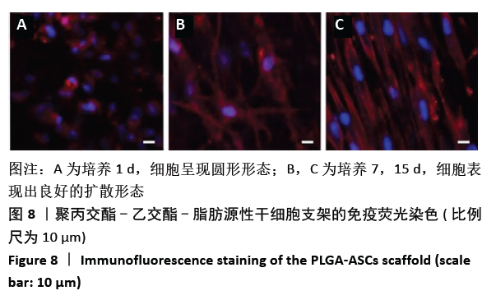
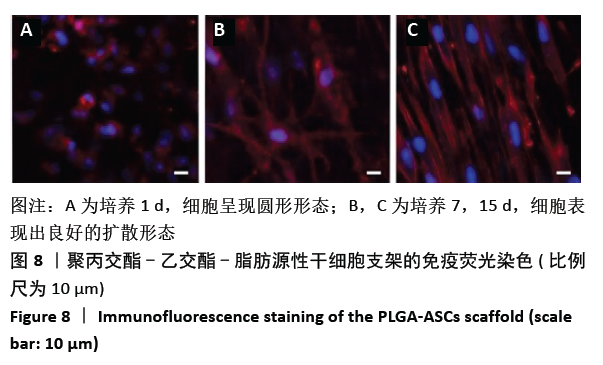
Chinese Journal of Tissue Engineering Research ›› 2021, Vol. 25 ›› Issue (34): 5484-5489.doi: 10.12307/2021.243
Previous Articles Next Articles
Poly(lactide-co-glycolide) scaffold combined with adipose derived stem cells in tissue-engineered urethral reconstruction
Li Zuowei, Fu Qiang, Song Lujie, Li Yonghui, Tian Binqiang
- Department of Urinary, Shanghai Jiao Tong University Affiliated Sixth People’s Hospital, Shanghai 200233, China
-
Received:2020-06-28Revised:2020-07-03Accepted:2020-08-15Online:2021-12-08Published:2021-07-27 -
Contact:Tian Binqiang, MD, Associate chief physician, Department of Urinary, Shanghai Jiao Tong University Affiliated Sixth People’s Hospital, Shanghai 200233, China -
About author:Li Zuowei, MD, Attending physician, Department of Urinary, Shanghai Jiao Tong University Affiliated Sixth People’s Hospital, Shanghai 200233, China -
Supported by:Science Research Fund of Shanghai Health and Family Planning Commission, No. 201440310 (to LYH)
CLC Number:
Cite this article
Li Zuowei, Fu Qiang, Song Lujie, Li Yonghui, Tian Binqiang. Poly(lactide-co-glycolide) scaffold combined with adipose derived stem cells in tissue-engineered urethral reconstruction[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(34): 5484-5489.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
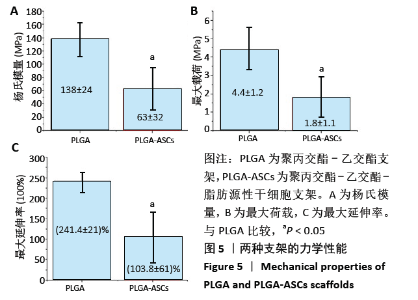
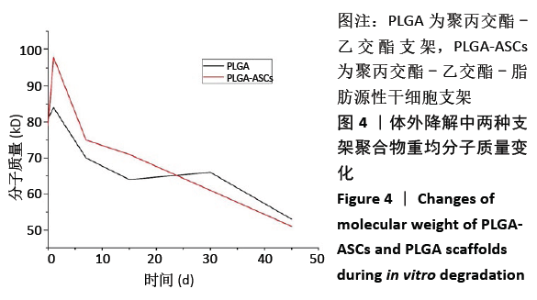
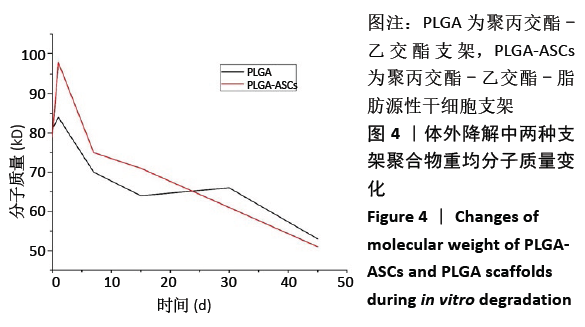
2.2.3 机械性能 支架的杨氏模量、最大荷载和最大伸长如图5所示,PLGA支架的力学性能测试值大于PLGA-ASCs(P < 0.05)。结果表明与PLGA支架相比,PLGA-ASCs支架的力学性能变化更大,这意味着与细胞整合后支架的机械性能降低,但PLGA- ASCs支架的机械强度大于50 MPa,仍足以支持干细胞生长。 2.2.4 残留溶剂含量 在热重分析中,温度<200 ℃时的质量损失通常表示有机溶剂或水的挥发。PLGA-ASCs支架在暴露于该温度期间的质量损失为2.88%。根据该测量可以假设PLGA-ASCs支架中存在(0.35±0.76) mg(投入的总质量 12 mg×2.88%为损失的质量,损失的部分多为溶剂和水分)溶剂和/或湿度。"
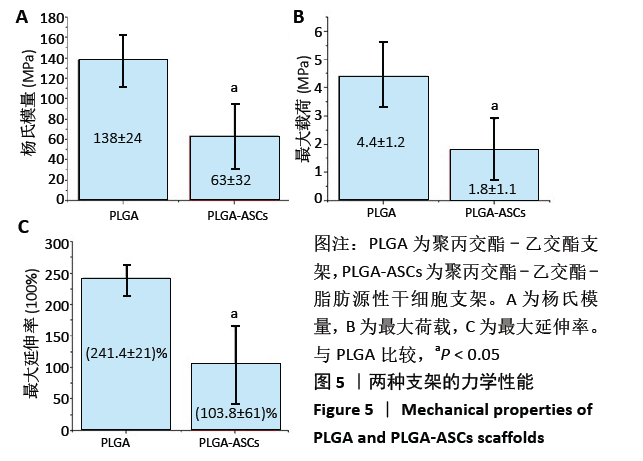
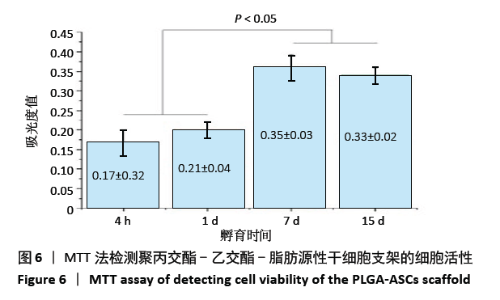
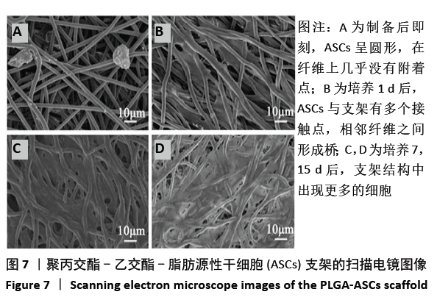
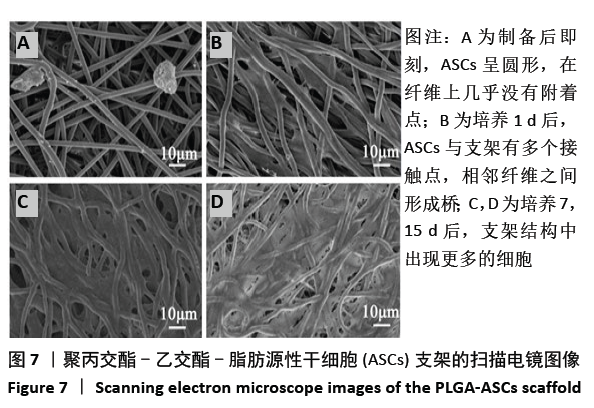
2.3 PLGA-ASCs支架的生物学特性 2.3.1 初始细胞数与细胞增殖 生物电喷雾(与纤维电纺丝无关)后细胞活力为(92.0±4.2)%。同时,经生物电喷雾和静电纺丝后的细胞存活率为(87.0±4.4)%。通过比较样品和标准品的吸光度值,使用MTT实验评估PLGA-ASCs支架结构中ASCs的初始密度(图6),纤维间融合的活细胞数平均为(5.123±1.012)×103/mm3。 PLGA-ASCs支架的总体积约为270 mm3,因此PLGA-ASCs总体积中的细胞数约为1.36×106。如前所述,对于生物电喷雾过程,使用2.60 mL/h流速和细胞浓度7.5×109 L-1的ASCs悬浮液,因此每个PLGA-ASCs支架的制备需0.65 mL的细胞悬液,对应于约4.88×106个细胞。 通过比较生物电喷雾的细胞数和PLGA-ASCs支架中的细胞数,评估支架的细胞整合效率。用该方法计算的细胞整合效率约为28%。PLGA-ASCs支架中ASCs的增殖如图6所示,在培养的第1-7天,活细胞数量显著增加(P < 0.001),同时在第7-15天细胞数量无明显变化。"
| [1] HAO Z, SONG Z, HUANG J, et al. The scaffold microenvironment for stem cell based bone tissue engineering. Biomater Sci. 2017;5(8):1382-1392. [2] ADEBIYI AA, TASLIM ME, CRAWFORD KD. The use of computational fluid dynamic models for the optimization of cell seeding processes. Biomaterials. 2011;32(34): 8753-8770. [3] KESTI M, MÜLLER M, BECHER J, et al. A versatile bio-ink for three-dimensional printing of cellular scaffolds based on thermally and photo-triggered tandem gelation. Acta Biomater. 2015;11:162-172. [4] LEE MC, SEONWOO H, GARG P, et al. Development of a bio-electrospray system for cell and non-viral gene delivery. Rsc Adv. 2018;8(12):6452-6459. [5] HONG N, YANG GH, LEE JH, et al. 3D bioprinting and its in vivo applications. J Biomed Mater Res B. 2018;106(1):444-459. [6] ASGHARI F, SAMIEI M, ADIBKIA K, et al. Biodegradable and biocompatible polymers for tissue engineering application: a review. Artif Cells Nanomed Biotechnol. 2017;45(2):185-192. [7] MARTINS C, SOUSA F, ARAÚJO F, et al. Functionalizing PLGA and PLGA derivatives for drug delivery and tissue regeneration applications. Adv Healthc Mater. 2018; 7(1):1701035. [8] NI R, MUENSTER U, ZHAO J, et al. Exploring polyvinylpyrrolidone in the engineering of large porous PLGA microparticles via single emulsion method with tunable sustained release in the lung: in vitro and in vivo characterization. J Control Release. 2017;249:11-22. [9] PALMA E, PASQUA A, GAGLIARDI A, et al. Antileishmanial activity of amphotericin B-loaded-PLGA nanoparticles: an overview. Materials. 2018;11(7):1167. [10] LIU J, HUANG J, LIN T, et al. Cell-to-cell contact induces human adipose tissue-derived stromal cells to differentiate into urothelium-like cells in vitro. Biochem Bioph Res Co. 2009;390(3):931-936. [11] ZHAO Z, YU H, FAN C, et al. Differentiate into urothelium and smooth muscle cells from adipose tissue-derived stem cells for ureter reconstruction in a rabbit model. Am J Transl Res. 2016;8(9):3757. [12] Zuk PA, Zhu M, Ashjian P, et al. Human adipose tissue is a source of multipotent stem cells. Mol Bio Cell. 2002;13(12):4279-4295. [13] ORABI H, ABOUSHWAREB T, ZHANG Y, et al. Cell-seeded tubularized scaffolds for reconstruction of long urethral defects: a preclinical study. Eur Urol. 2013; 63(3):531-538. [14] MOREY AF, WATKIN N, SHENFELD O, et al. SIU/ICUD consultation on urethral strictures: anterior urethra–primary anastomosis. Urology. 2014;83(3):S23-S26. [15] HAMPSON LA, MCANINCH JW, BREYER BN. Male urethral strictures and their management. Nat Rev Urol. 2014;11(1):43. [16] BENMERZOUG S, QUESNIAUX VFJ. Bioengineered 3D models for studying human cell–tuberculosis interactions. Trends Microbiol. 2017;25(4):245-246. [17] RIBEIRO-FILHO LA, SIEVERT KD. Acellular matrix in urethral reconstruction. Adv Drug Deliv Rev. 2015;82:38-46. [18] FENG C, XU Y, FU Q, et al. Reconstruction of three-dimensional neourethra using lingual keratinocytes and corporal smooth muscle cells seeded acellular corporal spongiosum. Tissue Eng Part A. 2011;17(23-24):3011-3019. [19] GUO H, SA Y, HUANG J, et al. Urethral reconstruction with small intestinal submucosa seeded with oral keratinocytes and TIMP-1 siRNA transfected fibroblasts in a rabbit model. Urol int. 2016;96(2):223-230. [20] ZOU XH, ZHI YL, CHEN X, et al. Mesenchymal stem cell seeded knitted silk sling for the treatment of stress urinary incontinence. Biomaterials. 2010;31(18):4872-4879. [21] Shi JG, Fu WJ, Wang XX, et al. Trans-differentiation of human adipose-derived stem cells into urothelial cells: potential for urinary tract tissue engineering. Cell Tissue Res. 2012;347(3):737-746. [22] ZANATTA G, STEFFENS D, BRAGHIROLLI DI, et al. Viability of mesenchymal stem cells during electrospinning. Braz J Med Biol Res. 2012;45(2):125-130. [23] HSIA HC, NAIR MR, MINTZ RC, et al. The fiber diameter of synthetic bioresorbable extracellular matrix influences human fibroblast morphology and fibronectin matrix assembly. Plast Reconstr Surg. 2011;127(6):2312-2320. [24] ZANATTA G, RUDISILE M, CAMASSOLA M, et al. Mesenchymal stem cell adherence on poly(D, L-lactide-co-glycolide) nanofibers scaffold is integrin-beta 1 receptor dependent. J Biomed Nanotechnol. 2012;8(2):211-218. [25] STANKUS JJ, GUAN J, FUJIMOTO K, et al. Micro-integrating smooth muscle cells into a biodegradable, elastomeric fiber matrix. Biomaterials. 2006;27(5):735-744. [26] CARVALHO MS, SILVA JC, UDANGAWA RN, et al. Co-culture cell-derived extracellular matrix loaded electrospun microfibrous scaffolds for bone tissue engineering. Mater Sci Eng C. 2019;99:479-490. [27] SANJAIRAJ V, KANNAN S, CAO T, et al. 3D-printed PCL/PPy conductive scaffolds as three-dimensional porous nerve guide conduits (ngcs) for peripheral nerve injury repair. Front Bioeng Biotechnol. 2019;7:266. [28] KANCZLER JM, WELLS JA, GIBBS DMR, et al. Bone tissue engineering and bone regeneration//Principles of Tissue Engineering. Academic Press. 2020:917-935. [29] LI S. Hydrolytic degradation characteristics of aliphatic polyesters derived from lactic and glycolic acids. J Biomed Mater Res. 1999;48(3):342-353. [30] ZHANG B, ZHANG P, WANG Z, et al. Tissue-engineered composite scaffold of poly (lactide-co-glycolide) and hydroxyapatite nanoparticles seeded with autologous mesenchymal stem cells for bone regeneration. J Zhejiang Univ Sci B. 2017;18(11):963-976. |
| [1] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [2] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [3] | Jiang Hongying, Zhu Liang, Yu Xi, Huang Jing, Xiang Xiaona, Lan Zhengyan, He Hongchen. Effect of platelet-rich plasma on pressure ulcers after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1149-1153. |
| [4] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [5] | Guan Qian, Luan Zuo, Ye Dou, Yang Yinxiang, Wang Zhaoyan, Wang Qian, Yao Ruiqin. Morphological changes in human oligodendrocyte progenitor cells during passage [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1045-1049. |
| [6] | Wang Zhengdong, Huang Na, Chen Jingxian, Zheng Zuobing, Hu Xinyu, Li Mei, Su Xiao, Su Xuesen, Yan Nan. Inhibitory effects of sodium butyrate on microglial activation and expression of inflammatory factors induced by fluorosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1075-1080. |
| [7] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [8] | Liao Chengcheng, An Jiaxing, Tan Zhangxue, Wang Qian, Liu Jianguo. Therapeutic target and application prospects of oral squamous cell carcinoma stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1096-1103. |
| [9] | Xie Wenjia, Xia Tianjiao, Zhou Qingyun, Liu Yujia, Gu Xiaoping. Role of microglia-mediated neuronal injury in neurodegenerative diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1109-1115. |
| [10] | Li Shanshan, Guo Xiaoxiao, You Ran, Yang Xiufen, Zhao Lu, Chen Xi, Wang Yanling. Photoreceptor cell replacement therapy for retinal degeneration diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1116-1121. |
| [11] | Jiao Hui, Zhang Yining, Song Yuqing, Lin Yu, Wang Xiuli. Advances in research and application of breast cancer organoids [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1122-1128. |
| [12] | Wang Shiqi, Zhang Jinsheng. Effects of Chinese medicine on proliferation, differentiation and aging of bone marrow mesenchymal stem cells regulating ischemia-hypoxia microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1129-1134. |
| [13] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [14] | Kong Desheng, He Jingjing, Feng Baofeng, Guo Ruiyun, Asiamah Ernest Amponsah, Lü Fei, Zhang Shuhan, Zhang Xiaolin, Ma Jun, Cui Huixian. Efficacy of mesenchymal stem cells in the spinal cord injury of large animal models: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1142-1148. |
| [15] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||