Chinese Journal of Tissue Engineering Research ›› 2025, Vol. 29 ›› Issue (6): 1176-1182.doi: 10.12307/2025.323
Previous Articles Next Articles
Mechanism of agomelatine alleviating anxiety- and depression-like behaviors in APP/PS1 transgenic mice #br#
#br#
Li Tian1, Ren Yuhua2, Gao Yanping2, Su Qiang1, 2
- 1Department of Physiology, School of Basic Medicine; Key Laboratory of Cellular Physiology, Ministry of Education; Shanxi Key Laboratory of Cell Physiology, Shanxi Medical University, Taiyuan 030001, Shanxi Province, China; 2Department of Medical Laboratory, Fenyang College of Shanxi Medical University, Fenyang 032200, Shanxi Province, China
-
Received:2024-02-07Accepted:2024-04-17Online:2025-02-28Published:2024-06-21 -
Contact:Li Tian, Department of Physiology, School of Basic Medicine; Key Laboratory of Cellular Physiology, Ministry of Education; Shanxi Key Laboratory of Cell Physiology, Shanxi Medical University, Taiyuan 030001, Shanxi Province, China Co-corresponding author: Su Qiang, Technician, Department of Physiology, School of Basic Medicine; Key Laboratory of Cellular Physiology, Ministry of Education; Shanxi Key Laboratory of Cell Physiology, Shanxi Medical University, Taiyuan 030001, Shanxi Province, China; Department of Medical Laboratory, Fenyang College of Shanxi Medical University, Fenyang 032200, Shanxi Province, China -
About author:Li Tian, PhD, Lecturer, Department of Physiology, School of Basic Medicine; Key Laboratory of Cellular Physiology, Ministry of Education; Shanxi Key Laboratory of Cell Physiology, Shanxi Medical University, Taiyuan 030001, Shanxi Province, China -
Supported by:the National Natural Science Foundation of China, No. 82301631 (to LT); the Natural Science Research Youth Program of Shanxi Provincial Department of Science and Technology, No. 20210302124086 (to LT)
CLC Number:
Cite this article
Li Tian, Ren Yuhua, Gao Yanping, Su Qiang. Mechanism of agomelatine alleviating anxiety- and depression-like behaviors in APP/PS1 transgenic mice #br#
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
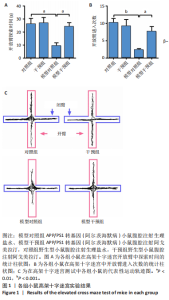
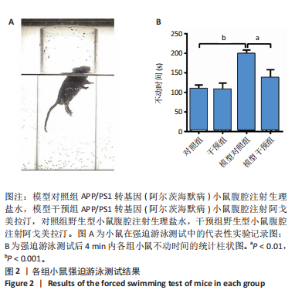
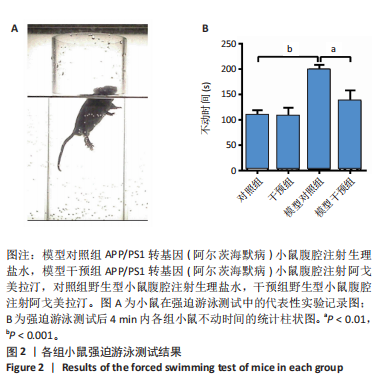
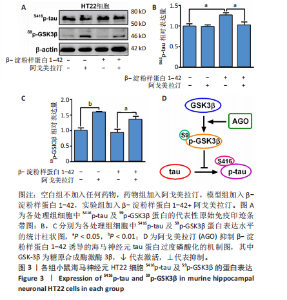
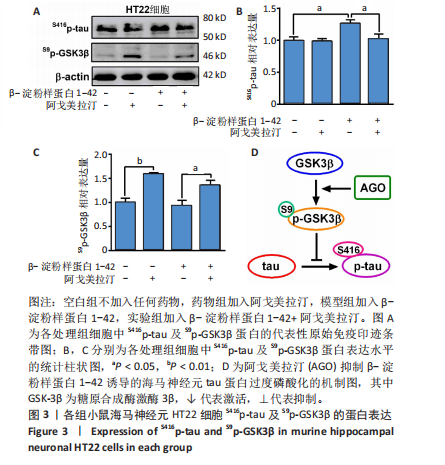
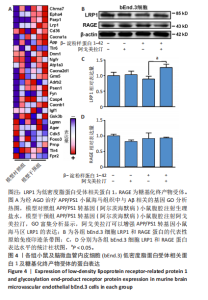
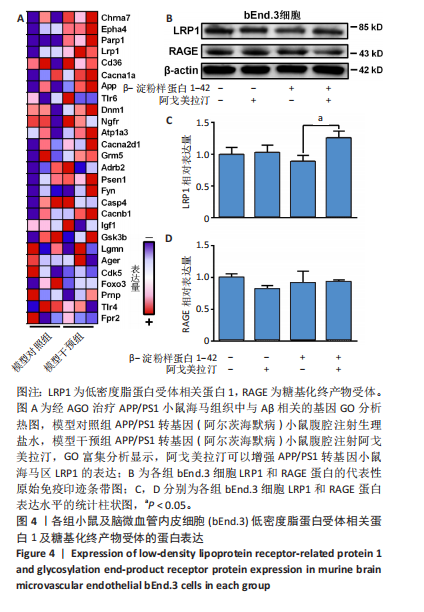
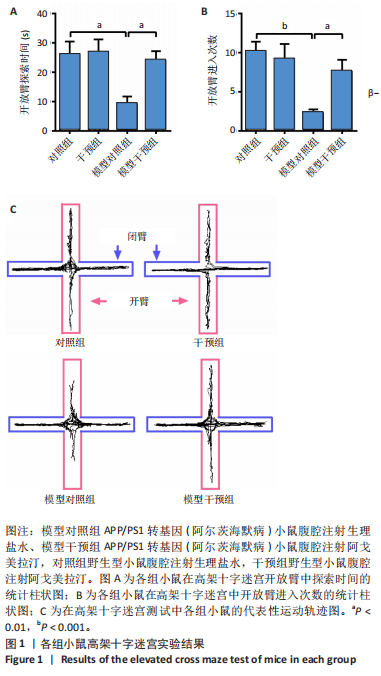
2.1 动物实验结果 2.1.1 实验动物数量分析 18只APP/PS1转基因小鼠与18只野生型小鼠全部进入结果分析。 2.1.2 阿戈美拉汀明显缓解了APP/PS1转基因小鼠的焦虑样行为 在高架十字迷宫测试中,与对照组小鼠相比,模型对照组小鼠开臂探索时间缩短[(26.156±4.315),(9.333±2.409) s,P < 0.01],提示9月龄APP/PS1转基因小鼠已伴有焦虑样行为出现;经阿戈美拉汀治疗后,模型干预组小鼠开臂探索时间长于模型对照组[(24.191±3.022),(9.333±2.409) s,P < 0.01],见图1A,表明阿戈美拉汀有效改善了APP/PS1转基因小鼠表现出的焦虑样行为。同时,模型对照组小鼠开放臂进入次数少于对照组(2.333±0.441,10.222±1.188,P < 0.001),模型干预组小鼠开放臂进入次数多于模型对照组(7.667±1.434,2.333±0.441,P < 0.01),见图1B。这些结果表明,APP/PS1转基因小鼠表现出焦虑样行为,而阿戈美拉汀有效改善了APP/PS1转基因小鼠表现出的焦虑样行为。 2.1.3 阿戈美拉汀有效改善了APP/PS1转基因小鼠的抑郁样行为 在强迫游泳测试中,模型对照组小鼠强迫游泳实验后4 min内的不动时间长于对照组[(199.222±9.334),(108.667±10.603) s,P < 0.001],提示9月龄APP/PS1转基因小鼠已伴有抑郁样行为出现;模型干预组小鼠强迫游泳实验后4 min内的不动时间短于模型对照组[(137.444±20.626),(199.222±9.334) s,P < 0.01],见图2,结果表明阿戈美拉汀可以明显减轻APP/PS1转基因小鼠的抑郁样行为。 2.2 阿戈美拉汀抑制β-淀粉样蛋白1-42诱导的HT22细胞tau蛋白过度磷酸化并促进S9p-GSK3β表达 阿尔茨海默病脑内神经元tau过度磷酸化病理与其异常的精神行为表现有关[13-14]。为此,此次实验进一步利用培养的HT22细胞进行了S416p-tau和S9p-GSK3β蛋白表达检测,以检测阿戈美拉汀对β-淀粉样蛋1-42所致神经元tau蛋白磷酸化的影响。如图3所示,模型组HT22细胞的S416p-tau蛋白表达高于空白组(P < 0.05),实验组HT22细胞的S416p-tau蛋白表达低于模型组(P < 0.05),表明阿戈美拉汀处理可有效抑制β-淀粉样蛋1-42诱导的HT22细胞tau蛋白过度磷酸化;药物组HT22细胞的S9p-GSK3β蛋白表达高于空白组(P < 0.01),实验组HT22细胞的S9p-GSK3β蛋白表达高于模型组(P < 0.05),提示阿戈美拉汀可能是通过促进S9p-GSK3β表达转而抑制β-淀粉样蛋白1-42诱导的海马神经元tau蛋白过度磷酸化,以拮抗β-淀粉样蛋白所致的神经元损伤,从而缓解神经元tau相关的精神行为异常。 2.3 阿戈美拉汀提高bEnd.3细胞LRP1蛋白的表达 通过对小鼠海马组织mRNA测序结果进一步分析(图4A),GO富集分析显示,阿戈美拉汀可以增强APP/PS1转基因小鼠海马区LRP1的蛋白表达。LRP1被认为是分布在血脑屏障上皮细胞上的一种转运受体,参与β-淀粉样蛋白和tau的向外周转运和清除[15-16]。相反,糖基化终产物受体蛋白是表达在血脑屏障上皮细胞上介导外周β-淀粉样蛋白进入脑内的关键受体[17]。因此,为了很好地模拟和检测APP/PS1转基因小鼠脑内微血管内皮细胞LRP1和糖基化终产物受体的蛋白表达情况,利用bEnd.3细胞和免疫印迹手段检测了β-淀粉样蛋白1-42存在的情况下阿戈美拉汀对bEnd.3细胞LRP1和糖基化终产物受体蛋白表达的影响。结果表明,β-淀粉样蛋白1-42处理可诱导bEnd.3细胞LRP1出现下调趋势,同时给予阿戈美拉汀和β-淀粉样蛋白1-42处理的bEnd.3细胞LRP1蛋白表达升高 (P < 0.05);各组bEnd.3细胞糖基化终产物受体蛋白表达比较差异无显著性意义(P > 0.05),见图4B-D。结果表明,阿戈美拉汀可能在不影响糖基化终产物受体蛋白表达的情况下诱导阿尔茨海默病小鼠脑内微血管内皮细胞LRP1蛋白表达升高,进而促进阿尔茨海默病脑内β-淀粉样蛋白和tau从中枢向外周转运和清除。"
| [1] CACABELOS R. What have we learnt from past failures in Alzheimer’s disease drug discovery? Expert Opin Drug Discov. 2022;17(4):309-323. [2] BUSCHE MA, HYMAN BT. Synergy between amyloid-β and tau in Alzheimer’s disease. Nat Neurosci. 2020;23(10):1183-1193. [3] CHEN Y, DANG M, ZHANG Z. Brain mechanisms underlying neuropsychiatric symptoms in Alzheimer’s disease: a systematic review of symptom-general and -specific lesion patterns. Mol Neurodegener. 2021;16(1):38. [4] GRACIA-GARCIA P, BUENO-NOTIVOL J, LIPNICKI DM, et al. Clinically significant anxiety as a risk factor for Alzheimer’s disease: Results from a 10-year follow-up community study. Int J Methods Psychiatr Res. 2023;32(3):e1934. [5] BOTTO R, CALLAI N, CERMELLI A, et al. Anxiety and depression in Alzheimer’s disease: a systematic review of pathogenetic mechanisms and relation to cognitive decline. Neurol Sci. 2022;43(7):4107-4124. [6] HASAN SMN, CLARKE C, STRAND T, et al. Putative pathological mechanisms of late-life depression and Alzheimer’s disease. Brain Res. 2023;1813:148423. [7] PATEL P, MASURKAR AV. The relationship of anxiety with Alzheimer’s disease: a narrative review. Curr Alzheimer Res. 2021;18(5):359-371. [8] CACABELOS R, CARRIL JC, CORZO L, et al. Pharmacogenetics of anxiety and depression in Alzheimer’s disease. Pharmacogenomics. 2023;24(1):27-57. [9] SU Q, LI T, LIU GW, et al. Agomelatine: a potential novel approach for the treatment of memory disorder in neurodegenerative disease. Neural Regen Res. 2023;18(4):727-733. [10] LI T, SU Q, ZHANG Z, et al. Ube2c-inhibition alleviated amyloid pathology and memory deficits in APP/PS1 mice model of AD. Prog Neurobiol. 2022;215:102298. [11] KRAEUTER AK, GUEST PC, SARNYAI Z. The elevated plus maze test for measuring anxiety-Like behavior in rodents. Methods Mol Biol. 2019; 1916:69-74. [12] YANKELEVITCH-YAHAV R, FRANKO M, HULY A, et al. The forced swim test as a model of depressive-like behavior. J Vis Exp. 2015(97):52587. [13] KRELL-ROESCH J, RAKUSA M, SYRJANEN JA, et al. Association between CSF biomarkers of Alzheimer’s disease and neuropsychiatric symptoms: Mayo Clinic Study of Aging. Alzheimers Dement. 2023;19(10):4498-4506. [14] LEYDER E, SURESH P, JUN R, et al. Depression-related phenotypes at early stages of Aβ and tau accumulation in inducible Alzheimer’s disease mouse model: Task-oriented and concept-driven interpretations. Behav Brain Res. 2023;438:114187. [15] HUANG Z, LIN HWK, ZHANG Q, et al. Targeting Alzheimer’s disease: the critical crosstalk between the liver and brain. Nutrients. 2022; 14(20):4298. [16] RAUCH JN, LUNA G, GUZMAN E, et al. LRP1 is a master regulator of tau uptake and spread. Nature. 2020;580(7803):381-385. [17] DEANE R, DU YAN S, SUBMAMARYAN RK, et al. RAGE mediates amyloid-beta peptide transport across the blood-brain barrier and accumulation in brain. Nat Med. 2003;9(7):907-913. [18] PASSERI E, ELKHOURY K, MORSINK M, et al. Alzheimer’s disease: treatment strategies and their limitations. Int J Mol Sci. 2022;23(22):13954. [19] LIVINGSTON G, HUNTLEY J, SOMMERLAD A, et al. Dementia prevention, intervention, and care: 2020 report of the Lancet Commission. Lancet. 2020;396(10248):413-446. [20] JIA L, DU Y, CHU L, et al. Prevalence, risk factors, and management of dementia and mild cognitive impairment in adults aged 60 years or older in China: a cross-sectional study. Lancet Public Health. 2020; 5(12):e661-e671. [21] ASMER MS, KIRKHAM J, NEWTON H, et al. Meta-analysis of the prevalence of major depressive disorder among older adults with dementia. J Clin Psychiatry. 2018;79(5):17r11772. [22] CHEN P, GUARINO PD, DYSKEN MW, et al. Neuropsychiatric symptoms and caregiver burden in individuals with Alzheimer’s disease: the TEAM-AD VA cooperative study. J Geriatr Psychiatry Neurol. 2018;31(4): 177-185. [23] LEUNG DKY, CHAN WC, SPECTOR A, et al. Prevalence of depression, anxiety, and apathy symptoms across dementia stages: A systematic review and meta-analysis. Int J Geriatr Psychiatry. 2021;36(9):1330-1344. [24] JOURDAN JP, BUREAU R, ROCHAIS C, et al. Drug repositioning: a brief overview. J Pharm Pharmacol. 2020;72(9):1145-1151. [25] SAVINO R, POLITO AN, MARSALA G, et al. Agomelatine: a potential multitarget compound for neurodevelopmental disorders. Brain Sci. 2023;13(5):734. [26] MILLAN MJ. Agomelatine for the treatment of generalized anxiety disorder: focus on its distinctive mechanism of action. Ther Adv Psychopharmacol. 2022;12:20451253221105128. [27] YU Y, CHEN Y, MA L, et al. Efficacy of agomelatine with cognitive behavioral therapy for delayed sleep-wake phase disorder in young adults: A randomized controlled study. Behav Sleep Med. 2023;21(5): 529-539. [28] CHEN Y, LI J, LIAO M, et al. Efficacy and safety of agomelatine versus SSRIs/SNRIs for post-stroke depression: a systematic review and meta-analysis of randomized controlled trials. Int Clin Psychopharmacol. 2024;39(3):163-173. [29] ARMSTRONG SM, MCNULTY OM, GUARDIOLA-LEMAITRE B, et al. Successful use of S20098 and melatonin in an animal model of delayed sleep-phase syndrome (DSPS). Pharmacol Biochem Behav. 1993;46(1):45-49. [30] YOUS S, ANDRIEUX J, HOWELL HE, et al. Novel naphthalenic ligands with high affinity for the melatonin receptor. J Med Chem. 1992;35(8):1484-1486. [31] MILLAN MJ, GOBERT A, LEJEUNE F, et al. The novel melatonin agonist agomelatine (S20098) is an antagonist at 5-hydroxytryptamine2C receptors, blockade of which enhances the activity of frontocortical dopaminergic and adrenergic pathways. J Pharmacol Exp Ther. 2003; 306(3):954-964. [32] STAHL SM. Mechanism of action of agomelatine: a novel antidepressant exploiting synergy between monoaminergic and melatonergic properties. CNS Spectr. 2014;19(3):207-212. [33] MENDEZ MF. The relationship between anxiety and Alzheimer’s disease. J Alzheimers Dis Rep. 2021;5(1):171-177. [34] DAFSARI FS, JESSEN F. Depression-an underrecognized target for prevention of dementia in Alzheimer’s disease. Transl Psychiatry. 2020;10(1):160. [35] SAIZ-VAZQUEZ O, GRACIA-GARCIA P, UBILLOS-LANDA S, et al. Depression as a risk factor for Alzheimer’s disease: a systematic review of longitudinal meta-analyses. J Clin Med. 2021;10(9):1809. [36] BABULAL GM, ROE CM, STOUT SH, et al. Depression is associated with tau and not amyloid positron emission tomography in cognitively normal adults. J Alzheimers Dis. 2020;74(4):1045-1055. [37] EGASHIRA N, IWASAKI K, TAKASHIMA A, et al. Altered depression-related behavior and neurochemical changes in serotonergic neurons in mutant R406W human tau transgenic mice. Brain Res. 2005;1059(1):7-12. |
| [1] | Zhang Di, Zhao Jun, Ma Guangyue, Sun Hui, Jiang Rong. Mechanism of depression-like behavior in chronic social defeat stress mice based on high-throughput sequencing [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1139-1146. |
| [2] | Leng Xiaoxuan, Zhao Yuxin, Liu Xihua. Effects of different neuromodulatory stimulation modalities on non-motor symptoms in Parkinson’s patients: a network meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1282-1293. |
| [3] | He Qiwang, , , Chen Bo, Liang Fuchao, Kang Zewei, Zhou Yuan, Ji Anxu, Tang Xialin, . Relationship between Alzheimer’s disease and sarcopenia and body mass index: analysis of GWAS datasets for European populations [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 1036-1046. |
| [4] | Xu Dongfang, Zhao Kun, Lu Changzhu, Wang Yuge, Bai Lianjie, Meng Fanmou, Wang Yang, , Yao Hongbo. m6A-related ferroptosis gene expression and its association with immune infiltration in Alzheimer’s disease: machine learning and molecular biology validation [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6421-6432. |
| [5] | Shi Gaolong, Ge Caijun, Chen Jianpeng, Wang Yuanbin, Fan Zelin, Yan Jun, Wang Qianliang. Mechanism by which the paraventricular nucleus of the hypothalamus is involved in chronic pain and anxiety in mice with lumbar disc herniation [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(22): 5707-5715. |
| [6] | Pei Xiaxia, Li Tian, Zhang Yanli, Gao Yanping, Su Qiang. A novel treadmill-based method for assessing learning and memory in mice [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(18): 4694-4701. |
| [7] | Wang Jiaying, Xu Chun, Mayila · Abudukelimu. Global research status, trends and hotspots of anxiety/depression in chronic obstructive pulmonary disease [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(11): 2920-2932. |
| [8] | Gao Wenyan, Zheng Zhaoyan, Pan Shang, Wang Peipei, Ji Chunhui, Lyu Shaoping. Bibliometric and visual analysis of Theta burst transcranial magnetic stimulation [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(20): 4389-4400. |
| [9] | Wei Wei, Li Jian, Huang Linhai, Lan Mindong, Lu Xianwei, Huang Shaodong. Factors affecting fall fear in the first movement of elderly patients after total knee or hip arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1351-1355. |
| [10] | Gao Ziqing, Ruan Sibei, Li Li, Ling Feng, Tang Xiaoqin, Kang Qingmei, Luo Siyi, Luo Jing, Tang Yaping, Tang Mingxi . Identification and survival analysis of tTA/tetO-CCKR-2 double transgenic mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(11): 1682-1687. |
| [11] | Gao Zhixiang, Jiang Yishan, Long Nengji, Xiao Cong. Risk factors for postoperative delirium in elderly patients after total hip arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(32): 5097-5102. |
| [12] | Liang Xin, Wang Heng, Li Xian-rong. Preoperative application of alprazolam for patients with anxiety and depression and pain after total knee arthroplasty: its safety and effectiveness [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(7): 985-992. |
| [13] | Song De-hua, Wei Li-li, Song Zhen-lan, Zhao Xiao-li. Psychological conditions of patients in different stages of in vitro fertilization-embryo transfer cycles [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(5): 809-814. |
| [14] | Su Jing-han, Wang Shu-yan, Liang Xue, Zhu Mei-ling, Qiao Shan, Yin Hong-ying. Mode and effect of cognitive-behavioral therapy for liver transplant recipients waiting for a liver transplant [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(5): 687-692. |
| [15] | Hou Yan-hong, Zhao Shu-yan, Qi Qinjiazi, Liu Li-hui, Chen Xiao-fei, Shi Bing, Zhu Ling, Xu Chen, Jia Rui-ming, Wang Wei-wei, Yang Jing, Liang Yong-qing, Tan Yi, Li Fu-xing, Ye Li-ping. Mental health status of relative donors versus unrelated donors before and after hematopoietic stem cell collection [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(28): 4524-4529. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||