Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (18): 2930-2937.doi: 10.12307/2022.705
Previous Articles Next Articles
Research progress of rheumatoid arthritis animal model with high expression of anti-citrullinated protein antibodies
Yang Chao1, Zhong Gejia2, Liu Chunfang1, Lin Na1
- 1Institute of Chinese Materia Medica, China Academy of Chinese Medical Sciences, Beijing 100700, China; 2Beijing Tibetan Hospital, China Tibetology Research Center, Beijing 100029, China
-
Received:2021-09-03Accepted:2021-11-04Online:2022-06-28Published:2022-01-30 -
Contact:Lin Na, Researcher, Institute of Chinese Materia Medica, China Academy of Chinese Medical Sciences, Beijing 100700, China Liu Chunfang, Associate researcher, Institute of Chinese Materia Medica, China Academy of Chinese Medical Sciences, Beijing 100700, China -
About author:Yang Chao, Doctoral candidate, Institute of Chinese Materia Medica, China Academy of Chinese Medical Sciences, Beijing 100700, China -
Supported by:National Natural Science Foundation of China (General Program), No. 81974529 (LCF); National Natural Science Foundation of China (General Program), No. 81974526 (to LN); Beijing Natural Science Foundation, No. 7192139 (to LN); Fundamental Scientific Research Expenses for Central Public Welfare Research Institute, No. ZXKT17041 (to LCF); the Science and Technology Innovation Project of China Academy of Chinese Medical Sciences, No. CI2021A03808 (to LN); National Major Financial Special Project (to ZGJ)
CLC Number:
Cite this article
Yang Chao, Zhong Gejia, Liu Chunfang, Lin Na. Research progress of rheumatoid arthritis animal model with high expression of anti-citrullinated protein antibodies[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(18): 2930-2937.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
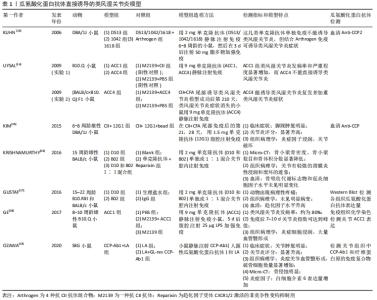
2.1 瓜氨酸化蛋白抗体单克隆抗体诱导模型 瓜氨酸化蛋白抗体是类风湿关节炎发病过程中一类关键的致病性自身抗体[8],其可通过活化多种免疫细胞,激活免疫活性因子发挥调控作用。一方面,这些免疫细胞的激活能够促进持续化炎症反应,导致病程反复、迁延不愈;另一方面,持续存在的炎症反应破坏局部组织,暴露大量自身抗原,促进自身免疫病理进程[9]。研究显示,瓜氨酸化蛋白抗体多克隆抗体无法诱导动物类风湿关节炎症状的产生[10],而瓜氨酸化蛋白抗体单克隆抗体不仅能促进炎症因子的表达[11-12],还诱导破骨细胞活化并介导骨丢失,进一步促进类风湿关节炎的病情发展[12],这为建立瓜氨酸化蛋白抗体高表达的类风湿关节炎动物模型提供了重要思路。 如表1所示,瓜氨酸化蛋白抗体单克隆抗体诱导的类风湿关节炎模型共有7篇文献报道[13-19],所采用的动物多选择DBA/1J,B10.Q,SKG以及BALB/c等小鼠;所采用的造模剂瓜氨酸化蛋白抗体单克隆抗体各文献报道的各不相同、不统一,基本来源于实验室自己制备,而且并不是所有报道的瓜氨酸化蛋白抗体单抗都可以直接诱导类风湿关节炎发生,如ACC1能直接诱导类风湿关节炎的发生,而ACC4则只能诱导类风湿关节炎复发并加重类风湿关节炎症状但不能直接诱导类风湿关节炎发生[14,18];造模方法主要有2种:一种是ACC1,D10和B02等单抗单独使用,通过静脉、腹腔或关节腔注射等诱导动物出现明显的类风湿关节炎症状[12,14,16-19],另一种是D513,ACC4等单抗结合Ⅱ型胶原(CII)或Arthrogen等免疫诱导动物类风湿关节炎发生[13-14]。"

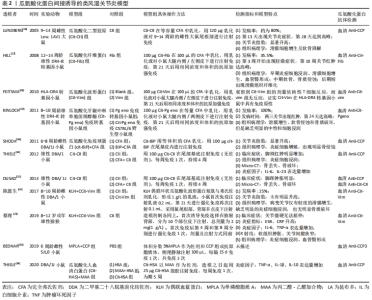
总之,通过动物注射瓜氨酸化蛋白抗体单抗,可直接诱导类风湿关节炎发生,或加重类风湿关节炎症状或诱导其复发,其中ACC1是研究较多的一类确有致病性的瓜氨酸化蛋白抗体单抗。值得注意的是:第一,瓜氨酸化蛋白抗体单抗单用诱导的类风湿关节炎临床症状普遍较轻;第二,由于动物种属、有无佐剂和所用造模方法的不同,不同实验条件下瓜氨酸化蛋白抗体单抗诱导所产生的类风湿关节炎疾病严重程度会出现差异;第三,由于单克隆抗体是由单一克隆B细胞杂交瘤产生,需经分离、纯化和内毒素检验等一系列复杂步骤后方可获得,成本高、难度大;第四,由于瓜氨酸化蛋白抗体阳性类风湿关节炎患者骨破坏并不是由某一特定的单抗导致,而更有可能是多种单抗的协同作用结果,所以用单一致病性单抗较难模拟患者体内复杂的病理状态。虽然这些不利因素可能会影响该模型的广泛使用(至今只有7篇文献报道),但由于瓜氨酸化蛋白抗体直接用于动物,能客观反映其对类风湿关节炎发生、发展的影响,因此该造模方法是未来研究瓜氨酸化蛋白抗体阳性类风湿关节炎的重要选择之一。 2.2 瓜氨酸化蛋白诱导模型 随着类风湿关节炎发病机制研究的深入,瓜氨酸化蛋白的重要程度也逐渐显现。虽然瓜氨酸化反应被认为是炎症过程中普遍存在的翻译后修饰过程,但是针对瓜氨酸化蛋白的耐受性丧失和相应产生的免疫反应几乎是类风湿关节炎所独有的[20]。甚至,一些学者认为瓜氨酸化蛋白就是诱导类风湿关节炎启动炎症反应的靶抗原,并且以瓜氨酸化蛋白为诱导剂成功建立瓜氨酸化蛋白抗体高表达的类风湿关节炎动物模型[21]。其造模方法为:首先,利用瓜氨酸化缓冲液,体外结合PAD酶催化蛋白发生瓜氨酸化反应后获取瓜氨酸化蛋白;进一步,利用瓜氨酸化蛋白免疫动物,通过适应性免疫反应诱导动物体内产生瓜氨酸化蛋白抗体。已知PAD酶有PAD 1,2,3,4,6共5种不同的亚型,分布在人体的各种组织中[22]。研究显示,在类风湿关节炎患者血清及关节滑膜内可检测到PAD2和PAD4,而未能检测到PAD1,PAD3和PAD6[23],证明 PAD2和PAD4 是催化 类风湿关节炎 免疫原蛋白瓜氨酸化最可能的亚型。据此,体外瓜氨酸化实验中常用PAD2,PAD4或PAD2/PAD4 cocktail来诱导反应。 采用瓜氨酸化蛋白诱导的瓜氨酸化蛋白抗体高表达类风湿关节炎动物模型于2005年被首次报道,至今研究论文共有11篇[24-34],是目前瓜氨酸化蛋白抗体高表达类风湿关节炎动物模型报道数量最多的一种,见表2。在该造模方法中,绝大多数(11篇中有9篇)采用瓜氨酸化蛋白与佐剂CFA、DDA、MPLA或MAA的乳化剂免疫动物,少数(仅2篇)单用瓜氨酸化蛋白免疫。在造模剂瓜氨酸化蛋白种类的选择上有多样,如瓜氨酸化CII最受欢迎有3篇文献报道,瓜氨酸化波形蛋白和瓜氨酸化波形蛋白短肽各有2篇文献报道,其他如瓜氨酸化纤维蛋白、氨酸化免疫球蛋白、瓜氨酸化人软骨蛋白聚糖、瓜氨酸化人血清白蛋白、环瓜氨酸多肽和瓜氨酸化牙龈卟啉单胞菌烯醇酶等各1篇。由于瓜氨酸化蛋白需要PAD通过酶促反应体外催化相关蛋白,并经分子生物学技术鉴定确认方可,步骤繁琐,难以获得,因此在一定程度上影响了该模型的推广应用。不过,肖长虹团队报道通过特定氨基酸序列可以合成具有免疫原性的环瓜氨酸化波形蛋白短肽,这或许可以有效降低瓜氨酸化蛋白的制备难度,为造模剂的来源提供了新的思路,该团队通过DBA/1小鼠和成年猕猴探究了该类合成肽的免疫原性,结果表明猕猴出现了十分明显的类风湿关节炎症状而小鼠的发病率仅为25%,由此可见不同物种对特定瓜氨酸化蛋白的反应性有很大差异[24-25]。总体来说,该类方法在造模动物的选择上与瓜氨酸化蛋白抗体直接诱导法类似,以DBA/1小鼠为主,也有部分选择转基因小鼠或Lewis大鼠、以及一项研究选择了与人类行为表现与神经系统结构和功能都高度相似的非人灵长类动物猕猴等。关于模型的特点,一方面,除了血清可检测到高水平表达的瓜氨酸化蛋白抗体外,关节均可出现红肿症状表现和病理特征,且绝大部分动物关节炎症状明显;需要特别注意的是,由于瓜氨酸化蛋白种类、诱导方式、注射剂量及实验动物品种和品系的不同,不同的造模方法所得的瓜氨酸化蛋白抗体的类型和表达水平也存在差异。"

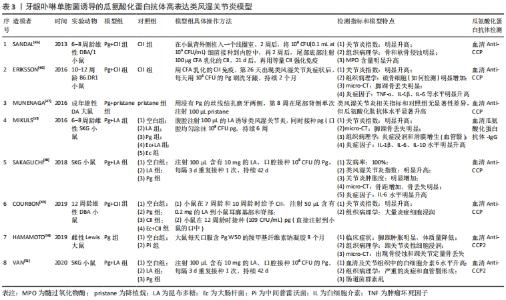
2.3 牙龈卟啉单胞菌诱导模型 类风湿关节炎与牙周病是两种具有许多共同特征的炎症性疾病,如均具有局部炎症引起的骨丢失等 [35]。牙龈卟啉单胞菌(Porphyromonas gingivalis,Pg)是一种革兰阴性厌氧球杆菌,是牙周病重要的致病菌之一[36],也是惟一已知的产生肽酰基精氨酸脱亚胺酶(Porphyromonas gingivalis peptidylarginine deiminase,PPAD)的细菌。PPAD具有与PAD相同的生理活性,可致蛋白质瓜氨酸化,机体识别瓜氨酸化抗原后通过适应性免疫产生瓜氨酸化蛋白抗体,从而进一步诱导破骨细胞活化[37-38],但其瓜氨酸化蛋白是片段化的,如纤维蛋白原和α-烯醇化酶被Pg产生的精氨酸特异性酶消化后的片段,不同于内源PPD产生的瓜氨酸化蛋白[39]。临床研究所证实,在类风湿关节炎患者的牙龈活检中发现牙龈卟啉单胞菌和牙龈瓜氨酸化蛋白,其血液中也含有高浓度的瓜氨酸化蛋白抗体和牙龈卟啉单胞菌抗体[40-41]。最近的一项研究证实PPAD能够瓜氨酸化天然人源蛋白质,包括纤维原蛋白、热休克蛋白、组蛋白、波形蛋白等[42]。同时,内源PAD相关的瓜氨酸化可被细菌来源的PPAD增强,并促进瓜氨酸化蛋白抗体的产生,且可能先于类风湿关节炎症状的出现[43-44],证实了临床瓜氨酸化蛋白抗体出现于类风湿关节炎临床症状之前的认识。虽然目前类风湿关节炎与pg的相关性并不完全清楚,但利用pg诱导瓜氨酸化蛋白抗体高表达的类风湿关节炎动物模型已经被多次应用,并可用于瓜氨酸化和关节炎发展相关的机制研究[45-51],见表3。"
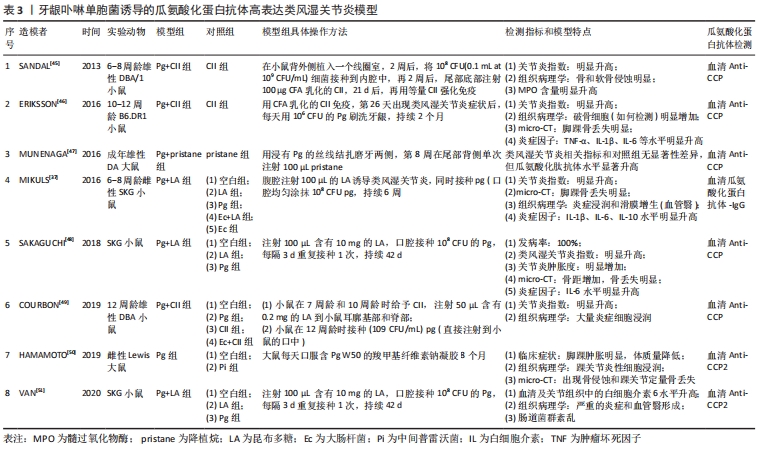

在表3的造模方法中,需要通过接种Pg并结合CII以及各类佐剂加强免疫,仅一篇文献报道了可以通过单独使用Pg感染建立模型,但实验周期长达8个月,这显然不利于该类模型的推广和应用[49]。Pg的接种方式包括口腔均匀涂抹、口腔注射、丝线结扎以及植入线圈等,其中口腔均匀涂抹操作简单,但常常需要每隔一两天操作1次,而后面3种方式的优缺点则恰恰相反。Pg的接种时间点上有类风湿关节炎造模剂注射之前、同时和之后3种情况,根据文献并没有发现不同的接种时间点对瓜氨酸化蛋白抗体表达量以及类风湿关节炎严重程度有明显影响。在造模动物的选择上,常选择DBA/1和SKG小鼠等,虽然也有研究人员选择DA或Lewis大鼠,其优点在于口腔接种Pg更加方便,但在造模成功率以及类风湿关节炎症状等方面,免疫易感小鼠更具优势。值得注意的是,在造模成功后,动物不仅具有关节炎临床症状特征外还有较明显的牙周炎症状,甚至还会诱导出现帕金森综合征[43],多种并发症可能会影响发病机制的深入研究。但是,相对于前述两种造模方法,Pg感染的模型能诱导出高表达水平的瓜氨酸化蛋白抗体,且所需要的造模材料更易获取和经济,或能成为瓜氨酸化蛋白抗体阳性类风湿关节炎研究的重要选择之一。 2.4 其他建模诱导方法 研究表明类风湿关节炎和HLA-DRB1等位基因有关,HLA-DR分子的作用是向辅助性T细胞呈递肽,帮助B细胞产生IgG抗体[51]。有学者选择PAD分别免疫C3H小鼠(C3H为健康小鼠,其IEβk链与β1链HLA-DRB1*04:01高度同源[52],β1链是与人类类风湿关节炎关联最强的等位基因)和DBA/2小鼠(其IEβd链类似于HLA-DRB1*04:02,一种与类风湿关节炎无关的HLADR4亚型),结果显示C3H小鼠产生了针对PAD的抗体和T细胞,以及针对瓜氨酸化纤维蛋白原肽的IgG抗体,而DBA/2小鼠不能。因此,研究者猜测T细胞对PAD蛋白的免疫可能通过半抗原或载体机制触发瓜氨酸化蛋白抗体,这种新的免疫方法或能构成类风湿关节炎高表达瓜氨酸化蛋白抗体小鼠模型的基础。 此外,爱泼斯坦-巴尔病毒(Epstein-Barr virus,EBV)和类风湿关节炎之间也存在关联。在类风湿关节炎患者的血清中存在抗瓜氨酸化EBV肽的抗体,这表明EBV感染可能会诱导瓜氨酸化蛋白抗体的产生[53]。已有研究报道将EBV阳性和阴性类风湿关节炎患者滑膜组织的异位淋巴结构双移植到重症联合免疫缺陷小鼠中,结果显示EBV感染与类风湿关节炎滑膜中产生瓜氨酸化蛋白抗体的B细胞原位激活密切相关[54],这或许也可以为建立瓜氨酸化蛋白抗体高表达的类风湿关节炎动物模型提供新思路。"

| [1] BRINK M, HANSSON M, MATHSSON-ALM L, et al. Rheumatoid factor isotypes in relation to antibodies against citrullinated peptides and carbamylated proteins before the onset of rheumatoid arthritis. Arthritis Res Ther. 2016;18:43. [2] KAPOOR S. Comment on The role of citrullinated protein antibodies in predicting erosive disease in rheumatoid arthritis: a systematic literature review and meta-analysis. Int J Rheumatol. 2015;2015: 175251. [3] ALETAHA D, NEOGI T, SILMAN AJ, et al. 2010 Rheumatoid arthritis classification criteria: an American College of Rheumatology/European League Against Rheumatism collaborative initiative. Arthritis Rheum. 2010;62:2569-2581. [4] BUGATTI S, BOGLIOLO L, VITOLO B, et al. Anti-citrullinated protein antibodies and high levels of rheumatoid factor are associated with systemic bone loss in patients with early untreated rheumatoid arthritis. Arthritis Res Ther. 2016;18:226. [5] KLARESKOG L, CATRINA AI, PAGET S. Rheumatoid arthritis. Lancet. 2009;373:659-672. [6] DAHA NA, TOES RE. Rheumatoid arthritis: are ACPA-positive and ACPA-negative RA the same disease? Nat Rev Rheumatol. 2011;7:202-203. [7] FERT-BOBER J, DARRAH E, ANDRADE F. Insights into the study and origin of the citrullinome in rheumatoid arthritis. Immunol Rev. 2020; 294:133-147. [8] DAI X, DAI X, GONG Z, et al. Disease-specific autoantibodies induce trained immunity in RA synovial tissues and its gene signature correlates with the response to clinical therapy. Mediat Inflamm. 2020; 2020:2109325. [9] DONG X, ZHENG Z, ZHAI Y, et al. ACPA mediates the interplay between innate and adaptive immunity in rheumatoid arthritis. Autoimmun Rev. 2018;17:845-853. [10] GE C, XU B, LIANG B, et al. Structural Basis of Cross-Reactivity of Anti-Citrullinated Protein Antibodies. Arthritis Rheumatol. 2019;71:210-221. [11] LU MC, LAI NS, YU HC, et al. Anti-citrullinated protein antibodies bind surface-expressed citrullinated Grp78 on monocyte/macrophages and stimulate tumor necrosis factor alpha production. Arthritis Rheum. 2010;62:1213-1223. [12] LU MC, LAI NS, YIN WY, et al. Anti-citrullinated protein antibodies activated ERK1/2 and JNK mitogen-activated protein kinases via binding to surface-expressed citrullinated GRP78 on mononuclear cells. J Clin Immunol. 2013;33:558-566. [13] KUHN KA, KULIK L, TOMOOKA B, et al. Antibodies against citrullinated proteins enhance tissue injury in experimental autoimmune arthritis. J Clin Invest. 2006;116:961-973. [14] UYSAL H, BOCKERMANN R, NANDAKUMAR KS, et al. Structure and pathogenicity of antibodies specific for citrullinated collagen type II in experimental arthritis. J Exp Med. 2009;206:449-462. [15] KIM Y, LEE J, JUNG H, et al. Development of synthetic anti-cyclic citrullinated peptide antibody and its arthritogenic role. J Clin Invest. 2015;4:e51. [16] KRISHNAMURTHY A, JOSHUA V, HAJ HENSVOLD A, et al. Identification of a novel chemokine-dependent molecular mechanism underlying rheumatoid arthritis-associated autoantibody-mediated bone loss. Ann Rheum Dis. 2016;75:721-729. [17] GUSTAF W, DUYGU BB, CÁTIA FC, et al. Autoantibodies to citrullinated proteins induce joint pain independent of inflammation via a chemokine-dependent mechanism Correction. Ann Rheum Dis. 2019; 78:865. [18] GE C, TONG D, LIANG B, et al. Anti-citrullinated protein antibodies cause arthritis by cross-reactivity to joint cartilage. JCI Insight. 2017; 2(13):e93688. [19] OZAWA T, OUHARA K, TSUDA R, et al. Physiological target, molecular evolution and pathogenic functions of a monoclonal ACPA obtained from an RA patient. Arthritis Rheumatol. 2020;72(12):2040-2049. [20] SCHELLEKENS GA, VISSER H, DE JONG BA, et al. The diagnostic properties of rheumatoid arthritis antibodies recognizing a cyclic citrullinated peptide. Arthritis Rheum. 2000;43:155-163. [21] KLARESKOG L, RöNNELID J, LUNDBERG K, et al. Immunity to citrullinated proteins in rheumatoid arthritis. Annu Rev Immunol. 2008;26:651-675. [22] PRATESI F, PANZA F, PAOLINI I, et al. Fingerprinting of anti-citrullinated protein antibodies (ACPA): specificity, isotypes and subclasses. Lupus. 2015;24:433-441. [23] FOULQUIER C, SEBBAG M, CLAVEL C, et al. Peptidyl arginine deiminase type 2 (PAD-2) and PAD-4 but not PAD-1, PAD-3, and PAD-6 are expressed in rheumatoid arthritis synovium in close association with tissue inflammation. Arthritis Rheum. 2007;56:3541-3553. [24] 陈恩生,崔明珠,赵晓峰,等.合成环瓜氨酸化蛋白短肽的免疫原性和致关节炎性研究[J]. 中国免疫学杂志,2017,33(1):25-30. [25] 蔡理,余焙佳,毕亚男,等.合成环瓜氨酸蛋白短肽诱导猕猴类风湿关节炎模型的研究[J].中国免疫学杂志,2019,35(16):1937-1943. [26] LUNDBERG K, NIJENHUIS S, VOSSENAAR ER, et al. Citrullinated proteins have increased immunogenicity and arthritogenicity and their presence in arthritic joints correlates with disease severity. Arthritis Res Ther. 2005;7:R458-R467. [27] HILL JA, BELL DA, BRINTNELL W, et al. Arthritis induced by posttranslationally modified (citrullinated) fibrinogen in DR4-IE transgenic mice. J Exp Med. 2008;205:967-979. [28] FEITSMA AL, VAN DER VOORT EI, FRANKEN KL, et al. Identification of citrullinated vimentin peptides as T cell epitopes in HLA-DR4-positive patients with rheumatoid arthritis. Arthritis Rheum. 2010;62:117-125. [29] KINLOCH A J, ALZABIN S, BRINTNELL W, et al. Immunization with Porphyromonas gingivalis enolase induces autoimmunity to mammalian α-enolase and arthritis in DR4-IE-transgenic mice. Arthritis Rheum. 2011;63:3818-3823. [30] SHODA H, FUJIO K, SHIBUYA M, et al. Detection of autoantibodies to citrullinated BiP in rheumatoid arthritis patients and pro-inflammatory role of citrullinated BiP in collagen-induced arthritis. Arthritis Res Ther . 2011;13:R191. [31] THIELE GM, DURYEE MJ, DUSAD A, et al. Citrullinated mouse collagen administered to DBA/1J mice in the absence of adjuvant initiates arthritis. Int Immunopharmacol. 2012;13:424-431. [32] DUSAD A, DURYEE MJ, SHAW AT, et al. Induction of bone loss in DBA/1J mice immunized with citrullinated autologous mouse type II collagen in the absence of adjuvant. Immunol Res. 2014;58:51-60. [33] BEDNAR KJ, NYCHOLAT CM, RAO TS, et al. Exploiting CD22 To Selectively Tolerize Autoantibody Producing B-Cells in Rheumatoid Arthritis. ACS Chem Biol. 2019;14:644-654. [34] THIELE GM, DURYEE MJ, HUNTER CD, et al. Immunogenic and inflammatory responses to citrullinated proteins are enhanced following modification with malondialdehyde-acetaldehyde adducts. Int Immunopharmacol. 2020;83:106433. [35] KADOWAKI T, NAKAYAMA K, OKAMOTO K, et al. Porphyromonas gingivalis proteinases as virulence determinants in progression of periodontal diseases. J Biochem. 2000;128:153-159. [36] YAMAKAWA M, OUHARA K, KAJIYA M, et al. Porphyromonas gingivalis infection exacerbates the onset of rheumatoid arthritis in SKG mice. Clin Exp Immunol. 2016;186:177-189. [37] MIKULS TR, PAYNE JB, YU F, et al. Periodontitis and Porphyromonas gingivalis in patients with rheumatoid arthritis. Arthritis Rheumatol. 2014;66:1090-1100. [38] MARESZ KJ, HELLVARD A, SROKA A, et al. Porphyromonas gingivalis facilitates the development and progression of destructive arthritis through its unique bacterial peptidylarginine deiminase (PAD). PLoS Pathog. 2013;9:e1003627. [39] JENNING M, MARKLEIN B, YTTERBERG J, et al. Porphyromonas gingivalisBacterial citrullinated epitopes generated by infection-a missing link for ACPA production. Ann Rheum Dis. 2020;79:1194-1202. [40] MAROTTE H. Tooth-brushing: an impact on rheumatoid arthritis. Joint Bone Spine. 2016;83:619-621. [41] GULLY N, BRIGHT R, MARINO V, et al. Porphyromonas gingivalis peptidylarginine deiminase, a key contributor in the pathogenesis of experimental periodontal disease and experimental arthritis. PLoS One. 2014;9:e100838. [42] DARRAH E. ANDRADE F. Rheumatoid arthritis and citrullination. Curr Opin Rheumatol. 2018;30:72-78. [43] SAKAI Y, SHIMAUCHI H, ITO H, et al. Porphyromonas gingivalis-specific IgG subclass antibody levels as immunological risk indicators of periodontal bone loss. J Clin Periodontol. 2001;28:853-859. [44] MARESZ KJ, HELLVARD A, SROKA A, et al. Porphyromonas gingivalis facilitates the development and progression of destructive arthritis through its unique bacterial peptidylarginine deiminase (PAD). PLoS Pathogens. 2013;9:e1003627. [45] SANDAL I, KARYDIS A, LUO J, et al. Bone loss and aggravated autoimmune arthritis in HLA-DRβ1-bearing humanized mice following oral challenge with Porphyromonas gingivalis. Arthritis Res Ther. 2016; 18:249. [46] ERIKSSON K, LöNNBLOM E, TOUR G, et al. Effects by periodontitis on pristane-induced arthritis in rats. J Transl Med. 2016;14:311. [47] MUNENAGA S, OUHARA K, HAMAMOTO Y, et al. The involvement of C5a in the progression of experimental arthritis with Porphyromonas gingivalis infection in SKG mice. Arthritis Res Ther. 2018;20:247. [48] SAKAGUCHI W, TO M, YAMAMOTO Y, et al. Detection of anti-citrullinated protein antibody (ACPA) in saliva for rheumatoid arthritis using DBA mice infected with Porphyromonas gingivalis. Arch Oral Biol. 2019;108:104510. [49] COURBON G, RINAUDO-GAUJOUS M, BLASCO-BAQUE V, et al. Porphyromonas gingivalis experimentally induces periodontis and an anti-CCP2-associated arthritis in the rat. Ann Rheum Dis. 2019;78:594-599. [50] HAMAMOTO Y, OUHARA K, MUNENAGA S, et al. Effect of Porphyromonas gingivalis infection on gut dysbiosis and resultant arthritis exacerbation in mouse model. Arthritis Res Ther. 2020;22:249. [51] VAN VW, PRUIJN GJ. Citrullination: a small change for a protein with great consequences for rheumatoid arthritis. Arthritis Res. 2000;2:249-251. [52] ARNOUX F, MARIOT C, PEEN E, et al. Peptidyl arginine deiminase immunization induces anticitrullinated protein antibodies in mice with particular MHC types. PNAS. 2017;114:E10169-E10177. [53] PRATESI F, TOMMASI C, ANZILOTTI C, et al. Deiminated Epstein-Barr virus nuclear antigen 1 is a target of anti-citrullinated protein antibodies in rheumatoid arthritis. Arthritis Rheum. 2006;54:733-741. [54] CROIA C, SERAFINI B, BOMBARDIERI M, et al. Epstein-Barr virus persistence and infection of autoreactive plasma cells in synovial lymphoid structures in rheumatoid arthritis. Ann Rheum Dis. 2013;72: 1559-1568. [55] SCHIFF M, WEINBLATT ME, VALENTE R, et al. Head-to-head comparison of subcutaneous abatacept versus adalimumab for rheumatoid arthritis: two-year efficacy and safety findings from AMPLE trial. Ann Rheum Dis. 2014;73:86-94. [56] HARROLD LR, LITMAN HJ, CONNOLLY SE, et al. Effect of Anticitrullinated Protein Antibody Status on Response to Abatacept or Antitumor Necrosis Factor-α Therapy in Patients with Rheumatoid Arthritis: A US National Observational Study. J Rheumatol. 2018;45(1):32-39. [57] COURVOISIER DS, CHATZIDIONYSIOU K, MONGIN D, et al. The impact of seropositivity on the effectiveness of biologic anti-rheumatic agents: results from a collaboration of 16 registries. Rheumatology (Oxford). 2021;60:820-828. [58] GENOVESE M C, FLEISCHMANN R, KIVITZ A, et al. Efficacy and safety of sarilumab in combination with csDMARDs or as monotherapy in subpopulations of patients with moderately to severely active rheumatoid arthritis in three phase III randomized, controlled studies. Arthritis Res Ther. 2020;22:139. [59] Lü S, WANG Q, LI G, et al. The treatment of rheumatoid arthritis using Chinese medicinal plants: from pharmacology to potential molecular mechanisms. J. Ethnopharmacol. 2015;176:177-206. [60] MAINI RN, ELLIOTT MJ, BRENNAN FM, et al. Monoclonal anti-TNF alpha antibody as a probe of pathogenesis and therapy of rheumatoid disease. Immunol Rev. 1995;144:195-223. [61] SAKAGUCHI N, TAKAHASHI T, HATA H, et al. Altered thymic T-cell selection due to a mutation of the ZAP-70 gene causes autoimmune arthritis in mice. Nature. 2003;426:454-460. |
| [1] | Tan Xinfang, Guo Yanxing, Qin Xiaofei, Zhang Binqing, Zhao Dongliang, Pan Kunkun, Li Yuzhuo, Chen Haoyu. Effect of uniaxial fatigue exercise on patellofemoral cartilage injury in a rabbit [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(在线): 1-6. |
| [2] | Zhang Lichuang, Xu Hao, Ma Yinghui, Xiong Mengting, Han Haihui, Bao Jiamin, Zhai Weitao, Liang Qianqian. Mechanism and prospects of regulating lymphatic reflux function in the treatment of rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1459-1466. |
| [3] | Xu Xinzhong, Wu Zhonghan, Yu Shuisheng, Zhao Yao, Xu Chungui, Zhang Xin, Zheng Meige, Jing Juehua. Biomechanical analysis of different ways of inserting Steinmann Pins into the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1313-1317. |
| [4] | Zhang Yufang, Lü Meng, Mei Zhao. Construction and verification of a full spine biomechanical model of adolescent scoliosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1351-1356. |
| [5] | Li Wei, Zhu Hanmin, Wang Xin, Gao Xue, Cui Jing, Liu Yuxin, Huang Shuming. Effect of Zuogui Wan on bone morphogenetic protein 2 signaling pathway in ovariectomized osteoporosis mice [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1173-1179. |
| [6] | Wang Baojuan, Zheng Shuguang, Zhang Qi, Li Tianyang. Miao medicine fumigation can delay extracellular matrix destruction in a rabbit model of knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1180-1186. |
| [7] | Lü Yiyan, Li Hanbing, Ma Xiaoqing, Zhang Han, Zhang Yuhang, Li Genlin. Establishment and characteristic analysis of interior heat and diabetes mouse model using compound factors [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1187-1193. |
| [8] | Chen Shijian, Li Ge, Zhang Yu, Guan Yalun, Li Xuejiao, Liu Shuhua, Li Yongchao, Li Yunfeng, Gao Jinfeng, Wei Xiaoyue, Zhao Yuhong. Comparison and evaluation of MPTP-induced subacute and chronic models of Parkinson’s disease in mice [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1247-1252. |
| [9] | Zhu Chan, Han Xuke, Yao Chengjiao, Zhang Qiang, Liu Jing, Shao Ming. Acupuncture for Parkinson’s disease: an insight into the action mechanism in animal experiments [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1272-1277. |
| [10] | Hu Wei, Xie Xingqi, Tu Guanjun. Exosomes derived from bone marrow mesenchymal stem cells improve the integrity of the blood-spinal cord barrier after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 992-998. |
| [11] | Wen Xiaoyu, Sun Yuhao, Xia Meng. Effects of serum containing Wuzang Wenyang Huayu Decoction on phosphorylated-tau protein expression in Alzheimer’s disease cell model [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1068-1073. |
| [12] | Wang Xinmin, Liu Fei, Xu Jie, Bai Yuxi, Lü Jian. Core decompression combined with dental pulp stem cells in the treatment of steroid-associated femoral head necrosis in rabbits [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1074-1079. |
| [13] | Liu Yuhang, Zhou Jianqiang, Xu Xuebin, Qu Xingyue, Li Ziyu, Li Kun, Wang Xing, Li Zhijun, Li Xiaohe, Zhang Shaojie. Establishment and validation of finite element model of lower cervical spine in 6-year-old children [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(6): 870-874. |
| [14] | Liu Yiyi, Qiu Junqiang, Yi Longyan, Zhou Cailiang. Effect of resistance training on interleukin-6 and C-reactive protein in middle-age and elderly people: a Meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 804-812. |
| [15] | Liu Jin, Li Zhen, Hao Huiqin, Wang Ze, Zhao Caihong, Lu Wenjing. Ermiao san aqueous extract regulates proliferation, migration, and inflammatory factor expression of fibroblast-like synovial cells in collagen-induced arthritis rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 688-693. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||