Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (34): 5523-5529.doi: 10.12307/2023.739
Previous Articles Next Articles
Biomechanical analysis of unilateral biportal endoscopic lumbar interbody fusion
Li Jiarui1, Yan Yang2, Wu Xiaogang2, Feng Haoyu1, He Liming1
- 1The Third Hospital of Shanxi Medical University (Shanxi Bethune Hospital, Shanxi Academy of Medical Sciences, Tongji Shanxi Hospital), Taiyuan 030032, Shanxi Province, China; 2College of Biomedical Engineering, Taiyuan University of Technology, Taiyuan 030024, Shanxi Province, China
-
Received:2022-09-27Accepted:2022-11-14Online:2023-12-08Published:2023-04-22 -
Contact:Feng Haoyu, Chief physician, The Third Hospital of Shanxi Medical University (Shanxi Bethune Hospital, Shanxi Academy of Medical Sciences, Tongji Shanxi Hospital), Taiyuan 030032, Shanxi Province, China He Liming, Associate chief physician, The Third Hospital of Shanxi Medical University (Shanxi Bethune Hospital, Shanxi Academy of Medical Sciences, Tongji Shanxi Hospital), Taiyuan 030032, Shanxi Province, China -
About author:Li Jiarui, Physician, The Third Hospital of Shanxi Medical University (Shanxi Bethune Hospital, Shanxi Academy of Medical Sciences, Tongji Shanxi Hospital), Taiyuan 030032, Shanxi Province, China -
Supported by:the Health Commission of Shanxi Province, Shanxi Medical Science and Technology Innovation Team Construction Plan, Science and Technology Training Team for Diagnosis and Treatment of Senile Spinal Degeneration and Injury Diseases, No. 2020TD13 (to FHY)
CLC Number:
Cite this article
Li Jiarui, Yan Yang, Wu Xiaogang, Feng Haoyu, He Liming. Biomechanical analysis of unilateral biportal endoscopic lumbar interbody fusion[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(34): 5523-5529.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
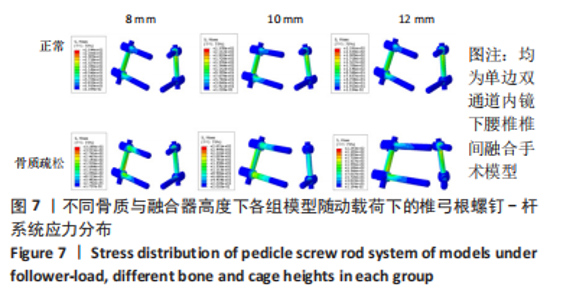
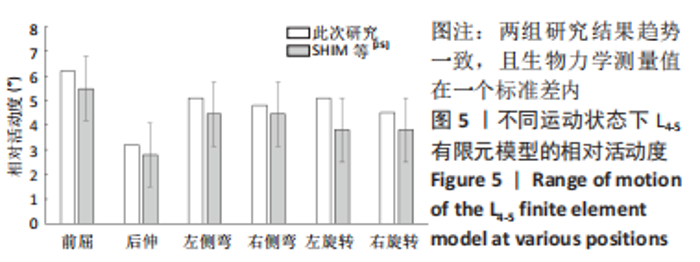
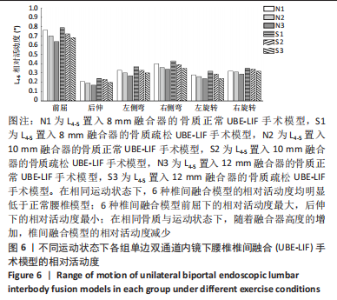
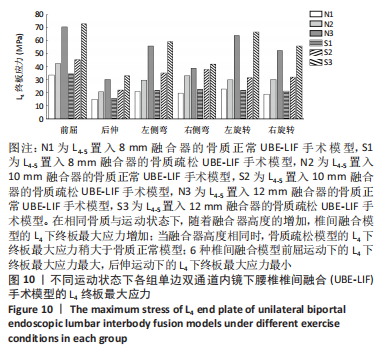
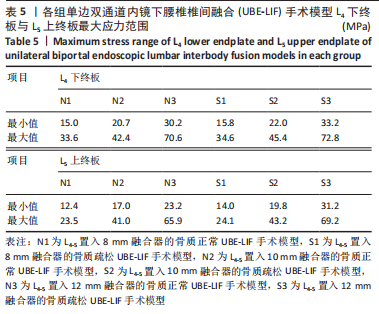
N0模型在前屈、后伸、左侧弯、右侧弯、左旋转、右旋转运动下的相对活动度分别为6.2°,3.2°,5.1°,5°,5°,5.1°;S0模型在前屈、后伸、左侧弯、右侧弯、左旋转、右旋转运动下的相对活动度分别为6.6°,3.5°,5.4°,5.2°,5.3°,5.3°,均远大于UBE-LIF手术模型的相对活动度。 6种UBE-LIF手术模型的相对活动度最大值均出现于前屈下,最小值出现于后伸运动下,且6种UBE-LIF模型右侧弯运动下的相对活动度大于左侧弯,右旋转运动下的相对活动度大于左旋转。在骨质正常模型中,相比N0模型,3种UBE-LIF手术模型L4-5节段的相对活动度均明显减少,N3模型相对活动度最小,其中,N2模型的相对活动度比N3模型增加7%,N1模型的相对活动度比N2模型增加10%。在骨质疏松模型中,S3模型的相对活动度最小,S2模型的相对活动度比S3模型增加10%,S1模型的相对活动度比S2模型增加7%。当融合器高度相同时,骨质疏松模型的相对活动度均稍大于骨质正常模型(约5.4%)。 2.2.2 椎弓根钉-杆系统的最大应力 见图7,8。"
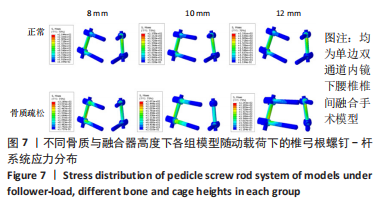
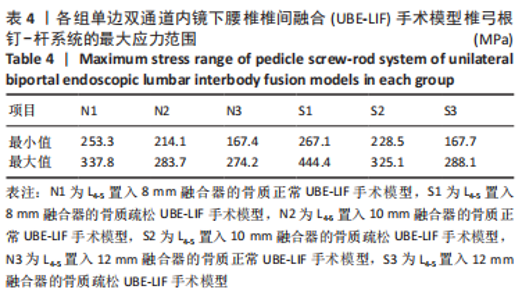
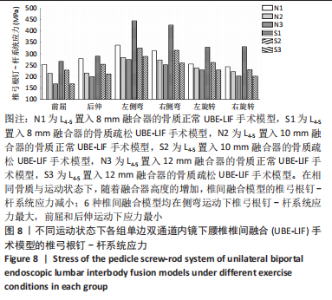
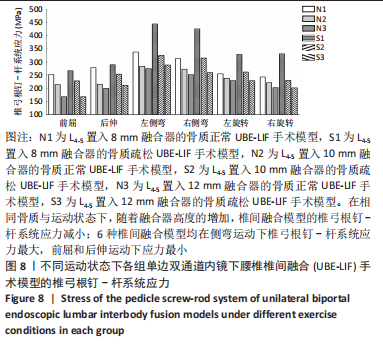
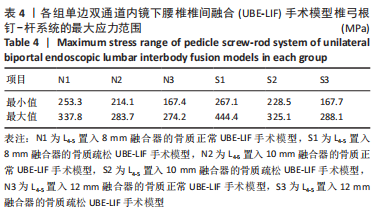
在骨质正常模型中,N3模型的椎弓根钉-杆系统最大应力值最小,N1模型的椎弓根钉-杆系统最大应力比N2大17%,N2模型的椎弓根钉-杆系统最大应力比N3模型大9%,N1模型的椎弓根钉-杆系统最大应力比N3模型大27%;在骨质疏松模型中,S3模型的的椎弓根钉-杆系统最大应力值最小,S1模型的椎弓根钉-杆系统最大应力比S2模型大19%,S2模型的椎弓根钉-杆系统最大应力比S3模型大29%,S1模型的椎弓根钉-杆系统最大应力比S3模型大53%,见表4。对于椎弓根钉-杆系统的最大应力,6种UBE-LIF模型均在模拟侧弯运动时应力达到最大,在前屈和后伸运动时最小,且应力值还表现为左侧弯运动大于右侧弯运动、左旋转运动大于右旋转运动、后伸运动大于前屈运动。当融合器的高度相同时,骨质疏松模型的椎弓根钉-杆系统最大应力均大于骨质正常模型(约13%)。"

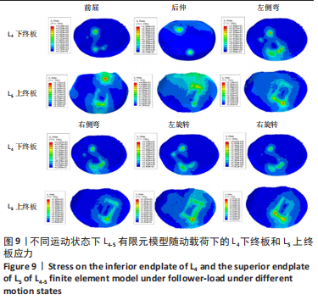
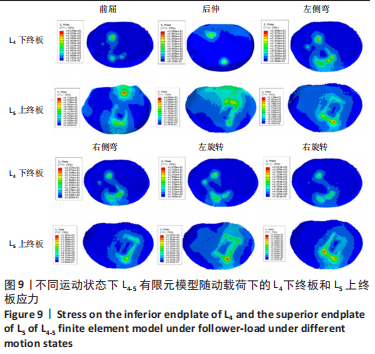
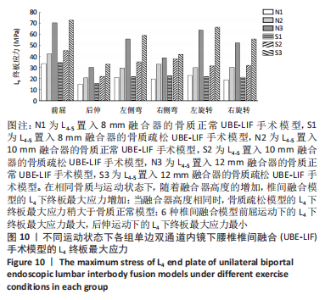
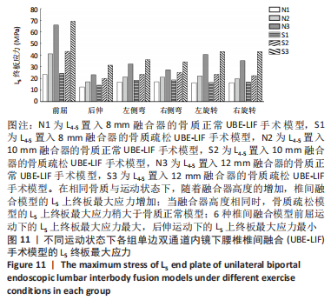
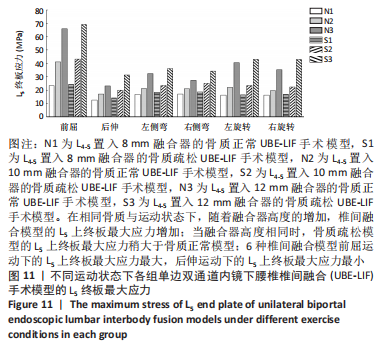
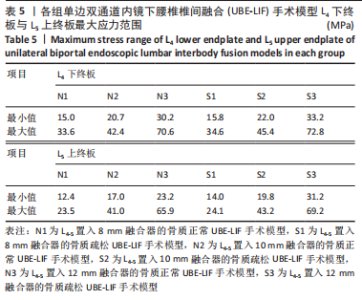
在骨质疏松模型中,S1模型的L4下终板最大应力最小,S2模型的L4下终板最大应力比S1模型增大47%,S3模型的L4下终板最大应力比S2模型增大61%,S3模型的L4下终板最大应力比S1模型增大139%;S1模型的L5上终板最大应力最小,S2模型的L5上终板最大应力比S1模型增大45%,S3模型的L5上终板最大应力比S2模型增大63%,S3模型的L5上终板最大应力比S1模型增大138%,见表5。 当融合器高度相同时,骨质疏松模型的L4下终板与L5上终板最大应力稍大于骨质正常模型(约7.5%)。在6种运动模式下,6种UBE-LIF模型的L4下终板应力均大于L5上终板(约24.1%),且在前屈下终板的最大应力最大,后伸运动时终板的最大应力最小。"

| [1] YASUHARA T, SASADA S, DATE I. [Lumbar Degenerative Disease:Key for Diagnosis]. No Shinkei Geka. 2021;49(6):1233-1245. [2] OPPENHEIMER JH, DECASTRO I, MCDONNELL DE. Minimally invasive spine technology and minimally invasive spine surgery: a historical review. Neurosurg Focus. 2009;27(3):E9. [3] MOBBS RJ, LI J, SIVABALAN P. Outcomes after decompressive laminectomy for lumbar spinal stenosis: comparison between minimally invasive unilateral laminectomy for bilateral decompression and open laminectomy: clinical article. J Neurosurg Spine. 2014;21(2):179-186. [4] HEO DH, SON SK, EUM JH. Fully endoscopic lumbar interbody fusion using a percutaneous unilateral biportal endoscopic technique: technical note and preliminary clinical results. Neurosurg Focus. 2017;43(2):E8. [5] YANG J, LIU C, HAI Y. Percutaneous Endoscopic Transforaminal Lumbar Interbody Fusion for the Treatment of Lumbar Spinal Stenosis: Preliminary Report of Seven Cases with 12-Month Follow-Up. Biomed Res Int. 2019;2019:3091459. [6] DU JY, KIELY PD, AL MAAIEH M. Lateral lumbar interbody fusion with unilateral pedicle screw fixation for the treatment of adjacent segment disease: a preliminary report. J Spine Surg. 2017;3(3):330-337. [7] YAO YC, CHOU PH, LIN HH. Risk Factors of Cage Subsidence in Patients Received Minimally Invasive Transforaminal Lumbar Interbody Fusion. Spine (Phila Pa 1976). 2020;45(19):E1279-e1285. [8] SINGHATANADGIGE W, SUKTHUAYAT A, TANAVIRIYACHAI T. Risk factors for polyetheretherketone cage subsidence following minimally invasive transforaminal lumbar interbody fusion. Acta Neurochir (Wien). 2021;163(9):2557-2565. [9] TAN JH, CHEONG CK, HEY HWD. Titanium (Ti) cages may be superior to polyetheretherketone (PEEK) cages in lumbar interbody fusion: a systematic review and meta-analysis of clinical and radiological outcomes of spinal interbody fusions using Ti versus PEEK cages. Eur Spine J. 2021;30(5):1285-1295. [10] IGARASHI H, HOSHINO M, OMORI K. Factors Influencing Interbody Cage Subsidence Following Anterior Cervical Discectomy and Fusion. Clin Spine Surg. 2019;32(7): 297-302. [11] CHOI WS, KIM JS, HUR JW. Minimally Invasive Transforaminal Lumbar Interbody Fusion Using Banana-Shaped and Straight Cages: Radiological and Clinical Results from a Prospective Randomized Clinical Trial. Neurosurgery. 2018;82(3):289-298. [12] JONES C, OKANO I, SALZMANN SN. Endplate volumetric bone mineral density is a predictor for cage subsidence following lateral lumbar interbody fusion: a risk factor analysis. Spine J. 2021;21(10):1729-1737. [13] HIYAMA A, SAKAI D, KATOH H. Comparative Study of Cage Subsidence in Single-Level Lateral Lumbar Interbody Fusion. J Clin Med. 2022;11(5):1374. [14] DERMAN PB, OHNMEISS DD, LAUDERBACK A, et al. Indirect Decompression for the Treatment of Degenerative Lumbar Stenosis. Int J Spine Surg. 2021;15(6):1066-1071. [15] OH KW, LEE JH, LEE JH. The Correlation Between Cage Subsidence, Bone Mineral Density, and Clinical Results in Posterior Lumbar Interbody Fusion. Clin Spine Surg. 2017;30(6):E683-e689. [16] FAN W, GUO LX, ZHAO D. Posterior Lumbar Interbody Fusion Versus Transforaminal Lumbar Interbody Fusion: Finite Element Analysis of the Vibration Characteristics of Fused Lumbar Spine. World Neurosurg. 2021;150:e81-e88. [17] LU T, REN J, SUN Z. Relationship between the elastic modulus of the cage material and the biomechanical properties of transforaminal lumbar interbody fusion: A logarithmic regression analysis based on parametric finite element simulations. Comput Methods Programs Biomed. 2022;214:106570. [18] HEINZ VON DER HOEH N, VILLA T, GALBUSERA F. Analysis of a Unilateral Bridging Cage for Lumbar Interbody Fusion: 2-Year Clinical Results and Fusion Rate with a Focus on Subsidence. World Neurosurg. 2018;116:e308-e314. [19] LIU X, MA J, PARK P. Biomechanical comparison of multilevel lateral interbody fusion with and without supplementary instrumentation: a three-dimensional finite element study. BMC Musculoskelet Disord. 2017;18(1):63. [20] YU Y, ZHOU Q, XIE YZ. Effect of Percutaneous Endoscopic Lumbar Foraminoplasty of Different Facet Joint Portions on Lumbar Biomechanics: A Finite Element Analysis. Orthop Surg. 2020;12(4):1277-1284. [21] LI J, XU W, ZHANG X. Biomechanical role of osteoporosis affects the incidence of adjacent segment disease after percutaneous transforaminal endoscopic discectomy. J Orthop Surg Res. 2019;14(1):131. [22] HEO DH, HONG YH, LEE DC. Technique of Biportal Endoscopic Transforaminal Lumbar Interbody Fusion. Neurospine. 2020;17(Suppl 1):S129-S137. [23] KIM JE, CHOI DJ. Unilateral biportal endoscopic decompression by 30° endoscopy in lumbar spinal stenosis: Technical note and preliminary report. J Orthop. 2018; 15(2):366-371. [24] VADAPALLI S, SAIRYO K, GOEL VK. Biomechanical rationale for using polyetheretherketone (PEEK) spacers for lumbar interbody fusion-A finite element study. Spine (Phila Pa 1976). 2006;31(26):E992-998. [25] SHIM CS, PARK SW, LEE SH. Biomechanical evaluation of an interspinous stabilizing device, Locker. Spine (Phila Pa 1976). 2008;33(22):E820-827. [26] HEO DH, LEE N, PARK CW. Endoscopic Unilateral Laminotomy with Bilateral Discectomy Using Biportal Endoscopic Approach: Technical Report and Preliminary Clinical Results. World Neurosurg. 2020;137:31-37. [27] PARK MK, PARK SA, SON SK. Clinical and radiological outcomes of unilateral biportal endoscopic lumbar interbody fusion (ULIF) compared with conventional posterior lumbar interbody fusion (PLIF): 1-year follow-up. Neurosurg Rev. 2019;42(3):753-761. [28] KIM JE, CHOI DJ. Biportal Endoscopic Transforaminal Lumbar Interbody Fusion with Arthroscopy. Clin Orthop Surg. 2018;10(2):248-252. [29] HEO DH, SHARMA S, PARK CK. Endoscopic Treatment of Extraforaminal Entrapment of L5 Nerve Root (Far Out Syndrome) by Unilateral Biportal Endoscopic Approach: Technical Report and Preliminary Clinical Results. Neurosurg. 2019;16(1):130-137. [30] KIM SK, KANG SS, HONG YH. Clinical comparison of unilateral biportal endoscopic technique versus open microdiscectomy for single-level lumbar discectomy: a multicenter, retrospective analysis. J Orthop Surg Res. 2018;13(1):22. [31] BERMEL EA, BAROCAS VH, ELLINGSON AM. The role of the facet capsular ligament in providing spinal stability. Comput Methods Biomech Biomed Engin. 2018;21(13): 712-721. [32] CLAESON AA, BAROCAS VH. Computer simulation of lumbar flexion shows shear of the facet capsular ligament. Spine J. 2017;17(1):109-119. [33] SEBAALY A, GROBOST P, MALLAM L. Description of the sagittal alignment of the degenerative human spine. Eur Spine J. 2018;27(2):489-496. [34] LÖFFLER MT, SOLLMANN N, MEI K. X-ray-based quantitative osteoporosis imaging at the spine. Osteoporos Int. 2020;31(2):233-250. [35] CHEN HC, WU JL, HUANG SC. Biomechanical evaluation of a novel pedicle screw-based interspinous spacer: A finite element analysis. Med Eng Phys. 2017;46:27-32. [36] BAE JS, LEE SH, KIM JS. Adjacent segment degeneration after lumbar interbody fusion with percutaneous pedicle screw fixation for adult low-grade isthmic spondylolisthesis: minimum 3 years of follow-up. Neurosurg. 2010;67(6):1600-1607. [37] YENI YN, HOU FJ, VASHISHTH D. Trabecular shear stress in human vertebral cancellous bone: intra- and inter-individual variations. J Biomech. 2001;34(10):1341-1346. [38] HANSEN U, ZIOUPOS P, SIMPSON R. The effect of strain rate on the mechanical properties of human cortical bone. J Biomech Eng. 2008;130(1):011011. [39] WU Y, LOAIZA J, BANERJI R, et al. Structure-function relationships of the human vertebral endplate. JOR spine. 2021;4(3):e1170. [40] FAN W, GUO LX. Biomechanical comparison of the effects of anterior, posterior and transforaminal lumbar interbody fusion on vibration characteristics of the human lumbar spine. Comput Methods Biomech Biomed Engin. 2019;22(5):490-498. [41] WESTBURY LD, SHERE C, EDWARDS MH. Cluster Analysis of Finite Element Analysis and Bone Microarchitectural Parameters Identifies Phenotypes with High Fracture Risk. Calcif Tissue Int. 2019;105(3):252-262. [42] POLIKEIT A, NOLTE LP, FERGUSON SJ. The effect of cement augmentation on the load transfer in an osteoporotic functional spinal unit: finite-element analysis. Spine (Phila Pa 1976). 2003;28(10):991-996. [43] FORMBY PM, KANG DG, HELGESON MD. Clinical and Radiographic Outcomes of Transforaminal Lumbar Interbody Fusion in Patients with Osteoporosis. Global Spine J. 2016;6(7):660-664. [44] KALIYA-PERUMAL AK, SOH TLT, TAN M. Factors Influencing Early Disc Height Loss Following Lateral Lumbar Interbody Fusion. Asian Spine J. 2020;14(5):601-607. [45] RASTEGAR S, ARNOUX PJ, WANG X. Biomechanical analysis of segmental lumbar lordosis and risk of cage subsidence with different cage heights and alternative placements in transforaminal lumbar interbody fusion. Comput Methods Biomech Biomed Engin. 2020;23(9):456-466. [46] CAPPELLETTO B, GIORGIUTTI F, BALSANO M. Evaluation of the effectiveness of expandable cages for reconstruction of the anterior column of the spine. J Orthop Surg (Hong Kong). 2020;28(1):2309499019900472. [47] PEKMEZCI M, TANG JA, CHENG L. Comparison of expandable and fixed interbody cages in a human cadaver corpectomy model, part I: endplate force characteristics. J Neurosurg Spine. 2012;17(4):321-326. [48] YAO N, WANG W, LIU Y. Percutaneous endoscopic lumbar discectomy and interbody fusion with B-Twin expandable spinal spacer. Arch Orthop Trauma Surg. 2011;131(6):791-796. |
| [1] | . Changes in Lumbosacral Sagittal Plane Parameters of L5/S1 Disc Herniation [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-6. |
| [2] | Jiang Xiaocheng, Shi Lu, Wang Yinbin, Li Qiujiang, Xi Chuangzhen, Ma Zefeng, Cai Lijun. Systematical evaluation of bone fusion rate after interbody fusion in patients with osteoporosis and lumbar degenerative disease treated with teriparatide [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1427-1433. |
| [3] | Zhong Yizheng, Huang Peizhen, Cai Qunbin, Zheng Liqin, He Xingpeng, Dong Hang. Microstructural indexes that determine the trabecular bone maximum stress of micro-finite element models [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1313-1318. |
| [4] | Li Xiaomin, Tian Xiangdong, Tan Yetong, Zhu Guangyu, Wang Rongtian, Wang Jian, Xue Zhipeng, Ma Sheng, Hu Yuanyi, Huang Ye, Ding Tiansong. Changes of lower limb force line and knee function after high tibial osteotomy in osteoporotic medial ventricular knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1325-1329. |
| [5] | Wu Taoguang, Nie Shaobo, Chen Hua, Zhu Zhengguo, Qi Lin, Tang Peifu. Biomechanical characteristics of a new multi-dimensional cross locking plate in the treatment of subtrochanteric nonunion [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1330-1334. |
| [6] | Peng Zhixin, Yan Wengang, Wang Kun, Zhang Zhenjiang. Finite element analysis and structural optimization design of 3D printed forearm braces [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1340-1345. |
| [7] | Wu Tianliang, Tao Xiuxia, Xu Hongguang. Influence of different bone mineral densities on cage subsidence after stand-alone oblique lateral interbody fusion: three-dimensional finite element analysis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1352-1358. |
| [8] | Liu Jinyu, Zhang Hanshuo, Cui Hongpeng, Pan Lingzhi, Zhao Boran, Li Fei, Ding Yu. Finite element biomechanical analysis of minimally invasive treatment of cervical spondylotic myelopathy and accurate exercise rehabilitation [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1359-1364. |
| [9] | He Yujie, Kang Zhijie, Xue Mingming, Jin Feng, Li Zhijun, Wang Xing, Xu Yangyang, Gao Mingjie, Li Jiawei, Li Xiaohe, Wang Haiyan. Finite element analysis of transarticular screw fixation of adolescent thoracic vertebra [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1365-1370. |
| [10] | Wen Xinghua, Ding Huanwen, Cheng Kai, Yan Xiaonan, Peng Yuanhao, Wang Yuning, Liu Kang, Zhang Huiwu. Three-dimensional finite element model analysis of intramedullary nailing fixation design for large femoral defects in Beagle dogs [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1371-1376. |
| [11] | You Zhengqiu, Zhang Zhongzu, Wang Qunbo. Early symptomatic intervertebral disc pseudocysts after discectomy detected on MRI [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1403-1409. |
| [12] | Li Chao, Zhang Peipei, Xu Mengting, Li Linlin, Ding Jiangtao, Liu Xihua, Bi Hongyan. Respiratory training improves morphological changes of the multifidus muscle in patients with chronic nonspecific lower back pain assessed by musculoskeletal ultrasound [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1417-1421. |
| [13] | Sun Jiajia, Zhu Haidi, Lu Yun, Zhang Kai. Comparison of bone metabolism markers between type 2 diabetes mellitus and non-type 2 diabetes mellitus patients with hip fracture [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1156-1160. |
| [14] | Liu Jiaxin, Jia Peng, Men Yutao, Liu Lu, Wang Yeming, Ye Jinduo. Design and optimization of bone trabecular structure with triply periodic minimal surfaces [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 992-997. |
| [15] | Zhu Lin, Gu Weiping, Wang Can, Chen Gang. Biomechanical analysis of All-on-Four and pterygomaxillary implants under different maxillary bone conditions [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 985-991. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||