Chinese Journal of Tissue Engineering Research ›› 2013, Vol. 17 ›› Issue (5): 777-784.doi: 10.3969/j.issn.2095-4344.2013.05.003
Previous Articles Next Articles
Application of mycophenolate mofetil combined with low-dose tacrolimus and corticosteroid regimen after renal transplantation
Ji Shu-ming1, Chen Jiang-hua2, Tan Jian-ming3, Zhang Xiao-dong4, Zhu Tong-yu5, Chen Li-zhong6,Liu Zhi-hong1
- 1 Research Institute of Nephrology, School of Medicine, Nanjing University/Nanjing General Hospital of Nanjing Military Command, PLA, Nanjing 210002, Jiangsu Province, China
2 The First Affiliated Hospital of Zhejiang University School of Medicine, Hangzhou 310012, Zhejiang Province, China3 Fuzhou General Hospital of Nanjing Military Command, Fuzhou 350025, Fujian Province, China4 Beijing Chaoyang Hospital, Beijing 100020, China5 Zhongshan Hospital, Fudan Univsersity, Shanghai 200032, China6 The First Affiliated Hospital, Sun Yat-sen University, Guangzhou 510630, Guangdong Province, China
-
Received:2012-07-29Revised:2012-11-09Online:2013-01-29Published:2013-03-08 -
Contact:Liu Zhi-hong, Academician, Doctoral supervisor, Research Institute of Nephrology, School of Medicine, Nanjing University/Nanjing General Hospital of Nanjing Military Command, PLA, Nanjing 210002, Jiangsu Province, China zhihong--liu@hotmail.com -
About author:Ji Shu-ming, Chief physician, Research Institute of Nephrology, School of Medicine, Nanjing University/Nanjing General Hospital of Nanjing Military Command, PLA, Nanjing 210002, Jiangsu Province, China jishuming@medmail.com.cn -
Supported by:The National Natural Science Foundation of China, No.81270834
CLC Number:
Cite this article
Ji Shu-ming1, Chen Jiang-hua2, Tan Jian-ming3, Zhang Xiao-dong4, Zhu Tong-yu5, Chen Li-zhong. Application of mycophenolate mofetil combined with low-dose tacrolimus and corticosteroid regimen after renal transplantation[J]. Chinese Journal of Tissue Engineering Research, 2013, 17(5): 777-784.
share this article
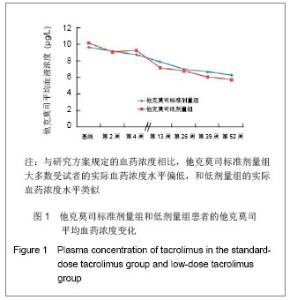
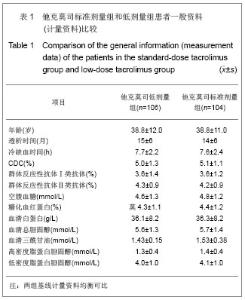
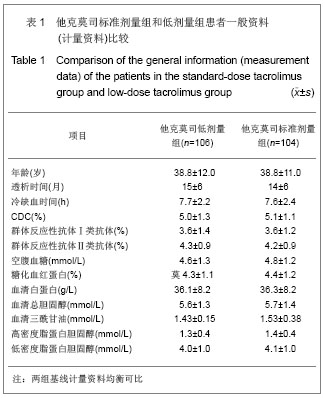
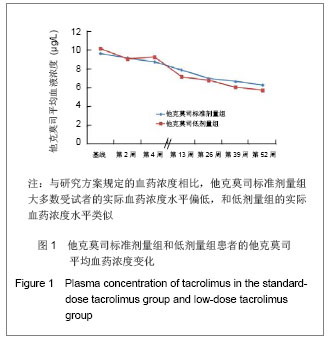
肾移植后不同时间点他克莫司标准剂量组和低剂量组他克莫司实际平均血药浓度水平类似,具体变化情况见图1。他克莫司标准剂量组患者基线、移植后2,4,13,26,39和52周的血药浓度低于方案要求范围的比例分别为59.0%,59.4%,68.0%,79.3%,70.5%,75.0%和83.9%。 在皮质激素使用方面,他克莫司标准剂量组基线、肾移植后2,4,13,26,39和52周,平均给药剂量分别为450.87,23.97,20.77,11.88,9.44,8.60和 8.10 mg/d,而对应的他克莫司低剂量组则分别为431.48,22.93,19.05,11.83,9.83,8.59和7.76 mg/d。"
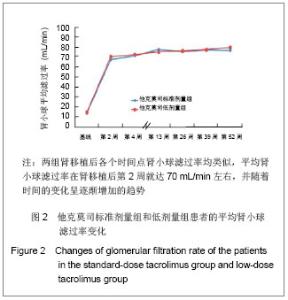
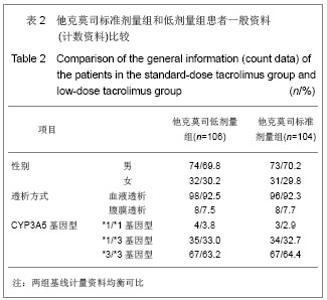
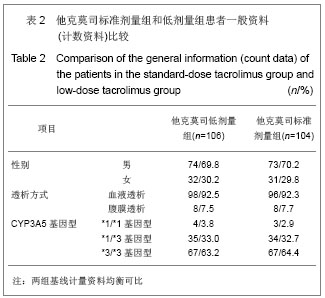
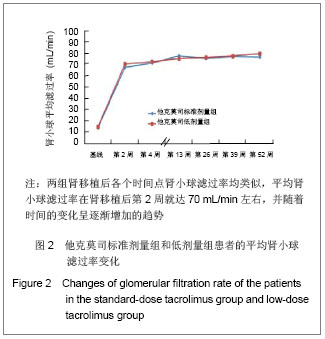
2.3 临床疗效评价 肾移植第12个月肾脏病理改变的CADI评分平均1.97分,其中他克莫司标准剂量组的平均CADI评分为1.82分,他克莫司低剂量组的平均CADI评分为2.13分,两组比较差异无显著性意义(P=0.081 3)。 肾移植第12个月的平均肾小球滤过率为 78.56 mL/min,其中他克莫司标准剂量组平均肾小球滤过率为77.08 mL/min,他克莫司低剂量组平均肾小球滤过率为80.12 mL/min,两组比较差异无显著性意义(P=0.794 9)。肾移植后各个时间点平均肾小球滤过率的变化见图2。 肾移植后6个月和12个月急性排斥反应发生率都仅为3.9%,他克莫司标准剂量组和低剂量组分别为2.6%和5.2%,两组差异无显著性意义(P=0.681 2)。 肾移植后6个月内和12个月内均未发生移植物丢失。移植后6个月内未出现死亡病例,而移植后12个月内,他克莫司低剂量组1例患者死亡。患者和移植物存活率均为99.5%,其中他克莫司标准剂量组和低剂量组人存活率分别为100%和99.1%,两组差异无显著性意义(P=1.000)。 17例患者(8.1%)肾移植后12个月内治疗失败,其中他克莫司标准剂量组为10例(9.6%),他克莫司低剂量组为7例(6.6%),两组差异无显著性意义(P=0.458 6)。在治疗失败的患者中,1例患者死亡,16例患者使用了研究方案对该治疗组规定以外的其他维持治疗的免疫抑制药物,其中8例使用了环孢素,6例使用了咪唑立宾,1例使用了来氟米特和1例使用了麦考酚钠肠溶片。 2.4 安全性评价 仅有8.6%的患者(他克莫司标准剂量组8.7%,他克莫司低剂量组8.5%)出现白细胞计数减少,2.4%的患者(他克莫司标准剂量组2.9%,他克莫司低剂量组1.9%)新发糖尿病,3.3%的患者(他克莫司标准剂量组2.9%,他克莫司低剂量组3.8%)新发高血脂,3.8%的患者出现感染(他克莫司标准剂量组3.8%,他克莫司低剂量组3.8%),0.5%的患者出现机会性感染(他克莫司标准剂量组无,他克莫司低剂量组0.9%),所有患者均未出现贫血、中性粒细胞减少、新发血管疾病、新发高血压、新发恶性肿瘤以及移植后最初6个月需要干预的囊状淋巴瘤。 出现不良事件的发生率为34.8%(他克莫司标准剂量组31.7%,他克莫司低剂量组37.7%),最常见的治疗出现不良事件为谷丙转氨酶升高(10.5%),白细胞计数减少(8.6%)和腹泻(5.7%)。绝大多数治疗出现不良事件为轻度或中度。 6例受试者(2.9%)[他克莫司标准剂量组1例(1.0%),他克莫司低剂量组5例(4.7%)]报告发生严重不良事件,分别为心血管意外1例、移植肾破裂1例、急性肾移植排斥2例、移植肾输尿管梗阻1例和巨细胞病毒感染1例,其中3例严重不良事件被研究者认为与研究药物相关,分别为急性肾移植排斥2例和巨细胞病毒感染1例。他克莫司低剂量组1例受试者因发生心血管意外严重不良事件导致死亡。本研究临床实验室检查和生命体征均未发现临床显著意义的变化。"
| [1] Li L, Sun Q. Renal transplantation in China: ten years of experience at Nanjing Jinling Hospital. Clin Transpl. 2006: 71-77.[2] Lodhi SA, Meier-Kriesche HU. Kidney allograft survival: the long and short of it. Nephrol Dial Transplant. 2011;26(1):15-17.[3] Morales JM, Marcén R, Del Castillo D, et al. Risk factors for graft loss and mortality after renal transplantation according to recipient age: a prospective multicentre study.Nephrol Dial Transplant. 2012;27 Suppl 4:iv39-iv46.[4] Phelan PJ, O'Kelly P, Tarazi M,et al. Renal allograft loss in the first post-operative month: causes and consequences.Clin Transplant. 2012;26(4):544-549.[5] Ekberg H, Bernasconi C, Nöldeke J, et al. Cyclosporine, tacrolimus and sirolimus retain their distinct toxicity profiles despite low doses in the Symphony study. Nephrol Dial Transplant. 2010;25(6):2004-2010.[6] Arend SM, Mallat MJ, Westendorp RJ,et al. Patient survival after renal transplantation; more than 25 years follow-up. Nephrol Dial Transplant. 1997;12(8):1672-1679.[7] Ekberg H, Tedesco-Silva H, Demirbas A, et al. Reduced exposure to calcineurin inhibitors in renal transplantation. N Engl J Med. 2007;357(25):2562-2575.[8] Matas AJ. Chronic progressive calcineurin nephrotoxicity: an overstated concept. Am J Transplant. 2011;11(4):687-692.[9] Tan TC,Robinson PJ.Mechanisms of calcineurin inhibitor-induced neurotoxicity.Transplantation Reviews.2006; 20(1):49-60.[10] Djamali A, Reese S, Hafez O, et al. Nox2 is a mediator of chronic CsA nephrotoxicity.Am J Transplant. 2012;12(8): 1997-2007.[11] Matas AJ.Chronic progressive calcineurin nephrotoxicity: an overstated concept.Am J Transplant. 2011;11(4):687-692.[12] Pascual M, Theruvath T, Kawai T, et al. Strategies to improve long-term outcomes after renal transplantation. N Engl J Med. 2002;346(8):580-590.[13] Burkhalter F, Oettl T, Descoeudres B, et al. High incidence of rejection episodes and poor tolerance of sirolimus in a protocol with early steroid withdrawal and calcineurin inhibitor-free maintenance therapy in renal transplantation: experiences of a randomized prospective single-center study. Transplant Proc. 2012;44(10):2961-2965.[14] Roberts DM, Jiang SH, Chadban SJ.The treatment of acute antibody-mediated rejection in kidney transplant recipients-a systematic review.Transplantation. 2012;94(8):775-783.[15] Isoniemi HM, Krogerus L, von Willebrand E,et al. Histopathological findings in well-functioning, long-term renal allografts. Kidney Int. 1992;41(1):155-160.[16] Cockcroft DW, Gault MH. Prediction of creatinine clearance from serum creatinine. Nephron. 1976;16(1):31-41.[17] Poggio ED, Wang X, Greene T, et al. Performance of the modification of diet in renal disease and Cockcroft-Gault equations in the estimation of GFR in health and in chronic kidney disease. J Am Soc Nephrol. 2005;16(2):459-466.[18] Qi J,Min ZL,Chang JW,et al. Zhonghua Qiguan Yizhi Zazhi. 2000;21(5):288-291.齐隽,闵志廉,常继伟,等.肾移植后免疫抑制用药方案与移植肾长期存活的关系[J].中华器官移植杂志,2000,21(5):288-291.[19] Xu D,Liu Y,Wang XH,et al. Zhonghua Qiguan Yizhi Zazhi. 2000; 21(5):285-287.徐达,刘永,王祥慧,等.用于肾移植的几种免疫抑制方案的对比研究[J]. 中华器官移植杂志,2000,21(5):285-287.[20] Morris RE.The future of immunosuppression: a personal view.Transplant Proc. 2004;36(2 Suppl):577S-579S.[21] Hardinger KL, Schnitzler MA, Koch MJ, et al. Cyclosporine minimization and cost reduction in renal transplant recipients receiving a C2-monitored, cyclosporine-based quadruple immunosuppressive regimen.Transplantation. 2004;78(8): 1198-1203.[22] Pang GX,Lu GC. Zhongguo Yaofang. 2008;19(16):1211-1213.庞国勋,陆国椿.我国2004-2006年免疫抑制剂市场状况分析[J].中国药房, 2008,19(16):1211-1213.[23] Nankivell BJ, Wavamunno MD, Borrows RJ,et al. Mycophenolate mofetil is associated with altered expression of chronic renal transplant histology.Am J Transplant. 2007; 7(2):366-376.[24] Boudjema K, Camus C, Saliba F, et al. Reduced-dose tacrolimus with mycophenolate mofetil vs. standard-dose tacrolimus in liver transplantation: a randomized study. Am J Transplant. 2011;11(5):965-976.[25] Cortesini R.Cyclosporine--lessons from the first 20 years. Transplant Proc. 2004;36(2 Suppl):158S-162S.[26] Burdmann EA, Andoh TF, Yu L, et al. Cyclosporine nephrotoxicity. Semin Nephrol. 2003;23(5):465-476.[27] Bakker RC,Scholten EM,de Fijter JW,et al. Chronic cyclosporine nephrotoxicity in renal transplantation. Transplantation Reviews.2004;18(1):54-64.[28] Maes BD, Vanrenterghem YF.Cyclosporine: advantages versus disadvantages vis-à-vis tacrolimus.Transplant Proc. 2004;36(2 Suppl):40S-49S.[29] Lucan M, Iacob G, Lucan C, et al.Ten years of cyclosporine use in renal transplantation: a single-center experience with 479 renal transplants.Transplant Proc. 2004;36(2 Suppl): 177S-180S.[30] Weimer R, Deisz S, Dietrich H, et al. Impact of maintenance immunosuppressive regimens--balance between graft protective suppression of immune functions and a near physiological immune response.Transpl Int. 2011;24(6): 596-609. [31] Naesens M, Lerut E, Sarwal M, et al. Balancing efficacy and toxicity of kidney transplant immunosuppression.Transplant Proc. 2009;41(8):3393-3395.[32] Jørgensen KA, Povlsen JV, Poulsen JH. Optimal time for determination of blood tacrolimus level.Transplant Proc. 2001;33(7-8):3164-3165.[33] Ji SM,Chen JS,Sun QQ,et al. Yixue Yanjiusheng Xuebao. 2011;24(1):34-38.季曙明,陈劲松,孙启全,等.环孢素转换至他克莫司治疗慢性移植肾肾病的临床和病理观察[J].医学研究生学报,2011,24(1): 34-38.[34] Zhu YH,Min ZL,Wang LM,et al. Zhonghua Qiguan Yizhi Zazhi. 2000;21(2):119-121.朱有华,闵志廉,王立明,等.普乐可复在肾移植中的临床应用[J].中华器官移植杂志, 2000,21(2):119-121.[35] Roberts DM, Jiang SH, Chadban SJ.The treatment of acute antibody-mediated rejection in kidney transplant recipients-a systematic review.Transplantation. 2012;94(8):775-783.[36] Ji SM, Liu ZH, Chen JS, et al. Rescue therapy by immunoadsorption in combination with tacrolimus and mycophenolate mofetil for C4d-positive acute humoral renal allograft rejection.Transplant Proc. 2006;38(10):3459-3463.[37] Ji SM, Wen JQ, Cheng DR, et al.Conversion from Calcineurin Inhibitors to Sirolimus Maintenance Therapy in Renal Allograft Recipients with Risk Factors. Open Journal of Organ Transplant Surgery. 2011;1(1): 8-13. [38] Ji SM, Liu ZH, Wu D,et al.Surveillance Renal Allograft Biopsy on Diagnosis of BK Virus Nephropathy in Chinese Renal Transplant Recipients. Open Journal of Organ Transplant Surgery. 2012;2(4):62-68.[39] John R, Herzenberg AM. Our approach to a renal transplant biopsy. J Clin Pathol. 2010;63(1):26-37.[40] Ji SM,Wen JQ,Cheng DR,et al. Qiguan Yizhi. 2012;3(2): 95-101.季曙明,文吉秋,程东瑞,等.采用程序化肾活检评估肾移植后低水平和标准水平他克莫司的疗效[J].器官移植,2012,3(2):95-101.[41] Matas AJ. Chronic progressive calcineurin nephrotoxicity: an overstated concept. Am J Transplant. 2011;11(4):687-692.[42] Ji SM, Li LS, Sun QQ,et al. Immunoregulation of thymosin alpha 1 treatment of cytomegalovirus infection accompanied with acute respiratory distress syndrome after renal transplantation.Transplant Proc. 2007;39(1):115-119.[43] Zu Q,Yao B,Yu LX. Shenzangbing yu Touxi Shenyizhi Zazhi. 2000;9(3):251-253.祖强,姚冰,于立新.FK506的肾毒性[J].肾脏病与透析肾移植杂志,2000,9(3):251-253.[44] Kidokoro K, Satoh M, Nagasu H,et al.Tacrolimus Induces Glomerular Injury via Endothelial Dysfunction Caused by Reactive Oxygen Species and Inflammatory Change.Kidney Blood Press Res. 2012;35(6):549-557.[45] Gijsen VM, Madadi P, Dube MP, et al. Tacrolimus-induced nephrotoxicity and genetic variability: a review.Ann Transplant. 2012;17(2):111-121.[46] Horike K, Takeda A, Yamaguchi Y, et al. Is arteriolar vacuolization a predictor of calcineurin inhibitor nephrotoxicity. Clin Transplant. 2011;25 Suppl 23:23-27.[47] Pirsch JD, Miller J, Deierhoi MH, et al. A comparison of tacrolimus (FK506) and cyclosporine for immunosuppression after cadaveric renal transplantation. FK506 Kidney Transplant Study Group.Transplantation. 1997;63(7):977-983.[48] Weir MR, Ward MT, Blahut SA, et al. Long-term impact of discontinued or reduced calcineurin inhibitor in patients with chronic allograft nephropathy. Kidney Int. 2001;59(4): 1567-1573.[49] Duijnhoven EM, Boots JM, Christiaans MH, et al. Influence of tacrolimus on glucose metabolism before and after renal transplantation: a prospective study. J Am Soc Nephrol. 2001; 12(3):583-588.[50] First MR, Gerber DA, Hariharan S,et al. Posttransplant diabetes mellitus in kidney allograft recipients: incidence, risk factors, and management.Transplantation. 2002;73(3): 379-386.[51] Campistol JM. Long-term maintenance therapy with calcineurin inhibitors: an update.Transplant Proc. 2010;42 (9 Suppl):S21-24.[52] Undre NA, Stevenson P, Schäfer A. Pharmacokinetics of tacrolimus: clinically relevant aspects.Transplant Proc. 1999; 31(7A):21S-24S.[53] Marcén R. Immunosuppressive drugs in kidney transplantation: impact on patient survival, and incidence of cardiovascular disease, malignancy and infection. Drugs. 2009;69(16):2227-2243. [54] Meier-Kriesche HU, Merville P, Tedesco-Silva H,et al. Mycophenolate mofetil initiation in renal transplant patients at different times posttransplantation: the TranCept Switch study. Transplantation. 2011;91(9):984-990.[55] Sollinger HW. Mycophenolate mofetil for the prevention of acute rejection in primary cadaveric renal allograft recipients. U.S. Renal Transplant Mycophenolate Mofetil Study Group. Transplantation. 1995;60(3):225-232.[56] European Mycophenolate Mofetil Cooperative Study Group. Placebo-controlled study of mycophenolate mofetil combined with cyclosporin and corticosteroids for prevention of acute rejection. Lancet. 1995;345(8961):1321-1325.[57] The Tricontinental Mycophenolate Mofetil Renal Transplantation Study Group. A blinded, randomized clinical trial of mycophenolate mofetil for the prevention of acute rejection in cadaveric renal transplantation. Transplantation. 1996;61(7):1029-1037. [58] Stegall MD, Park WD, Larson TS,et al. The histology of solitary renal allografts at 1 and 5 years after transplantation. Am J Transplant. 2011;11(4):698-707. |
| [1] | Jiang Jun, Zhang Bin. Analyzing current status of stem cell research based on National Natural Science Foundation of China's completed projects from 2018 to 2022 [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(19): 4145-4150. |
| [2] | Ma Shuwei, He Sheng, Han Bing, Zhang Liaoyun. Exosomes derived from mesenchymal stem cells in treatment of animals with acute liver failure: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1137-1142. |
| [3] | Chen Qian, Zhang Yang, Li Gaofeng. Effects of mycophenolate mofetil on the development of rat embryonic auricle [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(24): 3818-3823. |
| [4] | Wang Yaoquan, Xuan Juanjuan, Zhang Jixiang, Chen Kaige, Chen Guoyong, Wei Sidong. Mechanism of Kupffer cells in immune tolerance after liver transplantation [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(24): 3901-3909. |
| [5] | Fu Xinya, Ma Jinjin, Li Meimei, Wang Xingran, Xie Jile, Saijilafu. Establishment of an in vitro model of H2O2-induced neuronal senescence [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(11): 1733-1738. |
| [6] | Huang Chuwen, Jiang Hua, Li Minqing. Complications and death causes of peripheral blood stem cell transplantation in the treatment of thalassemia major [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(1): 42-48. |
| [7] | Xuan Juanjuan, Bai Hongtai, Zhang Jixiang, Wang Yaoquan, Chen Guoyong, Wei Sidong. Role of regulatory T cell subsets in liver transplantation and progress in clinical application [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1143-1148. |
| [8] | Yang Zhiwei, Liu Junchang, Gao Xiaolin, Jiang Taimao. Relationship between tacrolimus metabolic rate and early BK virus infection after kidney transplantation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 712-716. |
| [9] | Xu Yuan, Niu Yulin, Yuan Zhihui, Jia Lei, Pan Guanghui. Up-regulating follicular regulatory T cells for treating antibody-mediated rejection after kidney transplantation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(26): 4118-4122. |
| [10] | Zhang Huabin, Zhang Muchen, Liu Chang, Xu Manman, Yin Qichuan, Zhang Aijun. Viability of different fat derivatives in vitro and the outcome after transplantation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(24): 3779-3784. |
| [11] | Min Keting, Zhang Yiguo, Yang Hao, Lü Xin. Effect of mesenchymal stem cells on negative regulation of immune response of myeloid-derived suppressor cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(19): 3084-3089. |
| [12] | Zhu Dongming, Zhang Zhen, Zhang Jie, Yan Lianqi. Topical application of mycophenolate mofetil prevents epidural fibrosis by inhibiting the proliferation and migration of fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(16): 2551-2556. |
| [13] | Jiang Xin, Qiao Liangwei, Sun Dong, Li Ming, Fang Jun, Qu Qingshan. Expression of long chain non-coding RNA PGM5-AS1 in serum of renal transplant patients and its regulation of human glomerular endothelial cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 741-745. |
| [14] | Yang Xin, Jin Zhe, Feng Xu, Lu Bing. The current situation of knowledge and attitudes towards organ, eye tissue, body donation of residents in Shenyang [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 779-784. |
| [15] | Gu Honglin, Zheng Xiaoqing, Liang Changxiang, Zeng Shixing, Zhan Shiqiang, Chang Yunbing. Long-term survival rate and reoperation of Wallis interspinous dynamic stabilization system for treatment of lumbar degenerative disc disease [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(30): 4863-4869. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||