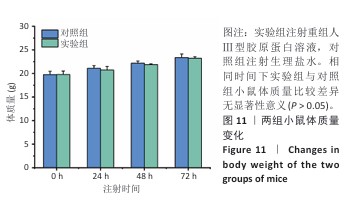
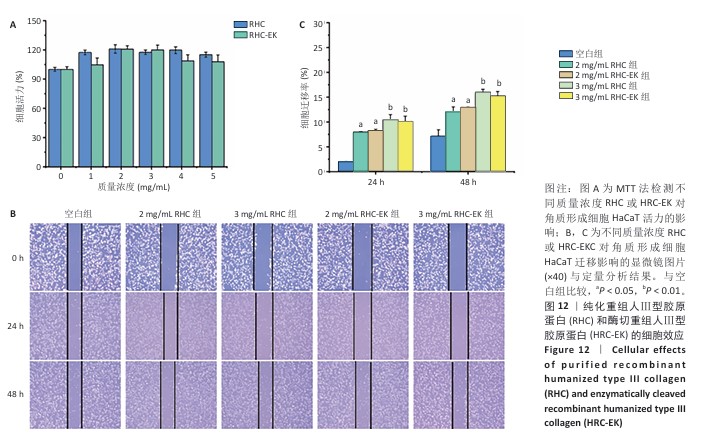
[1] RICARD-BLUM S. The collagen family. Cold Spring Harb Perspect Biol. 2011;3(1): a004978.
[2] SHOULDERS MD, RAINES RT. Collagen structure and stability. Annu Rev Biochem. 2009;78:929-958.
[3] RICARD-BLUM S, RUGGIERO FVAN DER REST M. The Collagen Superfamily//BRINCKMANN J, NOTBOHM H, MÜLLER PK, Editors. Collagen: Primer in Structure, Processing and Assembly. 2005, Springer Berlin Heidelberg: Berlin, Heidelberg, 2005:35-84.
[4] 祝洁,朱嘉慧,刘尧.胶原蛋白类医疗器械产业发展及监管现状[J].中国药事,2025,39(3):306-316.
[5] SORUSHANOVA A, DELGADO LM, WU Z, et al. The Collagen Suprafamily:From Biosynthesis to Advanced Biomaterial Development. Adv Mater. 2019;31(1): e1801651.
[6] 朱永真.重组胶原蛋白的生物制造及其应用[J].中外食品工业,2024(16): 30-32.
[7] LIU T, HAO J, LEI H, et al. Recombinant collagen for the repair of skin wounds and photo-aging damage. Regen Biomater. 2024;11:rbae108.
[8] LI Y, XIA Y, LIU X, et al. Rational design of bioengineered recombinant collagen-like protein enhances GelMA hydrogel for diabetic wound healing. Int J Biol Macromol. 2024;280(Pt 3):136012.
[9] DE MOURA CAMPOS S, DOS SANTOS COSTA G, KARP SG, et al. Innovations and challenges in collagen and gelatin production through precision fermentation. World J Microbiol Biotechnol. 2025;41(2):63.
[10] CAO L, ZHANG Z, YUAN D, et al. Tissue engineering applications of recombinant human collagen:a review of recent progress. Front Bioeng Biotechnol. 2024;12: 1358246.
[11] MATINONG AME, CHISTI Y, PICKERING KL, et al. Collagen Extraction from Animal Skin. Biology (Basel). 2022;11(6):905.
[12] SALIM NV, MADHAN B, GLATTAUER V, et al. Comprehensive review on collagen extraction from food by-products and waste as a value-added material. Int J Biol Macromol. 2024;278(Pt 1):134374.
[13] 中国食品药品检定研究院,四川省药品检验研究院,山东省医疗器械和药品包装检验研究院,等. YY/T 1849-2022 重组胶原蛋白.行业标准.2022:20.
[14] 复旦大学,四川大学,中国科学院生物物理研究所,等.YY/T 1888-2023 重组人源化胶原蛋白.行业标准.2023:20.
[15] FANG J, MA Z, LIU D, et al. Co-expression of recombinant human collagen α1(III) chain with viral prolyl 4-hydroxylase in Pichia pastoris GS115. Protein Expr Purif. 2023;201:106184.
[16] 夏慧敏,杜明会,唐雯,等.含重组胶原蛋白二类医疗器械产品的审评共性问题思考及对策探讨[J].中国医疗器械信息,2025,31(1):1-4,76.
[17] 刘迪,郭君慧,李其昌,等.重组类胶原蛋白的表达纯化及性质研究[J].食品与发酵工业,2025,51(6):127-133.
[18] 滕飞,柯博文,全冰华,等.重组Ⅰ型人源化胶原蛋白在大肠杆菌中的可溶性表达及纯化[J].现代食品科技,2024,40(12):41-48.
[19] WANG J, XIANG ZX, LUAN MF, et al. High-level secretory expression of recombinant type III human-like collagen α1 in Pichia pastoris via multilevel systematic optimization. Int J Biol Macromol. 2025;313:144270.
[20] QI Q, YAO L, LIANG Z, et al. Production of human type II collagen using an efficient baculovirus-silkworm multigene expression system. Mol Genet Genomics. 2016;291(6):2189-2198.
[21] 李斐,侯宇曦,饶犇,等.整合素结合序列N端Gly错义突变对重组胶原蛋白结构与功能的影响[J]. 生物工程学报,2025,41(4):1573-1587.
[22] 夏煌慧,黄建忠.重组胶原蛋白的生物合成研究进展[J].微生物学报,2025, 65(5):1939-1957.
[23] FERTALA A. Three Decades of Research on Recombinant Collagens:Reinventing the Wheel or Developing New Biomedical Products? Bioengineering (Basel). 2020;7(4):155.
[24] 傅容湛,范代娣,杨婉娟,等.重组胶原蛋白的产业发展历程和生物医学应用前景展望[J].生物工程学报,2022,38(9):3228-3242.
[25] LIU W, LIN H, ZHAO P, et al. A regulatory perspective on recombinant collagen-based medical devices. Bioact Mater. 2022;12:198-202.
[26] YUAN JJ, GENG SL, WANG TY. Research Progress Fusion Tags for Recombinant Protein Production. Biotechnol Appl Biochem. 2025;72(5):1477-1487.
[27] OJIMA-KATO T. Advances in recombinant protein production in microorganisms and functional peptide tags. Biosci Biotechnol Biochem. 2024;89(1):1-10.
[28] LEONHARDT F, GENNARI A, PALUDO GB, et al. A systematic review about affinity tags for one-step purification and immobilization of recombinant proteins:integrated bioprocesses aiming both economic and environmental sustainability. 3 Biotech. 2023;13(6):186.
[29] BOOTH WT, SCHLACHTER CR, POTE S, et al. Impact of an N-terminal Polyhistidine Tag on Protein Thermal Stability. ACS Omega. 2018;3(1):760-768.
[30] MISHRA V. Affinity Tags for Protein Purification. Curr Protein Pept Sci. 2020;21(8): 821-830.
[31] GRÄSLUND S, NORDLUND P, WEIGELT J, et al. Protein production and purification. Nat Methods. 2008;5(2):135-146.
[32] LIU F. Safety assessment of drug impurities for patient safety:A comprehensive review. Regul Toxicol Pharmacol. 2024;153:105715.
[33] WANG X, MORGAN DM, WANG G, et al. Residual DNA analysis in biologics development:Review of measurement and quantitation technologies and future directions. Biotechnol Bioeng. 2012;109(2):307-317.
[34] YANG H. Establishing acceptable limits of residual DNA. PDA J Pharm Sci Technol. 2013;67(2):155-163.
[35] HIGASHIYAMA K, YUAN Y, HASHIBA N, et al. Quantitation of Residual Host Cell DNA in Recombinant Adeno-Associated Virus Using Droplet Digital Polymerase Chain Reaction. Human Gene Therapy. 2023;34(11-12):578-585.
[36] SMITH JB, THÉVENON-EMERIC G, SMITH DL, et al. Elucidation of the primary structures of proteins by mass spectrometry. Analytical Biochemistry. 1991; 193(1):118-124.
[37] WEN J, ARTHUR K, CHEMMALIL L, et al. Applications of Differential Scanning Calorimetry for Thermal Stability Analysis of Proteins:Qualification of DSC. J Pharm Sci. 2012;101(3):955-964.
[38] KAPUST RB, TÖZSÉR J, FOX JD, et al. Tobacco etch virus protease:mechanism of autolysis and rational design of stable mutants with wild-type catalytic proficiency. Protein Eng. 2001;14(12):993-1000.
[39] UHLÉN M, FORSBERG G, MOKS T, et al. Fusion proteins in biotechnology. Curr Opin Biotechnol. 1992;3(4):363-369.
[40] MAJOREK KA, KUHN ML, CHRUSZCZ M, et al. Double trouble-Buffer selection and His-tag presence may be responsible for nonreproducibility of biomedical experiments. Protein Sci. 2014;23(10):1359-1368.
[41] PANEK A, PIETROW O, FILIPKOWSKI P, et al. Effects of the polyhistidine tag on kinetics and other properties of trehalose synthase from Deinococcus geothermalis. Acta Biochim Pol. 2013;60(2):163-166.
[42] SABATY M, GROSSE S, ADRYANCZYK G, et al. Detrimental effect of the 6 His C-terminal tag on YedY enzymatic activity and influence of the TAT signal sequence on YedY synthesis. BMC Biochem. 2013;14:28.
[43] WATLY J, HECEL A, WIECZOREK R, et al. Uncapping the N-terminus of a ubiquitous His-tag peptide enhances its Cu2+ binding affinity. Dalton Trans. 2019;48(36):13567-13579.
|