Chinese Journal of Tissue Engineering Research ›› 2014, Vol. 18 ›› Issue (32): 5214-5219.doi: 10.3969/j.issn.2095-4344.2014.32.022
Previous Articles Next Articles
Endothelial progenitor cells for diabetic vascular complications: is it a new therapeutic strategy?
Gao Cai-yan, Mai Chao-ping, Ha Xiao-qin
- Department of Laboratory Medicine, General Hospital of Lanzhou Military Region of PLA, Lanzhou 730050, Gansu Province, China
-
Received:2014-07-04Online:2014-08-06Published:2014-09-18 -
Contact:Ha Xiao-qin, M.D., Chief physician, Department of Laboratory Medicine, General Hospital of Lanzhou Military Region of PLA, Lanzhou 730050, Gansu Province, China -
About author:Gao Cai-yan, Studying for master’s degree, Department of Laboratory Medicine, General Hospital of Lanzhou Military Region of PLA, Lanzhou 730050, Gansu Province, China -
Supported by:the National Natural Science Foundation of China, No. 81273568; the Natural Science Foundation of Gansu Province, No. 1107RJZA114
CLC Number:
Cite this article
Gao Cai-yan, Mai Chao-ping, Ha Xiao-qin. Endothelial progenitor cells for diabetic vascular complications: is it a new therapeutic strategy?[J]. Chinese Journal of Tissue Engineering Research, 2014, 18(32): 5214-5219.
share this article
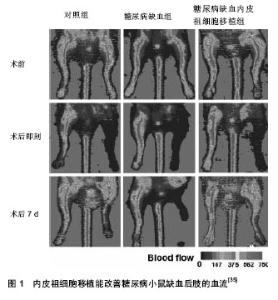
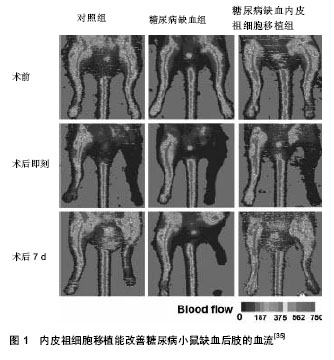
2.1 内皮祖细胞的生物学特性 内皮祖细胞可从骨髓、脐血、胚胎肝和外周血中分离培养获得,而骨髓中所占比例最大(四者之比为15∶10∶2∶1)。内皮祖细胞既表达造血干细胞(CD34+与CD133+)的表面标志物,又表达内皮细胞(KDR、CD31、vWf)的表面标志物,但是晚期内皮祖细胞主要表达CD34和KDR,而ACl33很快就消失了[3]。 近期有研究表明CD34+、KDR+的内皮祖细胞与缺血性疾病有更高的相关性[4]。在组织缺血缺氧和血管损伤的状态下,某些细胞因子、趋化因子如血管内皮生长因子、血小板源性生长因子[5-6]、粒-巨噬细胞集落刺激因子等的刺激下,骨髓源性内皮祖细胞能被动员至外周血,低氧诱导因子1α表达上调,并调节其下游的基质细胞衍生因子通过与内皮祖细胞表面的趋化因子受体CXCR-4结合,促进内皮祖细胞归巢到缺血部位,从而促进缺血组织的血管新生。白细胞介素8也参与了内皮祖细胞的运输过程,与其受体CXCR2和CXCR1相互作用介导了静脉注射的CD34+祖细胞向缺血组织的聚集[7]。 内皮祖细胞促进血管新生的机制包括两方面,一方面是通过自身的分化增殖而形成新生血管,无需依赖原来的血管系统,即血管新生,另一方面为血管发生,血管在原有基础上以出芽方式生长,首先在蛋白分解酶的作用下血管基底膜瓦解,细胞外基质降解,血管内皮细胞游离,通过黏附定位、增殖,形成新基底膜,出现管腔结构,最终成为新的血管分支与原有的血管相连接,形成血管网络[8]。 2.2 内皮祖细胞损伤和糖尿病血管并发症的发生机制 内皮祖细胞数量减少和功能损伤,使受损血管内皮修复和血管新生障碍,是导致糖尿病血管内皮功能紊乱和血管并发症发生的共同通路[9]。无论是1型糖尿病还是2型糖尿病,均有内皮祖细胞数量和功能的异常。Jung等[10]通过检测19例无糖尿病的血管疾病患者和20例糖尿病血管疾病患者的不同内皮祖细胞亚群的数量、凋亡及内皮功能,发现糖尿病血管疾病患者CD34−/CD133+/ KDR+的内皮祖细胞数量减少,凋亡增加,而祖细胞计数与外周动脉功能测试无关,推测凋亡增加可能是糖尿病血管病变中内皮祖细胞数量减少的主要机制。糖尿病及其高血糖、胰岛素抵抗、脂代谢紊乱对内皮祖细胞的迁移、分化为成熟内皮细胞、黏附、增殖以及整合入血管的所有功能均有损害[11]。内皮祖细胞的数量减少和功能受损主要有以下机制: 2.2.1 活性氧簇生成增加 氧化应激的增加,活性氧簇生成增多是糖尿病并发症发展过程中的主要代谢异常。活性氧簇生成增多会损伤内皮祖细胞从骨髓动员,并且,由于谷胱甘肽过氧化物酶水平的下降,使内皮祖细胞对氧化应激更为敏感[12],且凋亡增加。而在糖尿病患者,高血糖诱导的晚期糖基化终产物生成,多元醇途径活化,蛋白激酶C(PKC)/二酰甘油(DAG)途径和氨基己糖途径均可引起活性氧簇生成增多[13]。有研究表明,高糖条件下,静息状态的内皮祖细胞不断累积时,增殖状态的内皮祖细胞数量在不断减少[11]。 2.2.2 一氧化氮合成减少 选择性的胰岛素抵抗[14-15],胰岛素受体底物(IRS)-1/磷脂酰肌醇(PI)3-激酶途径受损,丝裂原活化蛋白激酶(MAPK)途径过度活化,特别是p38活化,减少了一氧化氮的生成、降低了基质细胞衍生因子1α的水平及其与受体CXCR4的反应性,最终减少内皮祖细胞的动员、增殖和分化并促进其凋亡[16]。残粒样脂蛋白(RLPs),富含三酰甘油的脂蛋白脂解产物能够通过增加氧化应激和减弱一氧化氮合成酶的活力来损伤内皮祖细胞的功能[17]。 2.2.3 抗氧化系统损害 血红素氧化酶1/脂联素轴应答不足。血红素氧化酶1, 能够使不稳定的血红蛋白降解为具有强大抗氧化作用和细胞保护作用的胆绿素和胆红素[18],它在糖尿病的氧化侵袭中起着保护作用。血红素氧化酶1还能促进脂联素的合成和释放[19],研究表明,脂联素在胰岛素抵抗,动脉粥样硬化和炎症发展过程中有保护作用[20]。而糖尿病患者体内血红素氧化酶-1/脂联素轴应答不足,损害了体内抗氧化、抗炎症系统,最终导致内皮祖细胞数量减少和功能障碍[21]。 而高密度脂蛋白胆固醇(HDL)和过氧化物酶体增殖物受体(PPARδ)通过PI3K/Akt通路介导能够促进内皮祖细胞的增殖和功能[22],PPARδ可通过低氧诱导因子1/基质细胞衍生因子1通路促进内皮祖细胞的动员和迁移,并对于低氧诱导的内皮祖细胞凋亡起到保护作用[23]。人apoA-1可增加循环内皮祖细胞的数量,提高其整合到血管内皮的能力,从而促进内皮祖细胞对内皮的修复。 2.2.4 低氧诱导因子1表达不稳定 高血糖使低氧诱导因子1表达不稳定,低氧诱导因子1α表达降低[24],从而使糖尿病患者心肌、伤口愈合、肾、视网膜等的细胞和组织的低氧应答减弱[25],进一步使内皮祖细胞动员减少,功能障碍。低氧诱导因子1由α和β两个亚单位组成,低氧诱导因子1α是低氧诱导的,受低氧调节,低氧诱导因子1β为结构亚单位,不受低氧诱导。低氧诱导因子1在低氧适应的调解中发挥着重要作用,可调节多个下游元件,其典型靶基因为血管内皮生长因子。低氧诱导因子1不仅可诱导血管内皮生长因子表达上调[26],还可诱导一氧化氮合成酶,血红素氧化酶1,和内皮素的表达上调[27-28]。一氧化氮和血管内皮生长因子能够促进内皮祖细胞从骨髓动员,而一氧化氮和血红素氧化酶1能保护内皮祖细胞抵御氧化侵袭。此外,低氧诱导因子1还能使基质细胞衍生因子1/CXCR4蛋白表达上调[29],基质细胞衍生因子1/CXCR4能够促进内皮祖细胞向缺血损伤部位迁移并修复损伤血管[30]。糖尿病高血糖使低氧诱导因子1α表达降低,不能调节其下游元件正常表达,使各组织低氧应答减弱,减少了内皮祖细胞的动员,趋化和对损伤血管的修复,促使糖尿病血管并发症的发生。 综以上各因素通过不同机制使内皮祖细胞从骨髓动员减少,迁移、黏附和成血管功能损害,以及存活数量减少,使糖尿病患者受损血管修复异常,血管新生障碍,最终导致糖尿病血管并发症的发生。 2.3 内皮祖细胞在治疗糖尿病患者血管并发症过程中的应用 由于糖尿病血管并发症患者内皮祖细胞的动员减少,功能受损,数量减少,不能正常修复损伤血管,因此,增进内皮祖细胞的动员,趋化和正常内皮祖细胞的移植则成为糖尿病血管并发症新的治疗策略,在糖尿病血管病变治疗中至关重要。 2.3.1 内皮祖细胞在糖尿病大血管并发症治疗中的应用 糖尿病大血管病变主要表现为糖尿病性心脏病、高血压、大动脉粥样硬化、糖尿病足等。动脉粥样硬化是糖尿病大血管并发症的病理基础,是以血管内皮细胞完整性破坏,巨噬细胞迁移,平滑肌细胞和成纤维细胞增生,以及细胞内外脂质积聚的病理过程。Fadini认为外周血循环内皮祖细胞水平能够替代累积心血管风险指数[31],指出内皮祖细胞计数是糖尿病患者外周动脉粥样硬化的重要生物标志,他的研究表明CD34+,KDR+的内皮祖细胞数量与糖尿病外周血管疾病的严重程度呈负相关。踝肱指数是一种最客观的远端动脉疾病诊断和预后指标,而内皮祖细胞数量和踝肱指数显著相关[32]。从健康动物体内提取内皮祖细胞植入糖尿病动物体内能减轻其侧支循环障碍;相反,糖尿病动物的内皮祖细胞不能刺激血管新生,甚至对抗血管新生。将从骨髓或外周血分离的正常内皮祖细胞移植到糖尿病患者伤口,可明显促进伤口愈合[33-34]。脐血来源的内皮祖细胞可通过诱导低氧诱导因子1α/白细胞介素8的表达,有效地恢复股动脉结扎后的糖尿病小鼠后肢的血流和功能。内皮祖细胞移植能促进糖尿病小鼠缺血后肢的血管新生(图1)[35],此外,经载有低氧诱导因子1基因的腺病毒转染后的内皮祖细胞体内成血管能力显著增强[36]。 2.3.2 内皮祖细胞在糖尿病微血管并发症治疗中的应用 糖尿病最常见的微血管并发症是糖尿病视网膜病变、糖尿病肾病、糖尿病神经病变。缺氧是糖尿病微血管病变的始动机制, 也可导致病变持续不断加重。 糖尿病心肌病:糖尿病性心肌病是一种复杂的微血管并发症,内皮祖细胞缺乏可能是导致微循环血管稀疏从而显著减少心肌灌注的根本原因。有研究表明,2型糖尿病患者心肌功能异常与内皮祖细胞数量减少和氧化应激的增强有关[37]。Reinhard等[38]对缺血性心肌病大鼠采用粒-巨噬细胞集落刺激因子动员、基质细胞衍生因子趋化内皮祖细胞,能够增加心肌灌注、改善细胞缺血状态、增加心肌动力,从而增强血管稳定性和心肌功能。 糖尿病肾病:干细胞或者祖细胞起源的细胞可以分化成肾小球系膜细胞和肾小管上皮细胞,骨髓起源的单核细胞参与肾脏内皮细胞损伤后修复[39]。Chade等[40]将肾动脉狭窄猪模型移植内皮祖细胞后,发现肾动脉狭窄肾血流量得到改善,肾小球滤过率增加,肾皮质外层毛细血管密度增加,另外,肾动脉狭窄模型的内皮祖细胞的趋化因子基质细胞衍生因子1、血管生成素1、促红细胞生成素及其受体表达也增强,他们还发现内皮祖细胞移植可以改善肾皮质外层肾小球硬化肾小管间质纤维化和血管周围纤维化,刺激内皮祖细胞归巢参与肾脏血管修复,对肾脏具有保护作用。 糖尿病视网膜病变:糖尿病视网膜病变分为增值型糖尿病视网膜病变和非增殖型糖尿病视网膜病变。大量研究证明内皮祖细胞的数量和功能改变对糖尿病视网膜病变的发生起着重要作用。外源性促红细胞生成素能够通过促红细胞生成素/促红细胞生成素受体系统来调节内皮祖细胞,从而减轻非增殖型糖尿病视网膜病变的损伤血管再生,但由于缺血相关的促红细胞生成素受体+内皮祖细胞的反弹性增多,可能会加重糖尿病视网膜病变的视网膜血管新生[41]。Fadini等[31]对60例2型糖尿病患者合并外周血管疾病和糖尿病视网膜病变患者分别计数CD34+KDR+的内皮祖细胞数目,结果发现糖尿病视网膜病变患者比外周血管疾病患者CD34+KDR+的内皮祖细胞细胞数目增多。此外神经营养因子可能通过刺激和动员内皮祖细胞诱导血管形成,从而导致糖尿病患者病理性视网膜血管新生[42]。而健康人外周血内皮祖细胞能够有效地修复损伤的视网膜血管,糖尿病患者外周血中内皮祖细胞功能障碍则不能有效地对损伤血管进行修复[43]。 糖尿病神经病变:糖尿病神经病变是由血管病变和代谢异常造成的。糖尿病神经病变均伴有神经微循环营养血管的缺血缺氧。糖尿病周围神经病变发病机制尚未完全明确,目前主要是由与高血糖相关的经典的多元醇途径、糖激化终产物途径、蛋白激酶途径和己糖胺途径等激活,导致氧化应激产物增多而增加了脂质、DNA、蛋白的过氧化,从而诱导细胞的凋亡并减少了神经血供。Jeong等[44]通过局部注射骨髓源性的内皮祖细胞于STZ所致的糖尿病神经病变大鼠的后肢,发现大鼠坐骨神经的感觉和运动神经传导速度,血流以及毛细血管密度较注射前均得到了恢复。"
| [1] Hristov M, Schmitz S, Nauwelaers F,et al.A flow cytometric protocol for enumeration of endothelial progenitor cells and monocyte subsets in human blood. J Immunol Methods. 2012; 381(1-2):9-13. [2] Ma XL,Sun XL,Wan CY,et al.Significance of circulating endothelial progenitor cells in patients with fracture healing process.J Orthop Res. 2012;30(11):1860-1866. [3] Fadini GP, Avogaro A. Potential manipulation of endothelial progenitor cells in diabetes and its complications. Diabetes,obesity & metabolism. 2010;12(7):570-583. [4] Fadini GP, Losordo D, Dimmeler S.Critical reevaluation of endothelial progenitor cell phenotypes for therapeutic and diagnostic use. Circulation research.2012;110(4):624-637. [5] Wang H, Huang H, Yin Y,et al. [Platelet derived growth factor receptor beta over-expression in endothelial progenitor cells promote reendothelialization after vascular injury]. Zhonghua xin xue guan bing za zhi.2014;42(3):214-218. [6] Tang B, Gong JP, Sun JM, et al.Construction of a plasmid for expression of rat platelet-derived growth factor C and its effects on proliferation, migration and adhesion of endothelial progenitor cells.Plasmid. 2013;69(3):195-201. [7] Medina RJ, O'Neill CL, O'Doherty TM, et al.Myeloid angiogenic cells act as alternative M2 macrophages and modulate angiogenesis through interleukin-8. Molecular medicine.2011;17(9-10):1045-1055. [8] Stitt A, Medina R. Harnessing Endothelial Progenitor Cells for Therapeutic Angiogenesis.//Mehta JL, Dhalla NS, eds. Biochemical Basis and Therapeutic Implications of Angiogenesis. Vol 6: Springer New York; 2013:445-458. [9] Madonna R,De Caterina R.Cellular and molecular mechanisms of vascular injury in diabetes--part II: cellular mechanisms and therapeutic targets.Vascul Pharmacol. 2011; 54(3-6):75-79. [10] Jung C,Rafnsson A,Shemyakin A,et al.Different subpopulations of endothelial progenitor cells and circulating apoptotic progenitor cells in patients with vascular disease and diabetes. Int J Cardiol.2010;143(3):368-372. [11] Westerweel PE TM, Rafii S, Jaspers JE,et al. Impaired endothelial progenitor cell mobilization and dysfunctional bone marrow stroma in diabetes mellitus. PLoS One. 2013; 8(3):e60357. [12] Galasso G, Schiekofer S, Sato K,et al.Impaired angiogenesis in glutathione peroxidase-1-deficient mice Is associated with endothelial progenitor cell dysfunction. Circ Res. 2006;98(2): 254-261. [13] Felice F, Lucchesi D, di Stefano R, et al.Oxidative stress in response to high glucose levels in endothelial cells and in endothelial progenitor cells: evidence for differential glutathione peroxidase-1 expression.Microvasc Res. 2010; 80(3):332-338. [14] Cubbon RM,Kahn MB,Wheatcroft SB.Effects of insulin resistance on endothelial progenitor cells and vascular repair. Clinical science (London, England: 1979).2009;117(5): 173-190. [15] Cubbon RM, Rajwani A, Wheatcroft SB. The impact of insulin resistance on endothelial function, progenitor cells and repair. Diab Vasc Dis Res. 2007;4(2):103-111. [16] Gallagher KA, Liu ZJ, Xiao M, et al. Diabetic impairments in NO-mediated endothelial progenitor cell mobilization and homing are reversed by hyperoxia and SDF-1 alpha. J Clin Invest. 2007;117(5):1249-1259. [17] Thorin E.Vascular disease risk in patients with hypertriglyceridemia: endothelial progenitor cells, oxidative stress, accelerated senescence, and impaired vascular repair. Can J Cardiol. 2011;27(5):538-540. [18] Issan Y, Kornowski R, Aravot D, et al.Heme oxygenase-1 induction improves cardiac function following myocardial ischemia by reducing oxidative stress.PloS one.2014; 9(3):1-12. [19] Kim DH, Vanella L, Inoue K,et al.Epoxyeicosatrienoic acid agonist regulates human mesenchymal stemcell–derived adipocytes through activation of HO-1-pAKT signaling and a decrease in PPARg. Stem Cells Dev. 2010;19(12): 1863-1873. [20] Shibata R,Sato K, Pimentel DR, et al.Adiponectin protects against myocardial ischemia-reperfusion injury through AMPK- and COX-2-dependent mechanisms. Nature medicine. 2005;11(10):1096-1103. [21] Issan Y, Hochhauser E, Kornowski R, et al.Endothelial progenitor cell function inversely correlates with long-term glucose control in diabetic patients: association with the attenuation of the heme oxygenase-adiponectin axis. Can J Cardiol. 2012;28(6):728-736. [22] Zhang HF, Wang L, Yuan HJ, et al. PPAR-gamma agonist pioglitazone prevents apoptosis of endothelial progenitor cells from rat bone marrow. Cell biology international.2013; 37(5): 430-435. [23] Wang Z, Moran EP, Ding L,et al. PPARalpha Regulates Mobilization and Homing of Endothelial Progenitor Cells through the HIF-1/SDF-1 Pathway. Invest Ophthalmol Vis Sci. 2014;55(6):3820-3832. [24] 赵湜,王红祥,毛红,等.葡萄糖对内皮祖细胞表达低氧诱导因子1α的影响[J].中国糖尿病杂志,2010,18(8):582-584. [25] Bento CF, Pereira P. Regulation of hypoxia-inducible factor 1 and the loss of the cellular response to hypoxia in diabetes.Diabetologia.2011;54(8):1946-1956. [26] Kimura H, Weisz A, Ogura T, et al. Identification of hypoxia-inducible factor 1 ancillary sequence and its function in vascular endothelial growth factor gene induction by hypoxia and nitric oxide. J Biol Chem. 2001;276(3): 2292-2298. [27] Caprara V,Scappa S,Garrafa E,et al.Endothelin-1 regulates hypoxia-inducible factor-1alpha and -2alpha stability through prolyl hydroxylase domain 2 inhibition in human lymphatic endothelial cells. 2014 Mar 6. pii: S0024-3205 (14)00287-2. [28] Miao RZ, Liu LQ, Chen L, et al. Activity of heme oxygenase-1 affects expression levels of hypoxia inducible factor-1 gene in vitro. Chin Med J (Engl). 2012 ;125(7):1310-1315. [29] Oh YS, Kim HY, Song IC, et al. Hypoxia induces CXCR4 expression and biological activity in gastric cancer cells through activation of hypoxia-inducible factor-1alpha. Oncology reports.2012;28(6):2239-2246. [30] Mao L,Huang M,Chen SC,et al.Endogenous endothelial progenitor cells participate in neovascularization via CXCR4/SDF-1 axis and improve outcome after stroke. CNS neuroscience & therapeutics.2014;20(5):460-468. [31] Fadini GP, Sartore S, Albiero M, et al. Number and function of endothelial progenitor cells as a marker of severity for diabetic vasculopathy. Arteriosclerosis, thrombosis,and vascular biology. 2006;26(9):2140-2146. [32] Cheung N, Wang JJ, Klein R,et al.Diabetic retinopathy and the risk of coronary heart disease: the Atherosclerosis Risk in Communities Study. Diabetes care.2007;30(7):1742- 1746. [33] Albiero M, Menegazzo L, Boscaro E,et al.Defective recruitment, survival and proliferation of bone marrow-derived progenitor cells at sites of delayed diabetic wound healing in mice. Diabetologia.2011;54(4):945-953. [34] Cheng Y, Guo S, Liu G, et al.Transplantation of bone marrow-derived endothelial progenitor cells attenuates myocardial interstitial fibrosis and cardiac dysfunction in streptozotocin-induced diabetic rats.Int J Mol Med. 2012; 30(4):870-876. [35] Shen WC, Liang CJ, Wu VC, et al. Endothelial Progenitor Cells Derived from Wharton's Jelly of the Umbilical Cord Reduces Ischemia-Induced Hind Limb Injury in Diabetic Mice by Inducing HIF-1α/IL-8 Expression. Stem Cells and Development. 2013;22(9):1408-1418. [36] Jiang M,Wang B,Wang C,et al.In vivo enhancement of angiogenesis by adenoviral transfer of HIF-1alpha-modified endothelial progenitor cells (Ad-HIF-1alpha-modified EPC for angiogenesis). The international journal of biochemistry & cell biology.2008;40(10):2284-2295. [37] Zhao CT, Wang M, Siu CW, et al. Myocardial dysfunction in patients with type 2 diabetes mellitus: role of endothelial progenitor cells and oxidative stress. Cardiovasc Diabetol. 2012;11:147. [38] Reinhard H, Jacobsen PK, Lajer M, et al. Multifactorial treatment increases endothelial progenitor cells in patients with type 2 diabetes. Diabetologia.2010;53(10):2129-2133. [39] Wang S, Li Y, Zhao J,et al. Mesenchymal stem cells ameliorate podocyte injury and proteinuria in a type 1 diabetic nephropathy rat model. Biology of blood and marrow transplantation : journal of the American Society for Blood and Marrow Transplantation.2013;19(4):538-546. [40] Chade AR, Zhu XY, Krier JD, et al.Endothelial progenitor cells homing and renal repair in experimental renovascular disease. Stem cells.Jun 2010;28(6):1039-1047. [41] Hu LM, Lei X, Ma B, et al.Erythropoietin Receptor Positive Circulating Progenitor Cells and Endothelial Progenitor Cells in Patients with Different Stages of Diabetic Retinopathy. Chin Med Sci J. 2011;26(2):69-76. [42] Liu X, Li Y, Liu Y, et al.Endothelial progenitor cells (EPCs) mobilized and activated by neurotrophic factors may contribute to pathologic neovascularization in diabetic retinopathy. The American journal of pathology.2010; 176(1):504-515. [43] Tan K, Lessieur E, Cutler A, et al. Impaired function of circulating CD34(+) CD45(-) cells in patients with proliferative diabetic retinopathy. Experimental eye research.2010;91(2): 229-237. [44] Jeong JO, Kim MO, Kim H, et al.Dual angiogenic and neurotrophic effects of bone marrow-derived endothelial progenitor cells on diabetic neuropathy. Circulation.2009; 119(5):699-708. |
| [1] | Sun Jiajia, Zhu Haidi, Lu Yun, Zhang Kai. Comparison of bone metabolism markers between type 2 diabetes mellitus and non-type 2 diabetes mellitus patients with hip fracture [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1156-1160. |
| [2] | Wang Ji, Zhang Min, Yang Zhongya, Zhang Long. A review of physical activity intervention in type 2 diabetes mellitus with sarcopenia [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1272-1277. |
| [3] | Ou Hangjun, Zhao Guangjian, Pan Yujia, Gong Caiwei, Zhao Quanwei, Liu Danan. Construction of a lentiviral vector overexpressing fibronectin type III domain containing 5 to inhibit apoptosis of endothelial cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(2): 216-222. |
| [4] | Zhu Biwen, Wang Dongzhi, Wu Di, Gong Tiancheng, Pan Haopeng, Lu Yuhua, Guo Yibing, Wang Zhiwei, Huang Yan. Biomimetic microenvironment constructed from gelatin methacrylamide/platelet-rich plasma hydrogel promotes the function of insulinoma cell line MIN6 in mice [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(12): 1824-1831. |
| [5] | Gao Lei, Qin Xinyuan, Nie Xin, Wang Lei, Wang Jiangning. Extracorporeal circulation compression perfusion in the reconstruction of limb microcirculation from the mechanism of mechanical and chemical signal transduction [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1334-1340. |
| [6] | Zhang Lichuang, Xu Hao, Ma Yinghui, Xiong Mengting, Han Haihui, Bao Jiamin, Zhai Weitao, Liang Qianqian. Mechanism and prospects of regulating lymphatic reflux function in the treatment of rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1459-1466. |
| [7] | Lü Yiyan, Li Hanbing, Ma Xiaoqing, Zhang Han, Zhang Yuhang, Li Genlin. Establishment and characteristic analysis of interior heat and diabetes mouse model using compound factors [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1187-1193. |
| [8] | Chen Xianghe, Liu Bo, Yang Kang, Lu Pengcheng, Yu Huilin. Treadmill exercise improves the myocardial fibrosis of spontaneous type 2 diabetic mice: an exploration on the functional pathway [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1210-1215. |
| [9] | Zhu Bingbing, Deng Jianghua, Chen Jingjing, Mu Xiaoling. Interleukin-8 receptor enhances the migration and adhesion of umbilical cord mesenchymal stem cells to injured endothelium [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1045-1050. |
| [10] | Zhang Yujie, Yang Jiandong, Cai Jun, Zhu Shoulei, Tian Yuan. Mechanism by which allicin inhibits proliferation and promotes apoptosis of rat vascular endothelial cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1080-1084. |
| [11] | Shui Xiaoping, Li Chunying, Li Shunchang, Sun Junzhi, Su Quansheng . Effects of aerobic and resistance exercises on brain-derived neurotrophic factor, nuclear factor-kappa B and inflammatory cytokines in skeletal muscle of type II diabetic rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 669-675. |
| [12] | Li Kongmei, Luo Yicai, Li Hao. Effect of inhibiting miR-203-3p expression on bone formation around dental implants in diabetic mice [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(32): 5102-5106. |
| [13] | Ye Ting, Li Jing, Xu Lijuan, Ma Li. Skin administration of Wentong Huoxue Cream can relieve inflammation in diabetic peripheral neuropathy rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(32): 5091-5096. |
| [14] | Tian Tian, Ouyang·Juyan, Li Yu, Miyesai·Ainiwaer, He Juanli, Li Zhenhua, Wang Hong. Mechanism by which echinacoside delays the senescence of human umbilical vein endothelial cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(31): 5014-5019. |
| [15] | Lin Bo, Chen Xinyu, Jin Qiu, Zhu Zhiman, Zhao Wenhui. Effects of miR-126-3p from adipose-derived mesenchymal stem cell released exosomes on human umbilical vein endothelial cell glucolipotoxicity [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(30): 4773-4779. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||