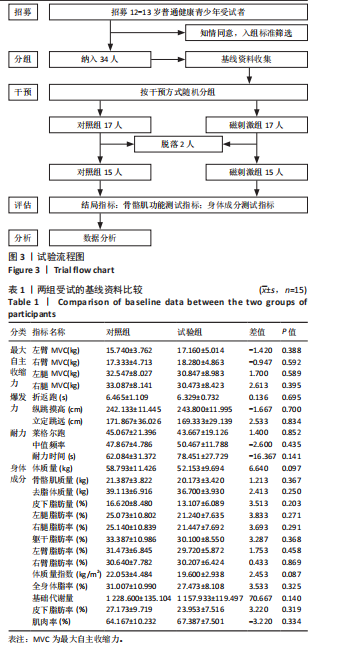
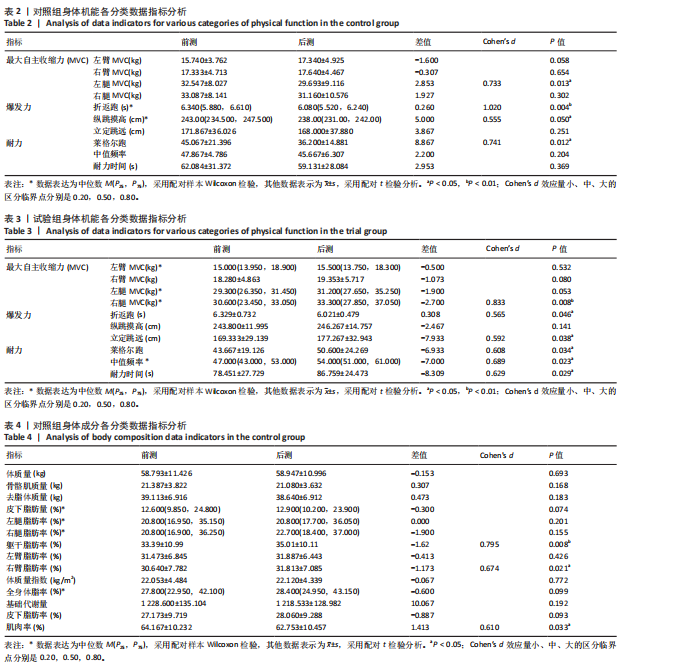
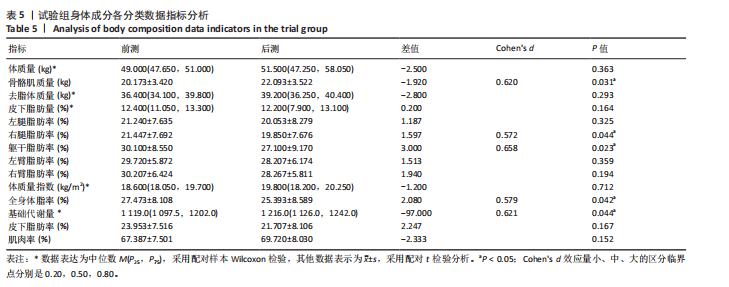
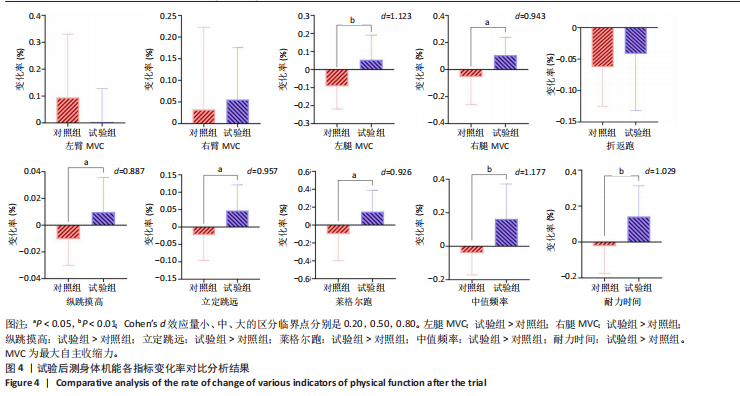
[1] CHENG X, GUO Q, JU L, et al. Association between sedentary behavior, screen time and metabolic syndrome among Chinese children and adolescents. BMC Public Health. 2024;24(1):1715.
[2] HU SS, Writing Committee of the Report on Cardiovascular Health and Diseases in China. Influencing Factors on Cardiovascular Health in China. J Geriatr Cardiol. 20248;21(1):4-33.
[3] 彭惠媛,李佳川,汤庆华.青少年学生体育锻炼坚持性现状调查与分析[J].体育科技文献通报,2020,28(1):37-40.
[4] YAP JLY, TAI YK, FRÖHLICH J, et al. Ambient and supplemental magnetic fields promote myogenesis via a TRPC1-mitochondrial axis: evidence of a magnetic mitohormetic mechanism. FASEB J. 2019;33(11):12853-12872.
[5] FRANCO-OBREGÓN A. Harmonizing Magnetic Mitohormetic Regenerative Strategies: Developmental Implications of a Calcium-Mitochondrial Axis Invoked by Magnetic Field Exposure. Bioengineering (Basel). 2023;10(10):1176.
[6] 厉中山,白石,王春露,等. 低频脉冲磁场诱导TRPC1技术对力量素质影响及训练应用展望[C].2022年全国第七届“中国体能训练科学大会”,2022: 1642-1648.
[7] 厉中山,王春露,刘洁,等. 短期低频脉冲磁场诱导经典瞬时感受器电位通道1对肱二头肌最大自主收缩力与力量耐力的影响[J]. 中国组织工程研究 2023,27(11):1796–1804.
[8] STEPHENSON MC, KRISHNA L, PANNIR SELVAN RM, et al. Magnetic field therapy enhances muscle mitochondrial bioenergetics and attenuates systemic ceramide levels following ACL reconstruction: Southeast Asian randomized-controlled pilot trial. J Orthop Translat. 2022;35:99-112.
[9] KIVILUOTO S, DECUYPERE JP, DE SMEDT H, et al. STIM1 as a key regulator for Ca2+ homeostasis in skeletal-muscle development and function. Skelet Muscle. 2011;1(1):16.
[10] SABOURIN J, COGNARD C, CONSTANTIN B. Regulation by Scaffolding Proteins of Canonical Transient Receptor Potential Channels in Striated Muscle. J Muscle Res Cell Motil. 2009;30(7–8):289-297.
[11] ZHANG BT, YEUNG SS, CHEUNG KK, et al. Adaptive Responses of Trpc1 and Trpc3 during Skeletal Muscle Atrophy and Regrowth. Muscle Nerve. 2014;49(5):691-699.
[12] GIROLAMI B, SERANO M, DI FONSO A, et al. Searching for Mechanisms Underlying the Assembly of Calcium Entry Units: The Role of Temperature and pH. Int J Mol Sci. 2023;24(6):5328.
[13] 张星杰,周家颖.青少年身体素质发展“敏感期”特性与训练干预“溢出效应”机理研究[J].西安体育学院学报,2024,41(2):169-175.
[14] THYAGARAJAN B, FOSTER MT. Beiging of white adipose tissue as a therapeutic strategy for weight loss in humans. Horm Mol Biol Clin Investig. 2017;31(2):/j/hmbci.2017.31.issue-2/hmbci-2017-0016/hmbci-2017-0016.xml.
[15] 魏佳,李博,冯连世,等.血流限制训练的方法学因素及潜在安全性问题[J].中国体育科技,2019,55(3):3-12.
[16] RAMSEY KA, ROJER AGM, D’ANDREA L, et al. The Association of Objectively Measured Physical Activity and Sedentary Behavior with Skeletal Muscle Strength and Muscle Power in Older Adults: A Systematic Review and Meta-Analysis. Ageing Res Rev. 2021;67:101266.
[17] PARK JH, MOON JH, KIM HJ, et al. Sedentary Lifestyle: Overview of Updated Evidence of Potential Health Risks. Korean J Fam Med. 2020;41(6):365-373.
[18] MEAR E, GLADWELL VF, PETHICK J. The Effect of Breaking up Sedentary Time with Calisthenics on Neuromuscular Function: A Preliminary Study. Int J Environ Res Public Health. 2022;19(21):14597.
[19] 厉中山,白石,刘洁,等.短期低频脉冲磁场诱导经典瞬时感受器电位通道1对局部肌肉肌力提升后的保持与衰减变化轨迹[J].中国组织工程研究, 2023,27(23):3721-3727.
[20] FREY N, FRANK D,LIPPL S, et al. Calsarcin-2 Deficiency Increases Exercise Capacity in Mice through Calcineurin/NFAT Activation. J Clin Invest. 2008;118(11): 3598-3608.
[21] ZHANG BT, YEUNG SS, CHEUNG KK, et al. Adaptive Responses of TRPC1 and TRPC3 during Skeletal Muscle Atrophy and Regrowth. Muscle Nerve. 2014;49(5): 691-699.
[22] 周加仙.敏感期的神经可塑性机制研究[J].华东师范大学学报(教育科学版), 2010,28(3):50-53+67.
[23] MIRZOEV TM. Mechanotransduction for Muscle Protein Synthesis via Mechanically Activated Ion Channels. Life (Basel Switz). 2023;13(2):341.
[24] 李伟,厉中山,白石.TRPC1钙离子通道在骨骼肌收缩功能中的作用及磁诱导激活效应在训练中的应用探索方向[J].体育科技文献通报,2023,31(2): 257-261.
[25] XIA L, CHEUNG KK, YEUNG SS, et al. The Involvement of Transient Receptor Potential Canonical Type 1 in Skeletal Muscle Regrowth after Unloading-Induced Atrophy. J Physiol. 2016;594(11):3111-3126.
[26] ZANOU N, SCHAKMAN O, LOUIS P, et al. Trpc1 ion channel modulates phosphatidylinositol 3-kinase/Akt pathway during myoblast differentiation and muscle regeneration. J Biol Chem. 2012;287(18):14524-14534.
[27] FRANCO-OBREGÓN A, TAI YK, WU KY, et al. The Developmental Implications of Muscle-Targeted Magnetic Mitohormesis: A Human Health and Longevity Perspective. Bioengineering (Basel). 2023;10(8):956.
[28] FRAYSSE B, DESAPHY JF, PIERNO S, et al. Decrease in resting calcium and calcium entry associated with slow-to-fast transition in unloaded rat soleus muscle. FASEB J. 2003;17(13):1916-1918.
[29] ZANOU N, SHAPOVALOV G, LOUIS M, et al. Role of TRPC1 channel in skeletal muscle function. Am J Physiol Cell Physiol. 2010;298(1):C149-C162.
[30] MORALES S, DIEZ A, PUYET A, et al. Calcium controls smooth muscle TRPC gene transcription via the CaMK/calcineurin-dependent pathways. Am J Physiol Cell Physiol. 2007;292(1):C553-C563.
[31] LIN J, WU H, TARR P, et al. Transcriptional Co-Activator PGC-1 Alpha Drives the Formation of Slow-Twitch Muscle Fibres. Nature. 2002;418(6899):797-801.
[32] LOUZADA RA, BOUVIERE J, MATTA LP, et al. Redox Signaling in Widespread Health Benefits of Exercise. Antioxid Redox Signal. 2020. doi: 10.1089/ars.2019.7949.
[33] TAI YK, NG C, PURNAMAWATI K, et al. Magnetic fields modulate metabolism and gut microbiome in correlation with Pgc-1α expression: Follow-up to an in vitro magnetic mitohormetic study. FASEB J. 2020;34(8):11143-11167.
[34] CALVO JA, DANIELS TG, WANG X, et al. Muscle-specific expression of PPARgamma coactivator-1alpha improves exercise performance and increases peak oxygen uptake. J Appl Physiol (1985). 2008;104(5):1304-1312.
[35] EKMARK M, GRØNEVIK E, SCHJERLING P, et al. Myogenin induces higher oxidative capacity in pre-existing mouse muscle fibres after somatic DNA transfer. J Physiol. 2003;548(Pt 1):259-269.
[36] VAN WESSEL T, DE HAAN A, VAN DER LAARSE WJ, et al. The muscle fiber type-fiber size paradox: hypertrophy or oxidative metabolism? Eur J Appl Physiol. 2010;110(4):665-694.
[37] HUGHES DC, ELLEFSEN S, BAAR K. Adaptations to Endurance and Strength Training. Cold Spring Harb Perspect Med. 2018;8(6):a029769.
[38] BERTERO E, POPOIU TA, MAACK C. Mitochondrial Calcium in Cardiac Ischemia/Reperfusion Injury and Cardioprotection. Basic Res. Cardiol. 2024;119(4):569,585.
[39] 杨景慧,王海霞,耿喜臣,等. 基本空战动作“检查6点钟”姿势腰背训练方案的表面肌电图特征研究[J]. 航天医学与医学工程,2020,33(3):196-202.
[40] WAGATSUMA A, SAKUMA K. Mitochondria as a potential regulator of myogenesis. ScientificWorldJournal. 2013;2013:593267.
[41] 宋淑华,熊开宇.离子浓度改变对骨骼肌膜电位和骨骼肌疲劳的研究进展[J].北京体育大学学报,2009,32(1):84-87.
[42] 厉中山,包义君,刘洁,等.低频脉冲磁场诱导TRPC1改善COVID-19患者康复期下肢的肌肉无力症状[J].中国组织工程研究,2024,28(16):2605-2612.
[43] 洪金涛,王晶晶,李岩松,等.久坐与社区老年人下肢肌力:跌倒恐惧与年龄的中介和调节作用[J].中国组织工程研究,2025,29(17):3566-3571.
[44] 苍穹.老年人下棋莫久坐[J].家庭科技,2016(10):25.
[45] 黄秋玉.不同强度功率自行车运动对久坐大学生心肺耐力、下肢力量和心理健康的影响[D].北京: 首都体育学院,2023.
[46] WONG CJK, TAI YK, YAP JLY, et al. Brief exposure to directionally-specific pulsed electromagnetic fields stimulates extracellular vesicle release and is antagonized by streptomycin: A potential regenerative medicine and food industry paradigm. Biomaterials. 2022;287:121658.
[47] WANG FS, WU RW, CHEN YS, et al. Biophysical Modulation of the Mitochondrial Metabolism and Redox in Bone Homeostasis and Osteoporosis: How Biophysics Converts into Bioenergetics. Antioxidants (Basel). 2021;10(9):1394.
[48] LAWLER JM, RODRIGUEZ DA, HORD JM. Mitochondria in the middle: exercise preconditioning protection of striated muscle. J Physiol. 2016;594(18):5161-5183.
[49] HÉNIQUE C, MANSOURI A, VAVROVA E, et al. Increasing mitochondrial muscle fatty acid oxidation induces skeletal muscle remodeling toward an oxidative phenotype. FASEB J. 2015;29(6):2473-2483.
[50] SUPRUNIUK E, MIKŁOSZ A, CHABOWSKI A. The Implication of PGC-1α on Fatty Acid Transport across Plasma and Mitochondrial Membranes in the Insulin Sensitive Tissues. Front Physiol. 2017;8:923.
[51] PEDERSEN BK, FEBBRAIO MA. Muscles, exercise and obesity: skeletal muscle as a secretory organ. Nat Rev Endocrinol. 2012;8(8):457-465..
[52] GONZALEZ-GIL AM, ELIZONDO-MONTEMAYOR L. The Role of Exercise in the Interplay between Myokines, Hepatokines, Osteokines, Adipokines, and Modulation of Inflammation for Energy Substrate Redistribution and Fat Mass Loss: A Review. Nutrients. 2020;12(6):1899.
[53] LIU S, CUI F, NING K, et al. Role of irisin in physiology and pathology. Front Endocrinol (Lausanne). 2022;13:962968.
[54] MULYA A, KIRWAN JP. Brown and Beige Adipose Tissue: Therapy for Obesity and Its Comorbidities? Endocrinol Metab Clin North Am. 2016;45(3):605-621.
[55] GÓRNICKA M, HAMULKA J, WADOLOWSKA L, et al. Activity-Inactivity Patterns, Screen Time, and Physical Activity: The Association with Overweight, Central Obesity and Muscle Strength in Polish Teenagers. Report from the ABC of Healthy Eating Study. Int J Environ Res Public Health. 2020;17(21):7842.
[56] KRAUSE MP, MILNE KJ, HAWKE TJ. Adiponectin-Consideration for its Role in Skeletal Muscle Health. Int J Mol Sci. 2019;20(7):1528.
[57] WALOWSKI CO, HERPICH C, ENDERLE J, et al. Determinants of bone mass in older adults with normal- and overweight derived from the crosstalk with muscle and adipose tissue. Sci Rep. 2023;13(1):5030.
[58] PARATE D, KADIR ND, CELIK C, et al. Pulsed electromagnetic fields potentiate the paracrine function of mesenchymal stem cells for cartilage regeneration. Stem Cell Res Ther. 2020;11(1):46.
|