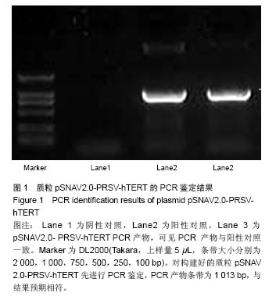
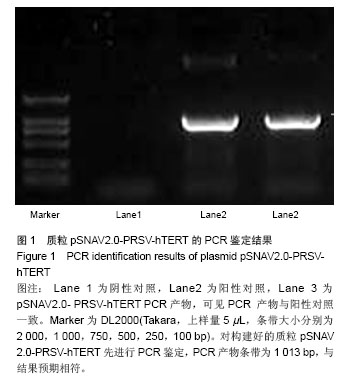
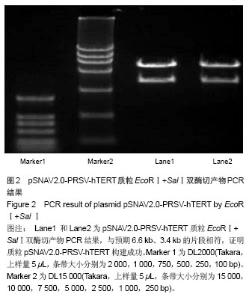
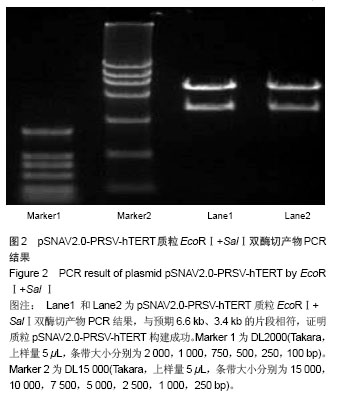
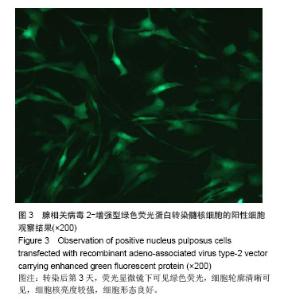
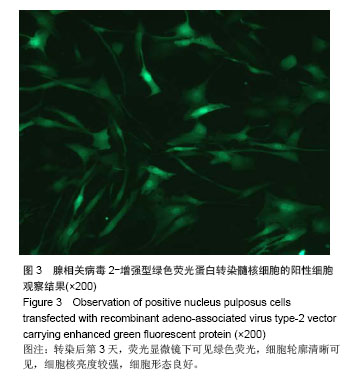
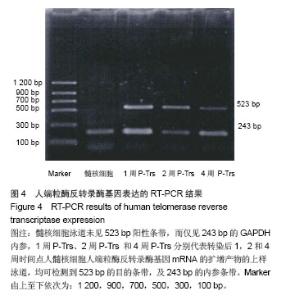
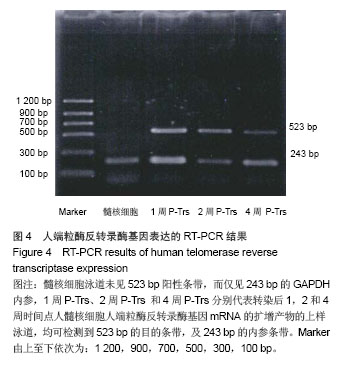
| [1]Meisel HJ, Siodla V, Ganey T, et al. Clinical experience in cell-based therapeutics: disc chondrocyte transplantation A treatment for degenerated or damaged intervertebral disc. Biomol Eng. 2007;24(1):5-21.
[2]Halloran DO, Grad S, Stoddart M, et al. An injectable cross-linked scaffold fornucleus pulposus regeneration. Biomaterials. 2008;29(4):438-447.
[3]Campisi J, Sahn-Ho K, Chang-Su L, et al. Cellular senescence,cancer and aging;the telomere connection.Exp Gerontol. 2001;36:1619.
[4]Greider CW. Telomere length regulation. Annu Rev Biochem. 1996;65:337.
[5]Blackburn EH. Telomere states and cell fates. Nature. 2000; 408(6808):53-56.
[6]Yan P, Bosman FT, Benhattar J. Tissue quanlity is an important determinant of telomerase activity as measured by TRAP assay. Biotechniques. 1998;25(4):660-662.
[7]Wieser M, Stadler G, Jennings P, et al. hTERT alone immortalizes epithelial cells of renal proximal tubules without changing their functional characteristics. Am J Physiol Renal Physiol. 2008;295(5):F1365-1375.
[8]Lee KM, Choi KH, Ouellette MM. Use of exogenous hTERT to immortalize primary human cells. Cytotechnology. 2004; 45(1-2):33-38.
[9]Gao K, Lu YR, Wei LL, et al. Immortalization of mesenchymal stem cells from bone marrow of rhesus monkey by transfection with human telomerase reverse transcriptase gene. Transplant Proc. 2008;40(2):634-637.
[10]Chung SA, Wei AQ, Connor DE, et al. Nucleus pulposus cellular longevity by telomerase gene therapy, Spine. 2007; 32(11):1188-1196.
[11]Matsumura T, Takesue M, Westerman KA, et al. Establishment of an immortalized human-liver endothelial cell line with SV40T and hTERT. Transplantation. 2004; 77(9):1357-1365.
[12]Wei LL, Gao K, Liu PQ, et al. Mesenchymal stem cells from Chinese Guizhou minipig by hTERT gene transfection, Transplant Proc. 2008;40(2):547-550.
[13]Sakai D, Mochida J, Yamamoto Y, et al. Immortalization of human nucleus pulposus cells by a recombinant SV40 adenovirus vector: establishment of a novel cell line for the study of human nucleus pulposus cells.Spine. 2004;29(14):
1515-1523.
[14]李连华,翁习生,邱贵兴,等. 2型重组腺相关病毒转染新西兰兔关节软骨细胞的体外研究[J]. 中华医学杂志,2006, 86(21):
1489-1492.
[15]张银刚,郭雄,李天清,等. 髓核内注射骨髓间质干细胞对椎间盘基质的影响[J]. 西安交通大学学报,2007,28(6)675-679.
[16]孙浩林, 李淳德. 原代培养差速贴壁法分离纯化大鼠腰椎间盘髓核细胞[J]. 中国组织工程研究与临床康复,2011,15(46):
8569-8573.
[17]林顺新,徐振华,汪平,等. 兔椎间盘髓核细胞体外培养自然退变的观察[J]. 医学新知杂志,2009,19(3):145-147.
[18]余磊,熊绍虎,戴景兴,等. 兔髓核内细胞的培养及生长曲线的测定[J]. 中华创伤骨科杂志, 2004,6(2): 184-186.
[19]肖剑,赵剑,潘欣,等. 兔腰椎间盘髓核细胞的培养及形态观察[J]. 中国矫形外科杂志, 2001,8(8): 793-795.
[20]张传志,周跃,李长青. 兔髓核细胞体外最佳培养营养条件的探索[J]. 中国矫形外科杂志, 2005,13(12): 1093-1094.
[21]陆森,李国强,王学浩,等.腺相关病毒载体介导的CTLA4Ig表达对大鼠肝移植免疫抑制作用的实验研究[J]. 南京医科大学学报(自然科学版),2005,25(4):224-227.
[22]杨民,马庆军,郭均, 等.重组人骨形态发生蛋白7的腺相关病毒载体的构建[J]. 中国骨与关节损伤杂志,2005,20(7): 458-461.
[23]邰升,苏志雷. 腺相关病毒在肝脏移植应用的研究进展[J]. 世界华人消化杂志,2006,14(22):2155-2158.
[24]黄云剑,赵景宏,杨唐俊, 等.Smad6和Smad7基因腺相关病毒载体的构建及其表达[J].细胞与分子免疫学杂志,2004, 20(3): 274-277.
[25]贺民,鞠延,毛伯镛. 腺相关病毒载体与中枢神经系统疾病的基因治疗[J].华西医学,2000,15(3):379-380.
[26]刘希民. 腺相关病毒载体基础研究及其在造血系统的应用进展[J].白血病•淋巴瘤,2002,11(1):59-61.
[27]李晨,安云庆. 腺相关病毒载体及其在基因治疗中的应用[J].
中国康复理论与实践,2006,12(1):51-52.
[28]王凯,彭建强,陈方平,等. rAAV2-GPⅡb/GPⅢa载体构建和体外表达、功能实验[J].中国实验血液学杂志,2006,14(2):369-374.
[29]徐世平,吴本俨,王孟薇,等. 胃癌相关基因GCRG 213正反义表达重组腺相关病毒载体的构建[J]. 华南国防医学杂志,2007, 21(3):1-5.
[30]Miller DG, Petek LM, Russell DW. Adeno-associated virus vectors integrate at chromosome breakage sites, Nat Genet. 2004;36(7):767-773.
[31]Iizuka T, Kanzaki S, Mochizuki H, et al. Noninvasive In Vivo Delivery of Transgene via Adeno-Associated Virus into Supporting Cells of the Neonatal Mouse Cochlea. Hum Gene Ther. 2008;19(4):384-390.
[32]丛敏,白艳锋,王萍,等. 腺相关病毒和慢病毒作为小干扰RNA转运载体的比较[J]. 国际外科学杂志,2009,36(9):585-589.
[33]董小岩. 新型腺相关病毒载体及应用研究[J]. 复旦大学,2010.
[34]吕颖慧, 王启钊, 肖卫东, 等. 自身互补型腺相关病毒载体发展趋势[J]. 生物工程学报,2009,25(5): 658-664.
[35]舒静波,刁勇,肖卫东,等. 腺病毒-腺相关病毒嵌合载体的研发趋势[J]. 华侨大学学报(自然科学版),2008,29(2):172-176. |