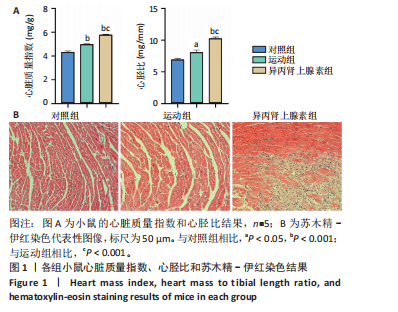
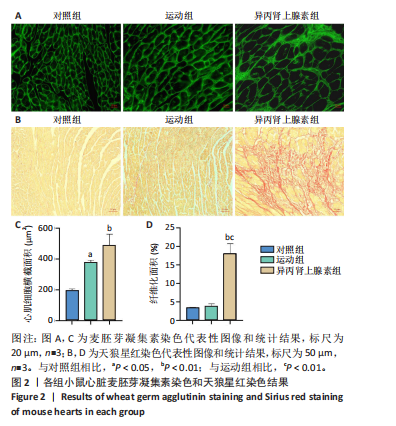
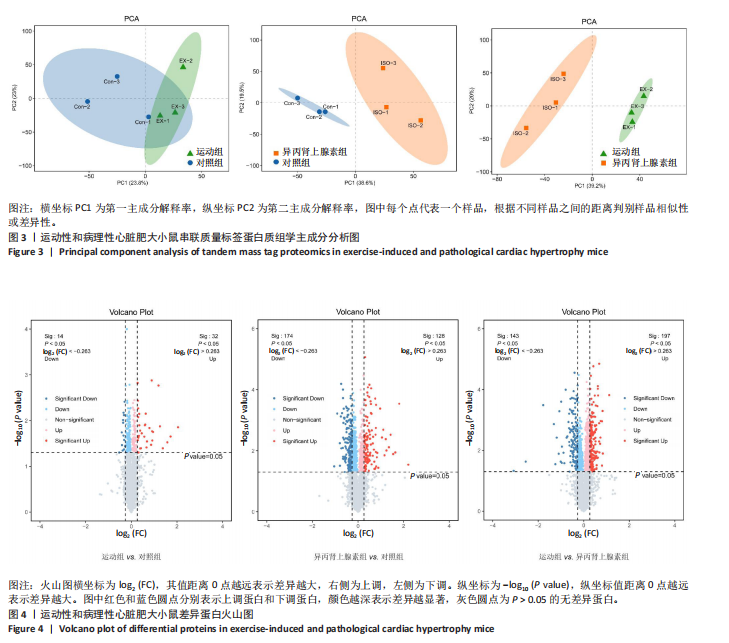
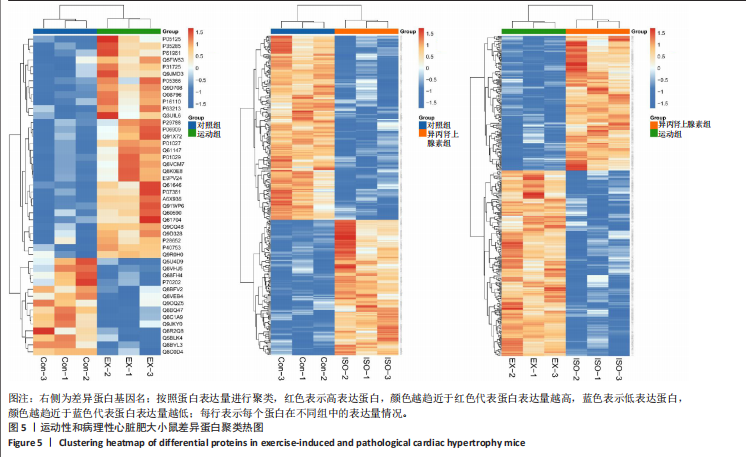
[1] AGABITI-ROSEI E, MUIESAN ML. Cardiac hypertrophy and hypertension. Curr Opin Nephrol Hypertens. 1998;7(2):211-216.
[2] MARTIN TG, JUARROS MA, LEINWAND LA. Regression of cardiac hypertrophy in health and disease: mechanisms and therapeutic potential. Nat Rev Cardiol. 2023;20(5):347-363.
[3] WEINER RB, BAGGISH AL. Exercise-induced cardiac remodeling. Prog Cardiovasc Dis. 2012;54(5):380-386.
[4] HENNING RJ. The differentiation of the competitive athlete with physiologic cardiac remodeling from the athlete with cardiomyopathy. Curr Probl Cardiol. 2024;49(9):102473.
[5] CARRENO JE, APABLAZA F, OCARANZA MP, et al. [Cardiac hypertrophy: molecular and cellular events]. Rev Esp Cardiol. 2006;59(5):473-486.
[6] NAKAMURA M, SADOSHIMA J. Mechanisms of physiological and pathological cardiac hypertrophy. Nat Rev Cardiol. 2018;15(7):387-407.
[7] BAI Y, ZHANG X, LI Y, et al. Protein Kinase A Is a Master Regulator of Physiological and Pathological Cardiac Hypertrophy. Circ Res. 2024;134(4):393-410.
[8] LIU X, LI H, HASTINGS MH, et al. miR-222 inhibits pathological cardiac hypertrophy and heart failure. Cardiovasc Res. 2024;120(3):262-272.
[9] SUOMINEN A, SUNI A, RUOHONEN S, et al. Melanocortin 1 Receptor Regulates Pathological and Physiological Cardiac Remodeling. J Am Heart Assoc. 2025; 14(4):e037961.
[10] BOOTH FW, ROBERTS CK, LAYE MJ. Lack of exercise is a major cause of chronic diseases. Compr Physiol. 2012;2(2):1143-1211.
[11] MOTR PACSG, LEAD A. Temporal dynamics of the multi-omic response to endurance exercise training. Nature. 2024;629(8010):174-183.
[12] MI S, JIANG H, ZHANG L, et al. Regulation of Cardiac-Specific Proteins Expression by Moderate-Intensity Aerobic Exercise Training in Mice With Myocardial Infarction Induced Heart Failure Using MS-Based Proteomics. Front Cardiovasc Med. 2021;8:732076.
[13] 彭勇, 史绍蓉, 黄思敏, 等. 4周中等强度有氧运动诱导大鼠心房肌蛋白质组差异表达的研究[J]. 中国应用生理学杂志,2018,34(5):450-456+463.
[14] 彭勇, 史绍蓉, 黄思敏, 等. 基于蛋白质组学的中等强度有氧运动对大鼠心房肌物质能量代谢蛋白差异表达的研究[J]. 中国康复医学杂志,2019,34(5): 521-526.
[15] DAVIS RT, 3RD, SIMON JN, UTTER M, et al. Knockout of p21-activated kinase-1 attenuates exercise-induced cardiac remodelling through altered calcineurin signalling. Cardiovasc Res. 2015;108(3):335-347.
[16] CHEN H, PEREZ JN, CONSTANTOPOULOS E, et al. A method to study the impact of chemically-induced ovarian failure on exercise capacity and cardiac adaptation in mice. J Vis Exp. 2014;86:51083.
[17] 秦娣, 秦学林, 郑一苇, 等. 有氧运动和恩格列净抑制铁死亡减轻异丙肾上腺素诱导的心脏重构[J]. 中国实验动物学报,2024,32(10):1281-1294.
[18] 强睿, 姚子昂, 韩东, 等. 基于TMT标记联合LC-MS/MS阐释茵芪三黄解毒汤抗肝纤维化的药效作用机制[J]. 中华中医药杂志,2023,38(12):5987-5992.
[19] COHN JN, FERRARI R, SHARPE N. Cardiac remodeling--concepts and clinical implications: a consensus paper from an international forum on cardiac remodeling. Behalf of an International Forum on Cardiac Remodeling. J Am Coll Cardiol. 2000;35(3):569-582.
[20] SHIMIZU I, MINAMINO T. Physiological and pathological cardiac hypertrophy. J Mol Cell Cardiol. 2016;97:245-262.
[21] MARON BJ, PELLICCIA A. The heart of trained athletes: cardiac remodeling and the risks of sports, including sudden death. Circulation. 2006;114(15):1633-1644.
[22] LIMYATI Y, LUCRETIA T, GUNADI JW, et al. Chronic moderate‑intensity exercise can induce physiological hypertrophy in aged cardiomyocytes through autophagy, with minimal Yap/Taz involvement. Biomed Rep. 2025;22(3):44.
[23] VAN BILSEN M, VAN NIEUWENHOVEN FA, VAN DER VUSSE GJ. Metabolic remodelling of the failing heart: beneficial or detrimental? Cardiovasc Res. 2009; 81(3):420-428.
[24] NEUBAUER S. The failing heart--an engine out of fuel. N Engl J Med. 2007; 356(11):1140-1151.
[25] MORA C, PINTADO C, RUBIO B, et al. Central leptin regulates heart lipid content by selectively increasing PPAR beta/delta expression. J Endocrinol. 2018;236(1): 43-56.
[26] ZHANG H, SHEN Y, KIM IM, et al. The Impaired Bioenergetics of Diabetic Cardiac Microvascular Endothelial Cells. Front Endocrinol (Lausanne). 2021;12:642857.
[27] GUTIERREZ-CUEVAS J, SANDOVAL-RODRIGUEZ A, MONROY-RAMIREZ HC, et al. Prolonged-release pirfenidone prevents obesity-induced cardiac steatosis and fibrosis in a mouse NASH model. Cardiovasc Drugs Ther. 2021;35(5):927-938.
[28] WANG Q, WANG X. The Effects of a Low Linoleic Acid/alpha-Linolenic Acid Ratio on Lipid Metabolism and Endogenous Fatty Acid Distribution in Obese Mice. Int J Mol Sci. 2023;24(15):12117.
[29] LIU L, NING X, WEI L, et al. Twist1 downregulation of PGC-1alpha decreases fatty acid oxidation in tubular epithelial cells, leading to kidney fibrosis. Theranostics. 2022;12(8):3758-3775.
[30] HU H, WANG J, PENG Z, et al. Dapagliflozin attenuates diabetes-induced podocyte lipotoxicity via ERRalpha-Mediated lipid metabolism. Free Radic Biol Med. 2025; 234:178-191.
[31] VERGARO G, PRUD’HOMME M, FAZAL L, et al. Inhibition of Galectin-3 Pathway Prevents Isoproterenol-Induced Left Ventricular Dysfunction and Fibrosis in Mice. Hypertension. 2016;67(3):606-612.
[32] XU GR, ZHANG C, YANG HX, et al. Modified citrus pectin ameliorates myocardial fibrosis and inflammation via suppressing galectin-3 and TLR4/MyD88/NF-kappaB signaling pathway. Biomed Pharmacother. 2020;126:110071.
[33] SUN JH, YANG HX, YAO TT, et al. Gentianella acuta prevents acute myocardial infarction induced by isoproterenol in rats via inhibition of galectin-3/TLR4/MyD88/NF-small ka, CyrillicB inflammatory signalling. Inflammopharmacology. 2021;29(1):205-219.
[34] XING Z, YANG C, FENG Y, et al. Understanding aconite’s anti-fibrotic effects in cardiac fibrosis. Phytomedicine. 2024;122:155112.
[35] YAKAR TULUCE S, TULUCE K, CIL Z, et al. Galectin-3 levels in patients with hypertrophic cardiomyopathy and its relationship with left ventricular mass index and function. Anatol J Cardiol. 2016;16(5):344-348.
[36] LI Y, ZHOU WW, SUN JH, et al. Modified citrus pectin prevents isoproterenol-induced cardiac hypertrophy associated with p38 signalling and TLR4/JAK/STAT3 pathway. Biomed Pharmacother. 2021;143:112178.
[37] KRUGER DN, BOSMAN M, VAN ASSCHE CXL, et al. Characterization of systolic and diastolic function, alongside proteomic profiling, in doxorubicin-induced cardiovascular toxicity in mice. Cardiooncology. 2024;10(1):40.
[38] WANG L, ZHANG Y, YU M, et al. Identification of Hub Genes in the Remodeling of Non-Infarcted Myocardium Following Acute Myocardial Infarction. J Cardiovasc Dev Dis. 2022;9(12):409.
[39] GAO J, LI Y, WANG T, et al. Analyzing gene expression profiles with preliminary validations in cardiac hypertrophy induced by pressure overload. Can J Physiol Pharmacol. 2018;96(8):701-709.
[40] CHEN H, TESIC M, NIKOLIC VN, et al. Systemic Biomarkers and Unique Pathways in Different Phenotypes of Heart Failure with Preserved Ejection Fraction. Biomolecules. 2022;12(10):1419.
[41] XU H, SHANG Q, CHEN H, et al. ITIH4: A New Potential Biomarker of “Toxin Syndrome” in Coronary Heart Disease Patient Identified with Proteomic Method. Evid Based Complement Alternat Med. 2013;2013:360149.
[42] SAMPIETRO T, NEGLIA D, BIONDA A, et al. Inflammatory markers and serum lipids in idiopathic dilated cardiomyopathy. Am J Cardiol. 2005;96(12):1718-1720.
[43] ZHOU LQ, LIU C, ZOU Y, et al. Development and verification of the nomogram for dilated cardiomyopathy gene diagnosis. Sci Rep. 2022;12(1):8908.
[44] XIAO Y, NI L, SHI H, et al. SAA1 deficiency alleviates cardiac remodeling by inhibiting NF-kappaB/p38/JNK and TGFbeta/Smad pathways. FASEB J. 2023; 37(5):e22911.
[45] WEN J, LI H, ZHOU Y, et al. Immunoglobin attenuates fulminant myocarditis by inhibiting overactivated innate immune response. Br J Pharmacol. 2025; 182(20):4984-5003.
|