中国组织工程研究 ›› 2026, Vol. 30 ›› Issue (26): 6907-6915.doi: 10.12307/2026.389
• 生物材料综述 biomaterial review • 上一篇 下一篇
纳米生物医学延缓骨关节炎进程的前沿与热点
伍宁缘1,韦智译2,丰 昊2,高 明1
- 1广西医科大学生命科学研究院,广西壮族自治区南宁市 530021;2广西医科大学第一附属医院,广西壮族自治区南宁市 530021
-
接受日期:2025-09-30出版日期:2026-09-18发布日期:2026-03-16 -
通讯作者:高明,博士,研究员,广西医科大学生命科学研究院,广西壮族自治区南宁市 530021 -
作者简介:伍宁缘,男,1998年生,贵州省毕节市人,穿青人,硕士,主要从事生物医学材料研究。 -
基金资助:国家自然科学基金项目(82160430),课题名称:线粒体靶向及功能调控的ROS响应型纳米探针用于骨关节炎诊疗的研究,项目负责人:高明
Frontiers and hot topics of nanobiomedicine in delaying the progression of osteoarthritis
Wu Ningyuan1, Wei Zhiyi2, Feng Hao2, Gao Ming1
- 1Life Sciences Institute, Guangxi Medical University, Nanning 530021, Guangxi Zhuang Autonomous Region, China; 2The First Affiliated Hospital of Guangxi Medical University, Nanning 530021, Guangxi Zhuang Autonomous Region, China
-
Accepted:2025-09-30Online:2026-09-18Published:2026-03-16 -
Contact:Gao Ming, MD, Researcher, Life Sciences Institute, Guangxi Medical University, Nanning 530021, Guangxi Zhuang Autonomous Region, China -
About author:Wu Ningyuan, MS, Life Sciences Institute, Guangxi Medical University, Nanning 530021, Guangxi Zhuang Autonomous Region, China -
Supported by:National Natural Science Foundation of China, No. 82160430 (to GM)
摘要:
文题释义:
纳米生物医学:是融合纳米技术与生物医学的交叉学科,借助纳米材料在纳米尺度的独特物理化学性质,开发靶向药物载体、生物检测传感器、组织工程支架等器件。纳米生物医学的核心是实现疾病的精准诊断、高效治疗及生物功能调控,推动医学向个体化、微创化发展,在癌症治疗、再生医学等领域前景广阔。
骨关节炎:是一种常见的关节疾病,主要由于关节软骨磨损、破坏及关节周围骨质增生等原因引起,常表现为关节疼痛、肿胀、僵硬、活动受限。骨关节炎的发病与年龄、遗传、肥胖、关节损伤、过度使用等多种因素有关。
背景:纳米生物医学的应用能够有效缓解骨关节炎的氧化应激、降低骨关节炎的炎症反应以及促进关节表面修复,从而延缓骨关节炎的发生与发展进程。
目的:综述纳米生物医学延缓骨关节炎进程的研究现状以及未来发展与挑战。
方法:由第一作者检索中国知网、PubMed、Scopus和Web of Science数据库中收录的文章,文献检索时限为各数据库建库至2025年4月,以“纳米生物医学,纳米复合水凝胶,工程化生物体,骨关节炎”为中文检索词,以“nanobiomedicine,nanocomposited hydrogel,engineered organisms,osteoarthritis”为英文检索词,最终选取符合标准的90篇文献进行综述。
结果与结论:目前纳米生物医学的研究进展正在不断更新,其中纳米生物材料成为当前骨关节炎治疗的主流研究方向。纳米生物医学材料具备抗炎抗氧化、软骨修复、精准递药、促进细胞分化和靶向治疗等功能,为骨关节炎的治疗提供了突破传统疗法局限(如药物短效、不良反应大、手术创伤大等缺点)的新方向。纳米生物医学的设计理念基于pH值、酶、温度等多响应机制,呈现智能响应与功能强化的集成特性,从而实现纳米载药体系的精准可控释药。
https://orcid.org/0009-0009-6008-2566 (伍宁缘)
中国组织工程研究杂志出版内容重点:生物材料;骨生物材料;口腔生物材料;纳米材料;缓释材料;材料相容性;组织工程
中图分类号:
引用本文
伍宁缘, 韦智译, 丰 昊, 高 明. 纳米生物医学延缓骨关节炎进程的前沿与热点[J]. 中国组织工程研究, 2026, 30(26): 6907-6915.
Wu Ningyuan, Wei Zhiyi, Feng Hao, Gao Ming. Frontiers and hot topics of nanobiomedicine in delaying the progression of osteoarthritis[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6907-6915.
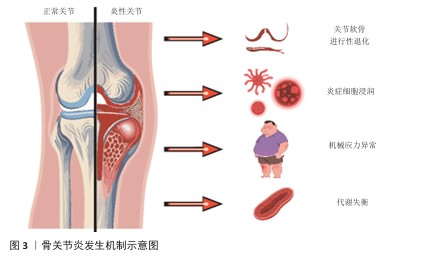
骨关节炎的发病机制是涉及多因素的复杂病理过程,主要包括关节结构的进行性退化、炎症反应、代谢失衡及机械应力等[11](图3)。骨关节炎的核心表现是关节软骨的进行性退化,表现为软骨细胞外基质中Ⅱ型胶原蛋白和蛋白聚糖的合成减少,从而导致软骨结构破
坏[12]。此外,滑膜组织中炎症细胞浸润及释放的炎性因子进一步加剧软骨分解代谢,并抑制合成代谢过程[13]。在分子层面,软骨细胞中活性氧和活性氮水平异常升高,导致线粒体功能障碍、DNA损伤及细胞凋亡,从而加速软骨退化[14]。软骨下骨重塑是骨关节炎的典型特征,成骨细胞过度活跃会形成骨硬化及骨赘[15],造成关节结构畸形和功能受限[16]。代谢失衡影响软骨细胞能量稳态,局部氧化应激环境持续促进细胞凋亡与细胞外基质降解[17-18],最终形成恶性循环。
2.2 骨关节炎的治疗方法 骨关节炎的传统治疗手段主要包括保守治疗和手术治疗两大类,二者协同构成了基于病因、疾病严重程度及影像学分级的阶梯化治疗方案。目前,临床药物治疗为保守治疗的核心,常用药物包括非类固醇抗炎药和皮质类固醇等。在疾病早期的治疗策略以保守干预为主,通过减少关节负荷、口服氨基葡萄糖及非类固醇抗炎药[19],同时联合物理治疗以有效控制症状[20]。当病情进展至中期,患者可采用支具固定或关节内注射治疗[21-22],旨在减轻滑膜炎症及降低机械性损伤。而对于晚期骨关节炎患者,由于关节结构已遭受严重破坏,手术治疗便成为必要手段,常见术式包括关节镜下清理术[23]、截骨术或关节置换术[24-25]。值得注意的是,在整个治疗过程中,肥胖患者的体质量管理将影响治疗全程[26]。
2.3 骨关节炎传统治疗方法的不足 在骨关节炎的保守治疗方法中,非类固醇抗炎药有明显的不良反应,涉及胃肠道、心脑血管等方面[27-28]。长期使用皮质类固醇则可能会导致骨质疏松症、高血压、糖尿病等并发症[29]。此外,该类药物口服时易被肝脏和血浆清除,为达到有效的血液药物浓度往往需要加大剂量,这进一步增加了不良反应风险。骨关节炎的手术治疗方面,常见的关节镜下清理术和关节置换术等虽能有效恢复关节功能,但却存在手术创伤大、术后康复周期长等问题[30],不仅增加了患者的生理痛苦,还带来了沉重的心理压力与经济负担。即便采用关节腔原位注射药物的保守治疗方式,仍面临药物易降解、作用时间短和不良反应大等诸多难题[31-32]。于是科研工作者们将纳米生物材料引入了关节炎治疗的基础研究当中,通过对不同的纳米材料体系、搭载的药物和响应机制进行探索,力求突破现有的治疗困境。
2.4.1 单一纳米材料体系 单一纳米材料体系是指由单一成分构成的纳米尺度材料,可简单分类为金属材料和非金属材料。单一纳米材料体系结构简单、性质稳定,是纳米生物医学研究的基础单元,凭借独特的物理化学性质及生物活性,在骨关节炎治疗中发挥了显著的抗炎及促修复作用。
在金属材料研究领域,人体所需必要金属元素成为研究热点。HUANG等[39]制备了一种新的单原子纳米酶,其中以碳纳米纤维限域的FeN5为活性中心,形成原子分散的类酶活性位点,该结构模拟细胞色素P450中轴向配位血红素的电子推送机制,表现出优异的氧化酶样活性。HU等[40]制备的铁基金属有机框架材料(Mil-88a)通过材料的类过氧化物酶活性清除过量活性氧调节,调控炎症微环境,功能上不仅下调基质金属蛋白酶13基因表达,还能同时促进Ⅱ型胶原合成,使小鼠关节组织OARSI评分得以改善,骨密度显著增加。在另外一项研究中,YU等[41]设计了一种铜基金属有机框架纳米酶用于骨关节炎治疗,该材料具有广谱抗氧化性能,表现类超氧化物歧化酶、类过氧化物氢酶及羟基自由基清除活性,其催化机制通过密度泛函理论计算得以验证,体内外实验均表明该纳米酶能有效降低细胞内活性氧水平、缓解滑膜巨噬细胞的缺氧、促进巨噬细胞从促炎的M1型向抗炎的M2型极化、抑制软骨基质降解,并且展现出良好的生物相容性。
除常见金属外,稀有金属纳米材料也显示出独特治疗潜力。例如,CHEN等[42]将钯单原子纳米酶嵌入介孔硒纳米酶中构建了具有自主靶向能力的双面结构纳米马达(Pd@MSe),利用钯单原子纳米酶中单原子效应增强电子转移能力,不仅大幅提升催化效率,还促进纳米酶扩散,迅速恢复组织氧平衡,从而阻断炎症级联反应、改善线粒体功能并缓解炎症。SHI团队[43]开发了一种氨基修饰的钽纳米颗粒(Ta-NH? NPs)用于治疗骨关节炎,该纳米酶具有类过氧化氢酶活性,可高效清除活性氧、保护软骨细胞,并抑制诱导型一氧化氮合酶表达,在大鼠骨关节炎模型中单次关节注射即实现持久疗效,效果优于天然酶,该纳米颗粒通过电荷调控策略有效延长药物在关节内的滞留时间。此外,一项研究采用近红外响应的钼基多金属氧酸盐纳米团簇,借助 Mo??/Mo??氧化还原循环实现活性氧清除,对超氧阴离子也表现出浓度依赖性清除能力,显著减轻小鼠骨关节炎模型的炎症和软骨破坏[44]。
非金属纳米材料体系具有机械强度高、化学特性稳定、生物相容性良好和自润滑性等特点,显著提升了治疗效果。有研究制备了一种氟化石墨烯量子点多功能纳米系统,不仅具备小尺寸和氟/氧官能团共存的特有结构,还表现出优异的水溶性、生物相容性与自润滑性能,经该体系处理的软骨细胞中Ⅱ型胶原和蛋白聚糖的mRNA表达显著上调,证实该材料具有延缓软骨降解的作用[45]。GAO等[46]研发的硒纳米酶可以清除活性氧、缓解软骨细胞衰老、恢复氧化还原平衡,从而减少细胞外损伤、减轻骨关节炎微环境中的炎症。YU等[47]研发了一种通过开环共聚将磷酸酯单元掺入聚氨基酸并与氧化石墨烯形成共聚物,其中磷酸酯单元的引入增强了细胞黏附特性,通过石墨烯进一步改善了材料的理化性能,最终获得的复合材料显示出良好的临床转化潜力。另有研究构建了基于单壁碳纳米管场效应晶体管的生物传感器,它能够超灵敏检测骨关节炎标志物——软骨酸性蛋白1,为疾病早期诊断与治疗监测提供了新途径[48]。LIU团队[49]近期开发的唾液酸修饰纳米富勒烯衍生物,可借助它与E-选择素的特异性结合实现向炎症关节的靶向分布,从而精准作用于病灶区域,增强抗炎疗效。近些年来,单一纳米材料体系凭借独特的尺寸、表面性质和物理化学特性正逐步改变骨关节炎诊断与治疗的格局,在生物医学领域展现出巨大的应用潜力。
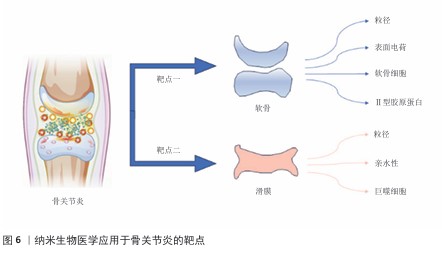
2.4.2 纳米复合型材料 近半个世纪以来,纳米技术在基础研究与应用转化领域发展迅速。其中,将单一纳米材料体系进行复合组装形成纳米复合材料,实现了功能的多样化,在骨关节炎研究中成为了热点方向。从材料构造角度而言,纳米复合材料的制备是通过共价交联或物理作用方式将纳米颗粒或纳米级材料引入搭载体系中[50-51],或二者相互修饰或装载,同时特殊化学基团修饰赋予纳米复合型材料独特的响应机制和精准靶向功能[52](图6)。
HU等[50]开发了一种负载铑和碳酸氢铵的pH值响应型纳米颗粒(Rh-PLGA-NPs@NH?)用于骨关节炎治疗,利用碳酸氢铵与低pH值溶液中的氢离子反应生成二氧化碳和铵根离子,触发纳米颗粒破裂并实现铑的靶向释放,这一pH值响应特性提高了药物在病变部位的生物利用度,同时降低了全身毒性。RONG等[51]通过甲基丙烯酸缩水甘油酯改性的透明质酸作为基质,以光聚合的方式制备一种新型多功能支架用于包封无机颗粒,这不仅实现外源血管内皮生长因子高效负载和控释,其中包封的Fe3O4纳米粒子还可以促进骨髓间充质干细胞的成骨分化,加速软骨缺损区域的新骨形成,还可以在骨关节炎修复过程中进行无创、无辐射的方式来跟踪新生骨组织。LUAN等[52]设计了一种上转换纳米复合材料,集诊断与治疗功能于一体,该体系以核壳结构上转换纳米颗粒与介孔二氧化硅为基础,通过硫缩酮连接体偶联马来酰亚胺-聚乙二醇-酮缩硫醇-N-羟基琥珀酰亚胺酯和β-环糊精,并负载黑洞猝灭剂,在高水平过氧化氢条件下,硫缩酮键断裂释放猝灭剂,恢复荧光以实时监测活性氧水平,同时β-环糊解离,从而诱导间充质干细胞向软骨方向分化。
关节腔在处于炎性环境下不仅pH值显著降低,炎性相关细胞因子表达也发生改变。LAN等[53]在pH值响应的基础上开发了一种基于基质金属蛋白酶13和pH值双响应的智能纳米诊疗平台(MRC-PPL@PSO),该平台以生物相容性聚合物构建pH值响应型胶束,能够在酸性微环境中可控释放中药活性成分补骨脂素;同时,通过偶联软骨靶向肽(WYRGRL)及基质金属蛋白酶13酶敏感肽(GPLGVRGC)实现了对病变软骨的特异性靶向与荧光信号激活,首次将酶响应诊断与pH值响应药物释放整合于同一纳米系统。LI等[54]巧妙地将温度响应与实时炎症检测相结合,开发温度响应型近红外Ⅱ型荧光与表面增强拉曼散射双模态纳米探针,用于长期监测大鼠关节炎温度变化,探针由负载近红外Ⅱ型荧光染料的温敏聚合物接枝多孔金纳米壳构成,通过比率信号(FL????/FL????与S????/S????)可逆响应温度变化,实现无辐射、实时动态的温度监测,为关节炎早期诊断与疗效评估提供了新型可视化工具。在机制方面的研究中,FAN团队[55]采用软骨细胞膜伪装并修饰三苯基溴化膦的空心普鲁士蓝纳米酶,构建了一种兼具细胞选择性、溶酶体逃逸及线粒体靶向功能的纳米系统,该体系可精确清除活性氧、减轻线粒体DNA泄漏,进而抑制环磷酸鸟苷-腺苷合成酶–干扰素刺激基因和核因子κB信号通路的激活,从免疫微环境调控角度延缓骨关节炎的进展。LI等[56]致力于实现持续可控的局部基因治疗,利用精氨酸-组氨酸-苯丙氨酸修饰的第5代聚酰胺树状大分子(G5-AHP)与miR-140形成复合物,构建多功能纳米递送系统,该系统可增强内吞作用、提高基因转染效率,通过维持软骨基质代谢平衡发挥治疗作用。目前纳米复合型材料被发现具有优化的治疗性能,为骨关节炎治疗提供了更多的临床选择。
2.4.3 纳米载药体系 如表1所示,新型的纳米载药体系同时兼具多种优点,例如增加药物递送的准确性、提高药物的稳定性、可控释放药物和降低生物毒性作用。YU等[57]将伊卡瑞林与单宁酸共同锚定于纳米颗粒表面,实现对骨关节炎的协同治疗,体外实验表明该体系可显著抑制基质金属蛋白酶3和环氧化酶2等促炎因子表达;体内实验进一步证实该材料有效缓解了大鼠模型的软骨退化与关节结构破坏。水凝胶用于药物的搭载释放一直是人们关注的研究方向,它通过调控药物物理状态或载体结构实现药物的长效释放,满足关节内治疗对持续药效的需求。HOU等[58]由此开发了一种独特且创新的治疗平台,这种载药体系通过将药物芦丁掺入黑磷纳米片中,再整合到透明质酸和聚乙烯醇基质中以形成纳米载药水凝胶,利用光热转换效应实现了近红外照射下的光热疗法,并与抗炎药物实现了协同治疗。这样的组合方式显著增强了原有药物的治疗效果,而且可以借助外源动力实现协同治疗。YAO等[59]通过点击化学将聚乙二醇、促软骨分化药物Kartogenin、润滑剂氢化大豆磷脂酰胆碱及荧光素结合于载药平台,构建了兼具荧光可视化、润滑与药物缓释功能的多功能载药体系,再利用微流控技术将其封装于透明质酸甲基丙烯酸酯水凝胶微球中,借助荧光信号可以实时监测软骨修复进程。同时,具备靶向性的纳米载药体系取得很大的研究进展,通过材料修饰或机制设计实现对病变部位的精准识别与药物释放,增强局部疗效的同时还减少了全身不良反应。HUANG等[60]利用叶酸特异性结合骨关节炎中M1型巨噬细胞表面过表达的叶酸受体β,将中药黄芩苷负载于金属有机框架合成纳米载药体系,借助独特的高孔隙率和pH值响应性在关节酸性环境中缓释药物,同时通过清除活性氧和调控巨噬细胞向M2方向极化缓解滑膜炎症与软骨退化,显著降低关节活性氧水平并改善软骨结构。
基于炎症条件下相关酶的表达差异,研究者开发了多种靶向递药策略以实现精准治疗。BEDINGFIELD等[61]制备了聚乳酸-羟基乙酸共聚物微米板靶向基质金属蛋白酶13,通过封装的小干扰RNA于pH值响应性纳米颗粒实现了靶向释放小干扰RNA,从而减少软骨降解与滑膜增生。除了酶靶向策略以外,特定的炎性微环境靶向也成为新的研究热点。有研究利用钙依赖膜修复机制实现了非侵入性细胞内靶向递送,利用光穿孔技术结合上转换纳米颗粒与金纳米颗粒,利用上转换纳米颗粒将近红外光转换为可见光激活金纳米颗粒等离子共振效应,在骨关节炎模型中靶向胞内高效递送巴瑞替尼,促进了软骨细胞分化及细胞外基质的表达[62]。纳米载药平台的加入弥补了治疗药物水溶性有限、半衰期短和生物利用度低等不足,这样的策略在纳米复合型材料的研究中具有深远的指导意义。
对于具体的机制通路研究中,ZHOU等[63]开发了一种抗炎超分子纳米纤维水凝胶(HAS2@DexP-Gel),通过负载编码透明质酸合成酶2的慢病毒和地塞米松磷酸钠将基因治疗与抗炎药物结合,该缓释系统显著延长了药物与病毒的作用时间,通过单细胞RNA测序证实该体系可调节滑膜成纤维细胞亚群比例、促进M2型巨噬细胞极化,并抑制磷脂酰肌醇3-激酶等信号通路,从而改善滑膜炎症微环境。WANG等[64]将甲氨蝶呤负载于乙酰化葡聚糖分子中构建了具有靶向性和pH值响应性的纳米药物系统,不仅降低了甲氨蝶呤的全身毒性,还增强其治疗效能,该体系可在炎症部位富集并释放药物,通过调节激酶信号传导与激活转录蛋白通路延缓骨关节炎的进展。在与中医学领域的结合研究中,XU等[65]创新性地将传统针灸与纳米递送结合,在针灸针表面修饰甲基水杨酸-环糊精复合物,靶向骨关节炎局部微环境,通过调节高迁移蛋白1表达的信号通路和抑制鸟苷三磷酸水解酶活性有效缓解疼痛并延缓软骨退化,验证了纳米药物和针灸协同治疗的潜力。REN等[66]构建了可靶向递送抗炎药并模拟针灸缓解疼痛的穴位给药系统,由雷公藤内酯、环戊基腺苷和人血清白蛋白纳米颗粒组成,相关实验结果表明此体系可特异性增强穴位镇痛效果,还可以调节炎症因子、恢复T细胞免疫的平衡,拥有更好的治疗效果和更低的药物毒性。此外,CHENGLONG等[67]利用依那西普竞争性抑制肿瘤坏死因子α与受体的结合,在炎症关键部位激发免疫调节反应,为关节炎治疗提供了新策略。随着纳米载药体系研究的深入,它在骨关节炎治疗上的应用价值正从单一药物递送平台向多功能治疗系统升级,从靶向递送、智能响应、协同治疗等多维度展开,既体现研究深度又衔接临床转化。

2.4.4 生物杂化材料 生物杂化材料是由生物材料与合成材料通过物理、化学或生物方法结合而成,兼具两者优势,其中包含细胞外泌体(细胞和细菌等来源,表2)和细胞疗法等,生物杂化材料不仅可以作为高效且精准的药物递送体系,还能经过工程化改造后为关节组织的再生治疗提供更多选择,在关节炎治疗领域展现出广阔前景。在脂质材料外泌体的研究中,通过化学修饰和装载内容物等方法进行功能的附加应用于骨关节炎治疗,例如,TANG等[68]提出了一种新型的固有抗炎外泌体,通过电穿孔法整合小鼠单核巨噬细胞衍生的外泌体和强大的抗炎免疫调节剂白细胞介素10,使用无创超声来增加该体系对血管的通透性,从而促进巨噬细胞极化为M2表型,其中抗炎和促消炎的联合机制为治疗骨关节炎和其他炎症性疾病提供了独特的见解。YANG等[69]利用改性天然多糖、透明质酸和硫酸软骨素组成微载体、改性的间充质干细胞外泌体作为搭载平台,内部封装的磁性纳米粒子能够捕获干细胞外泌体和封装药物双氯芬酸钠,微载体释放的双氯芬酸钠和外泌体在缓解骨关节炎症状和促进软骨修复方面展现了强大的协同治疗能力。YANG
等[70]探讨人脐带间充质干细胞分泌外泌体对骨关节炎大鼠模型的治疗作用,发现人脐带间充质干细胞外泌体促进骨关节炎大鼠的软骨再生,并且该组中滑液细胞因子、肿瘤坏死因子α、白细胞介素1β和白细胞介素6的水平均低于对照组;对于白细胞介素1β刺激的软骨细胞,外泌体处理可显著下调细胞中基质金属蛋白酶13和血小板反应蛋白解整合素金属肽酶5的表达、上调Ⅱ型胶原的表达。MENG等[71]研究脂肪组织来源基质细胞外泌体在骨关节炎治疗中的作用,发现软骨细胞与脂肪组织来源的基质细胞均可分泌外泌体,脂肪组织来源的基质细胞衍生外泌体可被软骨细胞吸收,而且相比软骨细胞外泌体可以分泌更高水平的微小RNA-429,通过荧光素酶测定证明微小RNA-429直接靶向束缚和伸长蛋白进而激活自噬通路,从而改善软骨损伤。
除动物细胞来源的外泌体外,植物源性外泌体同样成为骨关节炎治疗的研究热点。LIU等[72]从大蒜中提取出一种纳米颗粒,它的主要成分为大蒜衍生的外泌体样结构,体外实验表明该外泌体可显著促进Ⅱ型胶原和聚集蛋白聚糖的表达、抑制基质金属蛋白酶3和基质金属蛋白酶9的表达,机制研究进一步揭示该外泌体能够抑制丝裂原活化蛋白激酶的磷酸化,从而减轻软骨降解。LIANG等[73]从光螺旋藻中提取了细胞外泌体,为了促进细胞外泌体的持续递送,使用大黄酸水凝胶系统进行关节内注射,该体系在弱酸性条件下表现出pH值响应性和协同抗炎作用,减缓了炎症介导的氧化应激状态,并恢复软骨细胞中的线粒体膜电位,通过调节Janus激酶-信号传导和转录激活蛋白通路发挥协同抗炎作用。RASHIDI等[74]巧妙地从葡萄柚汁中分离外泌体作为骨关节炎新治疗方法,一方面,抑制环氧化酶 2、前列腺素内过氧化物合酶 2等炎症标志物表达,降低疼痛敏感性;另一方面,增强抗氧化基因活性、下调血小板反应蛋白解整合素金属肽酶5和肥厚型胶原表达、上调Ⅱ型胶原等软骨形成标志物表达,实现抗炎与促软骨再生的协同效应。这些研究标志着外泌体用于骨关节炎治疗的重大进步,将为未来研究提供全新的思路。
细菌与真菌来源的外泌体也在骨关节炎研究中展现出潜力。GONG等[75]将大鼠骨髓间充质干细胞与细菌来源的外泌体共培养制备了具有超声成像能力的材料体系,在与甲氨蝶呤的联合治疗进程中可显著增强甲氨蝶呤的疗效,使得关节炎指数评分降低、骨侵蚀和软骨破坏减少。为了寻找新的炎症作用靶点,HONG等[76]将肠道微生物群来源的外泌体注入到关节后其自身毒性引发局部炎症反应,作用于滑膜巨噬细胞后激活参与炎症途径的相关因子以及炎症递质的关键调节因子Y盒结合蛋白1,为临床治疗寻找新型干预靶点提供了选择。值得一提的是,细胞疗法应用于骨关节炎的研究较为成熟,很多研究成果已进入临床使用,例如,AO等[77]对14例接受反复关节内注射间充质干细胞的骨关节炎患者进行了为期3个月的随访评估,所有患者接受干细胞治疗后均表现出膝关节功能受限和疼痛的一定改善,其中有5例(35.7%)出现短暂性不良反应并自行消退。还有研究将10例骨关节炎患者自体骨髓间充质干细胞通过原位注射方法注入受损关节,随访评估发现这是一种安全且有效恢复关节功能和活动范围的治疗方法[78]。CAO等[79]将间充质干细胞注入502例骨关节炎患者的患病关节,同样发现间充质干细胞可以显著改善短期和长期治疗效果,亚组分析显示脂肪来源和高剂量的间充质干细胞疗效更加显著。在此类研究中,生物杂化材料融合细胞、植物及细菌真菌外泌体与细胞疗法等多类材料的优势,在未来基础医学研究与临床实践中发挥多手段治疗的作用,持续推动纳米智能诊疗体系的发展。

2.4.5 纳米复合水凝胶 纳米复合水凝胶在骨关节炎治疗中可通过多途径发挥作用:一方面,能够优化关节内力学微环境;另一方面,可实现靶向治疗药物的负载,同时还能诱导未分化细胞向软骨方向分化,以此加速软骨修复进程并调节关节内炎性微环境,最终达成治疗骨关节炎的目标。WU等[80]开发了一种臭氧富集的热响应纳米复合水凝胶,该材料通过将臭氧封装于全氟三丁胺与氟化透明质酸构建的纳米颗粒中,并与羟丙基几丁质的α-氨基进行偶联以增强稳定性,体外细胞实验表明该体系可显著降低血管内皮生长因子及白细胞介素1β、白细胞介素6和肿瘤坏死因子α等炎症因子水平,从而减轻滑膜炎症、抑制软骨破坏与软骨下骨异常重塑,有效缓解骨关节炎进展。ZHANG团队[81]设计了一种基于沸石咪唑酯框架8负载槲皮素的可注射水凝胶,该体系具备pH值与活性氧双重响应特性,可实现生物活性物质的控释并调节骨关节微环境,显著减轻滑膜炎性反应与软骨基质降解,促进软骨组织修复。这种开发的热敏可注射水凝胶可有效平衡润滑与生物活性物质的受控释放,代表了一种非常有前途的骨关节炎治疗方法。XU等[82]利用多巴胺中Mg2+与儿茶酚胺键之间的金属螯合作用成功构建了多功能纳米颗粒(DIC/Mg-PDA NPs),然后使用双氯芬酸进行酰胺化,再通过微流控技术制备成具有免疫调节和软骨修复能力的可注射水凝胶微球(DIC/Mg-PDA@HM),该微球可诱导巨噬细胞M1型转化为M2型,激活炎症的免疫治疗,并促进骨髓间充质干细胞的分化以实现软骨再生。在具体的治疗机制探索中,HALOI等[83]以异硫氰酸苯酯为基础制备了壳聚糖/普朗尼克F-127智能水凝胶,性能测试结果证明了该材料体系拥有足够的热敏性,注射入骨关节炎大鼠模型后显著降低了骨关节炎相关症状并激活抗炎活性通路。当前,纳米复合水凝胶在骨关节炎领域的研究仍存在广阔的探索空间,持续推进材料制备工艺的优化,并深入开展细胞水平及动物模型层面的实验研究,有望为骨关节炎的临床治疗开拓新的路径,它独特的理化性质和良好的生物相容性也展现出了在生物医学领域研究中广阔的发展前景。
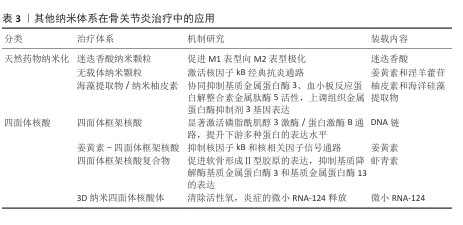
2.4.6 其他纳米体系 如表3所示,该章节主要讨论天然药物纳米化和四面体核酸的研究进展。在其他类型的纳米体系中,药物纳米化是将天然药物制成纳米级别的颗粒,纳米级别的尺寸让药物具有更大的比表面积,能显著提高药物的溶解速度和生物利用度,使得药物更容易穿透生物膜而被关节组织吸收。LU等[84]报告了一种独特的自组装策略,使用天然存在的多酚基化合物迷迭香酸构建独特的迷迭香酸纳米颗粒,它可促进巨噬细胞从M1表型向M2表型极化,从而抑制骨关节炎的发展。同样的思路,DAI等[85]将姜黄素与淫羊藿苷通过π-π堆积自组装形成无载体纳米颗粒,该材料的pH值响应释放特性在关节腔酸性环境中48 h累积释药率增加,姜黄素的自发荧光可实现体内实时追踪,显著抑制白细胞介素1β诱导的核因子kB典型抗炎通路激活,促进软骨细胞增殖。还有研究通过减小纳米柚皮素粒径至100 nm使其生物可利用度提升,与海洋硅藻提取物协同抑制基质金属蛋白酶3、血小板反应蛋白解整合素金属肽酶5活性、上调组织金属蛋白酶抑制剂3基因表达、减轻滑膜炎症及软骨胶原纤维破坏,效果与临床常用药物吲哚美辛相当,但无明显胃肠道不良反应[86]。
四面体核酸体是一种由4条 DNA 链自组装形成的纳米级核酸结构,具有高度的稳定性和独特的三维纳米结构。特殊结构赋予四面体核酸体多种功能,使它能够作为药物载体,通过表面修饰实现治疗分子的高效负载与靶向递送。FU等[87]提出使用四面体框架核酸(tFNA)作为体外培养环境中管理间充质干细胞行为的新方法,结合生物信息学方法探究了四面体框架核酸对间充质干细胞增殖、迁移和成软骨分化的影响,实验结果显示磷脂酰肌醇3激酶/蛋白激酶B通路被显著激活,提升了下游多种蛋白的表达水平,从而促进了软骨的分化。四面体核酸体还可以作为纳米载体搭载天然药物姜黄素,例如,ZHANG等[88]使用四面体框架核酸作为姜黄素的载体,它的载药效率较高,在溶液中分散良好且稳定性优于游离姜黄素,体外实验表明该复合物能显著提高小鼠单核巨噬细胞对姜黄素的摄取,通过调控核因子kB和核相关因子信号通路有效降低炎症因子的分泌,实现抗炎和抗氧化的协同作用。聚焦于骨关节炎治疗难题,还有实验研究了负载虾青素的四面体框架核酸复合物的治疗效果与机制,该复合物呈均匀分散的四面体结构,在血清中的稳定性良好,还能促进软骨形成Ⅱ型胶原的表达、抑制基质降解酶基质金属蛋白酶3和基质金属蛋白酶13的表达[89]。四面体框架核酸自组装三维立体DNA纳米结构具有优越的固有生物相容性、多功能性和强大的细胞内化能力。SHI等[90]成功合成了携带1个或3个微小RNA-124的3D纳米四面体核酸体,该材料凭借其强大的活性氧清除能力抑制了炎症环境中微小RNA-124的释放行为,证明了该体系在体外可有效保护软骨细胞免受白细胞介素1β损伤。
2.4.7 小结 该文系统综述了单一纳米体系、纳米复合型材料、纳米载药体系、纳米复合水凝胶、生物杂化材料以及其他纳米体系在骨关节炎治疗中的应用,深入分析了它们的独特优势,介绍了相关的工程化策略,包括化学修饰、内容物装载和化学修饰联合内容物装载等,这些策略赋予了纳米生物医学研究的多功能化,包括卓越的生物相容性、生物降解性、提高患者治疗体验、微创化和生物安全性,使纳米生物医学成为生物应用中有前途且有吸引力的治疗纳米平台。
| [1] TANIMURA C, TOKUSHIMA Y, YOSHIMURA J, et al. Effectiveness of an Instructional Design-Based Self-Management Program for Patients with Knee Osteoarthritis. Yonago Acta Med. 2025;68(1): 22-33. [2] 李远超,赵冰冰,杨畅然,等.聚焦式低强度脉冲超声波在膝骨关节炎治疗中的应用研究进展[J]. 华西医学,2023,38(1):134-139. [3] WOODELL-MAY J, STECKBECK K, KING W. Potential Mechanism of Action of Current Point-of-Care Autologous Therapy Treatments for Osteoarthritis of the Knee-A Narrative Review. Int J Mol Sci. 2021;22(5):2726. [4] LAEV SS, SALAKHUTDINOV NF. Anti-arthritic agents: progress and potential. Bioorg Med Chem. 2015;23(13):3059-3080. [5] XIAO S, TANG Y, LV Z, et al. Nanomedicine - advantages for their use in rheumatoid arthritis theranostics. J Control Release. 2019;316: 302-316. [6] RAO C, SHI S. Development of Nanomaterials to Target Articular Cartilage for Osteoarthritis Therapy. Front Mol Biosci. 2022;9:900344. [7] 赵勇,齐元玲.谈骨论筋,养护关节[J].中医健康养生,2025,11(3):35-37. [8] 刘维.痛风及高尿酸血症中西医结合诊疗指南[J].中医杂志,2023,64(1):98-106. [9] WAN Y, SHEN K, YU H, et al. Baicalein limits osteoarthritis development by inhibiting chondrocyte ferroptosis. Free Radic Biol Med. 2023;196:108-120. [10] REN X, ZHUANG H, LI B, et al. Gsmtx4 Alleviated Osteoarthritis through Piezo1/Calcineurin/NFAT1 Signaling Axis under Excessive Mechanical Strain. Int J Mol Sci. 2023;24(4):4022. [11] LIANG Q, DING Q, ZHAO L, et al. USP15-modified ADMSCs-Exo alleviates chondrocyte damage and effectively relieved osteoarthritis by inducing M2 polarization of macrophages through deubiquitinating FOXC1. J Orthop Surg Res. 2025; 20(1):336. [12] HOLZER LA, KRAIGER M, TALAKIC E, et al. Microstructural analysis of subchondral bone in knee osteoarthritis. Osteoporos Int. 2020; 31(10):2037-2045. [13] LIU L, HU F, WANG H, et al. Secreted Protein Acidic and Rich in Cysteine Mediated Biomimetic Delivery of Methotrexate by Albumin-Based Nanomedicines for Rheumatoid Arthritis Therapy. ACS Nano. 2019;13(5):5036-5048. [14] O’NEILL TW, FELSON DT. Mechanisms of Osteoarthritis (OA) Pain. Curr Osteoporos Rep. 2018;16(5):611-616. [15] FANG C, GUO JW, WANG YJ, et al. Diterbutyl phthalate attenuates osteoarthritis in ACLT mice via suppressing ERK/c-fos/NFATc1 pathway, and subsequently inhibiting subchondral osteoclast fusion. Acta Pharmacol Sin. 2022;43(5): 1299-1310. [16] SHEN J, LI S, CHEN D. TGF-β signaling and the development of osteoarthritis. Bone Res. 2014; 2:14002. [17] XIANG J, YANG X, TAN M, et al. NIR-enhanced Pt single atom/g-C(3)N(4) nanozymes as SOD/CAT mimics to rescue ATP energy crisis by regulating oxidative phosphorylation pathway for delaying osteoarthritis progression. Bioact Mater. 2024;36:1-13. [18] BENNETT CF, LATORRE-MURO P, PUIGSERVER P. Mechanisms of mitochondrial respiratory adaptation. Nat Rev Mol Cell Biol. 2022;23(12): 817-835. [19] BANNURU RR, OSANI MC, VAYSBROT EE, et al. OARSI guidelines for the non-surgical management of knee, hip, and polyarticular osteoarthritis. Osteoarthr Cartil. 2019;27(11):1578-1589. [20] KOLASINSKI SL, NEOGI T, HOCHBERG MC, et al. 2019 American College of Rheumatology/Arthritis Foundation Guideline for the Management of Osteoarthritis of the Hand, Hip, and Knee. Arthritis Care Res (Hoboken). 2020;72(2):149-162. [21] ROGOSCHIN J, PREISER T, HANSEN L, et al. Kinetic effects of unicompartmental unloader braces in medial knee osteoarthritis: A longitudinal study of joint load during level, stair, and slope walking. J Biomech. 2025;184:112593. [22] AYHAN E, KESMEZACAR H, AKGUN I. Intraarticular injections (corticosteroid, hyaluronic acid, platelet rich plasma) for the knee osteoarthritis. World J Orthop. 2014;5(3):351-361. [23] OZEKI N, SEIL R, KRYCH AJ, et al. Surgical treatment of complex meniscus tear and disease: state of the art. J Isakos. 2021;6(1):35-45. [24] PENG H, OU A, HUANG X, et al. Osteotomy Around the Knee: The Surgical Treatment of Osteoarthritis. Orthop Surg. 2021;13(5):1465-1473. [25] BEZERRA JCP, SANTOS M, ROCHA EAB, et al. Clinical effects of knee arthroplasty. Rev Assoc Med Bras(1992). 2023;69(6):e20230160. [26] BARROW DR, ABBATE LM, PAQUETTE MR, et al. Exercise prescription for weight management in obese adults at risk for osteoarthritis: synthesis from a systematic review. BMC Musculoskelet Disord. 2019;20(1):610. [27] BINDU S, MAZUMDER S, BANDYOPADHYAY U. Non-steroidal anti-inflammatory drugs (NSAIDs) and organ damage: A current perspective. Biochem Pharmacol. 2020;180:114147. [28] ROGOVEANU OC, STREBA CT, VERE CC, et al. Superior digestive tract side effects after prolonged treatment with NSAIDs in patients with osteoarthritis. J Med Life. 2015;8(4):458-461. [29] POFI R, CARATTI G, RAY DW, et al. Treating the Side Effects of Exogenous Glucocorticoids; Can We Separate the Good From the Bad? Endocr Rev. 2023;44(6):975-1011. [30] BAYLISS LE, CULLIFORD D, MONK AP, et al. The effect of patient age at intervention on risk of implant revision after total replacement of the hip or knee: a population-based cohort study. Lancet. 2017;389(10077):1424-1430. [31] TSCHOPP M, PFIRRMANN CWA, FUCENTESE SF, et al. A Randomized Trial of Intra-articular Injection Therapy for Knee Osteoarthritis. Invest Radiol. 2023;58(5):355-362. [32] 韩文英,阮海华.透明质酸治疗骨关节炎的研究进展[J].生物学杂志,2025,42(2): 99-106. [33] 郑珊珊,蔡悦,龚瑜贝,等.纳米载体用于包载生物活性多肽的研究进展[J].中国生物医学工程学报,2023,42(2):242-251. [34] MA L, ZHENG X, LIN R, et al. Knee Osteoarthritis Therapy: Recent Advances in Intra-Articular Drug Delivery Systems. Drug Des Devel Ther. 2022; 16:1311-1347. [35] FAUSTINO C, DUARTE N, PINHEIRO L. Triterpenes Drug Delivery Systems, a Modern Approach for Arthritis Targeted Therapy. Pharmaceuticals (Basel). 2023;17(1):54. [36] LIN C, WANG X, LIU N, et al. Characterization and Evaluation of HGF-Loaded PLGA Nanoparticles in a CCl4-Induced Acute Liver Injury Mouse Model. J Nanomater. 2019;2019(1):7936143. [37] KÜÇÜKTÜRKMEN B, ÖZ UC, BOZKIR A. In Situ Hydrogel Formulation for Intra-Articular Application of Diclofenac Sodium-Loaded Polymeric Nanoparticles. Turk J Pharm Sci. 2017;14(1):56-64. [38] YANG N, LI M, WU L, et al. Peptide-anchored neutrophil membrane-coated biomimetic nanodrug for targeted treatment of rheumatoid arthritis. J Nanobiotechnology. 2023;21(1):13. [39] HUANG L, CHEN J, GAN L, et al. Single-atom nanozymes. Sci Adv. 2019;5(5):eaav5490. [40] HU H, HUANG X, DAI Y, et al. Organic metal matrix Mil-88a nano-enzyme for joint repair in the osteoarthritis mouse model. Front Bioeng Biotechnol. 2023;11:1164942. [41] YU B, SUN W, LIN J, et al. Using Cu-Based Metal-Organic Framework as a Comprehensive and Powerful Antioxidant Nanozyme for Efficient Osteoarthritis Treatment. Adv Sci. 2024;11(13): 2307798. [42] CHEN X, YANG Y, CHEN J, et al. Dual-driven selenium Janus single-atom nanomotors for autonomous regulating mitochondrial oxygen imbalance to catalytic therapy of rheumatoid arthritis. Redox Biol. 2025;81:103574. [43] JIANG Y, LI T, YANG J, et al. Sustained intra-articular reactive oxygen species scavenging and alleviation of osteoarthritis by biocompatible amino-modified tantalum nanoparticles. Front Bioeng Biotechnol. 2023;11:1118850. [44] SHI G, JIANG H, YANG F, et al. NIR-responsive molybdenum (Mo)-based nanoclusters enhance ROS scavenging for osteoarthritis therapy. Pharmacol Res. 2023;192:106768. [45] GONG P, QI C, WANG D, et al. Fluorinated graphene quantum dots with long-term lubrication for visual drug loading and joint inflammation therapy. Friction. 2023;11(12): 2204-2220. [46] GAO W, LI S, MIAO Y, et al. Selenium nanozyme-crosslinked composite hydrogel for promoting cartilage regeneration in osteoarthritis via an integrated ‘outside-in’ and ‘inside-out’ strategy. J Colloid Interface Sci. 2025;693:137612. [47] YU Z, XING F, LI J, et al. 3D Printed Polycaprolactone/Phosphoester-Modified Poly (amino acids)-Graphene Oxide Scaffold for Meniscal Regeneration.J Mater Chem B. 2025. doi: 10.1039/d5tb00012b. [48] LV T, LIU J, LI F, et al. Label-Free and Ultrasensitive Detection of Cartilage Acidic Protein 1 in Osteoarthritis Using a Single-Walled Carbon Nanotube Field-Effect Transistor Biosensor. ACS Appl Mater Interfaces. 2024;16(28):36804-36810. [49] LIU L, LI X, WU Z, et al. Inflammation-targeting Fullerene Nanoparticles Dually Inhibit Macrophage and Osteoclast Differentiation for Mitigating Rheumatoid Arthritis. CCS Chem. 2024;6(9): 2275-2288. [50] HU B, GAO F, LI C, et al. Rhein laden pH-responsive polymeric nanoparticles for treatment of osteoarthritis. AMB Express. 2020;10(1):158. [51] RONG M, LIU D, XU X, et al. A Superparamagnetic Composite Hydrogel Scaffold as In Vivo Dynamic Monitorable Theranostic Platform for Osteoarthritis Regeneration. Adv Mater. 2024; 36(35):2405641. [52] LUAN Z, MA X, ZHAO Q, et al. Integrated diagnosis and treatment” of upconversion nanocomposite hydrogel for osteoarthritis treatment. Chem Eng J. 2024;488:150611. [53] LAN Q, LU R, CHEN H, et al. MMP-13 enzyme and pH responsive theranostic nanoplatform for osteoarthritis. J Nanobiotechnology. 2020; 18(1):117. [54] LI Q, XIAO S, GE X, et al. Temperature-Activated Near-Infrared-II Fluorescence and SERS Dynamic-Reversible Probes for Long-Term Assessment of Osteoarthritis In Vivo. Angew Chem Int Ed. 2024;63(35):e202408792. [55] FAN Y, NIU Z, YIN L, et al. Membrane biomimetic nanoenzyme-incorporated hybrid glycyrrhizic acid hydrogel for precise mitochondrial ROS scavenging for osteoarthritis treatment. Materials Today Bio. 2025;32:101778. [56] LI B, WANG F, HU F, et al. Injectable “nano-micron” combined gene-hydrogel microspheres for local treatment of osteoarthritis. NPG Asia Materials. 2022; 14(1):1. [57] YU Y, KIM SM, PARK K, et al. Therapeutic Nanodiamonds Containing Icariin Ameliorate the Progression of Osteoarthritis in Rats. Int Journal Mol Sci. 2023;24(21):15977. [58] HOU J, YIN S, JIAO R, et al. The combination of hydrogels and rutin-loaded black phosphorus nanosheets treats rheumatoid arthritis. Mater Today Bio. 2024;29:101264. [59] YAO Y, WEI G, DENG L, et al. Visualizable and Lubricating Hydrogel Microspheres Via NanoPOSS for Cartilage Regeneration. Adv Sci. 2023;10(15):2207438. [60] HUANG L, YAO Y, RUAN Z, et al. Baicalin nanodelivery system based on functionalized metal-organic framework for targeted therapy of osteoarthritis by modulating macrophage polarization. J Nanobiotechnology. 2024;22(1):221. [61] BEDINGFIELD SK, COLAZO JM, DI FRANCESCO M, et al. Top-Down Fabricated microPlates for Prolonged, Intra-articular Matrix Metalloproteinase 13 siRNA Nanocarrier Delivery to Reduce Post-traumatic Osteoarthritis. ACS Nano. 2021;15(9):14475-14491. [62] KIM HJ, CHO HB, KIM HR, et al. Upconverting-photon quenching-mediated perforation influx as an intracellular delivery method using posAuNP@UCNPs nanocomposites for osteoarthritis treatment. Nano Converg. 2024;11(1):1. [63] ZHOU F, CHEN M, QIAN Y, et al. Enhancing Endogenous Hyaluronic Acid in Osteoarthritic Joints with an Anti-Inflammatory Supramolecular Nanofiber Hydrogel Delivering HAS2 Lentivirus. Small. 2024;20(40):2400542. [64] WANG X, CAO W, SUN C, et al. Development of pH-sensitive dextran-based methotrexate nanodrug for rheumatoid arthritis therapy through inhibition of JAK-STAT pathways. Int J Pharmaceut. 2022;622:121874. [65] XU W, XIAO Y, ZHAO M, et al. Effective Treatment of Knee Osteoarthritis Using a Nano-Enabled Drug Acupuncture Technology in Mice. Adv Sci. 2023;10(28):2302586. [66] REN S, LIU H, WANG X, et al. Acupoint nanocomposite hydrogel for simulation of acupuncture and targeted delivery of triptolide against rheumatoid arthritis. J Nanobiotechnology. 2021;19(1):409. [67] CHENGLONG L, GUANTING L, YUE J, et al. A Novel Etanercept-loaded Nano-emulsion for Targeted Treatment of Inflammatory Arthritis via Draining Lymph Node. Curr Drug Deliv. 2024;21(8): 1106-1113. [68] TANG Y, WU Z, GUO R, et al. Ultrasound-augmented anti-inflammatory exosomes for targeted therapy in rheumatoid arthritis. J Mater Chem B. 2022;10(38):7862-7874. [69] YANG L, LI W, ZHAO Y, et al. Magnetic Polysaccharide Mesenchymal Stem Cells Exosomes Delivery Microcarriers for Synergistic Therapy of Osteoarthritis. ACS Nano. 2024;18(31): 20101-20110. [70] YANG H, ZHOU Y, YING B, et al. Effects of human umbilical cord mesenchymal stem cell-derived exosomes in the rat osteoarthritis models. Stem Cells Transl Med. 2024;13(8):803-811. [71] MENG C, NA Y, HAN C, et al. Exosomal miR-429 derived from adipose-derived stem cells ameliorated chondral injury in osteoarthritis via autophagy by targeting FEZ2. Int Immunopharmacol. 2023;120:110315. [72] LIU Y, NIE M, LI X, et al. Garlic-derived Exosomes Alleviate Osteoarthritis Through Inhibiting the MAPK Signaling Pathway. Appl Biochem Biotech. 2025;197(1):518-533. [73] LIANG F, ZHENG Y, ZHAO C, et al. Microalgae-Derived Extracellular Vesicles Synergize with Herbal Hydrogel for Energy Homeostasis in Osteoarthritis Treatment. ACS Nano. 2025;19(8): 8040-8057. [74] RASHIDI N, LIU C, GUILLOT PV, et al. Isolation, Characterization, and In Vitro Cell Studies of Plant-Based Exosome-like Nanovesicles for Treatment of Early Osteoarthritis. Int J Mol Sci. 2025;26(5): 2211. [75] GONG Z, HE Y, ZHOU M, et al. Ultrasound imaging tracking of mesenchymal stem cells intracellularly labeled with biosynthetic gas vesicles for treatment of rheumatoid arthritis. Theranostics. 2022;12(5):2370-2382. [76] HONG M, LI Z, LIU H, et al. <em>Fusobacterium nucleatum</em> aggravates rheumatoid arthritis through FadA-containing outer membrane vesicles. Cell Host Microbe. 2023;31(5):798-810.e7. [77] AO Y, DUAN J, XIONG N, et al. Repeated intra-articular injections of umbilical cord-derived mesenchymal stem cells for knee osteoarthritis: a phase I, single-arm study. BMC Musculoskel Dis. 2023;24(1):488. [78] MARDONES R, JOFRÉ CM, TOBAR L, et al. Mesenchymal stem cell therapy in the treatment of hip osteoarthritis. J Hip Preserv Surg. 2017; 4(2):159-163. [79] CAO M, OU Z, SHENG R, et al. Efficacy and safety of mesenchymal stem cells in knee osteoarthritis: a systematic review and meta-analysis of randomized controlled trials. Stem Cell Res Ther. 2025;16(1):122. [80] WU H, WANG J, LIN Y, et al. Injectable Ozone-Rich Nanocomposite Hydrogel Loaded with D-Mannose for Anti-Inflammatory and Cartilage Protection in Osteoarthritis Treatment. Small. 2024;20(25): 2309597. [81] ZHANG H, YUAN S, ZHENG B, et al. Lubricating and Dual-Responsive Injectable Hydrogels Formulated From ZIF-8 Facilitate Osteoarthritis Treatment by Remodeling the Microenvironment. Small. 2025;21(3):2407885. [82] XU Z, MA J, HU H, et al. Metal ion-crosslinking multifunctional hydrogel microspheres with inflammatory immune regulation for cartilage regeneration. Front Bioeng Biotechnol. 2025; 13:1540592. [83] HALOI P, CHAWLA S, KONKIMALLA VB. Thermosensitive smart hydrogel of PEITC ameliorates the therapeutic efficacy in rheumatoid arthritis. Eur J Pharma Sci. 2023;181:106367. [84] LU B, LI C, JING L, et al. Rosmarinic acid nanomedicine for rheumatoid arthritis therapy: Targeted RONS scavenging and macrophage repolarization. J Control Release. 2023;362: 631-646. [85] DAI W, JIN P, LI X, et al. A carrier-free nano-drug assembled via π–π stacking interaction for the treatment of osteoarthritis. Biomed Pharmacother. 2023;164:114881. [86] SHABAN NS, RADI AM, ABDELGAWAD MA, et al. Targeting Some Key Metalloproteinases by Nano-Naringenin and Amphora coffeaeformis as a Novel Strategy for Treatment of Osteoarthritis in Rats. Pharmaceuticals. 2023;16(2):260. [87] FU L, LI P, WU J, et al. Tetrahedral framework nucleic acids enhance the chondrogenic potential of human umbilical cord mesenchymal stem cells via the PI3K/AKT axis. Regen Biomater. 2023;10: rbad085. [88] ZHANG M, ZHANG X, TIAN T, et al. Anti-inflammatory activity of curcumin-loaded tetrahedral framework nucleic acids on acute gouty arthritis. Bioact Mater. 2022;8:368-380. [89] LI Y, LI J, XU S, et al. Tetrahedral Framework Nucleic Acid-Based Delivery of Astaxanthin Suppresses Chondrocyte Pyroptosis and Modulates Oxidative Stress for the Treatment of Osteoarthritis. Adv Healthc Mater. 2024;13(28):2401452. [90] SHI S, CHEN T, LU W, et al. Amelioration of Osteoarthritis via Tetrahedral Framework Nucleic Acids Delivering Microrna-124 for Cartilage Regeneration. Adv Funct Mater. 2023;33(46): 2305558. |
| [1] | 刘宏杰, 牟秋菊, 申玉雪, 梁 飞, 祝丽丽. 金属有机框架/羧甲基壳聚糖-氧化海藻酸钠/富血小板血浆水凝胶促糖尿病感染创面愈合[J]. 中国组织工程研究, 2026, 30(8): 1929-1939. |
| [2] | 杨学涛, 朱梦菡, 张宸熙, 孙一民, 叶 玲. 抗氧化纳米材料在口腔中的应用和不足[J]. 中国组织工程研究, 2026, 30(8): 2044-2053. |
| [3] | 刘大为, 崔颖颖, 王方辉, 王子轩, 陈宇涵, 李友瑞, 张荣和. 表没食子儿茶素没食子酸酯介导活性氧双向调控及在纳米材料中的应用[J]. 中国组织工程研究, 2026, 30(8): 2101-2112. |
| [4] | 许艺璇, 姚 俊, 刘旭璐, 李新莲, 刘志雄, 张志红. 含万古霉素的猪皮脱细胞外基质水凝胶促进皮肤感染创面愈合[J]. 中国组织工程研究, 2026, 30(20): 5214-5228. |
| [5] | 王宇航, 张 涵, 张超晶, 寇绪容, 井桐桐, 林日梅, 刘鑫宇, 娄石磊, 阎 慧, 孙 聪. 姜黄素提取及姜黄素纳米粒的制备及优化[J]. 中国组织工程研究, 2026, 30(2): 362-374. |
近年来随着对骨关节炎发病机制研究的深入,炎症反应、氧化应激、细胞外基质代谢失衡及细胞凋亡等关键病理因素被逐步揭示[3],为靶向治疗策略提供了重要的理论基础。在此背景下,纳米生物医学凭借其材料特性和功能多样性的优势迅速崛起,在骨关节炎治疗领域中展现出广阔的应用前景[4]。纳米生物材料可以通过多种方式发挥作用,如作为药物载体实现高效精准给药,增强药物在关节部位的疗效;材料自身也具有抗氧化、抗炎等特性,可调节关节内的炎性微环境,减轻炎症反应和氧化损伤;此外,某些材料还可以促进软骨细胞的增殖和细胞外基质的合成,有助于修复受损的关节组织[5-6]。
该文首先综述了目前骨关节炎现有的治疗方法,重点讨论了原位注射治疗的现状和局限,进而总结了纳米生物材料在该疾病治疗中的最新研究进展,包括单一纳米体系、纳米复合型材料、纳米载药体系、纳米复合水凝胶、生物杂化材料和其他纳米体系,最后对该领域的未来研究进行展望,旨在为骨关节炎的精准治疗和临床转化提供新的策略和思路。
中国组织工程研究杂志出版内容重点:生物材料;骨生物材料;口腔生物材料;纳米材料;缓释材料;材料相容性;组织工程
1.1.1 检索人及检索时间 由第一作者在2025年4月进行检索。
1.1.2 文献检索时限 各数据库建库至2025年4月。
1.1.3 检索数据库 中国知网、PubMed、Scopus和Web of Science数据库。
1.1.4 检索词 以“纳米生物医学,纳米复合水凝胶,工程化生物体,骨关节炎”为中文检索词,以“nanobiomedicine,nanocomposited hydrogel,engineered organisms,osteoarthritis”为英文检索词。
1.1.5 检索策略 采用主题词和自由词结合的方式进行检索,以PubMed数据库检索策略为例,如图1。
1.1.7 手工检索情况 手工检索26篇文献。
1.1.8 检索文献量 初步检索到2 000余篇文献。
1.2 入选标准
纳入标准:①已正式发表的期刊文献;②文献内容与“关键词”内容密切相关;③同类研究中质量、证据等级较高(文章影响因子较高,分区靠前,引用量较高,中文为中文核心期刊)的文献;④优先选择近期发表、相同领域中与主题相关的文献。
排除标准:①与研究目的不符的文献;②年代久远的文献;③研究内容落后、研究方法被淘汰、实验器材老化及重复性文献。
1.3 质量评估与数据提取 通过计算机初步检索得到与研究目的相关的中英文文献2 000余篇,经第一作者根据纳入与排除标准进一步筛选,选择与此次综述内容相符的300余篇文献,最终确定纳入90篇文献进行综述。文献筛选流程如图2。
3.2 该综述区别于他人他篇的特点 该文综述了纳米生物医学应用于骨关节炎的治疗,具体介绍了单一纳米体系、纳米复合型材料、纳米载药体系、生物杂化材料、纳米复合水凝胶和其他纳米体系在治疗骨关节炎中炎症反应、软骨再生、基因表达和细胞凋亡等方面的研究进展,同时简要介绍纳米生物医学应用于未来的研究方向,为纳米生物医学由基础研究转至临床应用提供新的思路。
3.3 该综述的局限性 文章虽然对大量相关文献进行了综合阐述,但纳米生物医学的研究方向极为丰富,该综述难以涵盖所有类型的研究方向及其在治疗关节炎方面的作用效果。在探讨未来研究方向时,提出的设想和发展理念可能过于理想化,在实际的研究过程中会面临诸多技术难题和资源有限方面的挑战。
3.4 该综述的重要意义 该文旨在对纳米生物医学在关节炎治疗中的研究现状进行全面综述,为骨关节炎治疗探寻一条全新的方向,同时为纳米生物在该领域未来的研究提供创新性的见解。
3.5 未来的发展建议 未来的研究应继续深入探讨纳米生物医学研究治疗骨关节炎的具体作用机制,包括它对细胞凋亡、炎症因子调控和软骨再生的影响,同时组建多学科的研究团队,以整合不同领域的知识和技术,这将有助于理解生物纳米材料的作用机制以及开发新型生物纳米材料,具体包括以下内容:①这些特定的纳米生物医学关于材料研究具有多种功能和特殊的固有性能,如光热、光动力学、化学动力学、光声、超声波、抗菌、磁性、机械和磁热性能是必要的设计,以赋予纳米生物医学领域扩展的多模式疗法;②应大力开发具有高生物相容性、良好的生物降解性或溶解性以及高载药率的纳米复合材料,设计成智能化和个性化的纳米治疗平台;③纳米生物医学研究对于治疗机制的探索不够深化,在进行基因测序、基因库分析和蛋白组学等检测后,后续验证实验应加以多种分析手段和验证方法,如基因敲除和构建类器官模型验证等,对于类器官模型验证的研究中,可以深入探究纳米药物在接近人体真实环境下的治疗机制、药物代谢过程以及药物对不同细胞类型的影响,不仅可以避免体外验证的实验难度,更符合科研活动中的动物伦理学,显著减少实验动物应用数量;④应更加重视纳米生物医学研究中的生物安全性、生物利用度和大规模合成,以促进治疗多种疾病的进一步发展和未来的临床应用;⑤在人工智能快速发展的大环境下,人工智能算法能够深度挖掘纳米生物医学研究的结构-性能关系,通过机器学习模型分析海量实验数据,比如可以从分子动力学模拟纳米材料的设计,从原子层面进行更详细的结构模拟,观察其实时的结构变化以及搭载药物的负载和释放过程,从而为优化纳米结构设计提供更有效的前期基础。人工智能与纳米生物医学的融合将催生智能诊疗一体化平台,实时动态调整纳米载药体系的释放策略,实现个性化的精准治疗。
该文具体综述单一纳米体系、纳米复合型材料、纳米载药体系、生物杂化材料、纳米复合水凝胶和其他纳米体系的纳米生物医学研究用于延缓骨关节炎的发展进程。单一纳米体系通过金属基的催化活性与非金属基的理化特性,实现活性氧清除与软骨修复的精准调控;纳米复合型材料借助pH值和酶等响应机制,将药物控释与病灶靶向诊疗一体化;纳米载药体系通过工程化策略,突破传统给药的短效性与全身毒性局限;生物杂化材料利用外泌体的天然靶向性,介导细胞内递送与免疫微环境重塑;纳米复合水凝胶通过三维网状结构,协同实现力学微环境调控与生长因子时空释放;其他纳米体系则从天然产物活性优化与基因编辑角度,拓展了纳米材料的生物相容性设计范式,后续还展望了纳米生物医学在后续研究中的发展前景,并给出具体的未来发展建议。
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||