Chinese Journal of Tissue Engineering Research ›› 2013, Vol. 17 ›› Issue (36): 6501-6507.doi: 10.3969/j.issn.2095-4344.2013.36.019
Previous Articles Next Articles
Platelet-rich plasma affects the proliferation and collagen production of mesenchymal stem cells
Rong Chun, Shen Yan-qing, Lü Ya-qing, Li Ming-yu, Xia Chang-suo
- The Affiliated Hospital of Medical College Qingdao University, Qingdao 266003, Shandong Province, China
-
Received:2012-11-06Revised:2012-12-13Online:2013-09-03Published:2013-09-03 -
About author:Rong Chun, Nurse in-charge, the Affiliated Hospital of Medical College Qingdao University, Qingdao 266003, Shandong Province, China Xcs009@163.com
CLC Number:
Cite this article
Rong Chun, Shen Yan-qing, Lü Ya-qing, Li Ming-yu, Xia Chang-suo. Platelet-rich plasma affects the proliferation and collagen production of mesenchymal stem cells[J]. Chinese Journal of Tissue Engineering Research, 2013, 17(36): 6501-6507.
share this article
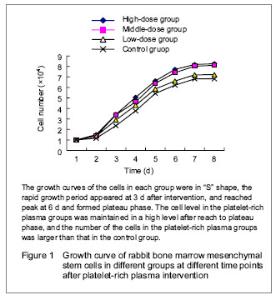
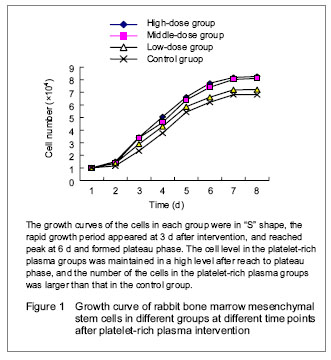
Culture of mesenchymal stem cells After primary cultured for 12 hours, most of the cells were adherent; after 2-3 days, the morphology of the cells was in polygons and fusiform; after 3-5 days, distinct colony formation was observed with rapid proliferation, and the clonies were close to each other; after 10-12 days, cell integration for about 90%, at this time, the cells were arranged in relatively uniform helical or spiral shape. The passaged mesenchymal stem cells adhered rapidly, and after 3 hours, most of the cells were adherent and evenly distributed in fusiform; that of the morphology of the passaged cells was more even than that of the primary cultured cells, the passaged cell growth well with rapid proliferation, obvious nucleus, clear nucleolus and a large proportion of nucleoplasm. Generally, after cultured for 7 days, the cells could reach 90% integration with even morphology and arranged in parallel or swirling spiral. There was no significant difference after continuously passaged to the 5th generation. However, with the increased frequency of passage, the cells appeared diversity; after passaged to the 10th generation, the cells were gradually aging, presented as increased cell debris, flattened cell morphology and decreased proliferation rate till stop. Expressions of surface antigens of mesenchymal stem cells Positive immunocytochemical staining of CD44 and CD90 of passage 3 cells was observed, the cytoplasm was brown or chocolate brown, and the CD34 staining was negative. Growth curve of the cells in each group The number of the incubated cells in each group was counted (mean n=6), then the data were recorded in the EXCEL table to generate the curve (growth curve) automatically. The growth curves of the cells in each group were in “S” shape (Figure 1). At day 1 after incubation, the cells growth in each group was relatively flat, and presented as the latent growth curve. At day 2 after incubation, the cells in the platelet-rich plasma groups had high proliferative activity and grew rapidly, and the growth curve was significantly increased, especially in the high-dose platelet-rich plasma group; then the rapid growth period of the growth curve was constructed. The proliferation of the cells was most significant in the high-dose platelet-rich plasma group with the shortest doubling time about 30 hours, and then reached to peak at 6 days, and formed plateau phase. The cells in the control group were grew rapidly at day 3, and showed the longest doubling time about 46 hours, and after 3 days, the rapid growth period of growth curve was constructed. The cell level in the platelet-rich plasma groups was maintained in a high level after reach to plateau phase, and the number of the cells in the platelet-rich plasma groups was larger than that in the control group."
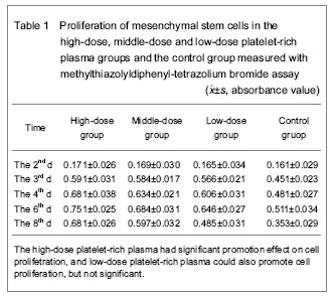
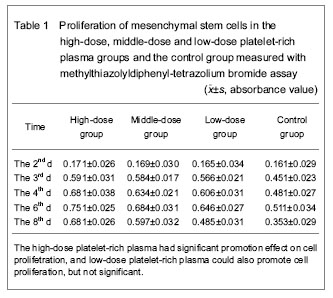
Survival and proliferation ability of mesenchymal stem cells in each group The survival and proliferation ability of mesenchymal stem cells in each group were showed in Tables 1 and 2. Methylthiazolyldiphenyl-tetrazolium bromide assay showed the absorbance values in the high-dose, middle-dose and low-dose platelet-rich plasma groups and the control group at 2 days after intervention were (0.171±0.026), (0.169±0.030), (0.165±0.034) and (0.161±0.029) respectively, and there was no significant difference (P > 0.05), that may be because of the cells growth in the stationary state, there was no significant difference in proliferation of cells. At day 3, there was significant difference in the absorbance value between platelet-rich plasma groups and the control group (P < 0.01), while there was no significant difference between platelet-rich plasma groups (P > 0.05), indicating that the platelet-rich plasma has certain effect in the proliferation of cells at 3 days after intervention without dose-dependent manner. There was significant difference in the absorbance value between high-dose platelet-rich plasma group and middle- and low-dose platelet-rich plasma groups at day 4 (P < 0.05 or P < 0.01), but there was no significant difference between middle-dose platelet-rich plasma group and the low-dose platelet-rich plasma group (P > 0.05), the results indicated that high-dose platelet-rich plasma had significant effect on cell proliferation, the low-dose platelet-rich plasma could also promote cell proliferation but not significant. At day 6, the platelet-rich plasma had significant promotion effect on cell proliferation and showed dose-dependent manner. At day 8, the platelet-rich plasma had significant promotion effect on cell proliferation and showed dose-dependent manner, but also found that the promotion effect was decreased when compared with that in the previous few day, and that may be saturated cell density. Statistical results showed that platelet-rich plasma had significant promotion effect on the proliferation of mesenchymal stem cells (since day 3), and the effect was in time-dependent manner. After cultured for several days, the effect had dose-dependent manner (since day 6), and the high-dose platelet-rich plasma had the most significant effect on cell proliferation."
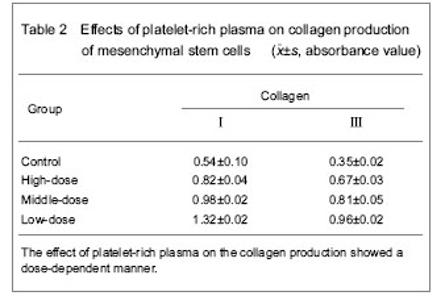
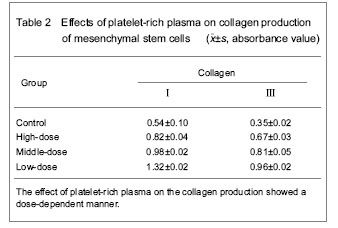
Effect of platelet-rich plasma on collagen production of mesenchymal stem cells Immunochemical staining results and enzyme-linked immunosorbent assay for quantitative determination of collagen showed that mesenchymal stem cells could produce the collagen type Ⅰ and Ⅲ platelet-rich plasma which could significantly promote the collagen production of mesenchymal stem cells, and there was significant difference when compared with the control group (P < 0.05). The effect of platelet-rich plasma on the collagen production showed a dose-dependent manner (Table 2)."
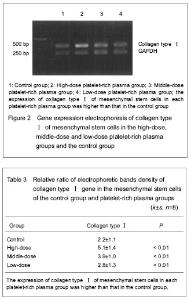
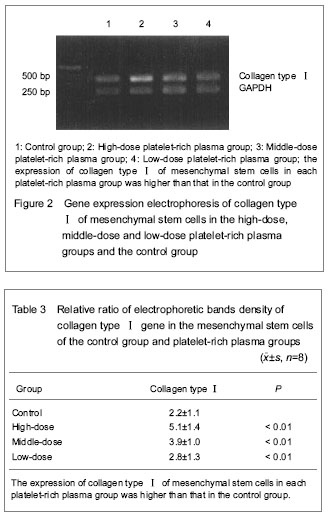
Gene expression of collagen typeⅠ The relative ratio of collagen type Ⅰ density is showed in Table 3. The Tband location of collagen type Ⅰ on the electropherogram was corresponded to the designed amplification length (Figure 2). The expression of collagen type Ⅰ of mesenchymal stem cells in each platelet-rich plasma group was increased when compared with that in the control group, and there was no significant difference between groups (P < 0.01)."
| [1] Mezey E, Mayer B, Németh K. Unexpected roles for bone marrow stromal cells (or MSCs): a real promise for cellular, but not replacement, therapy.Oral Dis. 2010;16(2):129-135. [2] Luo Q, Song G, Song Y, et al. Indirect co-culture with tenocytes promotes proliferation and mRNA expression of tendon/ligament related genes in rat bone marrow mesenchymal stem cells. Cytotechnology. 2009;61(1-2):1-10. [3] Chen X, Zou XH, Yin GL, et al. Tendon tissue engineering with mesenchymal stem cells and biografts: an option for large tendon defects? Front Biosci (Schol Ed). 2009;1:23-32. [4] Zhao L, Shen YQ, Rong C, et al. Characteristics and mechanisms of mesenchymal stem cell culture and induced differentiation. Zhongguo Zuzhi Gongcheng Yanjiu yu Linchuang Kangfu. 2010;14(40)7551-7554. [5] Liao MX, Ning LJ, Chen XH, et al. Strain-induced tenogenic differentiation of bone marrow mesenchymal stem cells. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2010;24(7): 817-821. [6] Chong AK, Chang J, Go JC. Mesenchymal stem cells and tendon healing Front Biosci. 2009 Jan 1;14:4598-605. [7] Zhang CL, Xia CS, Jiang ZY. Isolation of rabbit bone marrow mesenchymal stem cells using density gradient centrifugation and adherence screening methods. Zhongguo Zuzhi Gongcheng Yanjiu yu Linchuang Kangfu. 2009;6: 1181-1184. [8] Rosenbaum AJ, Grande DA, Dines JS. The use of mesenchymal stem cells in tissue engineering: A global assessment. Organogenesis. 2008;4(1):23-27. [9] Appel TR, Pitzsch B, Muller J, et al. Comparison of three different preparation of platelet concentrates for growth factor enrichment. Zhongguo Shouwaike Zazhi. 2002; 13(5):522-528. [10] Xu Y, Tang JB. Effect of basic fibroblast growth factor on matrix synthesis and NF-KB gene expression of tendoncyte. Zhonghua shouwaike zazhi. 2003;19:43-45. [11] Lynch SE, Genco RJ, Marx RE, et al. Tissue engineering: application in maxillofacial surgery and periodontics. Carol stream IL: Quintessence. 1999:11-15. [12] Nakayama N, Lee J, Chiu L. Vascular endothelial growth factor synergistically enhances bone morphogenetic protein-4-dependent lymphohematopoietic cell generation from embryonic stem cells in vitro. Blood. 2000; 95(7): 2275-2283. [13] Sanchez AR, Sheridan PJ, Kupp LI. Is platelet-rich plasma the perfect enhancement factor? A current review. The International journal of oral & maxillofacial implants. 2003; 18(1):93-103. [14] Lekovic V, Camargo PM, Weinlaender M, et al. Effectiveness of a combination of platelet-rich plasma, bovine porous bone mineral and guided tissue regeneration in the treatment of mandibular grade Ⅱ molar furcations in humans. Journal of clinical periodontology. 2003;30(8): 746-751. [15] Xia CS, Chen YH, Sun K, et al. Effects of platelet-rich plasma on BMSCs differentiation into SC in vitro. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2009;23(8): 997-1101. [16] Shan LC, Wang G, Zhang CQ. Effect of various concentrations of platelet-rich plasma on osteogenic differentiation of skeletal muscle-derived stem cells. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2009;23(8): 991-996 [17] Mishra A, Tummala P, King A, et al. Buffered platelet-rich plasma enhances mesenchymal stem cell proliferation and chondrogenic differentiation. Tissue Eng Part C Methods. 2009;15(3):431-435. [18] Jaibaji M. Advances in the biology of zone II flexor tendon healing and adhesion formation. Ann Plast Surg. 2000;45: 83-92. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [3] | Jiang Hongying, Zhu Liang, Yu Xi, Huang Jing, Xiang Xiaona, Lan Zhengyan, He Hongchen. Effect of platelet-rich plasma on pressure ulcers after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1149-1153. |
| [4] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [5] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [6] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [7] | Fan Quanbao, Luo Huina, Wang Bingyun, Chen Shengfeng, Cui Lianxu, Jiang Wenkang, Zhao Mingming, Wang Jingjing, Luo Dongzhang, Chen Zhisheng, Bai Yinshan, Liu Canying, Zhang Hui. Biological characteristics of canine adipose-derived mesenchymal stem cells cultured in hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1002-1007. |
| [8] | Geng Yao, Yin Zhiliang, Li Xingping, Xiao Dongqin, Hou Weiguang. Role of hsa-miRNA-223-3p in regulating osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1008-1013. |
| [9] | Lun Zhigang, Jin Jing, Wang Tianyan, Li Aimin. Effect of peroxiredoxin 6 on proliferation and differentiation of bone marrow mesenchymal stem cells into neural lineage in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1014-1018. |
| [10] | Zhu Xuefen, Huang Cheng, Ding Jian, Dai Yongping, Liu Yuanbing, Le Lixiang, Wang Liangliang, Yang Jiandong. Mechanism of bone marrow mesenchymal stem cells differentiation into functional neurons induced by glial cell line derived neurotrophic factor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1019-1025. |
| [11] | Duan Liyun, Cao Xiaocang. Human placenta mesenchymal stem cells-derived extracellular vesicles regulate collagen deposition in intestinal mucosa of mice with colitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1026-1031. |
| [12] | Pei Lili, Sun Guicai, Wang Di. Salvianolic acid B inhibits oxidative damage of bone marrow mesenchymal stem cells and promotes differentiation into cardiomyocytes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1032-1036. |
| [13] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [14] | Wang Shiqi, Zhang Jinsheng. Effects of Chinese medicine on proliferation, differentiation and aging of bone marrow mesenchymal stem cells regulating ischemia-hypoxia microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1129-1134. |
| [15] | Kong Desheng, He Jingjing, Feng Baofeng, Guo Ruiyun, Asiamah Ernest Amponsah, Lü Fei, Zhang Shuhan, Zhang Xiaolin, Ma Jun, Cui Huixian. Efficacy of mesenchymal stem cells in the spinal cord injury of large animal models: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1142-1148. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||