调节神经炎症[50]。神经细胞外泌体miR-155-5p在炎症中已被广泛研究,其跨免疫细胞类型的有效促炎作用奠定了在促炎miRNA中的独特地位。外泌体miR-155-5p的主要抗炎RNA靶点包括肌醇磷酸酶Ship1、转录因子Cebpb、信号转导和转录活化因子1抑制因子Socs1和抗炎受体白细胞介素13受体α1,因此,通过敲除或抑制miR-155-5p或者与抗炎miRNA联合使用可以有效改善神经系统的炎症反应,凸显了多种miRNA联合作用的重要性[51-54]。 2.4.4 外泌体miRNA抑制细胞凋亡 细胞凋亡是脑小血管病病理生理过程中的关键事件,脑组织由于各种危险因素导致血管病变,从而出现脑血流量减少,影响细胞内信号转导网络,激活了Bax、Caspase-3等凋亡因子表达,从而导致神经元死亡,是脑小血管病认知功能障碍的主要原因。研究表明,长链非编码RNA MALAT1通过抑制肿瘤细胞外泌体miR-7641靶向血栓素受体,降低Bax和裂解的Caspase-3 (c-Caspase-3)蛋白表达水平,减少了微血管内皮细胞损伤和细胞凋亡,缓解了脑小血管病引起的神经功能障碍[55]。体外和体内实验表明,巨噬细胞来源外泌体miR-503-5p通过靶向调节抗凋亡因子泛素特异性蛋白酶10下调了内质网应激生物标志物葡萄糖调节蛋白78和C/EBP同源蛋白的表达,抑制了神经元凋亡[56]。血浆外泌体miR-124通过靶向抑制信号转导和转录活化因子3的表达,增加了抗凋亡Bcl-2的蛋白表达,降低了促凋亡Bax的蛋白表达,增强大鼠缺血半暗带组织的抗凋亡能力,从而减少细胞凋亡[57]。血清外泌体miR-128-3p通过靶向促进Smurf2/YY1轴,提高了神经生长因子表达,改善了大鼠脑组织缺血缺氧状态,抑制了细胞凋亡[58]。肿瘤细胞来源外泌体miR-143-5p和miR-145-5通过调节Caspase-3/8/9、 Bax和Bcl-2蛋白表达协同增强细胞活力,抑制细胞凋亡[59]。神经细胞外泌体miR-193b-3p通过靶向降低细胞分裂周期42编码的鸟嘌呤核苷酸交换因子的表达,抑制喙腹外侧延髓神经元凋亡,导致神经元兴奋性和交感神经流出降低,从而避免血压升高导致的血管损伤[60]。在认知缺陷的老年人群中,血液中外泌体has-miR-128-3p、has-miR-144-5p、has-miR-146a-5p、has-miR-26a-5p、hsa-miR-29c-3p和hsa-miR-363-3p表达水平下调,可能通过参与细胞凋亡影响认知能力,是老年人认知能力下降的早期血液生物标志物[61]。脑小血管病往往累及颅内小血管,影响循环系统功能,出现慢性脑缺血缺氧状态,神经细胞是脑组织结构与功能的重要组成,具有缺血易感性,长期脑血流不足会导致神经细胞功能受损,细胞凋亡则是其主要表现形式。以上研究表明,细胞凋亡与其他信号通路之间的相互作用会加速疾病进程,因此,基于外泌体miRNA多靶点治疗的可行性,构建外泌体miRNA作用网络对疾病治疗具有重要意义。 2.5 外泌体miRNA在脑小血管病诊断中的应用 随着逐渐步入老龄化社会以及脑血管病相关危险因素的增加,脑小血管病发病率逐年上升,患病率随着年龄增长呈指数增加,同时会提高中老年人未来痴呆、脑卒中、残疾和死亡的风险[62]。目前脑小血管病的临床诊断主要依靠磁共振成像进行辅助,但由于脑小血管病疾病进程缓慢,具有隐匿性,前期多为静息状态,临床极易被忽视,早期常常出现漏诊、误诊等现象,一旦出现临床症状病情往往已发展严重。脑小血管病患者由于血脑屏障的存在而使脑脊液中的标志物浓度较低,且鉴于获取具有侵入性和检验价格高等缺陷,导致临床应用受限。外泌体miRNA可以相对稳定的存在于人体内各种体液中,避免受到组织中酶的分解,在脑小血管病患者尿液、血清、血液等体液中的表达水平具有特异性与显著性,鉴于其特有的稳定性与易得性,成为脑小血管病早期预防和疾病诊治的潜在生物学标记物和新型靶点[63]。研究表明,血浆外泌体miR-409-3p、miR-502-3p、miR-486-5p、miR-451a和miR-223-3p可用作鉴定脑小血管病的具有高敏感性和特异性的生物标志物[64]。此外,脑小血管病患者血浆外泌体miR-155与脑小血管病的影像学病灶数量呈正相关性,有可能成为预测脑小血管病的新型血清生物标志物[65]。 2.6 外泌体miRNA在脑小血管病治疗中的应用 目前脑小血管病的临床治疗尚无特异性药物,临床多参考缺血性脑卒中和血管性痴呆的防治方案,仍缺少针对性防治手段[66]。鉴于外泌体miRNA独特的结构特点,可以靶向多个信号通路在病理机制的不同发展阶段进行干预,可以有效针对脑小血管病的不同病理环节,见表2。已有研究证实,抑制血浆外泌体miR-320可以增加脑小血管病小鼠血管内皮生长因子的表达,减少炎症、氧化应激反应和内皮抑素,从而增强小鼠神经元细胞的发育,改善脑小血管病小鼠的学习和记忆能力相关的认知功能障碍[67]。miR-320e作为miR-320家族中的一员,通过调节脑白质负荷影响脑小血管病总体负荷,参与脑小血管病白质病变的进展[68]。血液中外泌体miR-29通过靶向调节Ⅳ型胶原α1表达,影响了磁共振成像表征,说明了外泌体miR-29在脑小血管病患者中的基因特异性[18]。此外脑脊液外泌体miR-27a-3p和血液外泌体miR-222-3p降低了环磷酸腺苷降解酶磷酸二酯酶3(PDE3)蛋白的相对表达,血液来源外泌体miR-221/miR-222和miR-27a/miR-27b/miR-128可能通过炎症反应、调节免疫应答、突触功能和血管内皮损伤参与脑小血管病的病理机制,改善认知功能障碍[69-70]。目前主要手段为注射miRNA模拟物或拮抗剂调节下游靶基因的表达,但如何发挥其最有效的治疗效果仍需深入研究。 "

| [1] SMITH EE. Clinical presentations and epidemiology of vascular dementia. Clin Sci (Lond). 2017;131(11):1059-1068. [2] XU Y, CHEN B, YI J, et al. Evidence-based evaluation of adjuvant therapy with Chinese medicine for cerebral small vessel disease: A systematic review and meta-analysis. Medicine (Baltimore). 2023;102(52): e36221. [3] WANG Z, CHEN Q, CHEN J, et al. Risk factors of cerebral small vessel disease: A systematic review and meta-analysis. Medicine (Baltimore). 2021;100(51):e28229. [4] DUPRÉ N, DRIEU A, JOUTEL A. Pathophysiology of cerebral small vessel disease: a journey through recent discoveries. J Clin Invest. 2024; 134(10): e172841. [5] YANG Q, WEI X, DENG B, et al. Cerebral small vessel disease alters neurovascular unit regulation of microcirculation integrity involved in vascular cognitive impairment. Neurobiol Dis. 2022;170: 105750. [6] CAMARDA C, TORELLI P, PIPIA C, et al. Association Between Atrophy of the Caudate Nuclei, Global Brain Atrophy, Cerebral Small Vessel Disease and Mild Parkinsonian Signs in Neurologically and Cognitively Healthy Subjects Aged 45-84 Years: A Crosssectional Study. Curr Alzheimer Res. 2018;15(11):1013-1026. [7] CUADRADO-GODIA E, DWIVEDI P, SHARMA S, et al. Cerebral Small Vessel Disease: A Review Focusing on Pathophysiology, Biomarkers, and Machine Learning Strategies. J Stroke. 2018;20(3):302-320. [8] LIAO Z, SUN H, CHANG Y, et al. The expression and clinical significance of miRNA-183 in cerebral ischemia-reperfusion injury patients with cerebral small vessel disease. Ann Transl Med. 2020;8(16):1005. [9] DIENER C, KELLER A, MEESE E. Emerging concepts of miRNA therapeutics: from cells to clinic. Trends Genet. 2022;38(6):613-626. [10] ZHANG J, SUN P, ZHOU C, et al. Regulatory microRNAs and vascular cognitive impairment and dementia. CNS Neurosci Ther. 2020; 26(12):1207-1218. [11] JOO HS, JEON HY, HONG EB, et al. Exosomes for the diagnosis and treatment of dementia. Curr Opin Psychiatry. 2023;36(2):119-125. [12] PAN BT, JOHNSTONE RM. Fate of the transferrin receptor during maturation of sheep reticulocytes in vitro: selective externalization of the receptor. Cell. 1983; 33(3):967-978. [13] JOHNSTONE RM, ADAM M, HAMMOND JR, et al. Vesicle formation during reticulocyte maturation. Association of plasma membrane activities with released vesicles (exosomes). J Biol Chem. 1987;262(19): 9412-9420. [14] VALADI H, EKSTRÖM K, BOSSIOS A, et al. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat Cell Biol. 2007; 9(6):654-659. [15] RATAJCZAK J, MIEKUS K, KUCIA M, et al. Embryonic stem cell-derived microvesicles reprogram hematopoietic progenitors: evidence for horizontal transfer of mRNA and protein delivery. Leukemia. 2006;20(5): 847-856. [16] WANG S, AURORA AB, JOHNSON BA, et al. The endothelial-specific microRNA miR-126 governs vascular integrity and angiogenesis. Dev Cell. 2008;15(2):261-271. [17] SONKOLY E, STÅHLE M, PIVARCSI A. MicroRNAs and immunity: novel players in the regulation of normal immune function and inflammation. Semin Cancer Biol. 2008; 18(2):131-140. [18] VERDURA E, HERVÉ D, BERGAMETTI F, et al. Disruption of a miR-29 binding site leading to COL4A1 upregulation causes pontine autosomal dominant microangiopathy with leukoencephalopathy. Ann Neurol. 2016; 80(5):741-753. [19] HE T, YANG GY, ZHANG Z. Crosstalk of Astrocytes and Other Cells during Ischemic Stroke. Life (Basel). 2022;12(6):910. [20] MARANGON D, RAFFAELE S, FUMAGALLI M, et al. MicroRNAs change the games in central nervous system pharmacology. Biochem Pharmacol. 2019;168:162-172. [21] NAGARAJ S, LASKOWSKA-KASZUB K, DĘBSKI KJ, et al. Profile of 6 microRNA in blood plasma distinguish early stage Alzheimer’s disease patients from non-demented subjects. Oncotarget. 2017;8(10):16122-16143. [22] MICELI V, RUSSELLI G, IANNOLO G, et al. Role of non-coding RNAs in age-related vascular cognitive impairment: An overview on diagnostic/prognostic value in Vascular Dementia and Vascular Parkinsonism. Mech Ageing Dev. 2020;191:111332. [23] LIM LP, LAU NC, GARRETT-ENGELE P, et al. Microarray analysis shows that some microRNAs downregulate large numbers of target mRNAs. Nature. 2005;433(7027): 769-773. [24] LIAO L, HUANG W, MA R, et al. Potential biomarkers for cerebral small vessel disease with cognitive impairment: a systematic review and meta-analysis. Front Aging Neurosci. 2025;16:1475571. [25] 王香元,佘瑞宁,方锐,等.脑小血管病实验动物模型及制备方法研究进展[J].中草药,2022,53(18):5851-5860. [26] DEY AK, STAMENOVA V, TURNER G, et al. Pathoconnectomics of cognitive impairment in small vessel disease: A systematic review. Alzheimers Dement. 2016;12(7):831-845. [27] HOU H, ZHANG G, WANG H, et al. High matrix metalloproteinase-9 expression induces angiogenesis and basement membrane degradation in stroke-prone spontaneously hypertensive rats after cerebral infarction. Neural Regen Res. 2014;9(11):1154-1162. [28] WANG Y, LIU Z. Research progress on the correlation between MRI and impairment caused by cerebral small vessel disease: A review. Medicine (Baltimore). 2023;102(40):e35389. [29] CANNISTRARO RJ, BADI M, EIDELMAN BH, et al. CNS small vessel disease: A clinical review. Neurology. 2019; 92(24):1146-1156. [30] SHARIF Y, JUMAH F, COPLAN L, et al. Blood brain barrier: A review of its anatomy and physiology in health and disease. Clin Anat. 2018;31(6):812-823. [31] CHEN JXY, VIPIN A, SANDHU GK, et al. Blood-brain barrier integrity disruption is associated with both chronic vascular risk factors and white matter hyperintensities. J Prev Alzheimers Dis. 2025;12(2):100029. [32] MA D, LU Y, YE H, et al. miR-4685-3p Alleviates Human Brain Microvascular Endothelial Cells Injury by Regulating MMP9. Ann Clin Lab Sci. 2024;54(2):179-189. [33] ZHU Y, ZOU W, SUN B, et al. Ginsenoside Rg1 Regulates the Activation of Astrocytes Through lncRNA-Malat1/miR-124-3p/Lamc1 Axis Driving PI3K/AKT Signaling Pathway, Promoting the Repair of Spinal Cord Injury. CNS Neurosci Ther. 2024;30(11):e70103. [34] TOMATIS F, ROSA S, SIMÕES S, et al. Engineering extracellular vesicles to transiently permeabilize the blood-brain barrier. J Nanobiotechnology. 2024; 22(1):747. [35] YANG Z, HUANG C, HUANG W, et al. Exacerbated ischemic brain damage in type 2 diabetes via methylglyoxal-mediated miR-148a-3p decline. BMC Med. 2024;22(1):557. [36] WEI JY, LIU H, LI Y, et al. Melatonin Protects Against Cocaine-Induced Blood-Brain Barrier Dysfunction and Cognitive Impairment by Regulating miR-320a-Dependent GLUT1 Expression. J Pineal Res. 2024;76(8):e70002. [37] KURMANN L, AZZARITO G, LEENERS B, et al. 17β-Estradiol Abrogates TNF-α-Induced Human Brain Vascular Pericyte Migration by Downregulating miR-638 via ER-β. Int J Mol Sci. 2024;25(21):11416. [38] 胡文立,杨磊,李譞婷,等.中国脑小血管病诊治专家共识2021[J].中国卒中杂志, 2021,16(7):716-726. [39] WANG L, LI M, LIU B, et al. miR-30a-5p mediates ferroptosis of hippocampal neurons in chronic cerebral hypoperfusion-induced cognitive dysfunction by modulating the SIRT1/NRF2 pathway. Brain Res Bull. 2024; 212:110953. [40] LI N, REN C, LI S, et al. Remote ischemic conditioning alleviates chronic cerebral hypoperfusion-induced cognitive decline and synaptic dysfunction via the miR-218a-5p/SHANK2 pathway. Prog Neurobiol. 2023; 230:102514. [41] PAN Q, WANG Y, LIU J, et al. MiR-17-5p Mediates the Effects of ACE2-Enriched Endothelial Progenitor Cell-Derived Exosomes on Ameliorating Cerebral Ischemic Injury in Aged Mice. Mol Neurobiol. 2023; 60(6):3534-3552. [42] ZHANG S, YAN ML, YANG L, et al. MicroRNA-153 impairs hippocampal synaptic vesicle trafficking via downregulation of synapsin I in rats following chronic cerebral hypoperfusion. Exp Neurol. 2020;332:113389. [43] FANG C, LI Q, MIN G, et al. MicroRNA-181c Ameliorates Cognitive Impairment Induced by Chronic Cerebral Hypoperfusion in Rats. Mol Neurobiol. 2017;54(10):8370-8385. [44] LOW A, MAK E, ROWE JB, et al. Inflammation and cerebral small vessel disease: A systematic review. Ageing Res Rev. 2019;53: 100916. [45] LI T, HUANG Y, CAI W, et al. Age-related cerebral small vessel disease and inflammaging. Cell Death Dis. 2020; 11(10):932. [46] LOU L, YU W, CHENG Y, et al. Quercetin can improve anesthesia induced neuroinflammation and cognitive dysfunction by regulating miR-138-5p/ LCN2. BMC Anesthesiol. 2025;25(1):21. [47] CHEN X, GAN Y, ZHANG K, et al. MicroRNA-204-5p Deficiency within the vmPFC Region Contributes to Neuroinflammation and Behavioral Disorders via the JAK2/STAT3 Signaling Pathway in Rats. Adv Sci (Weinh). 2025;12(10):e2403080. [48] LIU W, SHEN Y, PAN R, et al. mir-330-5p from mesenchymal stem cell-derived exosomes targets SETD7 to reduce inflammation in rats with cerebral ischemia-reperfusion injury. J Mol Histol. 2024;56(1):63. [49] HONG Z, ZUO Z, ZHAO Y, et al. Transcranial focused ultrasound stimulation alleviates NLRP3-related neuroinflammation induced by ischemic stroke via regulation of the Nespas/miR-383-3p/SHP2 pathway. Int Immunopharmacol. 2025;144:113680. [50] GAUDET AD, FONKEN LK, WATKINS LR, et al. MicroRNAs: Roles in Regulating Neuroinflammation. Neuroscientist. 2018; 24(3):221-245. [51] CARDOSO AL, GUEDES JR, PEREIRA DE ALMEIDA L,et al. miR-155 modulates microglia-mediated immune response by down-regulating SOCS-1 and promoting cytokine and nitric oxide production. Immunology. 2012;135(1):73-88. [52] MARTINEZ-NUNEZ RT, LOUAFI F, SANCHEZ-ELSNER T. The interleukin 13 (IL-13) pathway in human macrophages is modulated by microRNA-155 via direct targeting of interleukin 13 receptor alpha1 (IL13Ralpha1). J Biol Chem. 2011;286(3):1786-1794. [53] O’CONNELL RM, CHAUDHURI AA, RAO DS, et al. Inositol phosphatase SHIP1 is a primary target of miR-155. Proc Natl Acad Sci U S A. 2009;106(17):7113-7118. [54] WORM J, STENVANG J, PETRI A, et al. Silencing of microRNA-155 in mice during acute inflammatory response leads to derepression of c/ebp Beta and down-regulation of G-CSF. Nucleic Acids Res. 2009;37(17):5784-5792. [55] CHE F, HAN Y, FU J, et al. LncRNA MALAT1 induced by hyperglycemia promotes microvascular endothelial cell apoptosis through activation of the miR-7641/TPR axis to exacerbate neurologic damage caused by cerebral small vessel disease. Ann Transl Med. 2021;9(24):1762. [56] PENG Q, WANG S, HUANG S, et al. FTO/miR-503-5p/USP10 axis regulates neuronal endoplasmic reticulum stress-mediated apoptosis in ischemic stroke. Int Immunopharmacol. 2025;149:114150. [57] SONG W, TENG L, WANG H, et al. Exercise preconditioning increases circulating exosome miR-124 expression and alleviates apoptosis in rats with cerebral ischemia-reperfusion injury. Brain Res. 2025;1851:149457. [58] NIE X, CHENG R, HAO P, et al. MicroRNA-128-3p Affects Neuronal Apoptosis and Neurobehavior in Cerebral Palsy Rats by Targeting E3 Ubiquitin-Linking Enzyme Smurf2 and Regulating YY1 Expression. Mol Neurobiol. 2025;62(2):2277-2291. [59] JODEIRY ZAER S, AGHAMAALI M, AMINI M, et al. Cooperatively inhibition effect of miR-143-5p and miR-145-5p in tumorigenesis of glioblastoma cells through modulating AKT signaling pathway. Bioimpacts. 2024; 14(3):29913. [60] ZHANG S, WANG X, DAI T, et al. miR-193b-3p and miR-346 Exert Antihypertensive Effects in the Rostral Ventrolateral Medulla. J Am Heart Assoc. 2024;13(13):e034965. [61] SADLON A, TAKOUSIS P, EVANGELOU E, et al. Association of Blood MicroRNA Expression and Polymorphisms with Cognitive and Biomarker Changes in Older Adults. J Prev Alzheimers Dis. 2024; 11(1):230-240. [62] BORDES C, SARGURUPREMRAJ M, MISHRA A, et al. Genetics of common cerebral small vessel disease. Nat Rev Neurol. 2022;18(2): 84-101. [63] ALMUTAIRI MM, GONG C, XU YG, et al. Factors controlling permeability of the blood-brain barrier. Cell Mol Life Sci. 2016; 73(1):57-77. [64] PRABHAKAR P, CHANDRA SR, CHRISTOPHER R. Circulating microRNAs as potential biomarkers for the identification of vascular dementia due to cerebral small vessel disease. Age Ageing. 2017; 46(5):861-864. [65] GUO Y, LI D, LI J, et al. Expression and Significance of MicroRNA155 in Serum of Patients with Cerebral Small Vessel Disease. J Korean Neurosurg Soc. 2020; 63(4):463-469. [66] 彭丹涛,邵文.脑小血管病相关认知功能障碍中国诊疗指南(2019)[J].阿尔茨海默病及相关病,2019,2(3):405-407+403. [67] YU K, XU R, WU L, et al. Effects of microRNA-320 on learning and memory in mice with vascular cognitive impairment caused via cerebral ischemia. Cell Mol Biol (Noisy-le-grand). 2023;69(13):112-119. [68] GAO KJ, YIN RH, WANG Y, et al. Exosomal miR-320e as a Novel Potential Biomarker for Cerebral Small Vessel Disease. Int J Gen Med. 2023;16:641-655. [69] ZHAO W, SUN W, LI S, et al. Exosomal miRNA-223-3p as potential biomarkers in patients with cerebral small vessel disease cognitive impairment. Ann Transl Med. 2021;9(24):1781. [70] YASMEEN S, KAUR S, MIRZA AH, et al. miRNA-27a-3p and miRNA-222-3p as Novel Modulators of Phosphodiesterase 3a (PDE3A) in Cerebral Microvascular Endothelial Cells. Mol Neurobiol. 2019; 56(8):5304-5314. [71] LU Y, SHEN R, ZHU H, et al. Tandem Mass Tag (TMT) Quantitative Proteomic Analysis of Serum Exosomes in Cerebral Small-vessel Disease (CSVD) Patients With Depressive Symptoms. Curr Neurovasc Res. 2022;19(5):449-461. [72] FENG Q, LU Y, ZHANG R, et al. Identification of differentially expressed exosome proteins in serum as potential biomarkers for cognitive impairments in cerebral small vessel disease. Neurosci Lett. 2024; 822:137631. |
| [1] | Han Teng, Ma Hong, Yang Ruoyi, Luo Yi, Li Chao. Oral squamous cell carcinoma-derived exosomal delivery of angiopoietin-2 is involved in tumor angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1755-1767. |
| [2] | Liu Anting, Lu Jiangtao, Zhang Wenjie, He Ling, Tang Zongsheng, Chen Xiaoling. Regulation of AMP-activated protein kinase by platelet lysate inhibits cadmium-induced neuronal apoptosis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1800-1807. |
| [3] | Cao Yong, Teng Hongliang, Tai Pengfei, Li Junda, Zhu Tengqi, Li Zhaojin. Interactions between cytokines and satellite cells in muscle regeneration [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1808-1817. |
| [4] | Huang Jiawen, Pan Zhiyi, Xue Wenjun, Lian Yuanpei, Xu Jianda. Plant-derived vesicles and malignant tumor therapy: cross-species communication and modulation of host cell responses [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1828-1838. |
| [5] | Wang Baiyan, Yang Shu, Wang Yiming, Wu Mengqing, Xiao Yu, Guo Zixuan, Zhang Boyi, Feng Shuying. Exosome-delivered CRISPR/Cas system enables gene editing in target cells [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1839-1849. |
| [6] | Wang Zhenze, Liu Fende, Zhang Rui, Li Wujun. Mesenchymal stem cells in treatment of arteriosclerosis obliterans of lower extremities: systematic review and meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1869-1876. |
| [7] | Song Puzhen, Ma Hebin, Chen Hongguang, Zhang Yadong. Effect of bone marrow mesenchymal stem cell-derived exosomes combined with transforming growth factor beta 1 on macrophages [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1616-1623. |
| [8] | Cai Ziming, Yu Qinghe, Ma Pengfei, Zhang Xin, Zhou Longqian, Zhang Chongyang, Lin Wenping. Heme oxygenase-1 alleviates lipopolysaccharide-induced inflammatory response in nucleus pulposus mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1624-1631. |
| [9] | Yuan Xiaoshuang, Yang Xu, Yang Bo, Chen Xiaoxu, Tian Ting, Wang Feiqing, Li Yanju, Liu Yang, Yang Wenxiu. Effect of conditioned medium of diffuse large B-cell lymphoma cells on proliferation and apoptosis of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1632-1640. |
| [10] | He Jiale, Huang Xi, Dong Hongfei, Chen Lang, Zhong Fangyu, Li Xianhui. Acellular dermal matrix combined with adipose-derived stem cell exosomes promotes burn wound healing [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1699-1710. |
| [11] | Xia Linfeng, Wang Lu, Long Qianfa, Tang Rongwu, Luo Haodong, Tang Yi, Zhong Jun, Liu Yang. Human umbilical cord mesenchymal stem cell-derived exosomes alleviate blood-brain barrier damage in mice with septic encephalopathy [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1711-1719. |
| [12] | Cui Lianxu, Li Haomin, Xu Junrong, Tan Baodong, Lu Dahong, Peng Siwei, Wang Jinhui. Effect of umbilical cord mesenchymal stem cell conditioned medium on tissue repair after traumatic craniocerebral injury in miniature pigs [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1730-1735. |
| [13] | Chen Yulin, He Yingying, Hu Kai, Chen Zhifan, Nie Sha Meng Yanhui, Li Runzhen, Zhang Xiaoduo , Li Yuxi, Tang Yaoping. Effect and mechanism of exosome-like vesicles derived from Trichosanthes kirilowii Maxim. in preventing and treating atherosclerosis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1768-1781. |
| [14] | Jia Jinwen, Airefate·Ainiwaer, Zhang Juan. Effects of EP300 on autophagy and apoptosis related to allergic rhinitis in rats [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1439-1449. |
| [15] | Hou Chaowen, Li Zhaojin, Kong Jianda, Zhang Shuli. Main physiological changes in skeletal muscle aging and the multimechanism regulatory role of exercise [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1464-1475. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||