中国组织工程研究 ›› 2026, Vol. 30 ›› Issue (26): 6760-6767.doi: 10.12307/2026.766
• 水凝胶材料Hydrogel materials • 上一篇 下一篇
载人参皂苷Rg3脂质体水凝胶促进干细胞成软骨分化
赵清兰1,2,3,周新婷2,王华军3,钟佳轩2,3,郑力恒4,谭文成5,杨新春6,黄树森3,吴婷婷2,郑小飞3,洪劲松1,3
- 1广州中医药大学,广州市正骨医院,广东省广州市 510006;2国家医疗保健器具工程技术研究中心,广东省医用电子仪器及材料重点实验室,广东省科学院生物与医学工程研究所,广东省广州市 510316;3暨南大学附属第一医院运动医学科,广州市精准骨科与再生医学重点实验室,广东省速度能力研究重点实验室,广东省广州市 510630;4澳门医学科技研究协会,中国澳门特别行政区;5澳门卫生局仁伯爵综合医院骨科,中国澳门特别行政区;6泳江制药厂有限公司,岐英堂制药有限公司,中国澳门特别行政区
Ginsenoside Rg3-loaded liposome hydrogel promotes chondrogenic differentiation of stem cells
Zhao Qinglan1, 2, 3, Zhou Xinting2, Wang Huajun3, Zhong Jiaxuan2, 3, Zheng Liheng4, Tan Wencheng5, Yang Xinchun6, Huang Shusen3, #br# Wu Tingting2, Zheng Xiaofei3, Hong Jinsong1, 3
- 1Guangzhou University of Chinese Medicine, Guangzhou Orthopaedic Hospital, Guangzhou 510006, Guangdong Province, China; 2National Engineering Research Center for Healthcare Devices, Guangdong Provincial Key Laboratory of Medical Electronic Instruments and Materials, Institute of Biomedical and Health Engineering, Guangdong Academy of Sciences, Guangzhou 510316, Guangdong Province, China; 3Department of Sports Medicine, The First Affiliated Hospital of Jinan University, Guangdong Key Laboratory for Orthopaedic Precision and Regenerative Medicine, Guangdong Key Laboratory of Speed and Agility, Guangzhou 510630, Guangdong Province, China; 4IAN WO Medical Center, Macao Special Administrative Region, China; 5Department of Orthopedic Surgery, Centro Hospitalar Conde de São Januário, Macao Special Administrative Region, China; 6Yongjiang Pharmaceutical Factory Co., Ltd., China Qiyingtang Pharmaceutical Co., Ltd., Macao Special Administrative Region, China
Zhao Qinglan, MS candidate, Guangzhou University of Chinese Medicine, Guangzhou Orthopaedic Hospital, Guangzhou 510006, Guangdong Province, China; National Engineering Research Center for Healthcare Devices, Guangdong Provincial Key Laboratory of Medical Electronic Instruments and Materials, Institute of Biomedical and Health Engineering, Guangdong Academy of Sciences, Guangzhou 510316, Guangdong Province, China; Department of Sports Medicine, The First Affiliated Hospital of Jinan University, Guangdong Key Laboratory for Orthopaedic Precision and Regenerative Medicine, Guangdong Key Laboratory of Speed and Agility, Guangzhou 510630, Guangdong Province, China
摘要:
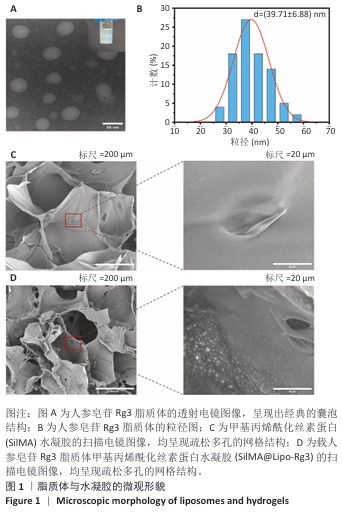
人参皂苷Rg3脂质体:此次研究中的人参皂苷Rg3脂质体系通过薄膜分散法制备,以磷脂和胆固醇为主要材料,负载人参皂苷Rg3。人参皂苷Rg3作为中药活性成分具有抗炎与促分化作用,但水溶性差、易降解限制了它的直接应用。脂质体包载方式有效提高了人参皂苷Rg3的稳定性与生物利用度,并且在水凝胶中呈现持续释放特性,为间充质干细胞的定向诱导提供稳定微环境,是此次研究复合水凝胶发挥成软骨功能的关键构件之一。
成软骨分化:是指骨髓间充质干细胞在特定诱导条件下向软骨样细胞表型转化的生物学过程。此次研究通过添加成软骨诱导培养基对骨髓间充质干细胞进行7 d的诱导培养,通过qPCR检测软骨标志基因的表达量化分化效果,通过细胞染色观察细胞外软骨基质合成情况。
背景:人参皂苷Rg3具有潜在的软骨保护及修复价值,但它的应用存在水溶性差、半衰期短、生物利用度低等缺点。为改善药物的药代动力学特性,将药物负载脂质体嵌入甲基化明胶、多糖类或丝素蛋白基水凝胶中已成为软骨组织工程研究的热点。
目的:制备载人参皂苷Rg3脂质体甲基丙烯酰化丝素蛋白水凝胶,进一步分析该水凝胶对小鼠骨髓间充质干细胞成软骨分化的作用。
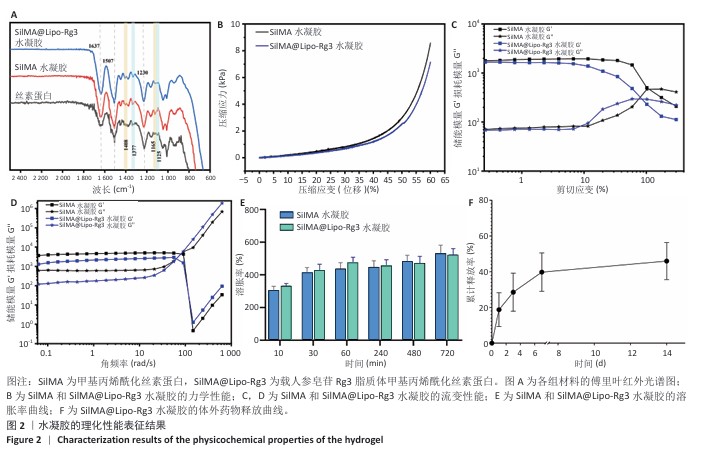
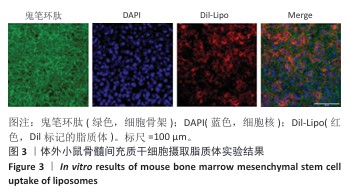
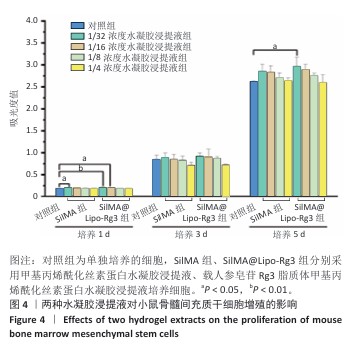
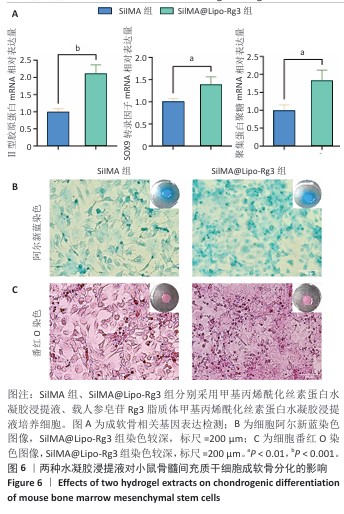
方法:①采用薄膜分散法制备人参皂苷Rg3脂质体,将Dil标记的脂质体与小鼠骨髓间充质干细胞共培养,鬼笔环肽染色检测细胞摄取脂质体情况。制备甲基丙烯酰化丝素蛋白(记为SilMA),将人参皂苷Rg3脂质体与SilMA混合,通过光交联制备复合水凝胶(记为SilMA@Lipo-Rg3),评估该水凝胶的微观形貌、力学性能、流变性能、溶胀性能与药物缓释性能。②采用不同浓度的SilMA水凝胶浸提液或SilMA@Lipo-Rg3水凝胶浸提液培养小鼠骨髓间充质干细胞,通过CCK-8检测与活死细胞染色评估材料的细胞相容性。分别采用1/8浓度的SilMA水凝胶浸提液、SilMA@Lipo-Rg3水凝胶浸提液培养小鼠骨髓间充质干细胞,成软骨诱导后,qPCR检测Ⅱ型胶原蛋白、SOX9、聚集蛋白聚糖mRNA表达,阿尔新蓝和番红O染色检测细胞内蛋白多糖、糖胺聚糖表达。
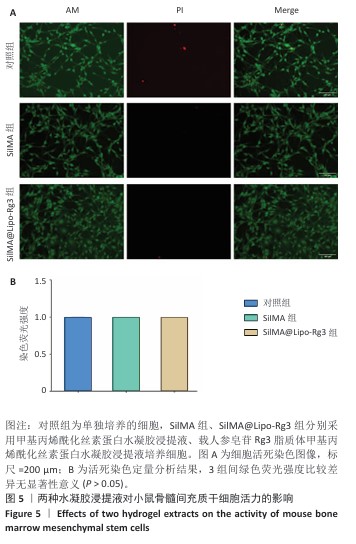
结果与结论:①鬼笔环肽染色显示,Dil标记的脂质体能够被小鼠骨髓间充质干细胞成功摄取。扫描电镜显示SilMA@Lipo-Rg3水凝胶呈现疏松多孔的网格结构。压缩实验与流变性能测试显示,与SilMA水凝胶相比,SilMA@Lipo-Rg3水凝胶的压缩刚度略有下降;两种水凝胶的溶胀性能无明显差异。SilMA@Lipo-Rg3水凝胶具有良好的缓释性能,可持续释放人参皂苷Rg3 14 d以上。②CCK-8检测与活死细胞染色显示,SilMA、SilMA@Lipo-Rg3水凝胶具有良好的细胞相容性。qPCR检测显示,SilMA@Lipo-Rg3组细胞内Ⅱ型胶原蛋白、SOX9、聚集蛋白聚糖mRNA表达高于SilMA组(P < 0.05);阿尔新蓝和番红O染色显示,SilMA@Lipo-Rg3组细胞内蛋白多糖、糖胺聚糖表达多于SilMA组。结果表明,SilMA@Lipo-Rg3水凝胶可促进小鼠骨髓间充质干细胞的成软骨分化。
https://orcid.org/0009-0003-2870-8182(赵清兰)
中国组织工程研究杂志出版内容重点:生物材料;骨生物材料;口腔生物材料;纳米材料;缓释材料;材料相容性;组织工程
中图分类号: