Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (15): 2394-2399.doi: 10.12307/2022.597
Previous Articles Next Articles
Correlation between body composition and bone mineral density in middle-aged and elderly people
Yuan Jiayao1, Lin Yanping1, Lin Xiancan1, Huang Jiachun2, Chen Tongying1, Lin Shi1, Lian Xiaohang1, Wan Lei2, Huang Hongxing2
- 1Guangzhou University of Chinese Medicine, Guangzhou 510000, Guangdong Province, China; 2the Third Affiliated Hospital of Guangzhou University of Chinese Medicine, Guangzhou 510000, Guangdong Province, China
-
Received:2021-06-16Revised:2021-06-17Accepted:2021-07-26Online:2022-05-28Published:2022-01-06 -
Contact:Huang Hongxing, MD, Professor, Chief physician, the Third Affiliated Hospital of Guangzhou University of Chinese Medicine, Guangzhou 510000, Guangdong Province, China -
About author:Yuan Jiayao, Master candidate, Guangzhou University of Chinese Medicine, Guangzhou 510000, Guangdong Province, China -
Supported by:the National Natural Science Foundation of China (General Project), No. 81973886 (to HHX); “Double First Class” and High-level University Discipline Collaborative Innovation Team Project of Guangzhou University of Chinese Medicine, No. 2021XK21 (to HHX); Subject Research Key Project of Guangzhou University of Chinese Medicine, No. XK2019028 (to HHX)
CLC Number:
Cite this article
Yuan Jiayao, Lin Yanping, Lin Xiancan, Huang Jiachun, Chen Tongying, Lin Shi, Lian Xiaohang, Wan Lei, Huang Hongxing. Correlation between body composition and bone mineral density in middle-aged and elderly people[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(15): 2394-2399.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
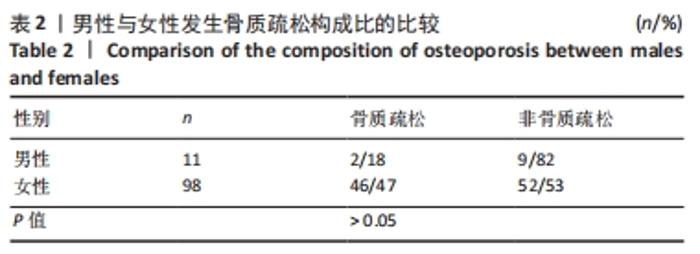
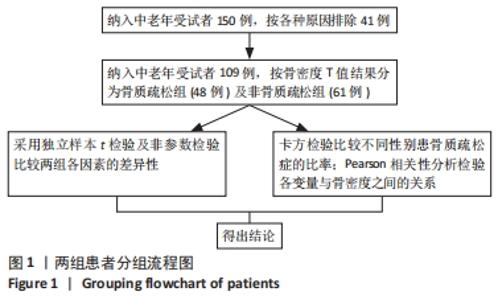
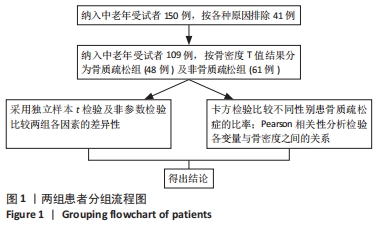
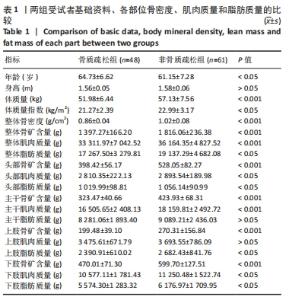
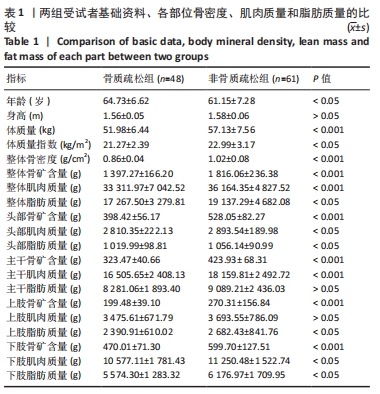
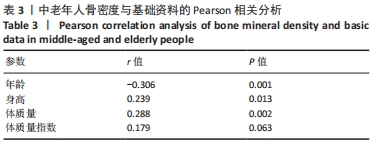
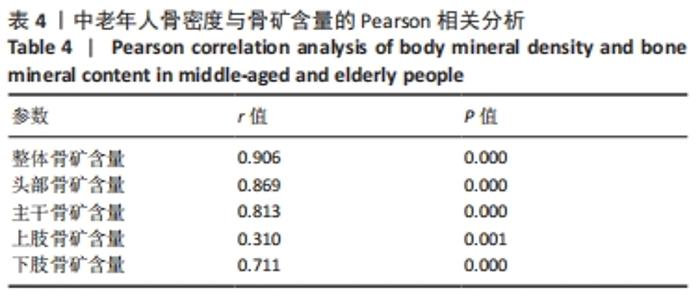
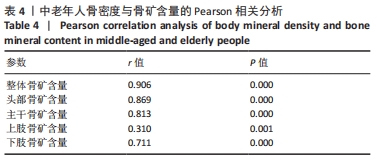
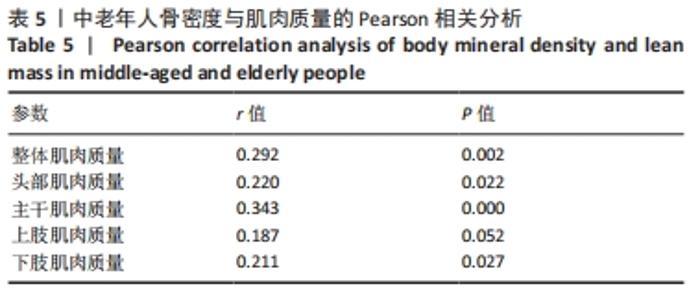
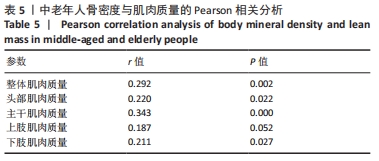
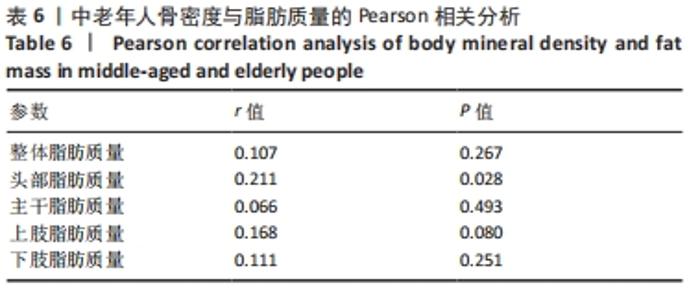
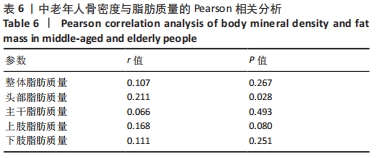
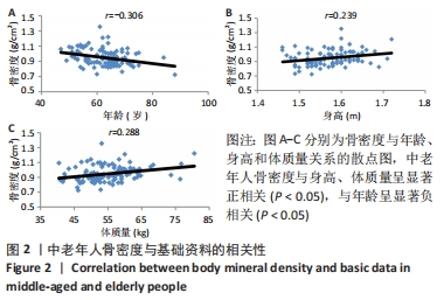
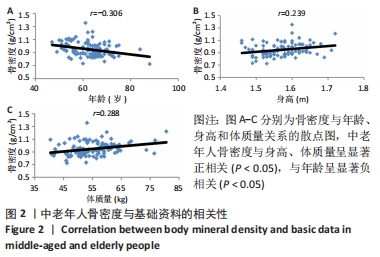
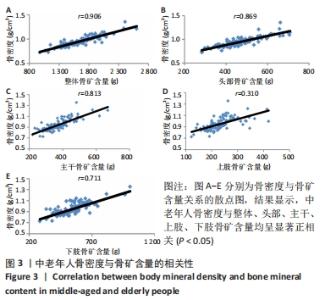
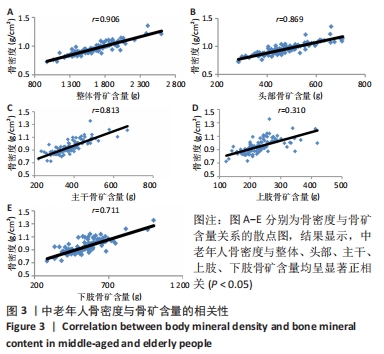
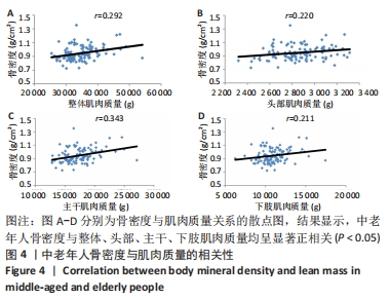
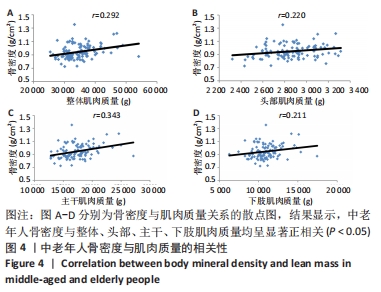
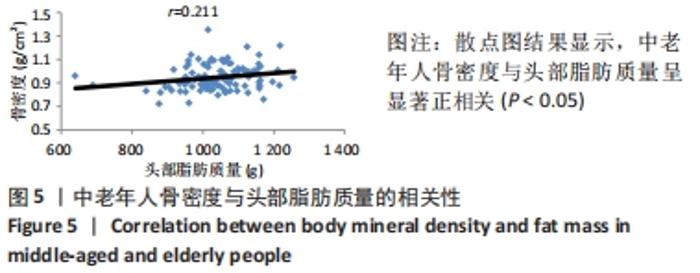
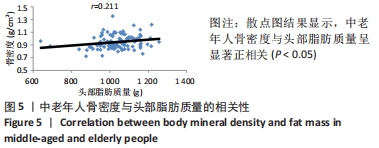
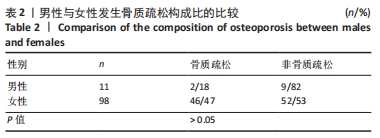
如表1所示,在基础资料方面,骨质疏松组年龄显著高于非骨质疏松组,差异有显著性意义(P < 0.05);身高低于非骨质疏松组,差异无显著性意义(P > 0.05);体质量和体质量指数显著低于非骨质疏松组,差异有显著性意义(P < 0.05)。在骨密度及骨矿含量对比方面,骨质疏松组的整体、头部、主干、上肢、下肢骨矿含量和整体骨密度显著低于非骨质疏松组,差异有显著性意义(P < 0.05)。在肌肉质量方面,骨质疏松组的整体、头部、主干、下肢肌肉质量显著低于非骨质疏松组,差异有显著性意义(P < 0.05);而骨质疏松组上肢肌肉质量也低于非骨质疏松组,但差异无显著性意义(P > 0.05)。在脂肪质量方面,骨质疏松组的整体、头部、上肢、下肢脂肪质量显著低于非骨质疏松组,差异有显著性意义(P < 0.05);骨质疏松组主干脂肪质量也低于非骨质疏松组,但差异无显著性意义(P > 0.05)。 2.4 不同性别患骨质疏松症构成比的比较 纳入的受试者中,男11例,女98例,平均年龄(62.72±7.19)岁,男性有18%的受试者发生骨质疏松,而女性中47%存在骨质疏松的情况,女性发生骨质疏松的比例大于男性,但差异无显著性意义(P > 0.05),见表2。"
| [1] YANG TL, SHEN H, LIU A, et al. A road map for understanding molecular and genetic determinants of osteoporosis. Nat Rev Endocrinol. 2020; 16(2):91-103. [2] SOZEN T, OZISIK L, CALIK BASARAN N. An overview and management of osteoporosis. Eur J Rheumatol. 2017;4(1):46-56. [3] 梁玉柱,郭洪刚.老年骨质疏松性髋部骨折:昨天、今天及未来[J].中国组织工程研究,2017,21(15):2438-2443. [4] 中华医学会骨质疏松和骨矿盐疾病分会.中国骨质疏松症流行病学调查及“健康骨骼”专项行动结果发布[J]. 中华骨质疏松和骨矿盐疾病杂志,2019,12(4):317-318. [5] ZHU Y, LIU S, CHEN W, et al.Epidemiology of low-energy fracture in Chinese postmenopausal women:changing trend of incidence since menopause and associated risk factors, a national population-based survey. Menopause. 2019;26(3):286-292. [6] PENG K,YAO P,KARTSONAKI C, et al. Menopause and risk of hip fracture in middle-aged Chinese women: a 10-year follow-up of China Kadoorie Biobank. Menopause. 2020;27(3):311-318. [7] ANAGNOSTIS P, SIOLOS P, GKEKAS NK, et al. Association between age at menopause and fracture risk: a systematic review and meta-analysis. Endocrine. 2019;63(2):213-224. [8] SULLIVAN SD,LEHMAN A,NATHAN NK, et al. Age of menopause and fracture risk in postmenopausal women randomized to calcium + vitamin D, hormone therapy, or the combination: results from the Women’s Health Initiative Clinical Trials. Menopause. 2017;24(4):371-378. [9] LUO X, CHENG R, ZHANG J, et al. Evaluation of body composition in POF and its association with bone mineral density and sex steroid levels. Gynecol Endocrinol. 2018;34(12):1027-1030. [10] ST-ONGE M, GALLAGHER D. Body composition changes with aging: The cause or the result of alterations in metabolic rate and macronutrient oxidation? Nutrition. 2010;26(2):152-155. [11] 夏维波,章振林,林华,等.原发性骨质疏松症诊疗指南(2017)[J].中国骨质疏松杂志,2019,25(3):281-309. [12] BIGAARD J, FREDERIKSEN K, TJØNNELAND A, et al. Body Fat and Fat-Free Mass and All-Cause Mortality. Obesity (Silver Spring, Md.). 2004; 12(7):1042-1049. [13] LI S. Relationship between soft tissue body composition and bone mass in perimenopausal women. Maturitas. 2004;47(2):99-105. [14] CUI L, SHIN M, KWEON S, et al. Relative contribution of body composition to bone mineral density at different sites in men and women of South Korea. J Bone Miner Metabol. 2007;25(3):165-171. [15] LUO X, CHENG R, ZHANG J, et al. Evaluation of body composition in POF and its association with bone mineral density and sex steroid levels. Gynecol Endocrinol. 2018;34(12):1027-1030. [16] DYTFELD J, IGNASZAK-SZCZEPANIAK M, GOWIN E, et al. Influence of lean and fat mass on bone mineral density (BMD) in postmenopausal women with osteoporosis. Arch Gerontol Geriatr. 2011;53(2): e237-e242. [17] SIDDIQUE N, FALLON N, CASEY MC, et al. Statistical analysis of fat and lean mass in osteoporosis in elderly population using total body DXA scans. Irish J Med Sci. 2020;189(3):1105-1113. [18] MOON S. Relationship of lean body mass with bone mass and bone mineral density in the general Korean population. Endocrine. 2014; 47(1):234-243. [19] FU X, MA X, LU H, et al. Associations of fat mass and fat distribution with bone mineral density in pre- and postmenopausal Chinese women. Osteoporos Int. 2011;22(1):113-119. [20] GJESDAL CG, HALSE JI, EIDE GE, et al. Impact of lean mass and fat mass on bone mineral density: The Hordaland Health Study. Maturitas. 2008; 59(2):191-200. [21] JIANG Y, ZHANG Y, JIN M, et al. Aged-Related Changes in Body Composition and Association between Body Composition with Bone Mass Density by Body Mass Index in Chinese Han Men over 50-year-old. PLOS ONE. 2015;10(6):e130400. [22] HANNAN MT, FELSON DT, ANDERSON JJ. Bone mineral density in elderly men and women: Results from the framingham osteoporosis study. J Bone Miner Res. 1992;7(5):547-553. [23] GARNETT SP, HOGLER W, BLADES B, et al. Relation between hormones and body composition, including bone, in prepubertal children. Am J Clin Nutr. 2004;80(4):966-972. [24] KAMEDA T, MANO H, YUASA T, et al. Estrogen inhibits bone resorption by directly inducing apoptosis of the bone-resorbing osteoclasts. J Exp Med. 1997;186(4):489-495. [25] OSHIMA K, NAMPEI A, MATSUDA M, et al. Adiponectin increases bone mass by suppressing osteoclast and activating osteoblast. Biochem Biophys Res Commun. 2005;331(2):520-526. [26] RITLAND LM, ALEKEL DL, MATVIENKO OA, et al. Centrally located body fat is related to appetitive hormones in healthy postmenopausal women. Eur J Endocrinol. 2008;158(6):889-897. [27] ZHANG P, PETERSON M, SU GL, et al. Visceral adiposity is negatively associated with bone density and muscle attenuation. Am J Clin Nutr. 2015;101(2):337-343. [28] SHEU Y, MARSHALL LM, HOLTON KF, et al. Abdominal body composition measured by quantitative computed tomography and risk of non-spine fractures: the Osteoporotic Fractures in Men (MrOS) study. Osteoporos Int. 2013;24(8):2231-2241. [29] AHN SH, LEE SH, KIM H, et al. Different Relationships Between Body Compositions and Bone Mineral Density According to Gender and Age in Korean Populations (KNHANES 2008-2010). J Clin Endocrinol Metab. 2014;99(10):3811-3820. [30] LIU-AMBROSE T, KRAVETSKY L, BAILEY D, et al. Change in Lean Body Mass Is a Major Determinant of Change in Areal Bone Mineral Density of the Proximal Femur: A 12-Year Observational Study. Calcif Tissue Int. 2006;79(3):145-151. [31] MARIN-MIO RV, MOREIRA LDF, CAMARGO M, et al. Lean mass as a determinant of bone mineral density of proximal femur in postmenopausal women. Arch Endocrinol Metab. 2018;62(4):431-437. [32] XIANG J, CHEN Y, WANG Y, et al. Lean Mass and Fat Mass as Mediators of the Relationship Between Physical Activity and Bone Mineral Density in Postmenopausal Women. J Women’s Health. 2017;26(5):461-466. [33] KAJI H. Linkage between muscle and bone. Current Opinion in Clinical Nutrition and Metabolic Care. 2013;16(3):272-277. [34] AHEDI H, AITKEN D, SCOTT D, et al. The Association Between Hip Muscle Cross-Sectional Area, Muscle Strength, and Bone Mineral Density. Calcif Tissue Int. 2014;95(1):64-72. [35] KARASIK D, COHEN-ZINDER M. The genetic pleiotropy of musculoskeletal aging. Front Physiol. 2012;3:303. [36] TERRACCIANO C, CELI M, LECCE D, et al. Differential features of muscle fiber atrophy in osteoporosis and osteoarthritis. Osteoporos Int. 2013; 24(3):1095-1100. [37] GENARO PS, PEREIRA GAP, PINHEIRO MM, et al. Influence of body composition on bone mass in postmenopausal osteoporotic women. Arch Gerontol Geriatr. 2010;51(3):295-298. [38] BLEICHER K, CUMMING RG, NAGANATHAN V, et al. The role of fat and lean mass in bone loss in older men: Findings from the CHAMP study. Bone. 2011;49(6):1299-1305. [39] 王春燕,何成奇.骨质疏松症治疗中的运动疗法[J].中国组织工程研究,2013,17(37):6657-6663. |
| [1] | Jiang Huanchang, Zhang Zhaofei, Liang De, Jiang Xiaobing, Yang Xiaodong, Liu Zhixiang. Comparison of advantages between unilateral multidirectional curved and straight vertebroplasty in the treatment of thoracolumbar osteoporotic vertebral compression fracture [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1407-1411. |
| [2] | Zhu Chan, Han Xuke, Yao Chengjiao, Zhou Qian, Zhang Qiang, Chen Qiu. Human salivary components and osteoporosis/osteopenia [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1439-1444. |
| [3] | Li Wei, Zhu Hanmin, Wang Xin, Gao Xue, Cui Jing, Liu Yuxin, Huang Shuming. Effect of Zuogui Wan on bone morphogenetic protein 2 signaling pathway in ovariectomized osteoporosis mice [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1173-1179. |
| [4] | Xiao Hao, Liu Jing, Zhou Jun. Research progress of pulsed electromagnetic field in the treatment of postmenopausal osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1266-1271. |
| [5] | Wu Min, Zhang Yeting, Wang Lu, Wang Junwei, Jin Yu, Shan Jixin, Bai Bingyi, Yuan Qiongjia. Effect of concurrent training sequences on body composition and hormone response: a Meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1305-1312. |
| [6] | Gao Yujin, Peng Shuanglin, Ma Zhichao, Lu Shi, Cao Huayue, Wang Lang, Xiao Jingang. Osteogenic ability of adipose stem cells in diabetic osteoporosis mice [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 999-1004. |
| [7] | Peng Kun. Improvement of the treatment effect of osteoporotic fractures: research status and strategy analysis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(6): 980-984. |
| [8] | Shen Song, Xu Bin. Diffuse distribution of bone cement in percutaneous vertebroplasty reduces the incidence of refracture of adjacent vertebral bodies [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 499-503. |
| [9] | Ou Liang, Kong Dezhong, Xu Daoqing, Ni Jing, Fu Xingqian, Huang Weichen. Comparative clinical efficacy of polymethyl methacrylate and self-solidifying calcium phosphate cement in vertebroplasty: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 649-656. |
| [10] | Zhong Yuanming, He Bingkun, Wu Zhuotan, Wu Sixian, Wan Tong, Zhong Xifeng. Meta-analysis of efficacy and safety of Jack kyphoplasty for osteoporotic vertebral compression fractures [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(3): 487-492. |
| [11] | Cai Feng, Yu Bo, Zeng Duo, Chen Qincan, Liao Qi. Cortical bone trajectory in elderly patients with osteoporosis of lumbar disease [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(3): 403-407. |
| [12] | Fan Danyang, Fu Runze, Mi Jiajing, Liu Chunyan. Expression and role of cannabinoid receptors during bone remodeling [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(2): 283-288. |
| [13] | Zhang Jinming, Tian Yingzhou, Zhao Ling, Xiong Lihua, Wang Qiuping, Song Wei, Wen Jianxuan. Icariin alleviates osteoporosis by promoting osteogenic differentiation of bone marrow mesenchymal stem cells in mice [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(19): 2991-2996. |
| [14] | Tang Zhihong, Duan Hao, Zhong Zongyu, Wang Weizhou, Chen Yongcheng, Li Xiaozhuang, Wang Yanghao, Liu Zhui, He Fei. Mechanism of mesenchymal stem cell transplantation in the treatment of osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(19): 3090-3094. |
| [15] | Huo Hua, Liu Guanjuan, Song Na, Zhou Qian, Cheng Yuting, Luo Shanshan, Liao Jian. Effects of zoledronic acid on alveolar bone metabolism and nucleotide-binding oligomerization domain-like receptor protein 3 inflammasome in the alveolar bone of ovariectomized osteoporosis rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(17): 2660-2666. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||