中国组织工程研究 ›› 2026, Vol. 30 ›› Issue (34): 9008-9016.doi: 10.12307/2026.857
• 组织构建综述 tissue construction review • 上一篇 下一篇
脑类器官区域特异性在缺血性脑卒中建模和药物开发中的应用
李 瀛1,王全玉1,冯崇义2,常 顺1,杨春爱3
- 1云南省第一人民医院/昆明理工大学附属医院,云南省昆明市 650032;2云南省迪庆州医院神经外科,云南省香格里拉市 674400;3云南大学附属医院,云南省昆明市 650021
-
收稿日期:2025-10-29修回日期:2026-01-17出版日期:2026-12-08发布日期:2026-04-14 -
通讯作者:常顺,博士,副主任医师,云南省第一人民医院/昆明理工大学附属医院神经外科,云南省昆明市 650032 并列通讯作者:杨春爱,硕士,副主任医师,云南大学附属医院,云南省昆明市 650021 -
作者简介:第一作者:李瀛,男,1972年生,副主任医师,主要从事神经系统功能障碍的中西医结合诊治研究。 -
基金资助:昆明理工大学医学联合专项-面上项目(KUST-KH2023032Y),项目负责人:常顺;云南省科技厅科技项目-面上项目(202401AT070057),项目负责人:常顺;云南省第一人民医院临床开放课题(2024SNKFKT-03),项目负责人:王全玉;昆明医科大学联合专项-面上项目(202301AY070001-213),项目负责人:杨春爱
Regional specificity of brain organoids and their application in ischemic stroke modeling and drug development
- 1The First People’s Hospital of Yunnan Province/Affiliated Hospital of Kunming University of Science and Technology, Kunming 650032, Yunnan Province, China; 2Department of Neurosurgery, Diqing Prefecture Hospital, Shangri-La 674400, Yunnan Province, China; 3Affiliated Hospital of Yunnan University, Kunming 650021, Yunnan Province, China
-
Received:2025-10-29Revised:2026-01-17Online:2026-12-08Published:2026-04-14 -
Contact:Chang Shun, PhD, Associate chief physician, Affiliated Hospital of Yunnan University, Kunming 650021, Yunnan Province, China -
About author:Li Ying, Associate chief physician, The First People’s Hospital of Yunnan Province/Affiliated Hospital of Kunming University of Science and Technology, Kunming 650032, Yunnan Province, China -
Supported by:Kunming University of Science and Technology Medical Joint Special Project - General Program, No. KUST-KH2023032Y (to CS); Yunnan Provincial Department of Science and Technology Project - General Program, No. 202401AT070057 (to CS); Yunnan Provincial First People’s Hospital Clinical Open Project, No. 2024SNKFKT-03 (to WQY); Kunming Medical University Joint Special Project - General Program, No. 202301AY070001-213 (to YCA)
摘要:
文题释义:
类器官:由干细胞在体外基质胶的3D环境培养形成的、具有自我更新和自我组织能力的细胞簇,能模拟体内器官的结构和功能。常见的细胞来源包括多能干细胞和成体干细胞。目前,科学家已成功培育出脑、肠、胃、肝、心、肺、肾、视网膜等多种类器官,主要应用于疾病建模、药物筛选与毒性测试、再生医学、发育生物学研究、个性化医疗等方面。
脑卒中:是一种由于脑血管突然破裂或因血管阻塞导致血液不能流入大脑,进而引起脑组织损伤的急性脑血管疾病。脑卒中类型主要分为缺血性脑卒中和出血性脑卒中,其中缺血性脑卒中占所有脑卒中的75%。缺血性脑卒中以溶栓、神经保护剂和抗血小板药物应用等治疗手段为主,出血性脑卒中的治疗以止血、降压为主。
背景:脑类器官技术构建及在缺血性脑卒中的应用是近年新型的研究热点。啮齿动物的大脑解剖结构和功能组织与人类大脑存在明显差异,决定了它们不能完全模仿人类大脑的生理、病理和解剖特征。同时,在临床上获取患者脑组织样本存在伦理问题,因此脑类器官在缺血性脑卒中研究中的重要意义在于可替代临床脑组织和动物模型构建更贴合人脑组织的体外研究模型,在疾病机制建模和药物开发中具有重要价值。
目的:对现阶段脑类器官的区域特异性研究进行综述总结,为脑类器官在缺血性脑卒中建模和药物开发中的应用提供新的技术手段和研究策略。
方法:通过中国知网和PubMed数据库进行有关类器官及缺血性脑卒中的文献检索,检索时限为各数据库建库至2025年5月,中文检索词为“缺血性脑卒中、脑卒中、类器官、缺血再灌注损伤、糖氧剥夺/复氧、前脑类器官、海马类器官、丘脑类器官、中脑类器官”,英文检索词为“ischemic stroke,stroke,organoids,oxygen-glucose deprivation/reoxygenation,middle cerebral artery occlusion,forebrain organoids,hippocampal organoids,thalamic organoids,midbrain organoids”。所有检索文献为研究原著和相关综述,最终共筛选98篇文献进行分析总结。
结果与结论:①背侧前脑类器官模拟皮质神经上皮,分化成星形胶质细胞、兴奋性神经元和少突胶质细胞等神经细胞,腹侧前脑类器官模仿神经节隆起,发育为基底神经节和纹状体,表现出广泛的神经元迁移并保持γ-氨基丁酸能特性;②人诱导多能干细胞产生的海马类器官移植可与小鼠海马体神经回路功能整合,接受兴奋性输入,形成具有突触连接的神经元网格;③丘脑/下丘脑类器官模拟人类下丘脑弓状核的发育、细胞多样性并探究多巴胺能神经元特异性;④中脑类器官广泛应用于帕金森病的建模、基因编辑和药物开发的研究中;⑤糖氧剥夺可诱导脑类器官体外模拟缺血性脑卒中,进而通过凋亡、坏死性凋亡、自噬和铁死亡等死亡途径参与神经损伤;糖氧剥夺处理后的脑类器官基因表达谱发生显著变化;应用鼠尾草酸等神经保护药物或在大脑中动脉闭塞大鼠体内移植脑类器官可改善大鼠的神经功能损伤,减少梗死体积,在移植再生医学领域发挥重要作用。
https://orcid.org/0009-0006-2398-0189(李瀛)
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
中图分类号:
引用本文
李 瀛, 王全玉, 冯崇义, 常 顺, 杨春爱. 脑类器官区域特异性在缺血性脑卒中建模和药物开发中的应用[J]. 中国组织工程研究, 2026, 30(34): 9008-9016.


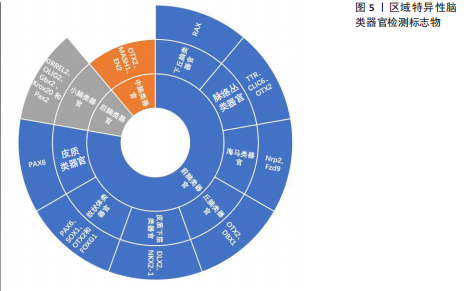
诱导剂的类型和组合、治疗时间和浓度,可以产生模拟不同大脑区域的类神经器官。非引导方法是利用干细胞固有的自组织能力,使用不含模式因子的最小分化培养基制备脑类器官,用于制备具有多脑区域多样性特征的全脑类器官,包括整个脑组织的发育,以及各个大脑区域的结构和生理特征[21]。而引导方法生成的区域特异性脑类器官具有更好的均匀性和可重复性[22]。引导脑类器官生成的方法是由Eiraku小组首创的[23],开发了基于EB样聚集体无血清培养的3D分化方案[24]。他们利用高效的三维聚集培养证明了胚胎干细胞可自组织形成顶端基部极化的皮质组织。所生成的皮质神经元是功能性的、可移植的,并且能够在体内和体外形成适当的远程连接。通过成纤维细胞生长因子、WNT和骨形态发生蛋白等分泌的模式因子,可以选择性地控制生成的白质组织的区域特征(进入嗅球、吻侧和尾侧皮质、下丘脑和脉络丛),不同皮质神经元的体内模拟出生顺序允许通过定时诱导细胞周期退出选择性地产生特定层特异性神经元。Matrigel基质诱导正确的极性刺激,促进大型、顶基神经上皮芽体的复杂生长。这些芽体在不断扩展中获得各种脑部特性和形成类似于脑室的充满液体的腔隙。结合干细胞自身的自组织能力,使得单个组织内部形成了多种大脑区域,包括后脑、中脑、前脑,甚至还有视网膜组织的特征[25]。前脑包括端脑和间脑,内侧皮质和海马类器官模型分配给背侧前脑类器官,纹状体类器官模型和神经节隆起类器官模型分配给腹侧前脑(皮质下)类器官。这些大脑结构起源于人类神经发育过程中的端脑。间脑类器官包括丘脑、下丘脑和垂体类器官,而脑桥、延髓和小脑则组成后脑结构[26](图5)。
2.1.1 前脑类器官
(1)背侧前脑类器官:区域特异性的脑类器官背侧前脑类器官(苍白球,大脑皮质)可模拟皮质神经上皮,可通过抑制WNT信号传导进行诱导,WNT抑制剂使用后可清晰显示大脑皮质的特征性分层结构,以及构成该结构的各种细胞(室管膜放射状胶质细胞、外侧放射状胶质细胞、神经祖细胞、兴奋性神经元[27]、星形胶质细胞和少突胶质细胞[28])。使用源自多能干细胞的人类大脑皮质球体在三维细胞结构中生成星形胶质细胞,发现大脑皮质球体衍生的胶质细胞与人类胎儿的原始星形胶质细胞非常相似,并且随着时间的推移,它们从主要的胎儿星形胶质细胞状态转变为日益成熟的星形胶质细胞状态,同时转录变化伴随着吞噬能力的改变和对神经元钙信号的影响[29]。在人类诱导多能干细胞衍生的皮质球体中生成了少突胶质前体细胞和髓鞘少突胶质细胞。人诱导多能干细胞分化产生包含少突胶质细胞以及神经元
和星形胶质细胞的三维神经球体,称为人少突胶质细胞球体,它衍生的少突胶质细胞谱系的发育阶段类似于原代人少突胶质细胞,并且谱系细胞的迁移及其对溶卵磷脂暴露的易感性可以通过实时成像捕获[30]。将在WNT抑制剂存在下的小鼠胚层干细胞作为体外定向分化研究的起点,快速、稳健地定向分化为确定的内胚层和前脑类器官[31]。
海马体作为神经系统的一部分,起源于神经外胚层谱系。在原肠胚形成过程中,形成3个胚层——内胚层、中胚层和外胚层。从外胚层诱导神经谱系,产生神经板,然后在神经形成的过程中向内折叠形成神经管[32]。海马体是大脑中仅有的几个在出生后继续产生神经元的区域之一,这与大脑中被认为不可再生的大部分区域形成了鲜明对比[33]。海马体对于塑造人类的体验至关重要,它通过在学习和记忆过程中的作用调节认知和情绪来发挥作用[34]。人诱导多能干细胞形成的海马类器官首先通过双Sma和Mad相关蛋白抑制(转化生长因子β/WNT)诱导前神经上皮细胞,之后通过骨形态发生蛋白4和CHIR诱导后背区表征,最终通过神经元成熟阶段产生[35]。人胚胎干细胞衍生的海马神经祖细胞产生海马齿状回颗粒神经元,将海马神经祖细胞移植到出生后第12天的非肥胖型重性糖尿病合并免疫缺陷小鼠的海马体中,移植6周后观察到绿色荧光蛋白阳性细胞广泛整合到海马齿状回的颗粒细胞层中,体细胞大小、总树突长度和分支明显增加,瞬时钠向内电流和持续钾向外电流,自发的突触后电流,并在穿孔路径刺激后诱导兴奋性突触后电流,表明移植细胞与局部神经回路的功能整合[36]。构建产生多种CA3锥体神经元亚型的人诱导多能干细胞类器官,将hCA3移植到小鼠海马体中,与邻近的小鼠神经元相比,hCA3表现出相似的膜特性和兴奋性,并在海马回路中接受功能性兴奋性输入,可实现海马连接的体外建模[37]。人胚胎干细胞从自组织的背体端脑组织中生成功能性海马颗粒状和锥体状神经元,该体外模型概述了人类海马的发育。培养的神经元具有电压依赖性Na-K电流、注射去极化电流后的动作电位和自发兴奋性突触后电流,表明神经元已经足够成熟,可以形成功能性突触,通过单个神经元表现出自发性钙瞬变表明神经元形成了一个具有突触连接的神经元网络[38]。有研究应用人诱导多能干细胞快速生成自由漂浮的海马球体用于阿尔茨海默病的研究,APP和非典型PS1变异海马球体显示出增加的β-淀粉样蛋白42/β-淀粉样蛋白40肽比率和降低的突触蛋白水平[39]。另有学者开发了一种高度标准化、可重复、快速(5周)的小鼠脑类器官模型,从胚胎神经干细胞开始逐渐分化并自组织成具有前脑背侧表型的功能性神经元3D网络,通过添加形态发生因子WNT3A,生成了具有特定海马区的脑类器官[40]。WU 等[35]基于液态金属-聚合物导体的网状神经接口与人类海马类器官的耦合构建了海马半机械人类器官,成功检测到海马类器官的神经活动。
脉络丛是一种高度血管化的器官,是位于大脑外侧、第三和第四脑室内的中枢神经系统的重要组成部分,脉络丛的主要功能是产生脑脊液,脑脊液充满脑室并围绕大脑和脊髓[41]。脉络丛组织由一层称为脉络丛室管膜细胞的特化细胞组成,这些细胞与血管密切相关,共同形成血液-脑脊液屏障。研究人员用人诱导多能干细胞衍生出脉络丛器官样结构,这些器官样结构形成了清晰界定的囊状结构,内部充满了类似于人类脑脊液的液体,脉络丛室管膜细胞标志物转甲状腺素蛋白和氯离子细胞内通道蛋白6上调,用于预测神经活性药物通过这种脉络丛屏障的渗透性。将脉络丛器官样结构中提取的液体进行蛋白质组学分析,结果显示其与体内产生的脑脊液样本具有显著的相似性[42]。与未添加WNT激动剂CHIR相比,CHIR+衍生的脉络丛类器官对β-淀粉样蛋白42低聚物刺激的反应更强,如促炎标志物肿瘤坏死因子α、白细胞介素6及基质金属蛋白酶2/9的上调,这可能与疾病建模和药物筛选研究的生理相关[43]。在端脑,脉络膜丛上皮起源于表达WNT的皮质边缘,β-catenin等典型WNT信号通路分子在小鼠和人胚胎脉络膜丛上皮中表达,表明该通路具有活性,已知人β-catenin的点突变可导致典型WNT信号的组成性激活,脉络膜丛上皮身份的丧失和该组织向神经元身份的明显转变。在暴露于骨形态发生蛋白4和低水平CHIR的混合物后,人胚胎干细胞衍生的类器官中会形成脉络丛上皮样结构,而经典WNT通路的高激活导致脉络丛上皮样结构标志物的抑制,表明经典的WNT信号通路必须以精确且受调控的方式发挥作用,才能保证哺乳动物大脑中脉络丛的正常发育[44]。
(2)腹侧(皮质下)前脑类器官:皮质下结构包括大脑皮质腹侧的端脑吻侧区域,形态诱导信号会形成神经节隆起的暂时性皮质下结构,神经节隆起对于产生大部分皮质中间神经元、嗅球中间神经元以及建立纹状体起着至关重要的作用。在神经节突起的最前端,即内侧神经节隆起,最终将产生约70%的皮质中间神经元群体;而位于更背侧位置的尾侧神经节隆起,则贡献了约30%的皮质中间神经元[45]。神经节隆起中的皮质命运决定的中间神经元会沿着广泛定义的浅层迁移通道或深层迁移通道向大脑皮质迁移。外侧神经节隆起位于尾部神经节隆起的吻侧,构成了纹状体,外侧神经节隆起中的特异性中间神经元会沿着前侧迁移通道迁移到嗅球处。前脑类器官方案利用双重Sma和Mad相关蛋白抑制进行神经诱导,通过抑制WNT信号传导而形成,同时也激活刺猬因子信号通路,在发育的前脑中刺猬因子表现出腹侧高到背侧低的梯度[46]。在类器官培养中,通常使用重组刺猬因子蛋白和/或靶向小分子的平滑激动剂激活刺猬因子通路模仿内源性的皮质下层信号,诱发腹侧端脑模式[47]。刺猬因子蛋白在神经管下端从腹侧方向表达,并向背侧扩散,形成浓度梯度,有助于神经管背腹侧轴的定形[48]。人类内侧神经节隆起是产生皮质中间神经元和相关谱系的关键脑腹侧区域,有研究用多能干细胞分化产生类似内侧神经节隆起结构域的人脑类器官来检测人类中间神经元迁移。嵌入在基质液滴内的内侧神经节隆起类器官中有广泛的神经元迁移,通常是单个神经元迁移到嵌入基质的表面,将 hsyn-GCamp6s标记的内侧神经节隆起类器官与hsyn-RFP标记的类器官融合生成脑类器官,在类器官侧观察到强劲的钙瞬变,说明迁移的中间神经元具有正常的神经元活动,此外,来自人内侧神经节隆起类器官的神经元在迁移过程中保持γ-氨基丁酸能的身份,迁移的中间神经元形态正常,在胞体和神经突区均形成兴奋性突触后密度,以上研究表明融合脑类器官可以作为人类神经元在三维细胞环境中迁移的模型[49]。有研究开发了具有空间分离的背侧和腹侧区域以及层状的顶基底细胞组织的背-腹侧微流控前脑样结构,分别模拟了人类前脑皮质和皮质下层的发育,这些基于微流体的神经发育模型共同提供了具有体内时空细胞分化和组织的三维管腔组织结构[50]。
纹状体是基底神经节的门户,接收来自大脑皮质和丘脑的输入,同时与中脑的黑质和苍白球形成复杂的投射关系。皮质纹状体投射是调节动机行为的前脑回路的关键组成部分。有学者利用发育中的人类纹状体的转录组轨迹,确定了一种调节视黄酸受体途径的小分子,它与WNT和激活素结合,产生3D人纹状体球。将纹状体类器官与大脑皮质类器官在三维培养物中组装在一起,形成皮质纹状体组合体。皮质神经元将轴突投射发送到纹状体类器官并形成突触连接。中等棘神经元在组装后电生理成熟,并在皮质神经元的光遗传学刺激后显示钙活性[51]。另有研究应用多能干细胞生成早产类器官解离细胞液移植在SCID小鼠纹状体中构建体内纹状体类器官,表现出强大的存活率和侧脑室室下区样结构,在其附近经常可以发现被绿色荧光蛋白阳性ChP上皮样细胞包围的大腔,观察到神经元和星形胶质细胞分化,单细胞转录组分析表现出比人脑类器官更高的细胞类型多样性,产生更多的支持细胞。表达纹状体标记物锌指和BTB结构域含量20和纹状蛋白3[52]。由于纹状体是神经退行性疾病中最脆弱的区域之一,因此纹状体类器官常用于帕金森病的多巴胺能神经元投射、突触连接[53],亨廷顿舞蹈症等棘神经元投射及精神分裂症的研究中[54-55]。
(3)间脑类器官:丘脑是间脑的最大分支,由前体的鼻翼组成。是来自大脑皮质的感觉-运动输入的主要传递中心。成熟的丘脑神经元会向初级视觉皮质发送远距离的神经信号,并通过依赖神经活动的机制来调节皮质的连接性。丘脑的神经信号主要被归类为谷氨酸能输入驱动信号[56]。丘脑/下丘脑类器官是通过诱导相同的神经上皮细胞生成的,刺猬因子和成纤维细胞生长因子8是驱动脑干中丘脑命运形成的主要外源性因子。用刺猬因子信号通路以及适当的附加因子(PD0325901、骨形态发生蛋白7/WNT3A)对其进行处理,通过融合代表发育中的丘脑或皮质的两个不同区域特定类器官来创建丘脑和皮质之间的相互投影,以实现详细的区域特征分析。早期人类丘脑类器官的scRNAseq显示存在神经元祖细胞、中间前体和未成熟神经元。在晚期丘脑类器官中,大多数细胞是星形胶质细胞和成熟的谷氨酸能和氨基丁酸能神经元,与体内丘脑组织相似[57]。
下丘脑是调节食欲、睡眠、昼夜节律、应激反应和生殖行为等生理功能的中心。下丘脑通过漏斗与脑垂体形成整体连接,调节内分泌激素的释放[58]。在利用多能干细胞生成下丘脑神经元方面已经取得了一些进展。利用骨形态发生蛋白、WNT、Notch抑制剂和刺猬因子激动剂指导分化,几个小组在体外产生了表达下丘脑样神经肽的神经元,其形态和遗传特性与体内神经元相似。这些下丘脑神经元在体外表现出对代谢信号的反应,其中一些可以整合到新生小鼠的大脑中[59-60]。HUANG等[61]报告了人诱导多能干细胞产生弓状类器官以模拟人类下丘脑弓状核的发育,这些类器官表现出类似于普拉德-威利综合征患者下丘脑的异常分化和转录组失调,可用于研究人类早期弓状发育过程中的细胞核特异性特征和疾病相关机制。有学者应用多能干细胞衍生的下丘脑类器官对下丘脑细胞多样性进行了模拟,利用带酪氨酸羟化酶-TdTomato报告基因的多能干细胞系检测多巴胺能神经元和其他表达酪氨酸羟化酶的细胞,RNA测序揭示了成熟下丘脑类器官中不同的神经元和非神经元细胞类型,探究了电生理活性下丘脑类器官中多巴胺能神经元特异性途径和功能[62]。刺猬因子处理的人胚胎干细胞分化(d14-22)的丘脑类器官与腹侧丘脑前核、丘脑核和内侧膝状核相似,称为腹侧丘脑类器官,单细胞RNA测序分析在腹侧丘脑类器官发育过程中形成的丘脑网状核样细胞簇表现出与体内丘脑网状核相似的异质性,腹侧丘脑类器官包含具有腹侧身份的多样化丘脑核,神经元中钙活性的频率和平均振幅显著升高。疾病相关基因修补结构域包含1或受体酪氨酸蛋白激酶的扰动会损害腹侧丘脑类器官中的神经元功能,但不会影响整体丘脑谱系发育[63]。KASAI等[64]使用3D培养的人诱导多能干细胞在体外重现了下丘脑对垂体功能的贡献,诱导垂体分泌促肾上腺皮质激素的能力达到与成年小鼠垂体相当的水平,在相同的聚集体内与下丘脑神经元同时成熟。下丘脑-垂体类器官通过促肾上腺皮质激素释放激素-促肾上腺皮质激素途径对体外环境低血糖状况做出反应,模拟体内危及生命的情况,并成功增加促肾上腺皮质激素的分泌。
2.1.2 中脑类器官 中脑介于间脑与脑桥之间,它从胚胎早期的中脑泡发展而来。它与许多生理功能有关,包括视觉、听觉、睡眠/觉醒和温度调节[9]。中脑类器官是一种基于干细胞技术构建的3D体外模型,用于模拟人类中脑的结构与功能,尤其在神经退行性疾病(如帕金森病)研究中具有重要价值[65]。中脑类器官通过成纤维细胞生长因子和刺猬因子信号通路的调控以及神经元的成熟过程生成,该过程旨在通过诱导神经上皮细胞后对WNT激活剂的处理和浓缩,促使神经外胚层分化形成底板结构,随后再形成中脑[66]。中脑类器官包含具有电生理活性的多巴胺能、γ-氨基丁酸能、谷氨酰胺能和血清素能神经元[67],在中脑类器官中检测到电活性和功能成熟的中脑多巴胺神经元和多巴胺的产生,中脑样器官产生的神经黑色素样颗粒在结构上与从人类黑质组织中分离出来的颗粒相似[68]。GERASIMOVA等[69]开发了由人诱导多能干细胞衍生的中脑类器官和外周血T细胞共培养的3D模型,在空间组织的微环境中研究了T细胞的运动性和与中脑组织的相互作用,证明T细胞浸润中脑类器官组织,导致神经细胞损失,可作为研究适应性免疫系统对中脑影响的有前途的工具。有研究优化了人胚胎干细胞生成富含多巴胺能神经元的中脑类器官的方案,引入了机械切割步骤,将大型类器官在立体显微镜下切割成直径小于300 μm的小块,这些碎片重组成第2代类器官,恢复生长并形成球形,表现出适当的神经活动和功能神经网络,这些类器官可以长期存活,允许重复和扩展评估,以推进大脑发育和疾病的基础研究[70]。用中脑类器官微生理系统模拟全氟辛烷磺酸盐暴露,每个中脑类器官微生理系统由一个来源于人类干细胞的中脑类器官和一个带有可灌注层的完全3D打印的支架装置组成,该模型可扩展,易于使用,并与传统的孔板兼容,可以轻松转移到多电极阵列系统上,用于即插即用的神经活动测量[71]。有研究用多能干细胞生成微中脑类器官,与典型的中脑类器官相比,微中脑类器官表现出均匀一致的形态、大小和基因表达模式。在分化后的30 d内表现出显著的加速成熟,如成熟的多巴胺能神经元均匀分布,包括胶质细胞在内的其他体内样细胞区室的存在,神经黑色素样颗粒的产生以及成熟的电生理特征[72]。人中脑类器官也可以由神经上皮干细胞产生,突出表现为星形胶质细胞和少突胶质细胞分化以及神经突髓鞘形成[73]。
脑干是大脑半球深部结构之间的大脑后部区域,它连接大脑和脊髓,分为中脑、脑桥和延髓3个部分,它们包含多个细胞核和广泛突出到大脑皮质、基底神经节和大脑其他部分的小纤维束。有学者开发了以一种生成人脑干类器官的方法,中脑、周围的脑干部分及其后面的神经嵴区域通过添加碱性成纤维细胞生长因子和表皮生长因子诱导神经元干细胞/祖细胞扩增,用脑源性神经营养因子和胶质细胞源性神经营养因子诱导多巴胺能神经元,这些类器官包含中脑/后脑祖细胞、去甲肾上腺素能和胆碱能神经元、多巴胺能神经元和神经嵴谱系细胞。单细胞RNA序列、蛋白质组学分析和电生理结果表明,这些类器官中的细胞群与人脑干的细胞群像相似[74]。
前脑和中脑区域特异性脑类器官研究现状见表1。
2.1.3 不同类器官之间的融合 脑类器官融合培养可以模拟不同大脑区域之间的复杂相互作用,融合指向背侧和腹侧前脑的类器官生成背腹轴,使用荧光报告基因,展示γ-氨基丁酸能中间神经元的迁移。中间神经元可以在融合的腹侧和背侧环状器官之间迁移,许多中间神经元在迁移到背侧区域后保持NKX2-1IWP-2可以成为成熟的神经元。同时,迁移的中间神经元在融合的脑类器官中产生各种背外侧神经节隆起/尾部神经节隆起和腹内侧神经节隆起衍生的皮质中间神经元亚型,绿色荧光蛋白阳性迁移细胞表现出不同迁移模式的特征,类似于迁移的皮质中间神经元的迁移模式[75]。不同类器官之间的自我模式化和融合能力允许产生特定的组织结构和功能性细胞连接[76]。人胚胎干细胞衍生的丘脑类器官与皮质类器官融合后形成互向突起[57]。ANDERSEN等[77]分化大脑皮质或后脑/脊髓类器官,将它们与人类骨骼肌球体组装在一起,生成3D皮质-运动组合体,发现皮质神经元投射并连接到脊髓球体,而脊髓源性运动神经元连接到肌肉,用谷氨酸释放或光遗传学刺激皮质球体可触发3D肌肉的强劲收缩,并在融合后10周内集合体在形态和功能上保持完整。神经上皮干细胞分化的中脑类器官和人诱导多能干细胞分化的纹状体类器官在共同培养基中培养形成中脑-纹状体组合模型,该模型概括了黑质纹状体途径的连接性,儿茶酚胺从中脑释放到纹状体,并在中脑多巴胺能神经元和纹状体神经元之间形成活性突触[78]。将中脑和非引导类器官被加载到Matrigel相邻通道中,形成来自两种不同类型类器官的

组装体,中脑类器官产生轴突突起,细胞从未引导的类器官迁移到基质胶中,朝中脑类器官迁移[79]。有研究用微流体技术将皮质、海马和丘脑类器官沉降到微孔阵列融合成脑组合体,脑组合体能够生长并发挥良好的功能,具有神经迁移和相互作用活跃的特征[80]。
2.2 脑类器官在缺血性脑卒中建模、药物开发及移植中的应用
2.2.1 脑类器官在缺血性脑卒中体外模型构建中的应用 从多能干细胞中提取的三维脑类器官能够模拟人体内大脑的细胞组成、皮质结构、脑神经连通性和表观遗传基因组学,在建立人源化缺血性脑卒中模型方面具有广阔的应用前景。一项关于脑类器官响应体外氧糖剥夺诱导的缺血性脑卒中损伤的变化及其在测试抗脑卒中药物中应用的报道表明,不同人胚胎干细胞体外培养时间的脑类器官具有不同的细胞组成,在分化85 d时的主要细胞成分Tuj1+神经元占73.6%,绿色荧光蛋白阳性星形胶质细胞占13.4%,巢蛋白阳性神经干细胞占14.5%,SOX2+细胞占21.2%,同时糖氧剥夺诱导的脑类器官损伤存在凋亡、坏死性凋亡、自噬和铁死亡等多种死亡方式[81]。人诱导多能干细胞分化的脑类器官经糖氧剥夺处理后基因表达谱结果显示,α-2-HS-糖蛋白下调、铁蛋白轻链抗体的表达不受糖氧剥夺/复氧严重缺氧的影响及肌肉丙酮酸激酶同工酶2的上调这些可能是人脑类器官特有的反应,而过氧化物酶体增殖物激活受体α的水平在脑类器官中没有发生变化,其原因可能是由于缺乏源自中胚层的小胶质细胞[82]。为了揭示活性依赖性转录因子神经元PAS结构域蛋白4是否在体外人缺血模型中被诱导表达,有学者从人诱导多能干细胞中生成脑类器官,在糖氧剥夺处理1 h后神经元PAS结构域蛋白4在人脑类器官中的表达量显著增加,表明人类和小鼠都有共同的缺血诱导神经元PAS结构域蛋白4 的机制[83]。器官型脑切片培养物适合模拟离体脑生理学/病理生理学。组成型光形态发生9信号复合体(constitutive photomorphogenesis 9 signalosome,CSN)是一种去NEDD氧化酶,它调控着cullin-RING-E3连接酶的泛素化活性,从而影响关键细胞蛋白的水平。利用相关脑驻留细胞类型的模型、离体器官型脑切片培养模型以及CRL neddylase状态修饰药物MLN4924和CSN5i-3,分别模拟和抑制CSN5 deNEDDylase活性,研究了CSN在神经炎症和缺血性神经元损伤中的作用。大鼠海马体制备的器官型脑切片培养物经MLN4924或CSN5i-3预处理后暴露于糖氧剥夺应激,结果表明,CSN5活性可减弱器官型脑切片培养中的缺血性神经元损伤,与小胶质细胞表型和炎症细胞因子的影响有关[84]。多能干细胞诱导的脑类器官在缺氧缺糖8 h和12 h,复氧24 h后黏液膜相关淋巴组织淋巴瘤转运蛋白1的水平显著升高,表明黏液膜相关淋巴组织淋巴瘤转运蛋白1参与了脑缺血再灌注损伤的过程[85]。
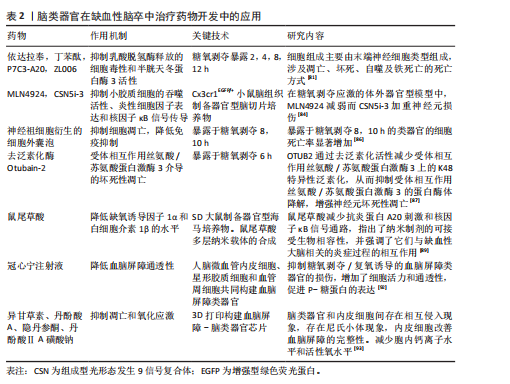
2.2.2 脑类器官在缺血性脑卒中治疗药物开发中的应用 有研究表明,由内部生成的诱导多能干细胞在胶原蛋白水凝胶中生成脑类器官,神经祖细胞衍生的细胞外囊泡保护脑类器官免受糖氧剥夺诱导的细胞死亡[86]。去泛素化酶Otubain-2抑制剂LN5P45处理显著降低了糖氧剥夺诱导的脑类器官中的混合谱系激酶结构域样蛋白磷酸化和神经元丢失[87]。有研究用小胶质细胞模拟脑类器官,将从人诱导多能干细胞分化的高纯度造血祖细胞掺入人诱导多能干细胞衍生的胚状体中,用小胶质细胞生长因子治疗可以增加跨膜蛋白119特异性小胶质细胞的数量,当用脂多糖处理时,可以观察到小胶质细胞活化的增加,表明成功制造具有小胶质细胞和神经元的脑类器官,用于研究先天免疫和神经系统发育之间的早期相互作用,并可能成为神经炎症和神经感染性疾病的模型[88]。KAMI?SKA等[89]制定了一种纳米治疗系统,将鼠尾草酸作为一种具有抗氧化和抗炎特性的神经保护剂递送到缺血脑组织,通过暴露于糖氧剥夺的器官型海马培养物进行模拟。单靶点的治疗方法不足以达到最佳的临床结果,有学者提出了一种双表型药物筛选策略,同时针对神经保护和抗炎机制,将神经元与小胶质细胞或脑类器官共培养模型用于验证药物对神经元存活和小胶质细胞激活的协同作用,为更精确地模拟疾病的复杂病理生理提供了平台,识别同时保护神经元、抑制神经炎症的化合物,提高治疗的有效性和安全性[90]。在小鼠或多能干细胞中重组脑类器官的基础上,BAKKER等[91]采用了基质辅助激光解吸电离质谱成像和荧光显微镜技术,建立了一个高度精确的器官组织培养平台,用于药物分析。重点是植入和收集类器官,同时还提供了针对电离质谱成像特定的数据采集、样本制备以及通过串联质谱进行分子鉴定的建议。因此,对脑类器官进行电离质谱成像研究为观察中枢神经系统发育过程中代谢物和蛋白质图谱的动态变化提供了宝贵的机会。有研究用人脑内皮细胞、周细胞和星形胶质细胞融合培养3D血脑屏障脑类器官,将血脑屏障脑类器官用糖氧剥夺/复氧处理模拟缺血再灌注血脑屏障损伤的体外模型,结果表明,糖氧剥夺/复氧诱导后脑类器官的细胞活力显著降低,体积缩小,紧密连接蛋白1、密蛋白5和闭合蛋白的蛋白含量显著降低,使用中药冠心宁注射液可抑制上述变化,保护血脑屏障脑类器官的功能[92]。异甘草素对缺血缺氧的脑类器官中乳酸脱氢酶的释放具有显著抑制作用,从而减轻了乳酸对脑类器官细胞的损伤,降低了类器官中半胱天冬蛋白酶3的表达,减轻了凋亡损伤。另外,用聚二甲基硅氧烷和聚甲基丙烯酸甲酯两种材料对脑类器官进行组装,和血管中的人脐静脉内皮细胞灌注芯片构建成微流控芯片脑类器官,结果显示细胞之间存在相互侵入的现象,类器官内部表达内皮细胞标志物CD31,同时,类器官成功分化为神经元谱系细胞,在缺血缺氧损伤后较人脐静脉内皮细胞参与的跨膜电位电阻值显著降低,表明人脐静脉内皮细胞参与了脑类器官血脑屏障的完整性。丹参类化合物丹酚酸A、丹酚酸ⅡA磺酸钠和隐丹参酮对缺血缺氧类器官具有抑制细胞凋亡、减少钙离子内流和活性氧释放的作用[93]。见表2。
2.2.3 脑类器官在缺血性脑卒中移植中的应用 通过胚层分化、神经诱导和在Matrigel液滴中形成极化神经上皮样结构,从人胚胎干细胞中产生脑类器官,将形成的脑类器官在大鼠大脑中动脉阻塞建模后6 h进行移植,移植14 d后脑梗死体积显著降低,在同侧皮质保留了更多的存活脑实质和更小的空洞,同侧脑容量比大脑中动脉阻塞组显著增加,观察到移植的脑类器官和宿主脑之间的血管形成。同时,来自移植脑类器官的细胞显示出多谱系分化的潜力,可以模拟体内皮质发育,支持运动皮质区域特异性重建,形成神经递质相关神经元,并通过原位分化和脑卒中后细胞置换实现与宿主脑的突触连接,还促进了脑卒中后髓鞘再生、轴突再生和少突胶质细胞生成[94]。
脑类器官在缺血性脑卒中大鼠中的成功移植为脑类器官治疗脑卒中的临床转化提供了可靠的实验基础,使其在临床中的应用成为可能。CAO等[95]诱导了光血栓性成年雄性小鼠局灶性皮质脑卒中,将多能干细胞培养的脑类器官移植到脑卒中非肥胖糖尿病-严重联合免疫缺陷小鼠的梗死核心和梗死周围区交界处。移植后第180天,梗死的核心完全被移植的类器官填充,在梗死核心区域的增强绿色荧光蛋白阳性组织神经元在受到电流脉冲引起的膜去极化以及自发性突触后电流的刺激时,会产生动作电位;大量带有绿色荧光蛋白的神经纤维出现在对侧和同侧的胼胝体、纹状体、海马体、同侧内囊、感觉皮质和腹侧后丘脑核中,说明移植的脑类器官在移植180 d后向脑卒中小鼠的特定脑区延伸长距离投射。同时,移植的脑类器官神经元接受来自宿主细胞的突触输入和来自脑类器官神经元到远端宿主神经元的突触输出,促进小鼠脑卒中后功能恢复。
区域特异性脑类器官人内侧神经节隆起类器官可以模拟腹前脑模式的早期阶段。将人内侧神经节隆起类器官移植到光血栓性脑卒中的非肥胖糖尿病-严重联合免疫缺陷小鼠的梗死核心和梗死周围感觉运动区之间的连接处。发现大多数移植的人内侧神经节隆起类器官存活良好,并主要分化为受损皮质的γ-氨基丁酸能中间神经元,有助于脑卒中后的恢复[96]。提高移植干细胞在受伤脑组织中的存活率和整合对于干细胞移植治疗脑卒中的成功至关重要。目前用于移植的脑类器官由人胚胎干细胞、人类多能干细胞和人诱导多能干细胞分化而成,这些细胞先形成胚胎体,在神经诱导培养基中进一步形成神经上皮细胞,最后形成神经管结构的脑类器官。在神经上皮细胞形成后还可加入刺猬信号激动剂引导内侧神经节隆起类器官形成,加入基质胶作为细胞外支架或在形成的类器官中转染慢病毒载体,验证移植的类器官在缺血性脑卒中鼠中的稳定性及发挥的重要作用。
以上研究结果显示,类器官在培养55 d时进行移植,一般移植数量为两三个类器官(直径600 μm,200 000个细胞),移植后28 d类器官显示出多分化的潜力,可以模拟体内皮质发育,实现与宿主大脑的突触连接,促进脑卒中后突触重建、轴突再生和血管生成。在移植后180 d脑卒中小鼠仍可长期存活并且移植物仍在分化,梗死的核心被移植物完全填塞,梗死核心中存在相当数量的宿主细胞,观察到STEM123+标记的星形胶质细胞和成熟的神经元,可以恢复脑卒中后的感觉运动功能[97]。以上研究为类器官重建梗死组织和促进脑卒中功能恢复提供了新的策略。今后研究方向可围绕类器官不同区域类型培养移植物,同时对移植物在缺血性脑卒中产生的炎症反应等作用机制进行深入探讨。
| [1] FEIGIN VL, BRAININ M, NORRVING B, et al. World Stroke Organization (WSO): Global Stroke Fact Sheet 2022. Int J Stroke. 2022;17(1):18-29. [2] PU L, WANG L, ZHANG R, et al. Projected Global Trends in Ischemic Stroke Incidence, Deaths and Disability-Adjusted Life Years From 2020 to 2030. Stroke. 2023;54(5):1330-1339. [3] CHAMORRO Á, DIRNAGL U, URRA X, et al. Neuroprotection in acute stroke: targeting excitotoxicity, oxidative and nitrosative stress, and inflammation. Lancet Neurol. 2016;15(8):869-881. [4] COOK DJ, TYMIANSKI M. Nonhuman primate models of stroke for translational neuroprotection research. Neurotherapeutics. 2012;9(2):371-379. [5] SOMMER CJ. Ischemic stroke: experimental models and reality. Acta Neuropathologica. 2017; 133(2):245-261. [6] GRENIER K, KAO J, DIAMANDIS P. Three-dimensional modeling of human neurodegeneration: brain organoids coming of age. Mol Psychiatry. 2020;25(2):254-274. [7] STILES J, JERNIGAN TL. The basics of brain development. Neuropsychol Rev. 2010;20(4):327-348. [8] MARTINEZ-FERRE A, MARTINEZ S. Molecular regionalization of the diencephalon. Front Neurosci. 2012;6:73. [9] GUY B, ZHANG JS, DUNCAN LH, et al. Human neural organoids: Models for developmental neurobiology and disease. Dev Biol. 2021;478:102-121. [10] KWAK T, PARK SH, LEE S, et al. Guidelines for Manufacturing and Application of Organoids: Brain. Int J Stem Cells. 2024;17(2):158-181. [11] WANG Z, WANG SN, XU TY, et al. Cerebral organoids transplantation improves neurological motor function in rat brain injury. CNS Neurosci Ther. 2020;26(7):682-697. [12] ARLOTTA P. Organoids required! A new path to understanding human brain development and disease. Nat Methods. 2018;15(1):27-29. [13] LUO C, LANCASTER MA, CASTANON R, et al. Cerebral Organoids RecapitulaeEpigenomic Signatures of the Human Fetal Brain. Cell Rep. 2016;17(12):3369-3384. [14] LANCASTER ML, RENNER M, MARTIN CAM, et al. Cerebral organoids model human brain development and microcephaly. Nature. 2013; 501(7467):373-379. [15] DEZONNE RS, SARTORE RC, NASCIMENTO JM, et al. Derivation of Functional Human Astrocytes from Cerebral Organoids. Sci Rep. 2017;7(1-4): 45091. [16] SCHUKKING M, MIRANDA HC,TRUJILLO CA, et al. Direct Generation of Human Cortical Organoids from Primary Cells. Stem Cells Dev. 2018;27(22): 1549-1556. [17] CAKIR B, XIANG YF, TANAKA Y, et al. Engineering of human brain organoids with a functional vascular-like system. Nat Methods. 2019;16(11):1169-1175. [18] SHI YC, SUN L, WANG MD, et al. Vascularized human cortical organoids (vOrganoids) model cortical development in vivo. PLoS Biol. 2020; 18(5):e3000705. [19] KOOK MG, LEE SE, SHIN N, et al. Generation of Cortical Brain Organoid with Vascularization by Assemblingwith Vascular Spheroid. Int J Stem Cells. 2022;15(1):85-94. [20] SCHAFER ST, MANSOUR AA, SCHLACHETZKI JCM, et al. An in vivo neuroimmune organoid model to study human microglia phenotypes. Cell. 2023; 186(10):2111-2126.e20. [21] PARK SH, SUN W. Toxicity assessment using neural organoids: innovative approaches and challenges. Toxicol Res. 2025;41(2):91-103. [22] QIAN XY, SONG HJ, MING GL. Brain organoids: advances, applications and challenges. Development. 2019;146(8):dev166074. [23] SASAI Y. Next-generation regenerative medicine: organogenesis from stem cells in 3D culture. Cell Stem Cell. 2013;12(5):520-530. [24] EIRAKU M, WATANABE K, MATSUO-TAKASAKI M, et al. Self-organized formation of polarized cortical tissues from ESCs and its active manipulation by extrinsic signals. Cell Stem Cell. 2008;3(5):519-532. [25] SMITS LM, SCHWAMBORN JC. Midbrain Organoids: A New Tool to Investigate Parkinson’s Disease. Front Cell Dev Biol. 2020;8:359. [26] MULDER LA, DEPLA JA, SRIDHAR A, et al. A beginner’s guide on the use of brain organoids for neuroscientists: a systematic review. Stem Cell Res Ther. 2023;14(1):87. [27] PAŞCA AM, SLOAN SA, CLARKE LE, et al. Functional cortical neurons and astrocytes from human pluripotent stem cells in 3D culture. Nat Methods. 2015;12(7):671-678. [28] MADHAVAN M, NEVIN ZS, SHICK HE, et al. Induction of myelinating oligodendrocytes in human cortical spheroids. Nat Methods. 2018; 15(9):700-706. [29] SLOAN SA, DARMANIS S, HUBER N, et al. Human Astrocyte Maturation Captured in 3D Cerebral Cortical Spheroids Derived from Pluripotent Stem Cells. Neuron. 2017;95(4):779-790.e6. [30] MARTON RM, MIURA Y, SLOAN SA, et al. Differentiation and maturation of oligodendrocytes in human three-dimensional neural cultures. Nat Neurosci. 2019;22(3):484-491. [31] MEDINA-CANO D, CORRIGAN EK, GLENN RA, et al. Rapid and robust directed differentiation of mouse epiblast stem cells into definitive endoderm and forebrain organoids. Development. 2022;149(20): dev200561. [32] NISHIMURA H, LI Y. Human pluripotent stem cell-derived models of the hippocampus. Int J Biochem Cell Biol. 2024;177:106695. [33] MORENO-JIMÉNEZ EP, FLOR-GARCÍA M, TERREROS-RONCAL J, et al. Adult hippocampal neurogenesis is abundant in neurologically healthy subjects and drops sharply in patients with Alzheimer’s disease. Nat Med. 2019;25(4):554-560. [34] BIDERMAN N, BAKKOUR A, SHOHAMY D. What Are Memories For? The Hippocampus Bridges Past Experience with Future Decisions. Trends Cogn Sci. 2020;24(7):542-556. [35] WU Y, CHENG J, QI J, et al. Three-dimensional liquid metal-based neuro-interfaces for human hippocampal organoids. Nat Commun. 2024;15(1):4047. [36] YU DX, DI GIORGIO FP, YAO J, et al. Modeling hippocampal neurogenesis using human pluripotent stem cells. Stem Cell Reports. 2014; 2(3):295-310. [37] SARKAR A, MEI A, PAQUOLA ACM, et al. Efficient Generation of CA3 Neurons from Human Pluripotent Stem Cells Enables Modeling of Hippocampal Connectivity In Vitro. Cell Stem Cell. 2018;22(5):684-697.e9. [38] SAKAGUCHI H, KADOSHIMA T, SOEN M, et al. Generation of functional hippocampal neurons from self-organizing human embryonic stem cell-derived dorsomedial telencephalic tissue. Nat Commun. 2015;6:8896. [39] POMESHCHIK Y, KLEMENTIEVA O, GIL J, et al. Human iPSC-Derived Hippocampal Spheroids: An Innovative Tool for Stratifying Alzheimer Disease Patient-Specific Cellular Phenotypes and Developing Therapies. Stem Cell Reports. 2020;15(1):256-273. [40] CIARPELLA F, ZAMFIR RG, CAMPANELLI A, et al. Murine cerebral organoids develop network of functional neurons and hippocampal brain region identity. iScience. 2021;24(12):103438. [41] SAUNDERS NR, DZIEGIELEWSKA KM, FAME RM, et al. The choroid plexus: a missing link in our understanding of brain development and function. Physiol Rev. 2023;103(1):919-956. [42] CHEW LH, MERCIER E, ROGALSKI JC, et al. Methods to extract and analyze fluid from human pluripotent stem cell-derived choroid plexus organoids. Front Mol Neurosci. 2023;16:1243499. [43] MUOK L, LIU C, CHEN X, et al. Inflammatory Response and Exosome Biogenesis of Choroid Plexus Organoids Derived from Human Pluripotent Stem Cells. Int J Mol Sci. 2023;24(8):7660. [44] PARICHHA A, SURESH V, CHATTERJEE M, et al. Constitutive activation of canonical Wnt signaling disrupts choroid plexus epithelial fate. Nat Commun. 2022;13(1):633. [45] LIM L, MI D, LLORCA A, et al. Development and Functional Diversification of Cortical Interneurons. Neuron. 2018;100(2):294-313. [46] CEDERQUIST GY, ASCIOLLA JJ, TCHIEU J, et al. Specification of positional identity in forebrain organoids. Nat Biotechnol. 2019;37(4):436-444. [47] PAVON N, SUN Y, PAK C. Cell type specification and diversity in subpallial organoids. Front Genet. 2024;15:1440583. [48] CHEN A, GUO Z, FANG L, et al. Application of Fused Organoid Models to Study Human Brain Development and Neural Disorders. Front Cell Neurosci. 2020;14:133. [49] XIANG Y, TANAKA Y, PATTERSON B, et al. Fusion of Regionally Specified hPSC-Derived Organoids Models Human Brain Development and Interneuron Migration. Cell Stem Cel. 2017;21(3): 383-398.e7. [50] XUE X, KIM YS, PONCE-ARIAS AI, et al. A Patterned Human Neural Tube Model Using Microfluidic Gradients. Nature. 2024;28(8007):391-399. [51] MIURA Y, LI MY, BIREY F, et al. Generation of human striatal organoids and cortico-striatal assembloids from human pluripotent stem cells. Nat Biotechnol. 2020;38(12):1421-1430. [52] HUANG S, HUANG F, ZHANG H, et al. In vivo development and single-cell transcriptome profiling of human brain organoids. Cell Prolif. 2022;55(3):e13201. [53] ZHENG X, HAN D, LIU W, et al. Human iPSC-derived midbrain organoids functionally integrate into striatum circuits and restore motor function in a mouse model of Parkinson’s disease. Theranostics, 2023;13(8):2673-2692. [54] WU S, HONG Y, CHU C, et al. Construction of human 3D striato-nigral assembloids to recapitulate medium spiny neuronal projection defects in Huntington’s disease. Proc Natl Acad Sci U S A. 2024;121(22):e2316176121. [55] SAWADA T, BARBOSA AR, ARAUJO B, et al. Recapitulation of Perturbed Striatal Gene Expression Dynamics of Donor’s Brains With Ventral Forebrain Organoids Derived From the Same Individuals With Schizophrenia. Am J Psychiatry. 2023;181(6):493-511. [56] SHERMAN SM. Thalamus plays a central role in ongoing cortical functioning. Nat Neurosci. 2016;19(4):533-541. [57] XIANG Y, TANAKA Y, CAKIR B, et al. hESC-Derived Thalamic Organoids Form Reciprocal Projections When Fused with Cortical Organoids. Cell Stem Cel. 2019;24(3):487-497.e7. [58] XIE Y, DORSKY RI. Development of the hypothalamus: conservation, modification and innovation. Development. 2017;144(9):1588-1599. [59] OGAWA J, PAO GM, SHOKHIREV MN, et al. Glioblastoma Model Using Human Cerebral Organoids. Cell reports, 2018;23(4):1220-1229. [60] MERKLE FT, MAROOF A, WATAYA T, et al. Generation of neuropeptidergic hypothalamic neurons from human pluripotent stem cells. Development. 2015;142(4):633-643. [61] HUANG WK, WONG SZH, PATHER SR, et al. Generation of hypothalamic arcuate organoids from human induced pluripotent stem cells. Cell Stem Cel. 2021;28(9):1657-1670.e10. [62] SARRAFHA L, NEAVIN DR, PARFITT GM, et al. Novel human pluripotent stem cell-derived hypothalamus organoids demonstrate cellular diversity. iScience. 2023;26(9):107525. [63] KIRAL FR, CAKIR B, TANAKA Y, et al. Generation of ventralized human thalamic organoids with thalamic reticular nucleus. Cell Stem Cel. 2023; 30(5):677-688.e5. [64] KASAI T, SUGA H, SAKAKIBARA M, et al. Hypothalamic Contribution to Pituitary Functions Is Recapitulated In Vitro Using 3D-Cultured Human iPS Cells. Cell Rep. 2020;30(1):18-24.e5. [65] CUI X, LI X, ZHENG H, et al. Human midbrain organoids: a powerful tool for advanced Parkinson’s disease modeling and therapy exploration. NPJ Parkinsons Dis. 2024;10(1):189. [66] QIAN X, NGUYEN HN, SONG MM, et al. Brain-Region-Specific Organoids Using Mini-bioreactors for Modeling ZIKV Exposure. Cell. 2016;165(5): 1238-1254. [67] SMITS LM, MAGNI S, KINUGAWA K, et al. Single-cell transcriptomics reveals multiple neuronal cell types in human midbrain-specific organoids. Cell Tissue Res. 2020;382(3):463-476. [68] JO J, XIAO Y, SUN AX, et al. Midbrain-like Organoids from Human Pluripotent Stem Cells Contain Functional Dopaminergic and Neuromelanin-Producing Neurons. Cell Stem Cel. 2016;19(2):248-257. [69] GERASIMOVA E, BEENEN AC, KACHKIN D, et al. Novel co-culture model of T cells and midbrain organoids for investigating neurodegeneration in Parkinson’s disease. NPJ Parkinsons Dis. 2025; 11(1):36. [70] WANG X, SUN G, TANG M, et al. Human Midbrain Organoids Enriched With Dopaminergic Neurons for Long-Term Functional Evaluation. Cell Prolif. 2025;58(7):e70005. [71] TIAN C, CAI H, AO Z, et al. Engineering human midbrain organoid microphysiological systems to model prenatal PFOS exposure. Sci Total Environ. 2024;947:174478. [72] YAO X, KANG JH, KIM KP, et al. Production of Highly Uniform Midbrain Organoids from Human Pluripotent Stem Cells. Stem Cells Int. 2023;2023:3320211. [73] MONZEL AS, SMITS LM, HEMMER K, et al. Derivation of Human Midbrain-Specific Organoids from Neuroepithelial Stem Cells. Stem Cell Reports. 2017;8(5):1144-1154. [74] EURA N, MATSUI TK, LUGINBÜHL J, et al. Brainstem Organoids From Human Pluripotent Stem Cells. Front Neurosci. 2020;14:538. [75] BAGLEY JA, REUMANN D, BIAN S, et al. Fused cerebral organoids model interactions between brain regions. Nat Methods. 2017;14(7):743-751. [76] FAUSTINO MARTINS JM, FISCHER C, URZI A, et al. Self-Organizing 3D Human Trunk Neuromuscular Organoids. Cell Stem Cel. 2020;26(2):172-186.e6. [77] ANDERSEN J, REVAH O, MIURA Y, et al. Generation of Functional Human 3D Cortico-Motor Assembloids. Cell. 2020;183(7):1913-1929.e26. [78] BARMPA K, SARAIVA C, LOPEZ-PIGOZZI D, et al. Modeling early phenotypes of Parkinson’s disease by age-induced midbrain-striatum assembloids. Commun Biol. 2024;7(1):1561. [79] CASSEL DE CAMPS C, ROSTAMI S, XU V, et al. Microfabricated dynamic brain organoid cocultures to assess the effects of surface geometry on assembloid formation. Biotechnol J. 2024;19(8):e2400070. [80] ZHU Y, ZHANG X, SUN L, et al. Engineering Human Brain Assembloids by Microfluidics. Adv Mater. 2023;35(14):e2210083. [81] WANG SN, WANG Z, WANG XY, et al. Humanized cerebral organoids-based ischemic stroke model for discovering of potential anti-stroke agents. Acta Pharmacol Sin. 2023;44(3):513-523. [82] IWASA N, MATSUI TK, IGUCHI N, et al. Gene Expression Profiles of Human Cerebral Organoids Identify PPAR Pathway and PKM2 as Key Markers for Oxygen-Glucose Deprivation and Reoxygenation. Front Cell Neurosci. 2021;15:605030. [83] TAKAHASHI H, ASAHINA R, FUJIOKA M, et al. Ras-like Gem GTPase induced by Npas4 promotes activity-dependent neuronal tolerance for ischemic stroke. Proc Natl Acad Sci U S A. 2021; 118(32):e2018850118. [84] TIAN Y, MILIC J, MONASOR LS, et al. The COP9 signalosome reduces neuroinflammation and attenuates ischemic neuronal stress in organotypic brain slice culture model. Cell Mol Life Sci. 2023; 80(9):262. [85] 曹光昭. 灯盏生脉胶囊对脑缺血再灌注后神经炎症的调控作用研究[D]. 北京: 中国中医科学院,2023. [86] ZHENG X, ZHANG L, KUANG Y, et al. Extracellular Vesicles Derived from Neural Progenitor Cells--a Preclinical Evaluation for Stroke Treatment in Mice. Transl Stroke Res. 2021;12(1):185-203. [87] MEI F, DENG D, CAO Z, et al. Deubiquitination of RIPK3 by OTUB2 potentiates neuronal necroptosis after ischemic stroke. EMBO Mol Med. 2025;17(4): 679-695. [88] WANG T, GASTFRIEND BD, MCDONALD V, et al. Derivation of a Human Brain Organoid with Microglia Development. J Vis Exp. 2025;(215). doi: 10.3791/67491. [89] KAMIŃSKA K, GRYGIER B, REGULSKA M, et al. Multilayered Nanocarriers as a New Strategy for Delivering Drugs with Protective and Anti-inflammatory Potential: Studies in Hippocampal Organotypic Cultures Subjected to Experimental Ischemia. Mol Neurobiol, 2025;62(5):6333-6351. [90] LIU XH, LIU HY, GU J, et al. Neuroprotective and Anti-inflammatory Dual-Phenotypic Drug Screening Strategies. ACS Chem Neurosci. 2025; 16(9):1631-1633. [91] BAKKER B, VAES RDW, ABERLE MR, et al. Preparing ductal epithelial organoids for high-spatial-resolution molecular profiling using mass spectrometry imaging. Nature protocols, 2022;17(4):962-979. [92] 杜宏英, 薛志峰, 夏忠庭, 等. 3D血脑屏障类器官氧糖剥夺模型的构建及冠心宁注射液的保护作用探究[J]. 药学学报,2022,57(10):3086-3094. [93] 刘凯. 3D打印脑类器官模型的芯片制备与中药活性成分治疗作用的研究[D].镇江: 江苏大学,2024. [94] WANG SN, WANG Z, XU TY, et al. Cerebral Organoids Repair Ischemic Stroke Brain Injury. Transl Stroke Res. 2020;11(5):983-1000. [95] CAO SY, YANG D, HUANG ZQ, et al. Cerebral organoids transplantation repairs infarcted cortex and restores impaired function after stroke. NPJ Regen Med. 2023;8(1):27. [96] CAO SY, TAO MD, LOU SN, et al. Functional reconstruction of the impaired cortex and motor function by hMGEOs transplantation in stroke. Biochem Biophys Res Commun. 2023;671:87-95. [97] LANCASTER MA. Brain organoids get vascularized. Nat Biotechnol. 2018;36(5):407-408. [98] MICHÓOR P, RENARDSON L, LI S, et al. Neurorestorative Approaches for Ischemic StrokeChallenges, Opportunities, and Recent Advances. Neuroscience. 2024;550:69-78. |
| [1] | 王 峥, 程 吉, 于金龙, 刘文红, 王召红, 周鲁星. 水凝胶材料在脑卒中治疗中的应用进展与未来展望[J]. 中国组织工程研究, 2026, 30(8): 2081-2090. |
| [2] | 高 峰, 张 俊, 余文君, 单于玉婧, 赵 乐, 胡雨婷, 王俊华, 刘永富. 佩戴腕手矫形器对脑卒中患者手功能障碍作用的Meta分析[J]. 中国组织工程研究, 2026, 30(8): 2124-2131. |
| [3] | 陶代菊, 苏海玉, 王宇琪, 沈志强, 何 波. 高/低表达miR-122-5p稳转PC12细胞株的构建和鉴定[J]. 中国组织工程研究, 2026, 30(7): 1790-1799. |
| [4] | 杨媛媛, 周珊珊, 成小菲, 冯露叶, 汤继芹. 非侵入性脑刺激治疗脑卒中后下肢运动功能障碍的网状Meta分析[J]. 中国组织工程研究, 2026, 30(4): 1008-1018. |
| [5] | 高震东, 张 理, 陈 攀, 尹希瑶, 李佳怡, 杨平娟, 张 敏, 廖淑馨, 石林林, 高社干. 患者源性食管胃结合部腺癌类器官模型的构建与鉴定[J]. 中国组织工程研究, 2026, 30(34): 8970-8977. |
| [6] | 何人大, 马 威, 孙永思, 莫雪妮. 脑类器官医学研究未来:跨学科融合培养与生物工程技术及优化模型成熟度[J]. 中国组织工程研究, 2026, 30(34): 9088-9094. |
| [7] | 唐 娅, 李 龙, 黄 杜, 黄朝露. 3D打印外骨骼对脑卒中患者手功能康复效果的系统综述[J]. 中国组织工程研究, 2026, 30(29): 7648-7653. |
| [8] | 王雪婷, 杨 巍, 王鹏琴. 脑卒中康复机器人:国内外研究现状及热点[J]. 中国组织工程研究, 2026, 30(28): 7404-7409. |
| [9] | 李思慧, 王 芹, 崔慎红, 成小菲, 冯梓芸, 王德花, 梁春婷, 冷 军. 非侵入性神经调控技术对脑卒中后失语患者言语功能影响的网状Meta分析[J]. 中国组织工程研究, 2026, 30(28): 7485-7493. |
| [10] | 范萌萌, 丁嘉莉, 万宇杰, 黄海量. 神经调控技术促进脑卒中后上肢运动功能的系统评价与网状Meta分析[J]. 中国组织工程研究, 2026, 30(28): 7494-7504. |
| [11] | 周 丽, 李 蕊, 陈 浩, 陈嘉琪, 刘玉红, 吴 娜. 构建基于类器官技术的小鼠体外溃疡性结肠炎模型[J]. 中国组织工程研究, 2026, 30(24): 6231-6238. |
| [12] | 曹新燕, 冷晓轩, 高世爱, 陈金慧, 刘西花. 脑卒中偏瘫患者痉挛程度的影响因素及风险预测模型分析[J]. 中国组织工程研究, 2026, 30(23): 6092-6098. |
| [13] | 牟健诚, 罗 杰, 刘皓天, 杨卓涛, 穆誉霄, 钱 达, 孟旭莉. 精准医疗背景下乳腺癌类器官药物治疗领域的应用与前景[J]. 中国组织工程研究, 2026, 30(19): 5033-5039. |
| [14] | 黄正波, 欧 敏, 李国顺, 段福慧, 刘建琦, 娄菊相, 赵艳修, 苏晓艳. 构建肾类器官在急性肾损伤中的创新性应用[J]. 中国组织工程研究, 2026, 30(19): 5040-5049. |
| [15] | 高洪梅, 张 坤, 肖东杰, 刘 华. 人脂肪多系分化应激耐受细胞治疗大鼠缺血性脑卒中[J]. 中国组织工程研究, 2026, 30(19): 4853-4859. |
人脑是一个高度组织化的结构,由各种细胞类型组成,包括神经元、神经胶质细胞和血管细胞。模拟人脑发育具有挑战性,因为它需要协调不同的生化和物理事件。在人类早期大脑发育过程中,神经管分为3个主要的脑泡:前脑、中脑和后脑。在神经管闭合或继发囊泡期后,2个初级脑囊泡进一步细分。前脑又分为端脑和间脑,视神经泡从端脑和间脑的边缘延伸[7]。在细胞层面,外源性和内源性因素的区域特异性定位能够促使细胞朝着特定的谱系方向分化,从而形成胚胎大脑的不同区域(图1)[8]。由于伦理原因以及胎儿组织难以获取,人类脑组织发育的研究一直颇具挑战性。尽管动物模型在探究发育神经生物学问题方面发挥了重要作用,但不同物种之间的根本差异限制了将这些模型中的研究成果应用于人类发育的能力。人类干细胞衍生的类神经器官为研究人类神经系统的基本问题提供了有吸引力的动物模型补充[9]。
最近开发的由人类多能干细胞诱导分化的三维脑类器官为脑卒中研究提供了新的视野。脑类器官是由造血干细胞衍生的三维多细胞聚集体,可以复制大脑的结构特征,再现神经系统发育、神经元网络的复杂结构和神经电生理学,反映不同细胞类型与各种微环境因素之间的相互作用,使其有望成为神经毒物筛选的模型[10]。体外培养的脑类器官包括神经祖细胞、神经元和神经胶质细胞等[11],具有细胞资源获得途径广泛、细胞数量大、三维组织结构以及可检测的神经连接和大脑功能等优势[12]。和人脑相比,脑类器官具有相似的细胞组成、皮质
结构和表观遗传基因组学[12]。由人胚胎干细胞H9分化的大脑类器官概括了人类胎儿大脑发育早期到中期的转录组特征和表观基因组,胎儿和发育的皮质显示出与脑类器官相似的完全甲基化状态,非CG甲基化富集在胎儿皮质和大脑类器官的超增强位点,在脑类器官中观察到的DNA甲基化谷超甲基化与在吻侧脑和原代脑细胞中观察到的发育程序一致。多孔类器官是一种改良的脑类器官方法,它在分化的早期阶段增加了双重Sma和Mad相关蛋白抑制,在脑类器官和多孔类器官中,具有突触传递和神经发生功能的基因出现了上调,同时,在脑类器官中保留了具有神经元功能的共表达基因模块[13],表观基因组分析为改进脑类器官方法提供了方向[13]。脑类器发展的时间历程见图2。 此综述对现阶段脑类器官的区域特异性研究进行综述总结,以期为脑类器官在缺血性脑卒中建模和药物开发中的应用提供新的技术手段和研究策略。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
1.1 资料来源
1.1.1 检索人及检索时间 由第一作者在2025年1-6月进行检索。
1.1.2 检索文献时限 各数据库建库至2025年6月。
1.1.3 检索数据库 PubMed数据库、中国知网。
1.1.4 检索词 英文检索词为“ischemic stroke,stroke,organoids,oxygen-glucose deprivation/reoxygenation,middle cerebral artery occlusion,forebrain organoids,hippocampal organoids,thalamic organoids,midbrain organoids”,中文检索词为“缺血性脑卒中、脑卒中、类器官、缺血再灌注损伤、糖氧剥夺/复氧、前脑类器官、海马类器官、丘脑类器官、中脑类器官”。
1.1.5 检索文献类型 研究原著及综述。
1.1.6 手工检索情况 无。
1.1.7 检索策略 以PubMed数据库检索策略为例,见图3。
1.1.8 检索文献量 共得到中英文文献1 300余篇。
1.2 入组标准
1.2.1 纳入标准 ①缺血性脑卒中相关文献;②脑类器官相关文献;③缺血性脑卒中与脑类器官结合应用的相关文献;④前脑类器官、海马类器官、丘脑类器官、纹状体和中脑类器官等区域特异性脑类器官相关文献;⑤同一领域选择发表年份较新的文献。
1.2.2 排除标准 ①与研究主题无关的文献;②重复性文献;③存在争议的文献。
1.3 数据的提取 根据排除标准剔除相关文献,通过泛读剩余文献的标题和摘要进行筛选,选取与主题相关的文章进行精读,最终纳入符合标准的文献98篇。文献检索流程见图4。
引导类器官在整个分化过程中,小分子和生长因子被用来指导多能干细胞形成代表大脑特定区域的细胞和组织,如大脑皮质、海马和中脑。这些定向类器官培养有时能够以相对一致的比例产生多种细胞类型的混合物,在批次和细胞系之间表现出较少的变化[29]。但与体内条件相比,它们仍然是一个简化的模型,因为它们仅包含来自神经谱系的组织,缺乏上覆的脑膜和脉管系统,作为循环基本要素脉管系统的缺乏限制了类器官的大小和生存能力。如在扩展培养过程中,中脑类器官通常会变得大小异质,其中一些会发展成形态不规则的超大球体。当它们超过一定大小时,这些类器官通常会停止生长并表现出核心坏死,这可能是由于类器官缺少充足的血供导致氧气或营养不足引起的[97]。另外,定向类器官通常包含相对较小的神经上皮结构,其细胞结构有时不明确,可能是由于过度使用外部因素干扰了多能干细胞的自组织和细胞-细胞相互作用。3D脑类器官能够模拟人脑的发育和主要功能,使其在药物筛选和疾病建模方面具有强大的潜力。它从人诱导多能干细胞发展而来,可克服研究人员在用动物模型研究疾病时发现的物种间差异,作为动物模型的可能替代品,可显著缩短药物筛选过程,同时可以避免动物模型使用时出现的伦理问题。
尽管脑类器官的优势尽显,但关于脑类器官在缺血性脑卒中的研究仍处于早期研究阶段,目前脑类器官在缺血性脑卒中建模中的应用多局限于糖氧剥夺/复氧诱导后细胞凋亡等常规表型的检测,常以此模型用于动物和细胞模型机制验证后的补充实验,并未充分发挥脑类器官接近于人脑组织的优势。可能存在的原因是一方面脑类器官缺乏血管化和高度变异性,另一方面是类器官培养中血脑屏障和非神经元细胞类型的缺失。未来这些方面都将纳入类器官培养中需要解决的关键问题。研究人员正在积极努力克服这些限制,通过类器官融合及芯片等工程技术创新逐步提高脑类器官的保真度。另外,脑类器官在抗卒中药物开发的应用中有一些新的探索,有研究创新的应用质谱成像、微流控芯片构建了药物分析平台,还有学者针对脑类器官提出双表型药物筛选策略,为研究神经保护、神经发育提供更可靠更贴合人脑的实验模型,为减少在再灌注过程中产生负面影响的新型抗氧化剂的研发提供了可能性。这些研究在一定程度上推动了缺血性脑卒中治疗药物研发的新思路,为抗卒中药物从开发到临床的过渡阶段提供了巨大的可能性。与此同时,脑类器官作为移植组织器官在缺血性脑卒中移植治疗中初步崭露头角,移植的脑类器官和宿主脑之间有血管形成,脑类器官显示出多谱系的分化潜力,支持大脑组织区域特异性重建,生成多种必须神经元及突触连接,缓解了缺血性脑卒中大鼠的神经损伤。未来有望通过多种区域特异性的脑类器官融合技术塑造更加贴合人脑组织的神经突触传递和血运网络的移植脑类器官,为缺血性脑卒中再生医学的发展提供新的治疗思路。总的来说,脑类器官技术在推进脑卒中研究和改善治疗方案方面有着巨大的潜力。
脑类器官为缺血性脑卒中研究提供了更接近人类大脑的体外模型,带来了新的机遇,然而,其发展仍受限于模型复杂性、标准化、功能成熟度、转化可行性以及成本技术门槛等诸多瓶颈。目前研究的困境主要有以下几个方面,首先,脑类器官的培养方案在不同实验室间存在差异,分化出类器官在大小、细胞组成、成熟度等方面各有不同,对不同实验室的实验结果相互比较验证难度较大。不能形成统一的类器官培育标准,临床应用标准化生产和可重复性面临了一定的困难。其次,尽管脑类器官能模拟人脑的某些特性,并显示出与人类大脑相似的细胞类型和部分结构,但目前构建的类器官通常缺乏血管系统,无法完全模拟缺血性脑卒中血管阻塞的核心病理,缺少免疫细胞浸润,难以模拟脑卒中后复杂的神经炎症反应以及机械应力、流体压力等微环境信号,与真实体内环境存在差异,这使得其在完全模拟缺血性脑卒中后的复杂病理生理过程方面存在局限。再次,虽然脑类器官为药物筛选提供了新策略,部分研究成功应用于抗缺血性脑卒中药物的验证,但类器官毕竟是简化的体外模型,药物在类器官上显示有效,不能等同于在复杂的人体环境中同样有效且安全,如血脑屏障缺乏直接影响药物的分布和抵达病灶的效率,这些因素限制了类器官用于药物筛选的评价标准和体系的统一。最后,由于建立和维持人类诱导多能干细胞来源的脑类器官培养体系的高成本,专业的细胞培养技术和知识,单细胞转录组、蛋白质组学等多维组学信息的整合,多重因素提高了技术的复杂性和成本,进一步限制了其在许多实验室的广泛应用[98]。未来应加强脑类器官与动物模型和临床研究的衔接,建立标准和质量控制体系,脑类器官技术有望在缺血性脑卒中乃至更广泛的神经科学和药物研发领域发挥更大的作用。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
脑类器官是主要由胚胎干细胞和人类诱导多能干细胞分化产生的一种自组织、三维、器官定向的组织模型。它的形成包括非引导和引导方案,非引导方案利用自组织和细胞间相互作用的特性来生成脑类器官。引导方案可以通过调节小分子来产生大脑区域特异性类器官。现阶段前脑类器官(包括大脑、背侧和腹侧、纹状体、神经节隆起、脉络丛、海马体、丘脑和下丘脑)、中脑和后脑(包括小脑和脑干)区域脑类器官均可以在体外产生。脑类器官主要应用在阿尔茨海默氏病、创伤性脑损伤等脑部疾病中。在缺血性脑卒中研究中的应用仅处于早期阶段。因此概括了脑类器官的细分类型和各分型在脑部疾病中研究的进展,同时主要介绍了缺血性脑卒中涉及的脑类器官研究方向,证明目前应用于缺血性脑卒中研究的脑类器官主要是非引导型和海马体类器官,在药物疗效验证、模型构建和移植再生治疗中均有涉猎,但都未进行深入探讨于研究。因此本综述为未来将不同类型的脑类器官大量应用于缺血性脑卒中的药物筛查、机制建模和再生医学的研究中提供了理论依据,为拓宽缺血性脑卒中的治疗窗口,修复卒中后梗死脑组织提供新的治疗策略。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||