Chinese Journal of Tissue Engineering Research ›› 2014, Vol. 18 ›› Issue (36): 5885-5890.doi: 10.3969/j.issn.2095-4344.2014.36.025
Previous Articles Next Articles
Cryopreservation of human ovarian tissue and the evaluation methods
Zhang Ling, Peng Tao, Yu Yan-ling
- Reproductive Medical Center, Maternal and Child Health Hospital of the Xinjiang Uygur Autonomous Region, Urumqi 830000, Xinjiang Uygur Autonomous Region, China
-
Revised:2014-06-07Online:2014-08-30Published:2014-08-30 -
Contact:Yu Yan-ling, Chief physician, Master’s supervisor, Reproductive Medical Center, Maternal and Child Health Hospital of the Xinjiang Uygur Autonomous Region, Urumqi 830000, Xinjiang Uygur Autonomous Region, China -
About author:Zhang Ling, Master, Reproductive Medical Center, Maternal and Child Health Hospital of the Xinjiang Uygur Autonomous Region, Urumqi 830000, Xinjiang Uygur Autonomous Region, China -
Supported by:High-Technology Research and Development Program of Xinjiang Uygur Autonomous Region, No. 201317105; Science and Technology Program of Urumqi, No. Y121320016
CLC Number:
Cite this article
Zhang Ling, Peng Tao, Yu Yan-ling. Cryopreservation of human ovarian tissue and the evaluation methods[J]. Chinese Journal of Tissue Engineering Research, 2014, 18(36): 5885-5890.
share this article
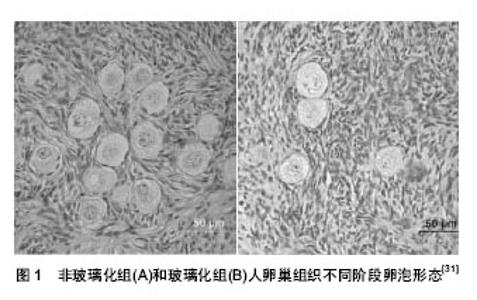
2.1 卵巢组织冷冻技术 卵巢组织冷冻技术即将离体的组织或器官保存在低温中并保持其活性。目前主要使用的卵巢组织冷冻方法主要有慢速冷冻、快速冷冻和玻璃化冷冻。 2.1.1 慢速冷冻 1998年,Oktay等[8]首次将取出的人卵巢皮质组织在-197 ℃液氮中保存。至此之后有很多报道这样的冷冻方法。2004年Donnez等[9]、2005年Meirow等[10]、2006年Demeestere等[11]以及2010年Serrano等[12]均报道慢速冷冻卵巢组织,解冻后自体移植,或进行辅助生殖技术获得自然妊娠或活婴分娩。这些研究证实了慢速冷冻法可使卵巢组织保存卵泡质量,并能获得具有生殖潜能的卵巢组织。 2.1.2 快速冷冻 快速冷送通常的方法是将卵巢组织置于含低浓度(1.5 mol/L)的冷冻保护剂中。在液氮蒸汽中放置> 12 h后投人液氮中保存[13]。目前对于该方法的报道不多。其有效性需进一步研究。 2.1.3 玻璃化冷冻 在20世纪90年代初期,有许多学者将卵巢组织进行玻璃化冷冻,并取得一系列的成功。在1996年出现第1例关于人卵巢组织低温保存的报道[14]。Wang等[15]研究表明,玻璃化冷冻较慢速冷冻法可以更好地保存人卵巢组织的间质细胞、胶原束、细胞间隙及始基卵泡。Keros等[16]报道人卵巢组织玻璃化冷冻后对卵泡的保存效果与慢速冷冻相当,但玻璃化冷冻后卵巢基质保存的结构完整性明显优于慢速冷冻组。玻璃化冷冻法使冷冻过程简化,费用低,效率高,具有巨大的发展潜力[17-19]。 2.2 影响冷冻化效果的因素 目前还没有对卵巢组织冷冻的统一方案,可能影响卵巢组织冷冻的因素,总结如下。 2.2.1 患者的选择 关于卵巢冻存的适应人群相关文献已进行了一些研究[20-23],在卵巢冷冻保存过程中,会损失7%的卵泡,移植后新血供建立前还会丧失60%以上的始基卵泡[24],因此,卵巢组织必须存有相当数量才能有良好的冷冻效果,必须考虑患者的疾病及年龄因素。不同种类的卵巢疾病,会造成卵巢组织不同程度的损伤。Catherine等[25]对卵巢组织冷冻的研究发现,随着患者年龄的增大,其卵巢中始基卵泡和初级卵泡的平均密度在不断下降。而现有文献对于卵巢疾病种类、患者年龄及患者其他因素等对冷冻效果的影响还不是很全面,有待进一步研究论证。 2.2.2 卵巢组织标本的大小 卵巢组织块的厚度可能会影响冷冻保护剂的渗透,从而影响冷冻剂的保护作用。目前文献报道大部分卵巢组织块都处理为厚度约1 mm,Meirow等[26]将人卵巢组织切至厚度为1 mm左右,得到较好的保存效果。李宇彬等[27]研究表明,0.5-1.0 mm的厚度,卵巢组织与新鲜组对照始基卵泡正常率没有显著的差异。最近,赵硕[28]对大块人卵巢组织的冻融进行研究表明,15 mm2×(2.0-3.0) mm的人卵巢组织冷冻复苏后卵泡形态及功能良好,冷冻大块卵巢组织具有可行性。 2.2.3 冷冻保护剂 Amorim等[29]的研究显示,二甲基亚砜与乙二醇效果最好。严晓南等[30]将二甲基亚砜与乙二醇联合应用,既促进玻璃化过程,提高冷冻效率,又能减少两者的用量,减轻冷冻保护剂的细胞毒性作用。Sheikhi等[31]联合二甲基亚砜、乙二醇和丙二醇作为冷冻保护剂对人卵巢组织进行玻璃化冷冻,得到形态正常卵泡的比率达为92.31%(图1)。 Isachenko等[32]报道单独用渗透性冷冻保护剂比混合应用蔗糖的冷冻保存效果好。Xiao等[33]研究发现,联合使用低浓度的二甲基亚砜,乙二醇和蔗糖既可以达到很好的冷冻效果,也降低了冷冻保护剂的毒性。因此,冷冻保护剂的选择及其浓度的确定,以及和冷冻方法的结合,需要进一步实验探讨,才能得到最佳冷送效果的保护剂配比。"
| [1] Meriow D, Biederman H, Anderson RA, et al. Toxicity of chemotherapy and radiation on femalc reproduction. Clin Obstet Gynecol.2010;53:727-739. [2] Schover LR. Rates of postcancer parenthood. J Clin Oncol. 2009;27:321-322. [3] Tomao F,Spinelli GP,Panici PB,et al.Ovarian funtion, reproduction and strategies for fertility preservation sfter brease cancer. Crit Rev Oncol Hematol.2010;76:1-12. [4] Blumenfeld Z. How to preserve fertility in young women exposed to chemotherapy? The role of GnRH agonist cotreatment in addition to cryopreservation of embrya, oocytes, or ovaries. Oncologist.2007;12:1044-1054. [5] Chen C. Pregnacy sfter human oocyte cryopreseration. Lancet.1986;1:884-886. [6] Behringer K, Wildt L,Mueller H,et al.No protection of the ovarian follicle pool with the use of GnRH-analogues or oral contraceptives in young women treated with escalated BEACOPP for advanced-stage Hodgkin lymphoma. Final results of a phase Ⅱtrial from the German Hodgkin Study Group. Ann Oncol.2010;21:2052-2060. [7] Gosden RG, Baird DT, Wade JC, et al. Restoration of fertility to oophorectmized sheep by ovarian autografts stored at -196 degrees C. Hum Reprod.1994;9:597-603. [8] Oktay K, Newton H, Aubard Y, et al.Cryopreservation of immature human oocytes and ovarian tissue an emerging technology. Fertil Steril.1998;69(1):1-7. [9] Donnez J, Dolmans MM, Demylle D, et al. Livebirth after orthotopic transplantion of crypreserved ovarian tissue. Lancet. 2004;364(9443):1405-1410. [10] Meirow D,Levron J,Eldar-Geva T,et al.Pregnancy after transplantation of cryopreserved ovarian tissue in patient with ovarian failure after chemotherapy. N Engl J Med.2005;353(3): 318-321. [11] Demeestere I,Simon P,Buxant F,et al.Ovarian function and spontaneous pregnancy after combined heterotopic and orthotopic cryopreserved ovarian tissue transplantation in a patient preciously treated with bone marrow transplantation: case report. Hum Reprod.2006;21(8):2010-2014. [12] Serrano MS, Crespo J, Mirabet V.Twins born after transplantation of ovarian cortical tissue and oocyte citrification. Fertil Steril.2010;93(1):268.e11-3. [13] Gook A, Edgar DH, Stern C. Effect of cooling rate and dehydration regimen on the histological appearance of human ovarian cortex following cryopreservation in 1,2-propanediol. Hum Reprod.1999;14(8):2061-2068. [14] Hovatta O. Methods for cryopreservation of human ovarian tissue. Reprod Biomed Online.2005;10(6):729-734. [15] Wang Y,Xiao Z,Li L,et al.Novel needle immersed vitrification: a practical and convent method with potential advantages in mouse and human ovarian tissue cryopreservation. Hum Reprod.2008;23(10):2256-2265. [16] Keros V,Xella S,Hultenby K,et al.Vitrification versus controlled-rate freezing in cryopreservation of human ovarian tissue. Hum Reprod.2009;24(7):1670-1683. [17] Gandolfi F, Paffoni A, Papasso Brambilla E, et al.Efficiency of equilibrium cooling and vitrification procedures for the cryopreservation of ovarian tissue: comparative analysis between human and animal models. Fertil Steril.2006;85(1): 1150-1156. [18] Lornage J, Salle B. Ovarian and oocyte cryopreservation. Curr Opin Obstet Gynecol.2007;19(4):390-394. [19] Fabbri R, Pasquinelli G, Bracone G, et al.Cryopreservation of human ovarian tissue. Cell Tissue Bank.2006;7(2): 123-133. [20] Poirot C,Vacher-Lavenu M,Helardot P,et al.Human ovarian tissue cryopreservation:Indications and feasibility. Hum Reprod. 2002;17:1447-1452. [21] Hovatta O. Cryobiology of ovarian and testicular tissue. Clin Obstet Gynecol. 2003;17:331-342. [22] Practice committee of ASRM and SART. Ovarian tissue and oocyte cryopreservation. Fert Steril.2004;86:142-147. [23] 徐玲玲,冒韵东. IVF-ET中获卵数与储备卵泡、年龄、卵巢体积及基础激素水平的相关性研究[J].实用临床医药杂志,2007, 11(4):50-52. [24] Aubard Y, Poirot C, Piver P. Cryopreservation of ovarian tissur. Gynecol Obstet Fertil.2002;30(5):358-366. [25] Poirot C, Vacher-Lavenu MC, Helardot P, et al. Human ovarian tissue cryopreservation: indications and feasibility. Hum Reprod. 2002;17(6):1447-1452. [26] Merrow D, Levron J, Eldar-Geva T, et al. Pregnancy after transplantation of vryopreserved ovarian tissue in a patient with ovarian failure after chemotherapy. N Engl J Med.2005; 353(3):318-321. [27] 李宇彬,周灿权,麦庆云,等.人类卵巢组织条厚度对超低温冷冻效果影响的研究[J].生殖医学杂志,2011,20(2):136-140. [28] 赵硕,赵淑琴,时庆,等.两种冷冻方法对人大块卵巢组织卵泡活性的影响[J].解剖学报,2013,44(3):427-432. [29] Amorim CA, Rondina D, Lueci CM, et al. Cryopreservation of sheep primordial follicles. Reprod Domest Anita.2007;42(1): 53-57. [30] 严晓南,刘平,张小为,等.玻璃化冷冻保存对小鼠卵巢组织的影响[J].中国比较医学杂志,2007,17(5):262-265. [31] Sheikhi M, Hultenby K, Niklasson B, et al. Clinical grade vitrification of human ovarian tissue: an ultrastructural analysis of follicles and stroma in oitrified tissue. Hum Reprod. 2011;26(3):594-603. [32] Lsachenko V, Isachenko E, Rahimi G, et al.Cryopreservation of human ovarian tissue by direct plunging into liquid nitrogen: negative effect of disaccharides in vitrification solution. Cryo Letters.2002;23(5):333-344. [33] Xiao Z, Wang Y, Li L, et al. Needle immersed vitrification can lower the concentration of cryoprotectant in human ovarian tissue cryopreservation. Fertil Steril.2010;94(6):2323-2328. [34] Babaei H, Derakhshanfar A, Kheradmand A. Vitrification of mouse ovaries using ethylene glucol and DMSO as cryoprotenctants: histopathological evaluation. Veterinarski Arhiv.2007;77(1):19-27. [35] Zhou XH, Wu YJ, Shi J, et al. Cryopreservation of human ovarian tissue: comparison of novel direct cover vitrification and conventional vitrification. Cryobiology.2010;50(2): 101-105. [36] Newton H, Fisher J, Arnold JRP, et al. Permeation of human ovarian tissue with cryoprotecrive agents in preparation for cryopreservation. Human Reproduction.1998;13(2):377-380. [37] Amorim CA, David A, Van Langendonckt A, et al. Vitrification of human ovarian tissue: effect of different solutions and procedures. Fertil Steril.2011;95(3):1094-1097. [38] Santos RR, tharasanit T, Van Haeften T, et al. Vitrification of goat preantral follicles enclosed in ovarian tissue by using conventional and solid-surface vitrification methods. Cell Tissue Res.2007;327(1):167-176. [39] Isachenko E, Isachenko V, Nawroth F, et al. Human ovarian tissue preservation: is vitrification acceptable method for assisted reproduction. Cryo Letters.2008;29(4):301-314. [40] 黄丽萨,谭世桥,屈清华.人类卵巢组织玻璃化冷冻及复苏后形态学及组织增殖活性观察[J].四川大学学报:医学版,2010, 41(2): 261-264. [41] 吕艳.自制冷冻环卵巢组织玻璃化冷冻的实验研究[D].武汉:华中科技大学,2009. [42] Donnez J, Dolmans MM. Cryopreservation and transplantation of ovarian tissue. Clin Obstet Gynecol.2010;53(4):787-796. [43] 彭艳,于丛一,李予,等.慢速冷冻和玻璃化冷冻对人类卵泡组织中卵泡形态的研究[J].中山大学学报,2007,28(1):70-74. [44] 李云秀,孙莹璞,唐莉,等.不同冷冻方案保存人类卵巢组织的活性比较[J].中国组织工程研究,2013,17(53):9125-9131. [45] 孙文枫,苏迎春,孙莹璞,等.冷冻保存对人卵巢组织雌二醇分泌功能的影响[J].生殖与避孕,2009,29(6):406-409. [46] Rimon E, Cohen T, Amsterdama A. Apoptosis in cryopreserved ovarian tissue-a tool of evaluation cryopreservation utility. Int J Oncol.2005;27(2):345-353. [47] Demirci B, Salle B, Frappart L, et al. Morphological alternations and DNA fragmentation in oocytes from primordial and primary follicles after freezing-thawing of ovarian cortex in sheep. Fertil Steril.2002;77(3):595-600. [48] Rimon E, Cohen T, Dantes A, et al. Apoptosis in cryopreserved human ovarian tissue obtained from cancer patients: a tool for evaluating cryopreservation utility. Int J Oncol.2005;27(2):345-353. [49] Gougeon A, Busso D. Morphologic and functional determinants of primordial and primary follocles in the monkey ovary. Mol Cell Endocrinol.2000;163(1-2):33-42. [50] Hussein MR, Bedaiwy MA, Falcone T.Analysis of apoptotic cell death, Bcl-2, and p53 protein exposure to warm ischemia. Fertil Steril.2006;85(suppl 1):1082-1092. [51] 傅晓艳,周颖,韩海艳,等.乙二醇、丙二醇对冻融人卵巢卵泡凋亡的影响[J].中国病理生理杂志,2009,25(3):513-518. [52] 黄丽丽,王文军,李琳,等.人卵巢组织冷冻对印迹基因IGF-2/H19甲基化程度及表达的影响[J].国际生殖健康/计划生育杂志,2012, 31(6):436-438. [53] Demeestere I, Simon P, Emiliani S, et al. Fertility preservation successful: transplantation of cryopreserved ovarian tissue in a young patient previously treated for Hodgkin’s disease. Oncologist.2007;12(12):1437-1442. [54] Meirow D,Levron J,Eldar-Geva T,et al.Monitoring the ovaries after autotransplantation of cryopreserved ovarian tissue: endocrine studies, in vitro fertilization cycles, and live birth. Fertil Steril.2007;87(2):418.e7-418.e15. [55] Kim SS, Hwang IT, Lee HC. Heterotopic autotransplantation of cryobanked human ovarian tissue as a strategy to restore ovarian function. Feril Steril.2004;82(4):930-932. [56] Oktay K, Buyuk E, Veeck L, et al. Embryo development after heterotopic transplantation of cryopreserved ovarian tissue. Lancet.2004;363(9412):837-840. [57] Wolner-Hanssen P,Hagglund L,Ploman F,et al. Autotransplantation of cryopreserved ovatian tissue to the right forearm 4(1/2) years after autologous stem cell transplantation. Acta Obstet Gynecol Scand.2005;84(7): 695-698. [58] Abir R,Orvieto R,Raanani H,et al.Parameters affecting successful transplantation of frozen-thawed human fetal ovaries into immunodeficient mice. Fertil Steril.2003;80(2): 421-428. [59] Aubard Y. Ovarian tissue xenografting. Eur J Obstet Gynecol Reprod Biol.2003;108(1):14-18. |
| [1] | Li Shanshan, Guo Xiaoxiao, You Ran, Yang Xiufen, Zhao Lu, Chen Xi, Wang Yanling. Photoreceptor cell replacement therapy for retinal degeneration diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1116-1121. |
| [2] | Jiang Xin, Qiao Liangwei, Sun Dong, Li Ming, Fang Jun, Qu Qingshan. Expression of long chain non-coding RNA PGM5-AS1 in serum of renal transplant patients and its regulation of human glomerular endothelial cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 741-745. |
| [3] | Yang Xin, Jin Zhe, Feng Xu, Lu Bing. The current situation of knowledge and attitudes towards organ, eye tissue, body donation of residents in Shenyang [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 779-784. |
| [4] | Zeng Xianghong, Liang Bowei. A new strategy for the treatment of osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 431-437. |
| [5] | Ruan Guangping, Yao Xiang, Liu-Gao Miyang, Cai Xuemin, Li Zian, Pang Rongqing, Wang Jinxiang, Pan Xinghua. Umbilical cord mesenchymal stem cell transplantation for traumatic systemic inflammatory response syndrome in tree shrews [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 3994-4000. |
| [6] | Chen Xiao, Guo Zhi, Chen Lina, Liu Xuanyong, Zhang Yihuizhi, Li Xumian, Wang Yueqiao, Wei Liya, Xie Jing, Lin Li. Factors affecting the mobilization and collection of autologous peripheral blood hematopoietic stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(19): 2958-2962. |
| [7] | Zhang Jianhui, Ma Heran, Tan Yi, Wang Zhihui. Knee injury repair using human adipose-derived mesenchymal stem cells-based scaffold-free three-dimensional gel-like construct in pigs [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(19): 2969-2975. |
| [8] | Zhang Hongqing, Xie Xufang, Wu Xiaomu. Effectiveness and clinical application limitations of stem cells in the treatment of multiple sclerosis and optic neuromyelitis spectrum diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(19): 3049-3056. |
| [9] | Zhao Shuangdan, Zheng Jiahua, Qi Wenbo, Huang Xianghua. Role and mechanism of exosomes derived from mesenchymal stem cells in reproductive system diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(19): 3097-3102. |
| [10] | Sun Weixing, Zhao Yongchao, Zhao Ranzun. Mesenchymal stem cell transplantation in the treatment of myocardial infarction: problems, crux and new breakthrough [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(19): 3103-3109. |
| [11] | Wang Xiaobo, Wang Changan, Han Jianle, Yang Qingyan, Yang Shuaiping, Yang Junwei. Influence of conversion from cyclosporine to tacrolimus on glucose metabolism and cardiovascular risk profiles in stable kidney transplant patients [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(14): 2236-2240. |
| [12] | Cao Linlin, Ding Kaiyang, Song Hao, Wu Guolin, Hu Maogui, Fan Dandan, Zhou Chenyang, Wang Cuicui, Feng Yuanyuan. Efficacy and influencing factors of autologous hematopoietic stem cell transplantation in the treatment of malignant lymphoma [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(13): 1993-1998. |
| [13] | Zhang Wenjian, Ma Lingfu, Wang Zhimin, Mo Wenjian, Zhou Ruiqing. Muscle mass evaluation and influencing factors of sarcopenia in allogeneic hematopoietic stem cell transplantation patients [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(13): 1999-2004. |
| [14] | Li Liqiang, Jiao Longxing, Zhang Wu, Yan Wentao, Li Jian, Li Minghao. Effect of immature dendritic cells derived from bone marrow on rejection of orthotopic liver transplantation in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(13): 2025-2029. |
| [15] | Wei Zhongling, Jiang Yizhi, Huang Laiquan, Yan Jiawei, Yu Zhengzhi, Wang Nana, Huang Chen, Wang Ran, Huang Dongping. Severe aplastic anemia treated with unrelated cord blood combined with matched sibling allogeneic hematopoietic stem cell transplantation [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(13): 2049-2054. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||